Beruflich Dokumente
Kultur Dokumente
Chemical Formula Worksheet: Write Cation-Anion Combos
Hochgeladen von
prabhu4321Originalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Chemical Formula Worksheet: Write Cation-Anion Combos
Hochgeladen von
prabhu4321Copyright:
Verfügbare Formate
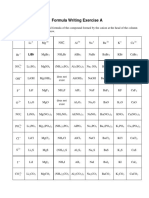
Chemical Formula Writing Worksheet
Determine the chemical formula for each cation and anion combination. Write your answers in each box. Set 1 (The combining power of silver is 1 and zinc is 2) Anions + chloride oxide iodide hydride sulfide nitride
Cations Sodium Potassium
Magnesium Calcium Copper(II) Iron(II) Iron(III) Silver Zinc Aluminum Set 2 Anions + bromide oxide fluoride astatide selenide phosphide
Cations Lithium Barium Cesium Strontium Copper(I) Copper(II) Lead(II) Lead(IV) Gallium Nickel(II)
May be used for educational purposes upon acknowledgement. www.chemicalformula.org
Chemical Formula Writing Worksheet
Determine the chemical formula for each cation and anion combination. Write your answers in each box. Brackets are only needed when the polyatomic group is greater than 1. Eg. Strontium phosphate, Sr3(PO4)2 Set 3 (The combining power of silver is 1 and zinc is 2. The formula for the ammonium ion is NH4+) Anions + nitrate NO3sulfate SO42hydroxide OHcarbonate CO32phosphate PO43hydrogen carbonate HCO3-
Cations Sodium Potassium
Magnesium Barium Iron(II) Iron(III) Silver Zinc Aluminum Ammonium Set 4 (The combining power of silver is 1 and zinc is 2. The formula for the ammonium ion is NH4+) Anions + nitrite NO2chromate CrO42sulfite, SO32dichromate Cr2O72chlorate ClO3acetate* CH3COO-
Cations Lithium Mercury(I) Tin(II) Silver Iron(II) Iron(III) Barium Zinc Aluminum
Ammonium
*The acetate group, CH3COO- is written first as this correctly shows the position of the ionic bond. Eg. CH3COO-Na+ May be used for educational purposes upon acknowledgement. www.chemicalformula.org
Chemical Formula Writing Worksheet - Answers
Set 1 (The combining power of silver is 1 and zinc is 2) Anions + Na+ K+ Ca2+ Cu2+ Fe2+ Fe3+ Ag+ Zn2+ Al3+ chloride ClNaCl KCl MgCl2 CaCl2 CuCl2 FeCl2 FeCl3 AgCl ZnCl2 AlCl3 oxide O2Na2O K2O MgO CaO CuO FeO Fe2O3 Ag2O ZnO Al2O3 iodide INaI KI MgI2 CaI2 CuI2 FeI2 FeI3 AgI ZnI2 AlI3 hydride HNaH KH MgH2 CaH2 CuH2 FeH2 FeH3 AgH ZnH2 AlH3 sulfide S2Na2S K2S MgS CaS CuS FeS Fe2S3 Ag2S ZnS Al2S3 nitride N3Na3N K3N Mg3N2 Ca3N2 Cu3N2 Fe3N2 FeN Ag3N Zn3N2 AlN
Cations Sodium Potassium Calcium Copper(II) Iron(II) Iron(III) Silver Zinc Aluminum Set 2
Magnesium Mg2+
Cations Lithium Barium Cesium Strontium Copper(I) Copper(II) Lead(II) Lead(IV) Gallium Nickel(II)
Anions + Li+ Ba2+ Cs+ Sr2+ Cu+ Cu2+ Pb2+ Pb4+ Ga3+ Ni2+
bromide BrLiBr BaBr2 CsBr SrBr2 CuBr CuBr2 PbBr2 PbBr4 GaBr3 NiBr2
oxide O2Li2O BaO Cs2O SrO Cu2O CuO Pb2O3 Pb2O Ga2O3 NiO
fluoride FLiF BaF2 CsF SrF2 CuF CuF2 PbF3 PbF4 GaF3 NiF2
astatide AtLiAt BaAt2 CsAt SrAt2 CuAt CuAt2 PbAt2 PbAt4 GaAt3 NiAt2
selenide Se2Li2Se BaSe Cs2Se SrSe Cu2Se CuSe PbSe PbSe2 Ga2Se3 NiSe
phosphide P3Li3P Ba3P2 Cs3P Sr3P2 Cu3P Cu3P2 Pb3P2 Pb3P4 GaP Ni3P2
May be used for educational purposes upon acknowledgement. www.chemicalformula.org
Chemical Formula Writing Worksheet - Answers
Brackets are only needed when the polyatomic group is greater than 1. Eg. Strontium phosphate, Sr3(PO4)2 Set 3 (The combining power of silver is 1 and zinc is 2. The formula for the ammonium ion is NH4+) Anions + Na+ K+ Ba2+ Fe2+ Fe3+ Ag+ Zn2+ Al3+ nitrate NO3LiNO3 KNO3 Mg(NO3)2 Ba(NO3)2 Fe(NO3)2 Fe(NO3)3 AgNO3 Zn(NO3)3 Al(NO3)3 NH4NO3 sulfate SO42Li2SO4 K2SO4 MgSO4 BaSO4 FeSO4 Fe2(SO4)3 Ag2SO4 ZnSO4 Al2(SO4)3 (NH4)2SO4 hydroxide OHLiOH KOH Mg(OH)2 Ba(OH)2 Fe(OH)2 Fe(OH)3 AgOH Zn(OH)2 Al(OH)3 NH4OH carbonate CO32Li2CO3 K2CO3 MgCO3 BaCO3 FeCO3 Fe2(CO3)3 Ag2CO3 ZnCO3 Al2(CO3)3 (NH4)2CO3 phosphate PO43Li3PO4 K3PO4 Mg3(PO4)2 Ba3(PO4)2 Fe3(PO4)2 FePO4 Ag3PO4 Zn3(PO4)2 AlPO4 (NH4)3PO4 hydrogen carbonate HCO3LiHCO3 KHCO3 Mg(HCO3)2 Ba(HCO3)2 Fe(HCO3)2 Fe(HCO3)3 AgHCO3 Zn(HCO3)2 Al(HCO3)3 NH4HCO3
Cations Sodium Potassium Barium Iron(II) Iron(III) Silver Zinc Aluminum
Magnesium Mg2+
Ammonium NH4+
Set 4 (The combining power of silver is 1 and zinc is 2. The formula for the ammonium ion is NH4+) Anions + Li+ Hg+ Sn2+ Ag+ Fe2+ Fe3+ Ba2+ Zn2+ Al3+ nitrite NO2LiNO2 HgNO2 Sn(NO2)2 AgNO2 Fe(NO2)2 Fe(NO2)3 Ba(NO2)2 Zn(NO2)2 Al(NO2)3 NH4NO2 chromate CrO42Li2CrO4 Hg2CrO4 SnCrO4 Ag2CrO4 FeCrO4 Fe2(CrO4)3 BaCrO4 ZnCrO4 Al2(CrO4)3 (NH4)2CrO4 sulfite, SO32Li2SO3 Hg2SO3 Sn(SO3)2 AgSO3 Fe(SO3)2 Fe2(SO3)3 Ba(SO3)2 Zn(SO3)2 Al2(SO3)3 NH4SO3 dichromate Cr2O72Li2Cr2O7 Hg2Cr2O7 SnCr2O7 Ag2Cr2O7 FeCr2O7 Fe2(Cr2O7)3 BaCr2O7 ZnCr2O7 Al2(Cr2O7)3 (NH4)2Cr2O7 chlorate ClO3LiClO3 HgClO3 Sn(ClO3)2 AgClO3 Fe(ClO3)2 Fe(ClO3)3 Ba(ClO3)2 Zn(ClO3)2 Al(ClO3)3 NH4ClO3 acetate* CH3COOCH3COOLi CH3COOHg (CH3COO)2Sn CH3COOAg (CH3COO)2 Fe (CH3COO)3 Fe (CH3COO)2 Ba (CH3COO)2 Zn (CH3COO)3 Al CH3COONH4
Cations Lithium Mercury(I) Tin(II) Silver Iron(II) Iron(III) Barium Zinc Aluminum
Ammonium NH4+
*The acetate group, CH3COO- is written first as this correctly shows the position of the ionic bond. Eg. CH3COO-Na+
May be used for educational purposes upon acknowledgement. www.chemicalformula.org
Das könnte Ihnen auch gefallen
- Balancing Equations 31 PDFDokument5 SeitenBalancing Equations 31 PDFIgnacio Jr. PaguyoNoch keine Bewertungen
- Balancing Equations Worksheet #2: © 2004 Cavalcade Publishing, All Rights ReservedDokument2 SeitenBalancing Equations Worksheet #2: © 2004 Cavalcade Publishing, All Rights ReservedkenotNoch keine Bewertungen
- Acid Bases and Salts Worksheet 1Dokument6 SeitenAcid Bases and Salts Worksheet 1Pooja Debnath100% (3)
- 13 CP Additional Word and Balancing With Key PDFDokument4 Seiten13 CP Additional Word and Balancing With Key PDFrohanNoch keine Bewertungen
- Limiting Reactant Worksheet #1Dokument2 SeitenLimiting Reactant Worksheet #1Brandon BaxterNoch keine Bewertungen
- Stoichiometry Worksheet SolutionsDokument2 SeitenStoichiometry Worksheet SolutionsQwert LimNoch keine Bewertungen
- Electrolysis Calculations AnswersDokument4 SeitenElectrolysis Calculations AnswersPersonnumberunoo100% (1)
- Making Salts Worksheet-1452167307Dokument3 SeitenMaking Salts Worksheet-1452167307R o a a A h m e dNoch keine Bewertungen
- Writing Formulas (Criss-Cross Method) WorksheetDokument1 SeiteWriting Formulas (Criss-Cross Method) WorksheetCarlton GrantNoch keine Bewertungen
- Ionic Puzzle ActivityDokument4 SeitenIonic Puzzle ActivityEngr Mumtaz0% (1)
- Molarity - Worksheet 1 Ans KeyDokument4 SeitenMolarity - Worksheet 1 Ans KeyThentamilselvi MNoch keine Bewertungen
- Elements Compounds and Mixtures WorksheetDokument2 SeitenElements Compounds and Mixtures WorksheetLiam PriceNoch keine Bewertungen
- Year 8 Chapter 1 How Plants GrowDokument32 SeitenYear 8 Chapter 1 How Plants GrowNicholas Leong100% (1)
- 141 Oxidation Reduction Worksheet KeyDokument2 Seiten141 Oxidation Reduction Worksheet KeyStephanie RendonNoch keine Bewertungen
- Nomenclature Practice ChemistryDokument6 SeitenNomenclature Practice ChemistryScott Wheeler100% (2)
- Chapter 13 Transition Elements ExerciseDokument6 SeitenChapter 13 Transition Elements Exerciseisqma100% (1)
- Activity Series WorksheetDokument2 SeitenActivity Series Worksheetapi-369711475% (4)
- Chemical Formula and Names WorksheetDokument4 SeitenChemical Formula and Names WorksheetAndrea Rio60% (5)
- Compounds PPT - Part 1Dokument37 SeitenCompounds PPT - Part 1mihad osman100% (1)
- Science Worksheet For Intervention Classes Grade 7 2nd TermDokument10 SeitenScience Worksheet For Intervention Classes Grade 7 2nd TermkainatNoch keine Bewertungen
- Acid and Base Worksheet 1-07-08 Ans KeyDokument4 SeitenAcid and Base Worksheet 1-07-08 Ans KeyShazia FarheenNoch keine Bewertungen
- Worksheet 10.6Dokument2 SeitenWorksheet 10.6SavithaBroonanNoch keine Bewertungen
- Balancing Chemical EquationDokument4 SeitenBalancing Chemical Equationamin_zaman100% (1)
- Worksheet - Empirical and Molecular FormulasDokument2 SeitenWorksheet - Empirical and Molecular FormulasWassachol Sumarasingha100% (2)
- Endothermic and Exothermic Reactions WorksheetDokument4 SeitenEndothermic and Exothermic Reactions Worksheetabdulhakim100% (1)
- Precipitation ReactionsDokument3 SeitenPrecipitation ReactionsborgiamatriceNoch keine Bewertungen
- Acids, Bases and Salts Worksheet 1Dokument2 SeitenAcids, Bases and Salts Worksheet 1Abhaydev KA100% (1)
- Formula Mass WorksheetDokument2 SeitenFormula Mass WorksheetJha DBestNoch keine Bewertungen
- 5070 Chemistry ECR v1.1 080316Dokument153 Seiten5070 Chemistry ECR v1.1 080316warisNoch keine Bewertungen
- Directions: Use Chapter 4 Section 2 and The Periodic Foldable To Complete This WorksheetDokument2 SeitenDirections: Use Chapter 4 Section 2 and The Periodic Foldable To Complete This WorksheetLeila Bawab71% (14)
- Interconversion of Mole, Number of Particles, Mass and Molar MassDokument1 SeiteInterconversion of Mole, Number of Particles, Mass and Molar MassMariana AhmadNoch keine Bewertungen
- Properties of Matter and Materials WorksheetDokument16 SeitenProperties of Matter and Materials Worksheetrashmi_harryNoch keine Bewertungen
- Worksheet - Balancing Chemical Equations With Type of ReactionDokument2 SeitenWorksheet - Balancing Chemical Equations With Type of Reactionapi-25147013850% (4)
- Calculating Moles and Mass in Chemical ReactionsDokument3 SeitenCalculating Moles and Mass in Chemical ReactionsFatema KhatunNoch keine Bewertungen
- Balancing Equations 12Dokument2 SeitenBalancing Equations 12Ignacio Jr. PaguyoNoch keine Bewertungen
- Types 20 of 20 Chemical 20 Reaction 20 Worksheet 20 AnswersDokument2 SeitenTypes 20 of 20 Chemical 20 Reaction 20 Worksheet 20 AnswersErvin CabangalNoch keine Bewertungen
- Sankey Diagram Efficiency + Energy TransferDokument4 SeitenSankey Diagram Efficiency + Energy TransferSelwah Hj AkipNoch keine Bewertungen
- Common Names of Chemical Compounds and Formula SSCDokument5 SeitenCommon Names of Chemical Compounds and Formula SSCSubin M MNoch keine Bewertungen
- Atomic Structure WorksheetDokument6 SeitenAtomic Structure WorksheetJanet UsherNoch keine Bewertungen
- Acids Bases and Salts Worksheet 1Dokument2 SeitenAcids Bases and Salts Worksheet 1api-251783882100% (3)
- Worksheet 1 Types of ChemDokument3 SeitenWorksheet 1 Types of ChemV-academy MathsNoch keine Bewertungen
- Collision Theory WorksheetDokument2 SeitenCollision Theory Worksheetsapiens123Noch keine Bewertungen
- Naming Chemical Compounds Worksheet: © 2004 Cavalcade Publishing, All Rights ReservedDokument5 SeitenNaming Chemical Compounds Worksheet: © 2004 Cavalcade Publishing, All Rights ReservedManohar GarimellaNoch keine Bewertungen
- Chemistry Worksheet 2Dokument8 SeitenChemistry Worksheet 2Marie BozemanNoch keine Bewertungen
- CBSE Class 10 Science Revision Notes Chapter - 2 Acids, Bases and SaltsDokument11 SeitenCBSE Class 10 Science Revision Notes Chapter - 2 Acids, Bases and Saltsmilind dhamaniyaNoch keine Bewertungen
- Types of Chemical Reaction WorksheetDokument2 SeitenTypes of Chemical Reaction WorksheetKenneth Myro GarciaNoch keine Bewertungen
- Elements, Compounds & Mixtures Worksheet Part 1: Read The Following Information On Elements, Compounds and Mixtures. Fill in ElementsDokument4 SeitenElements, Compounds & Mixtures Worksheet Part 1: Read The Following Information On Elements, Compounds and Mixtures. Fill in ElementsMariam JoumalNoch keine Bewertungen
- Limiting Reagents and YieldsDokument7 SeitenLimiting Reagents and Yieldss17m0582Noch keine Bewertungen
- Chemical Formula Writing Worksheet PDFDokument4 SeitenChemical Formula Writing Worksheet PDFkezia0% (1)
- Chemical Formula Writing WorksheetDokument4 SeitenChemical Formula Writing Worksheetapi-286295321Noch keine Bewertungen
- Naming & Balancing Chemical Formula - Sheet1Dokument1 SeiteNaming & Balancing Chemical Formula - Sheet1arseniy kraschenkoNoch keine Bewertungen
- Ionic Formula WorksheetDokument2 SeitenIonic Formula WorksheetKhondokar TarakkyNoch keine Bewertungen
- Chemical Formula Writing WorksheetDokument5 SeitenChemical Formula Writing WorksheetÂziz ShuvoNoch keine Bewertungen
- Activity 8 NomenclatureDokument2 SeitenActivity 8 NomenclatureCyruss MeranoNoch keine Bewertungen
- Chemical Formulas and NamesDokument8 SeitenChemical Formulas and Namesalbenis_batistaNoch keine Bewertungen
- Chemical Formula Writing Worksheet SolutionsDokument3 SeitenChemical Formula Writing Worksheet SolutionsReid Pineda Creencia100% (1)
- Chemical Formula Writing Exercise - Cations and AnionsDokument5 SeitenChemical Formula Writing Exercise - Cations and AnionsQusai Saify100% (3)
- Inorganic Compounds: Chemical Name Chemical FormulaDokument6 SeitenInorganic Compounds: Chemical Name Chemical FormulaFrendick LegaspiNoch keine Bewertungen
- Write The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Dokument2 SeitenWrite The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Bea Lha Zandra BesingaNoch keine Bewertungen
- howtowritechemicalformulacomplete-110302230114-phpapp01Dokument86 Seitenhowtowritechemicalformulacomplete-110302230114-phpapp01VladimirNoch keine Bewertungen
- Reactive Compensation & Transmission LossesDokument15 SeitenReactive Compensation & Transmission Lossesfredrick30100% (2)
- Power Semiconductor Device NptelDokument14 SeitenPower Semiconductor Device NptelMohammad Aminul IslamNoch keine Bewertungen
- Manidha Naeyam Ias & Ips Free Coaching Centre TNPSC Group I Answer Key For A SeriesDokument5 SeitenManidha Naeyam Ias & Ips Free Coaching Centre TNPSC Group I Answer Key For A Seriesprabhu4321Noch keine Bewertungen
- Final Year Projects 2011 Power ElectronicsDokument5 SeitenFinal Year Projects 2011 Power Electronicsprabhu4321Noch keine Bewertungen
- An Analytical Approach For DG Allocation - BalaDokument10 SeitenAn Analytical Approach For DG Allocation - Balaprabhu4321Noch keine Bewertungen
- Matlab GaDokument15 SeitenMatlab Gagreenday3Noch keine Bewertungen
- BooksDokument4 SeitenBooksprabhu43210% (1)
- Remote Control For Home ApplianceDokument1 SeiteRemote Control For Home ApplianceAkash MehtaNoch keine Bewertungen
- Re MeDokument1 SeiteRe Merohit11116235Noch keine Bewertungen
- Qustion TestDokument3 SeitenQustion Testprabhu4321Noch keine Bewertungen
- Oral Controlled Drug Delivery System Alteration To DoDokument12 SeitenOral Controlled Drug Delivery System Alteration To DoMallamma ReddyNoch keine Bewertungen
- Movement Into and Out of Cells: A.2.1 Matter and EnergyDokument13 SeitenMovement Into and Out of Cells: A.2.1 Matter and EnergyRovina Narayan DiasNoch keine Bewertungen
- Seasonal Variation and Water Quality Assessment of Bhairab River in Khulna PDFDokument9 SeitenSeasonal Variation and Water Quality Assessment of Bhairab River in Khulna PDFSheikh Serajul HakimNoch keine Bewertungen
- The Difference Between Total and Free ChlorineDokument1 SeiteThe Difference Between Total and Free ChlorineTimur SekerciogluNoch keine Bewertungen
- Answers To End-Of-Chapter Questions: 1 A B I C D I IIDokument2 SeitenAnswers To End-Of-Chapter Questions: 1 A B I C D I IIKenneth KnightNoch keine Bewertungen
- Halal Testing and Gelatin Screening Using Lc-Ms/Ms By: Tan Shin Jowi, PHDDokument2 SeitenHalal Testing and Gelatin Screening Using Lc-Ms/Ms By: Tan Shin Jowi, PHDSoeyanto DuckoNoch keine Bewertungen
- BS 3148-80 - Water For Making ConcreteDokument5 SeitenBS 3148-80 - Water For Making ConcreteMichael VeneerNoch keine Bewertungen
- Principles of Smoking 17392Dokument8 SeitenPrinciples of Smoking 17392Khadijah Zaimatun NisaNoch keine Bewertungen
- Elastomer Solutions For Pharmaceutical Packaging and Medical DevicesDokument27 SeitenElastomer Solutions For Pharmaceutical Packaging and Medical DevicesRonaldo CamargoNoch keine Bewertungen
- NEET Chemistry Chapter Wise Weightage 2024 1Dokument3 SeitenNEET Chemistry Chapter Wise Weightage 2024 1grosha mitraNoch keine Bewertungen
- Combating Green Oil PDFDokument5 SeitenCombating Green Oil PDFkulaspiroNoch keine Bewertungen
- CHAPTER 2 Extra CycloalkanesDokument13 SeitenCHAPTER 2 Extra Cycloalkanesellina safianNoch keine Bewertungen
- Rel XPR 6600 Iso Resin DatasheetDokument2 SeitenRel XPR 6600 Iso Resin DatasheetJainam ShahNoch keine Bewertungen
- Activated Carbon PDFDokument10 SeitenActivated Carbon PDFDavid JoeventusNoch keine Bewertungen
- FCCU Operation Monitoring and Problem Diagnosis - Catalyst RelatedDokument5 SeitenFCCU Operation Monitoring and Problem Diagnosis - Catalyst Relatedsaleh4060Noch keine Bewertungen
- What is Paper? A Concise GuideDokument13 SeitenWhat is Paper? A Concise GuideAHMAD HAZIQ BIN MOHD RAHMATNoch keine Bewertungen
- 02 X Controlling Polymer Stereochemistry in RingopeningDokument22 Seiten02 X Controlling Polymer Stereochemistry in Ringopeningmoine dorotheeNoch keine Bewertungen
- Heurísticas para la Síntesis de Procesos 19-27Dokument24 SeitenHeurísticas para la Síntesis de Procesos 19-27Camilo RamirezNoch keine Bewertungen
- Industrial Chemistry Lecture on Cement ProductionDokument4 SeitenIndustrial Chemistry Lecture on Cement ProductionShivani SharmaNoch keine Bewertungen
- Organic Chemistry (Part Two) NotesDokument17 SeitenOrganic Chemistry (Part Two) NotesahmedNoch keine Bewertungen
- Sutarno IPMC ITB2017 1.4974439Dokument11 SeitenSutarno IPMC ITB2017 1.4974439Samuel ArelianoNoch keine Bewertungen
- Dip B1-6.3.1 - Aircraft Materials - Composite and Non-Metallic SR 2017-07-14Dokument54 SeitenDip B1-6.3.1 - Aircraft Materials - Composite and Non-Metallic SR 2017-07-14Fahad AlDossari100% (1)
- Yay OFFLINEDokument6 SeitenYay OFFLINEmichellaineNoch keine Bewertungen
- CH101 - Chemistry: Module Overview and General AimsDokument9 SeitenCH101 - Chemistry: Module Overview and General AimsShresth SanskarNoch keine Bewertungen
- Cell - The Unit of Life - NCERT Based PYQsDokument9 SeitenCell - The Unit of Life - NCERT Based PYQsAkhil singhNoch keine Bewertungen
- Masterton Odd No - AnswersDokument257 SeitenMasterton Odd No - Answers박시후Noch keine Bewertungen
- Calcium Carbonate Lab ReportDokument2 SeitenCalcium Carbonate Lab ReportAlexander Weber0% (1)
- Mar M 247Dokument30 SeitenMar M 247sunsirNoch keine Bewertungen
- S Y B SC Syllabus 1Dokument71 SeitenS Y B SC Syllabus 1Dr. Mithil Fal DesaiNoch keine Bewertungen
- LysosomeDokument8 SeitenLysosomemir zaheeruddinNoch keine Bewertungen