Beruflich Dokumente
Kultur Dokumente
Corson I Search
Hochgeladen von
api-3216259060 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
74 Ansichten13 SeitenBiochemistry allows scientists to better understand how macromolecules affect humans. Macromolecules do things from making humans unique to providing energy. Biochemistry is important on global, local, and personal levels.
Originalbeschreibung:
Originaltitel
corson i search
Copyright
© © All Rights Reserved
Verfügbare Formate
DOCX, PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenBiochemistry allows scientists to better understand how macromolecules affect humans. Macromolecules do things from making humans unique to providing energy. Biochemistry is important on global, local, and personal levels.
Copyright:
© All Rights Reserved
Verfügbare Formate
Als DOCX, PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
74 Ansichten13 SeitenCorson I Search
Hochgeladen von
api-321625906Biochemistry allows scientists to better understand how macromolecules affect humans. Macromolecules do things from making humans unique to providing energy. Biochemistry is important on global, local, and personal levels.
Copyright:
© All Rights Reserved
Verfügbare Formate
Als DOCX, PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 13
Running head: MACROMOLECULES
AND YOU
Macromolecules and You
Roark Corson
Ocean Lakes High School: Math and Science Academy
MACROMOLECULES AND YOU
Introduction
I walk into Walmart, and a shiny container catches my eye. I move closer,
wanting to get a better look. The container is full of protein, and the label reads, Build
more muscle with this Whey Protein!. Having been through molecular biology, I knew
a fair amount about proteins: how they are made, used, and their composition, but I knew
almost nothing about what they do for the human body. Im left wondering if protein can
really turn me into the muscular monster on the protein container, and resolve to find out
more about protein, and macromolecules in general.
Macromolecules are larger and more complex molecules that are normally created
by the polymerization of smaller subunits; like using LEGO bricks to make a building.
Macromolecules do things from making humans unique to providing energy for a walk
around a building. For this reason, they are important to every human across the world,
and the study of biochemistry allows scientists to better understand how they affect
humans, and how to optimize them to do even more for us. Because the research can be
applied to any human, it is of local and global importance, and biochemical research is
important and needs to be done. Personally, this was important to me because I like both
chemistry and biology, and biochemistry is a marriage of not just the names, but of the
two disciplines. I also like to help people, and altering the basis of human life, the
genome, is the best way to cure genetic issues, and is making rapid progress and will
soon be implemented in everyday life. Biochemistry is important on global, local, and
personal levels, and deserves to be researched and furthered due to its widespread
applications.
MACROMOLECULES AND YOU
The question I set out to research was How do macromolecules affect the human
body, and how can this information be used to help people? I also had five subquestions that helped me to focus on smaller areas of this larger question before putting it
all together. The answer to this question does not have a definitive answer; instead it has
some answers that have not been discovered yet, as experiments and research are still
being done that will find more applications for macromolecules and further their use to
humans.
What I Know, Assume, and Imagine
Before I started this research project, I knew about macromolecules in a very
vague way. I knew what they were, and how they worked, but I knew nothing about their
applications and the more specific knowledge that would be needed for this project. I had
five sub-questions, which are:
1) What are the subsets of molecules that are part of the four larger macromolecule
groups?
2) How are each of the macromolecules created and ingested by humans?
3) What is the relationship between macronutrients and macromolecules?
4) How are macromolecules altered both organically and synthetically?
5) Why are macromolecules essential to life?
My knowledge mostly covered the first question, and I knew a little about the
second question, but my work was mostly cut out for me. I designed these questions with
a simple idea in mind: building up. I would start with the basics, like the types of
macromolecules, and then work up the applications and why they are so important. The
first question was mostly to set the groundwork for the rest of my research and make sure
MACROMOLECULES AND YOU
my current knowledge was accurate. The second and third question deal with how
humans use macromolecules, both as regular macromolecules, and as macronutrients,
which are energy providing nutrients for the human body. The penultimate question deals
with both inorganic and organic alteration of macromolecules; an important topic for
chemists, as some macromolecular structures need to be altered to be most effective. The
final question is the largest one, and deals with the why of organic molecules: why do
we need them? This is the answer to Does this research matter? because it proves that
humans need macromolecules, and altering them can help many people. These subquestions guided my research, and helped me answer my umbrella question after my
research was done.
Story of the Search
My search took about a month. I started by doing online research and referencing
my biochemistry textbook when I needed help on a topic or when I needed some more
information to flesh out my knowledge. This textbook became a great source of
knowledge, as I knew it was reliable and covered every macromolecule, as well as their
applications. I began by Googling macromolecules, and reading the simplest entries I
could find in order to make my first post and make sure I knew the basics about
macromolecules. When I found information that seemed like it was incorrect or I had not
learned it before, I would verify it in the textbook to make sure it was true.
Using the research, as well as the textbook to back up further research, I began to
amass more research using the sub-questions to guide me along. I mostly used web
sources, but I also used some journal articles that covered applications of the
macromolecules and biochemistry in general. I was able to gather enough research to
MACROMOLECULES AND YOU
answer all of my sub-questions, and I began to write my blog posts; one for each
question. I was getting closer and closer to being able to answer my umbrella question.
At the start of my search, I sent an email to Mr. Bala Ramjee, an organic chemist
by trade, to ask for an interview. He responded with a yes, but when I followed up, I got
no response. This was one of my biggest failures, because Mr. Ramjee specializes in the
applications of biochemistry, and he is currently working on nanomaterials. From this
experience, I have learned a few things.
First, I have learned that emailing several people a cover letter is much smarter
and efficient. If I had done this, I might have been able to conduct an interview, but due
to the lack of response so late into the timeline, I was not be able to do an interview. I
will be sure to send my request to several people to increase my chances next time. I also
learned how to make a cover letter that is appropriate to send to professionals.
Next, I learned that it is important to follow up within a day or two in order to
maintain contact with the person. I waited too long, and this could be the reason that I
missed the opportunity, as he could be on vacation right now, unable to check email.
Next time, I will follow up in a more punctual manner.
One of my larger successes was having the biochemistry textbook, as that allowed
me to fact check the research I got from the Internet, as well as gathering additional
information that I could not easily find online. Another success was my sub-questions.
They were broad enough to require a fair amount of research, but specific enough so I
would not have to do unnecessary research just to check a box. The search ended with
five blog posts, an interview post, and a whole host of research.
What I Discovered
MACROMOLECULES AND YOU
There are four types of macromolecules: lipids, nucleic acids,
carbohydrates, and proteins. Each of these macromolecules greatly affects the human
body, and resides in every cell in every one of us. First, the basics of each
macromolecule must be understood.
There are two types of proteins, simple and conjugated. According to Moulton,
simple proteins are only chains of any of the 20 amino acids. Conjugated proteins are
more complex, and function in interaction with other functional groups such as carboxyl
and carbonyl (Moulton, n.d.). Prosthetic groups are non-amino acids groups, and most
are vitamins (Moulton, n.d.).
There are two main nucleic acids, DNA and RNA, or deoxyribonucleic acid and
ribonucleic acid (Nucleic Acids, 2013). DNA is the genetic material of the cell, and the
sequence of nitrogenous bases creates variation in the human species (Nucleic Acids,
2013). RNA directs protein synthesis in the cell (Nucleic Acids, 2013). I learned all of
this information in molecular biology this year, but it is still important for the reader to
understand, as nucleic acids are arguably the most important macromolecule.
Carbohydrates fall into one of three groups: monosaccharides, disaccharides, or
polysaccharides (The Chemistry of Biology: Carbohydrates. , n.d.). Monosaccharides
have the generic formula (CH2O)n, where n can be 3, 5, or 6, and are called trioses,
pentoses, or hexoses (The Chemistry of Biology: Carbohydrates. , n.d.). Common
examples of monosaccharides are glucose, galactose, and fructose. Disaccharides are two
monosaccharides connected by a glycosidic bond, such as sucrose, lactose, or maltose,
and formed by condensation reactions (The Chemistry of Biology: Carbohydrates. ,
n.d.). Polysaccharides are formed by the polymerization of monosaccharides to form a
MACROMOLECULES AND YOU
chain, and examples include glycogen and cellulose (The Chemistry of Biology:
Carbohydrates., n.d.).
There are three types of lipids: steroids, triglycerides, and phospholipids (Reusch,
2013). Steroids control hormonal balances in the human body, and act as signal senders
and receptors (Reusch, 2013). Steroids also preserve the fluidity of the phospholipid
membranes (Reusch, 2013). Triglycerides store energy, and when the bonds are broken,
energy is released for the cell to perform actions (Reusch, 2013). Phospholipids form the
phospholipid bilayer of the cell membrane, and control what enters and leaves the cell
(Reusch, 2013).
Every day, humans use each of the four macromolecules in vastly different ways.
Part of my research involves applications of macromolecules, so it is important to
understand the basic consumption and use of macromolecules in daily life. For example,
humans use DNA from conception to death, as it stores the genetic information that
makes humans unique. Carbohydrates are stored in the body in limited amounts, and are
used for short-term energy, whereas fat is stored energy for later use. They are ingested
in food and digested into small sugars that are broken into glucose or glucose itself is
ingested (Gebel, 2011). Then, glucose is absorbed into the lining of the small intestine,
and the blood-sugar level rises (Gebel, 2011). The human body cannot make
carbohydrates. Fats are stored in special connective tissues called adipose tissue or fat
depots (Ophardt, 2003). These depots also serve as cushion and provide structural
support for organs like the heart, liver, and kidney (Ophardt, 2003). They insulate the
body from heat, and the body cannot make them by itself, just like carbs. Humans do not
ingest nucleic acids. RNA helps cells make proteins, and mRNA transfers genetic
MACROMOLECULES AND YOU
information outside the cell cells make proteins. DNA is passed onto children through
gametes, which are synthesized in meiosis (Hendrickson, 2015). For proteins, if the
amino acids aren't immediately used, the body can reconfigure them into glucose by
stripping the nitrogen from the acid and using it for a nucleic acid, then the rest of the
protein is turned into energy (Chernus and Skolnik, n.d.). This process uses water; so
high protein intake can cause dehydration. Humans cannot synthesize essential amino
acids, so they get them from proteins (Chernus and Skolnik, n.d.).
I also researched macronutrients as an application for macromolecules.
Macronutrients are nutrients that provide energy in the form of calories to the human
body. Nutrients are needed for growth, metabolism, and other bodily functions. The
prefix "macro" means large, so these macronutrients are needed in large quantities for the
body to function. There are three macronutrients: protein, fat, and carbohydrates.
Carbohydrates are needed as a main source of fuel for the body, and are easily
used by the body, as all cells and tissues can use glucose for energy (Mckinley Health
Center, 2014). Proteins are used for growth, tissue repair, creating essential enzymes and
hormones, and keeping lean muscle mass (Mckinley Health Center, 2014). Fats are used
for development and growth, and are the most concentrated energy source (Mckinley
Health Center, 2014). They also absorb vitamins, A, D, E, K, and carotenoids, provide
organ cushions, and maintain cell membranes (Mckinley Health Center, 2014).
The relationship between macromolecules and macronutrients is complicated.
The body cannot make macronutrients, but humans can make nucleic acids and
nonessential proteins organically. Macromolecules and macronutrients are both large
molecules, but there are more categories of macromolecules than macronutrients because
MACROMOLECULES AND YOU
the body is able to synthesize nucleic acids, and lipids other than fat are considered
macromolecules. The use of macromolecules to create macronutrients is obvious, as
glucose is used by every cell and tissue in the body. Both macromolecules and
macronutrients are essential to the human body and everyday functions.
The alteration of macromolecules is an important topic for biochemists. Because
macromolecules are essential to the function of the human body, their fragility must be
carefully understood. If a nucleic acid such as DNA is mutated, it can change the entire
sequence of nitrogenous bases, and create genetic defections.
Proteins can denature, which means a loss of protein structure and function
(Protein Folding, 2013). This can be caused by a misfolding in the protein, which does
not cause a complete loss of function, but enough to cause drastic issues (Protein Folding,
2013). For example, tau proteins are proteins that are very concentrated in the central
nervous system, and when they denature, Alzheimer's can be caused (Protein Folding,
2013). Cystic Fibrosis is also caused by cystic fibrosis transmembrane conductance
regulator (CFTR), a protein in the lungs, misfolding and causing Cystic Fibrosis (Protein
Folding, 2013). Also, a deletion of phenylalanine, an amino acid, at the 508th position in
CFTR is present in 70% of Cystic Fibrosis cases, another example of an organic
alteration of macromolecules (Protein Folding, 2013).
The coding of the nucleic acid is separated into codons, which are sequences of
three nucleotides of RNA or DNA (Eggling, n.d.). Several organic alterations can
happen, like Addition, Substitution, or Deletion, which are all organic alterations
(Eggling, n.d.).
Synthetic alteration of nucleic acids can be done by splicing genes
(Eggling, n.d.). Basically, this means opening a section of DNA of one organism, and
MACROMOLECULES AND YOU
inserting part of the DNA of another organism (Eggling, n.d.). Certain enzymes can also
break the phosphodiester bonds in the backbone of the DNA to open up the sequence
(Eggling, n.d.). This allows humans to create hybrids using genetic engineering, and is
very controversial, as it allows for large errors that could cause new diseases as bad as
Cystic Fibrosis or Alzheimer's (Eggling, n.d.).
Each of the macromolecules is required in large amounts for humans to perform
daily functions. Proteins can be enzymes that catalyze biochemical reactions and are
imperative to metabolism (Macromolecules Of Life, n.d.). They also have structural
function such as actin and myosin in muscle (Macromolecules Of Life, n.d.). Proteins
control immune responses, cell adhesion, and the cell cycle, and break into the essential
amino acids humans cannot make (Macromolecules Of Life, n.d.).
DNA is organized into chromosomes, and stores the genetic information that
makes people unique (Macromolecules Of Life, n.d.). It makes up the genome, which
can be analyzed to find defects and mutations for every individual (Macromolecules Of
Life, n.d.).
Carbohydrates and lipids store energy, short term and long term respectively
(Macromolecules Of Life, n.d.). Carbohydrates make up structural components like
chitin in fungi and cellulose in animals (Macromolecules Of Life, n.d.). The 5-carbon
monosaccharide ribose is an important component of coenzymes like NAD and FAD and
the backbone of RNA (Macromolecules Of Life, n.d.). Lipids form the cell membranes
phospholipid bilayer, as well as signaling with hormones, and storing nutrients in the
liver and other organs (Macromolecules Of Life, n.d.).
10
MACROMOLECULES AND YOU
In conclusion, macromolecules affect the human body in a multitude of ways.
Scientists have begun to use genetic engineering in order to cure diseases that arise from
mutations in the genome, which is made of DNA, a nucleic acid. People can be helped
using knowledge of macromolecules, such as providing food that has more carbohydrates
for marathon runners so they can use the energy quickly. Macromolecules deserve to be
researched, and can change the lives of people with genetic disease and improve our
manipulation of the human body. This research is valuable to every human, as well as
being a relatively undeveloped field for something with this many applications and the
potential to save lives. I am very interested in them, and I would consider researching
them further with possible experimentation for symposium and even my senior project.
11
MACROMOLECULES AND YOU
References
Bettelheim, F. A. (2007). Introduction to organic and biochemistry. Belmont, CA:
Brooks/Cole.
Eggling, S. (n.d.). Nucleic Acid Alterations. Retrieved May 25, 2016, from
http://dl.clackamas.edu/ch106-09/nucleic2.html
Gebel, E. (2011, March). How the Body Uses Carbohydrates, Proteins, and Fats.
Retrieved May 23, 2016, from http://www.diabetesforecast.org/2011/mar/howthe-body-uses-carbohydrates-proteins-and-fats.html
Hendrickson, K. (2015, October 12). Why Humans Need Nucleic Acids. Retrieved May
23, 2016, from http://www.livestrong.com/article/383411-why-humans-neednucleic-acids/
Macromolecules Of Life. (n.d.). Retrieved May 26, 2016, from
http://www.shomusbiology.com/macromolecules-of-life.html
McKinley Health Center - Macronutrients: The Importance of Carbohydrate, Protein, and
Fat - University of Illinois. (14, April 02). Retrieved May 24, 2016, from
http://www.mckinley.illinois.edu/handouts/macronutrients.htm
Moulton, G. E. (n.d.). The Chemistry of Biology. Retrieved May 23, 2016, from
http://www.infoplease.com/cig/biology/proteins.html
Nucleic Acids. (2013, May 05). Retrieved May 23, 2016, from
https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/nucacids.htm
Ophardt, C. E. (2003). Lipid Matabolism. Retrieved May 23, 2016, from
http://chemistry.elmhurst.edu/vchembook/620fattyacid.html
12
MACROMOLECULES AND YOU
Protein Folding. (2013). Retrieved May 25, 2016, from
http://chemwiki.ucdavis.edu/Core/Biological_Chemistry/Proteins/Protein_Structu
re/Protein_Folding
Reusch, W. (2013, May 05). Lipids. Retrieved May 23, 2016, from
https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/lipids.htm
The Chemistry of Biology: Carbohydrates. (n.d.). Retrieved May 23, 2016, from
http://www.infoplease.com/cig/biology/carbohydrates.html
M. Huang, C.-H. Hsu, J. Wang, S. Mei, X. Dong, Y. Li, M. Li, H. Liu, W. Zhang, T. Aida,
W.-B. Zhang, K. Yue, S. Z. D. Cheng. Selective assemblies of giant tetrahedra
via precisely controlled positional interactions. Science, 2015; 348 (6233): 424
DOI: 10.1126/science.aaa2421
13
Das könnte Ihnen auch gefallen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Boost Listening 1 Teacher S Edition PDFDokument96 SeitenBoost Listening 1 Teacher S Edition PDFHuy Lê QuangNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- Nato Code Numbers: Scope of ListDokument6 SeitenNato Code Numbers: Scope of ListRain HeinNoch keine Bewertungen
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- Wel-Come: Heat Treatment Process (TTT, CCT & CCR)Dokument14 SeitenWel-Come: Heat Treatment Process (TTT, CCT & CCR)atulkumargaur26Noch keine Bewertungen
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- 19.-Solid Waste TreatmentDokument108 Seiten19.-Solid Waste TreatmentShaira Dale100% (1)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- OxygendemandDokument12 SeitenOxygendemandAllenNoch keine Bewertungen
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- A Research Presented ToDokument28 SeitenA Research Presented ToAngeliePanerioGonzagaNoch keine Bewertungen
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- CBLMDokument37 SeitenCBLMDTVS Inc.Noch keine Bewertungen
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Excavation Trench Permit Ex 1 F0206Dokument5 SeitenExcavation Trench Permit Ex 1 F0206emeka2012Noch keine Bewertungen
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- Pathology SEQ Answers - Adaptive Responses & Cell InjuryDokument7 SeitenPathology SEQ Answers - Adaptive Responses & Cell InjurysugandiNoch keine Bewertungen
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- XII Biology Practicals 2020-21 Without ReadingDokument32 SeitenXII Biology Practicals 2020-21 Without ReadingStylish HeroNoch keine Bewertungen
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
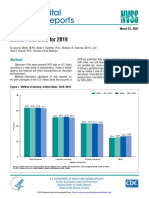
- National Vital Statistics Reports: Births: Final Data For 2019Dokument51 SeitenNational Vital Statistics Reports: Births: Final Data For 2019Simon ReichNoch keine Bewertungen
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- General Method For Cable Sizing - Electrical Installation GuideDokument10 SeitenGeneral Method For Cable Sizing - Electrical Installation Guidebhaskar_dandapathakNoch keine Bewertungen
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Granulation Process Basic UnderstandingDokument3 SeitenGranulation Process Basic UnderstandingRainMan75Noch keine Bewertungen
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Man S Bible 50 Essential Tips For Success With Your Mind Body and WomenDokument155 SeitenThe Man S Bible 50 Essential Tips For Success With Your Mind Body and WomenDonStemple100% (4)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- Eliasmith2012-Large-scale Model of The BrainDokument5 SeitenEliasmith2012-Large-scale Model of The Brainiulia andreeaNoch keine Bewertungen
- Modeling, Control and Simulation of A Chain Link Statcom in Emtp-RvDokument8 SeitenModeling, Control and Simulation of A Chain Link Statcom in Emtp-RvBožidar Filipović-GrčićNoch keine Bewertungen
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- CapsulesDokument60 SeitenCapsulesprinceamitNoch keine Bewertungen
- ASTM Standards For WoodDokument7 SeitenASTM Standards For WoodarslanengNoch keine Bewertungen
- Epigenetics & HomoeopathyDokument17 SeitenEpigenetics & HomoeopathyBishal100% (1)
- Di SilvioDokument47 SeitenDi SilviomaryroseengNoch keine Bewertungen
- Civil Aviation Authority of BangladeshDokument1 SeiteCivil Aviation Authority of BangladeshS.M BadruzzamanNoch keine Bewertungen
- Thai Cuisine: Reporters: Bantayan, Kenneth Samejon, Clarish Lovely Relevo, Mary GraceDokument47 SeitenThai Cuisine: Reporters: Bantayan, Kenneth Samejon, Clarish Lovely Relevo, Mary Gracemiralona relevoNoch keine Bewertungen
- Ocean StarDokument36 SeitenOcean Starrobertshepard1967Noch keine Bewertungen
- Skilled Labour Supply Proposal: Aiming To Be The Best Practice Skilled Labour Supplier For Construction in 2017Dokument7 SeitenSkilled Labour Supply Proposal: Aiming To Be The Best Practice Skilled Labour Supplier For Construction in 2017Abdul BaseerNoch keine Bewertungen
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- Disease PreventionDokument14 SeitenDisease PreventionJoan InsonNoch keine Bewertungen
- Brief Psychological Interventions For Borderline Personality Disorder. A Systematic Review and Meta-Analysis of Randomised Controlled TrialsDokument9 SeitenBrief Psychological Interventions For Borderline Personality Disorder. A Systematic Review and Meta-Analysis of Randomised Controlled TrialsFELIPE ROJAS TRAVERSONoch keine Bewertungen
- Victron Orion-Tr - Smart - DC-DC - Charger-Manual Non IsolatedDokument19 SeitenVictron Orion-Tr - Smart - DC-DC - Charger-Manual Non IsolatedThomist AquinasNoch keine Bewertungen
- Construction Regulations, 2014 PDFDokument58 SeitenConstruction Regulations, 2014 PDFbubele pamlaNoch keine Bewertungen
- Pulse Oximetry CircuitDokument19 SeitenPulse Oximetry Circuitنواف الجهنيNoch keine Bewertungen
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Textile Chemical Brochure 8.6.22 - 031Dokument1 SeiteTextile Chemical Brochure 8.6.22 - 031NIKESH PRAKASHNoch keine Bewertungen
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)