Beruflich Dokumente
Kultur Dokumente
Organic Compounds
Hochgeladen von
api-322767090 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
859 Ansichten10 SeitenOrganic compounds are those that contain carbon. Most organic molecules in the cell are: carbohydrates (sugars and starches) lipids (fats), proteins, nucleic acids (DNA and RNA) these molecules are usually in the form of polymers, long chains of similar subunits.
Originalbeschreibung:
Originaltitel
organic compounds
Copyright
© Attribution Non-Commercial (BY-NC)
Verfügbare Formate
PPT, PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenOrganic compounds are those that contain carbon. Most organic molecules in the cell are: carbohydrates (sugars and starches) lipids (fats), proteins, nucleic acids (DNA and RNA) these molecules are usually in the form of polymers, long chains of similar subunits.
Copyright:
Attribution Non-Commercial (BY-NC)
Verfügbare Formate
Als PPT, PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
859 Ansichten10 SeitenOrganic Compounds
Hochgeladen von
api-32276709Organic compounds are those that contain carbon. Most organic molecules in the cell are: carbohydrates (sugars and starches) lipids (fats), proteins, nucleic acids (DNA and RNA) these molecules are usually in the form of polymers, long chains of similar subunits.
Copyright:
Attribution Non-Commercial (BY-NC)
Verfügbare Formate
Als PPT, PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 10
Organic Compounds
t used to be thought that only
living things could synthesize the
complicated carbon compounds
found in cells
German chemists in the 1800's
learned how to do this in the lab,
showing that "organic
compounds can be created by
non-organic means.
Today, organic compounds are
those that contain carbon. (with a
few exceptions such as carbon
dioxide and diamonds)
Carbon's Bonding Pattern
Carbon has 4 electrons in its outer
shell. To satisfy the octet rule, it needs
to share 4 other electrons. This means
that each carbon atom forms 4 bonds.
The 4 bonds are in the form of a
tetrahedron, a triangular pyramid.
Carbon can form long chains and rings,
especially with hydrogens attached.
Compounds with just carbon and
hydrogen are "hydrocarbons: non-polar
compounds like oils and waxes.
unctional Groups
Most of the useful behavior of organic compounds
comes from functional groups attached to the carbons.
A functional group is a special cluster of atoms that
performs a useful function.
our Basic Types of Organic
Molecule
Most organic molecules in the cell are:
carbohydrates (sugars and starches), lipids
(fats), proteins, and nucleic acids (DNA and
RNA).
These molecules are usually in the form of
polymers, long chains of similar subunits.
Because they are large, these molecules are
called macromolecules. The subunits are called
monomers.
The cell also contains water, inorganic salts and
ions, and other small organic molecules.
Carbohydrates
Sugars and starches: "saccharides.
The name "carbohydrate comes from the
approximate composition: a ratio of 1 carbon to 2
hydrogens to one oxygen (CH
2
O). or instance
the sugar glucose is C
6
H
12
O
6
.
POLYSACCHARIDES
When several carbohydrates combine, it is
called a POLYsaccharide ("poly" means
many). Hundreds of sugars can be combined
in a chain. These chains are also known as
starches. You can find starches in foods
such as pasta and potatoes. They are very
good sources of energy for your body.
.
Carbohydrates are used for energy production
and storage, and for structure.
Lipids
Lipids are the main non-
polar component of cells.
Mostly hydrocarbons
carbon and hydrogen.
They are used primarily
as energy storage and
cell membranes.
4 main types: fats (energy
storage), phospholipids
(cell membranes), waxes
(waterproofing), and
steroids (hormones).
ats
Triglycerides are the main type of fat.
A triglyceride is composed of 3 fatty
acids attached to a molecule of
glycerol.
atty acids are long hydrocarbon
chains with an acid group at one end.
The chains pack together to make a
solid fat. n liquid fats, like vegetable
oils, double bonds kink the
hydrocarbon chain, which prevents the
chains from packing together nicely.
This lowers the melting temperature,
making them liquids.
Glycerol is a 3 carbon carbohydrate. t
has 3 alcohol (-OH) groups, which link
up with the acid groups in the fatty
acids.
ats store about twice as much energy
per weight as carbohydrates like
starch.
Proteins
The most important type of
macromolecule.
Roles:
Structure: collagen in skin, keratin
in hair, crystallin in eye.
Enzymes: all metabolic
transformations, building up,
rearranging, and breaking down of
organic compounds, are done by
enzymes, which are proteins.
Transport: oxygen in the blood is
carried by hemoglobin, everything
that goes in or out of a cell (except
water and a few gasses) is carried
by proteins.
Also: nutrition (egg yolk),
hormones, defense, movement
Amino Acids
Amino acids are used in every cell of your
body and are used to build the proteins you
need to survive.
All organisms need some proteins, whether
they are used in muscles or as simple
structures in the cell membrane.
Even though all organisms have differences,
they still have one thing in common, the
need for basic chemical building blocks.
Amino Acids are the subunits of proteins.
Proteins consist of long chains of amino
acids.
There are 20 different kinds of amino acids
in proteins.
The different properties of a protein come
from the arrangement of the amino acids.
The first thing you might be asking is, "What is an amino
acid?" There are over twenty, and each one of them is a
little different.
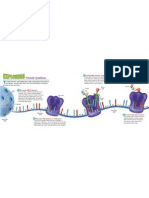
Nucleic Acids
The nucleic acids are the building
blocks of living organisms. You may
have heard of DNA described the
same way. Guess what? DNA is
just one type of nucIeic acid. Some
other types are RNA, mRNA, and
tRNA. All of these "Na's" work
together to help cells replicate and
build proteins. NA? Hold on. Might
that stand for nucleic acid? t might.
These nucleotides are made of three parts.
1. A five carbon sugar
2. A base that has a nitrogen (N) atom
3. An ion of phosphoric acid
Das könnte Ihnen auch gefallen
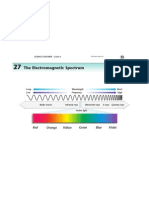
- Em SpectrumDokument1 SeiteEm Spectrumapi-32276709Noch keine Bewertungen
- Atmosphere Intro AssignmentDokument1 SeiteAtmosphere Intro Assignmentapi-32276709Noch keine Bewertungen
- Protein SynthesisDokument1 SeiteProtein Synthesisapi-32276709Noch keine Bewertungen
- Endocrine Reproductive Review SheetDokument3 SeitenEndocrine Reproductive Review Sheetapi-32276709Noch keine Bewertungen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- The Process of Manufacture of Urea in A Naphtha Based PlantDokument4 SeitenThe Process of Manufacture of Urea in A Naphtha Based PlantahmedkhidryagoubNoch keine Bewertungen
- 110 WS Solutions KeyDokument2 Seiten110 WS Solutions Keyshanmugam karthickNoch keine Bewertungen
- 8 Esters Have Many Uses Due To Their Characteristic Aromas and Often Have CommonDokument3 Seiten8 Esters Have Many Uses Due To Their Characteristic Aromas and Often Have CommonMohamed MuhajireenNoch keine Bewertungen
- Berlinite Alpo: Crystal DataDokument1 SeiteBerlinite Alpo: Crystal DataEr RutvikNoch keine Bewertungen
- Ethylene Oxide Product TreeDokument1 SeiteEthylene Oxide Product TreeManojNoch keine Bewertungen
- Hydroxyl Radical Scavenging ActivityDokument5 SeitenHydroxyl Radical Scavenging ActivityManish Kumar DubeyNoch keine Bewertungen
- Organic Synthesis TEMPLATEDokument30 SeitenOrganic Synthesis TEMPLATEKyle HallNoch keine Bewertungen
- Experiment 1 Ester SapnificationDokument14 SeitenExperiment 1 Ester SapnificationTajTaj100% (1)
- Sapcopan 2544Dokument2 SeitenSapcopan 2544gurinder pal singhNoch keine Bewertungen
- ASTM Reference Fluid For Coolant TestsDokument5 SeitenASTM Reference Fluid For Coolant TestsINEZ VILLANUEVANoch keine Bewertungen
- Actual 2006 STPM ChemistryDokument20 SeitenActual 2006 STPM ChemistryShin Yi100% (1)
- Azo DyeDokument21 SeitenAzo DyeFazal rahimNoch keine Bewertungen
- Chemistry of LipidsDokument88 SeitenChemistry of LipidsNovia EkaNoch keine Bewertungen
- Chem 27.1 Experiment 3 Determination of Mixed AlkaliDokument30 SeitenChem 27.1 Experiment 3 Determination of Mixed AlkaliAlvin Serafica0% (1)
- DF1 QuestionsDokument29 SeitenDF1 Questionspawico8232Noch keine Bewertungen
- Group 14 ElementsDokument10 SeitenGroup 14 ElementsWorld of ScienceNoch keine Bewertungen
- Tidy-Print Pn-Ad-Tds - EngDokument4 SeitenTidy-Print Pn-Ad-Tds - Engtendi soerjanagaraNoch keine Bewertungen
- Palm Oil and Palm Kernel OilDokument5 SeitenPalm Oil and Palm Kernel OilDHL123Noch keine Bewertungen
- Biochemistry and Pathology of Radical-Mediated Protein OxidationDokument18 SeitenBiochemistry and Pathology of Radical-Mediated Protein OxidationSams SriningsihNoch keine Bewertungen
- Cuaderno Trabajo1 - QU338 - 2022-2Dokument14 SeitenCuaderno Trabajo1 - QU338 - 2022-2cesar tineoNoch keine Bewertungen
- Naming Compounds and Balancing EquationsDokument18 SeitenNaming Compounds and Balancing EquationsjessjayleeNoch keine Bewertungen
- Outline: General Properties of Organometallic ComplexesDokument70 SeitenOutline: General Properties of Organometallic ComplexesikalailatulNoch keine Bewertungen
- Can20-172494-01 Ec 21183053 F PDFDokument6 SeitenCan20-172494-01 Ec 21183053 F PDFLavin ZengNoch keine Bewertungen
- MacacDokument9 SeitenMacacShawn NgNoch keine Bewertungen
- 12 Chemistry Exemplar Chapter 1Dokument403 Seiten12 Chemistry Exemplar Chapter 1chiragNoch keine Bewertungen
- Extraction Copper PDFDokument2 SeitenExtraction Copper PDFWilliamNoch keine Bewertungen
- PFD Andrussow Full Design NacnDokument278 SeitenPFD Andrussow Full Design NacnANGELA RIOJANoch keine Bewertungen
- Tricarboxylic Acid CycleDokument25 SeitenTricarboxylic Acid CycleJulia Mae LabajoNoch keine Bewertungen
- Stoichiometry ModuleDokument8 SeitenStoichiometry ModuleKaye NicolasNoch keine Bewertungen
- Processing Biomass in Conventional Oil RefineriesDokument10 SeitenProcessing Biomass in Conventional Oil RefineriesSpafiu Paula RalucaNoch keine Bewertungen