Beruflich Dokumente
Kultur Dokumente
Module 5 (Air Around Us)
Hochgeladen von
nor shafiniOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Module 5 (Air Around Us)
Hochgeladen von
nor shafiniCopyright:
Verfügbare Formate
29
MODULE 5: AIR AROUND US SECTION A
1. Figure 1 shows the component of gases in air.
1%
Figure 1 P, Q, R and S are represented by A B C D P Carbon dioxide Oxygen Nitrogen Nitrogen Q Inert gases Nitrogen Oxygen Oxygen R Oxygen Carbon dioxide Inert gases Carbon dioxide S Nitrogen Inert gases Carbon dioxide Inert gases
2. The information below is about the properties of gas X.
Odourless Dissolves in alkaline pyrogallol solution Does not change the colour of litmus paper
Which of the following is probably gas X? A B C D Carbon dioxide Hydrogen Nitrogen Oxygen
Modul Pengayaan Sains STF (PMR)
30
The characteristics in Table 1 below belong to a gas Test Solubility in water Solubility in sodium hydroxide solution Action on a lighted splinter Table 1 The gas is A B C D oxygen nitrogen hydrogen carbon dioxide Result Soluble Very soluble Extinguishes the lighted splinter
An experiment is set up as shown in Figure 2. At the end of the experiment the lime water turn chalky.
Figure 2 What conclusion can be drawn from the experiment? A B C D 5 Carbon element exists in fuel Heat is released when fuel is burning Soot is produced when fuel is burning Carbon dioxide is produced when fuel is burning
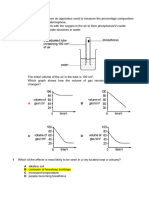
Figure 3 below shows a set-up of apparatus
Figure 3
Modul Pengayaan Sains STF (PMR)
31
A gas was collected in the test tube after a few hours. The gas was tested with a few tests. Which of the following is true? Test Effect on burning splinter Effect on lime water Effect on hydrogen carbonate indicator Effect on glowing splinter Changes Extinguished with a pop sound Turns cloudy No changes Relights
A B C D
Figure 4 below shows two candles. Each of them is left burning in different gas jars filled with inhaled air and the other with exhaled air.
Inhale air
Figure 4 Which of the following will be observed? A B C D 7
Exhaled air
Candle Y and Z continues to burn. Candle Y extinguishes before candle Z Candle Z extinguishes before candle Y. Candle Y and Z extinguishes at the same time.
What are the similarities between respiration and combustion? I II III IV A B C D Produce energy Uses oxygen Occurs inside living cells Produce water I and II only I, II and IV only I, III and IV only II, III and IV only
Modul Pengayaan Sains STF (PMR)
32
Which of the following pair is true about pollutants and their effects? Pollutants Carbon monoxide Haze Sulphur dioxide Oxides of nitrogen Effects Thinning of the ozone layer Soil become acidic Cause breathing problem Green house effect
A B C D
Which air pollutants is produced from combustion of fossil fuel? A B C D Nicotine Chlorofluorocarbon Carbon monoxide Nitrogen oxide
10 What is the phenomenon caused by excessive carbon dioxide in the air? A B C D Acid rain Soil erosion Green house effect The thinning of ozone layer
Modul Pengayaan Sains STF (PMR)
33
SECTION B 1. Diagram 1 shows a substance P which is allowed to burn in a gas jar containing lime water.
Cover
Burning spoon Air Substance P Lime water
Diagram1
1
(a) (i) Draw what you observe at the wall of the gas jar. [ 1 mark ] (ii) How to test the presence of the substance formed in (a) (i) .. [ 1 mark ] (b) Write a word equation if the substance P is charcoal.
+ Oxygen
Heat energy
Light energy
[ 2 mark ]
(c ) (i) What happen to the lime water in the gas jar ? [ 1 mark ] (ii) Explain your answer. [ 1 mark ]
Modul Pengayaan Sains STF (PMR)
34
(d) Complete the fire triangle by putting the factors needed for combustion.
(i) (ii) (iii)
P : .. Q: ... R : .
2.
Diagram 2 shows the relationship between respiration and photosynthesis.
R Photosynthesis S
Diagram 2 a. Name the gases R and S R: S: (2 marks).
Respiration
b. Name a living thing that absorbs gas R.
(1 mark).
c. Name 2 living organisms that release gas S.
i ii . (2 mark)
d. Name two processes that release gas S.
. (2 mark)
Modul Pengayaan Sains STF (PMR)
35
e. State two characteristics of gas R and gas S R i:. ii:.... S i:. ii:.... ( 4 marks) 3. Three sets of apparatus as shown in Diagram 3 below are set up and left aside in a horizontal position for three hours.
Diagram 3 The result of the experiment is shown in the Table 1 below. Test tube X Y Z Observation Indicator moves inwards towards the boiling tube Indicator moves inwards towards the boiling tube Indicator does not move Table 1 a What inference can you make from the observation of the indicator? i ii ii b c X: __________________________________________________________ Y : __________________________________________________________
Z: _________________________________________________________ ( 3 marks ) State the function of the cotton soaked with sodium hydroxide solution. ________________________________________________________________ ( 1 mark ) State the conclusion from the observation of the experiment . ___________________________________________________ _____________
Modul Pengayaan Sains STF (PMR)
36
( 1 mark )
Modul Pengayaan Sains STF (PMR)
Das könnte Ihnen auch gefallen
- 6CH01 01R Que 20140523Dokument28 Seiten6CH01 01R Que 20140523Celinne TehNoch keine Bewertungen
- Bgcse Double Award Paper 3 2017 SolutionsDokument15 SeitenBgcse Double Award Paper 3 2017 SolutionsCrystal Machipisa100% (2)
- How To Build Solar FarmDokument30 SeitenHow To Build Solar Farmfibref100% (3)
- HKDSE Chem FX ExamS5 2011 Set1 EngDokument27 SeitenHKDSE Chem FX ExamS5 2011 Set1 Eng12376590Noch keine Bewertungen
- F2 IS Exam 1 (13-14)Dokument9 SeitenF2 IS Exam 1 (13-14)羅天佑Noch keine Bewertungen
- Diagnostic Test in General Chemistry 1Dokument13 SeitenDiagnostic Test in General Chemistry 1Dearest Notes100% (1)
- Samsung Refrigerator and Whaser Repair 2011Dokument416 SeitenSamsung Refrigerator and Whaser Repair 2011nibble1974Noch keine Bewertungen
- Hot Dip GalvanizingDokument24 SeitenHot Dip GalvanizingKarthikeyan MuniaswamyNoch keine Bewertungen
- Aetheric Battery - Copy #1Dokument6 SeitenAetheric Battery - Copy #1Keresztúri FerencNoch keine Bewertungen
- Loading LPG CalculationsDokument12 SeitenLoading LPG CalculationsJohn Green67% (3)
- Practice Makes Perfect in Chemistry: Oxidation-ReductionVon EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionBewertung: 5 von 5 Sternen5/5 (1)
- Dual Plate Check ValveDokument38 SeitenDual Plate Check ValveVikas Kumar PathakNoch keine Bewertungen
- Paper 2 SPM Trial2007Dokument10 SeitenPaper 2 SPM Trial2007Saya Menang100% (1)
- Electron arrangement of element XDokument12 SeitenElectron arrangement of element XVictoria PetrusNoch keine Bewertungen
- Ss 3 Chemistry Mock 2 Exam First Term 2017-18Dokument8 SeitenSs 3 Chemistry Mock 2 Exam First Term 2017-18Elena SalvatoreNoch keine Bewertungen
- Set VDokument5 SeitenSet VChew Gee LanNoch keine Bewertungen
- 2nd Mid Term 12Dokument8 Seiten2nd Mid Term 12Nor RasyidahNoch keine Bewertungen
- DHA MOCK EXAMINATION FOR CHEMISTRY CLASS CIIIDokument12 SeitenDHA MOCK EXAMINATION FOR CHEMISTRY CLASS CIIIaNoch keine Bewertungen
- 1997 Paper 2Dokument14 Seiten1997 Paper 2api-3826629Noch keine Bewertungen
- IT Chem F5 SPM Model Paper (E)Dokument10 SeitenIT Chem F5 SPM Model Paper (E)Norzawati NoordinNoch keine Bewertungen
- 1998 Paper 2Dokument16 Seiten1998 Paper 2api-3826629Noch keine Bewertungen
- Air (TYS) Ashley MDL8D-2 I7ikrDokument12 SeitenAir (TYS) Ashley MDL8D-2 I7ikrashleytham89Noch keine Bewertungen
- St John Convent Secondary School Chemistry PaperDokument25 SeitenSt John Convent Secondary School Chemistry PaperChanda S MwambaNoch keine Bewertungen
- Practice Chap 1 and 2 - AnswerDokument11 SeitenPractice Chap 1 and 2 - AnswerNur Afiqah Mohd ZakiNoch keine Bewertungen
- RTS Chemistry SPM Question Bank Chapter 12Dokument8 SeitenRTS Chemistry SPM Question Bank Chapter 12dobbybibiNoch keine Bewertungen
- Holiday Homework f4Dokument27 SeitenHoliday Homework f4amin_zamanNoch keine Bewertungen
- Sns Paper 2 KimiaDokument16 SeitenSns Paper 2 KimiaDuong Han CalebNoch keine Bewertungen
- Chemistry SPM Forecast PapersDokument16 SeitenChemistry SPM Forecast Paperswhywhyq0% (1)
- Chemistry Worksheet 1 Year 11Dokument8 SeitenChemistry Worksheet 1 Year 11fatma.darghouth2Noch keine Bewertungen
- S.3chem 1Dokument8 SeitenS.3chem 1Lakogaharry BillclintonNoch keine Bewertungen
- March Test (F4) - 2009Dokument12 SeitenMarch Test (F4) - 2009Rozilah YunusNoch keine Bewertungen
- Pahang NEW STPM 2012 ChemistryDokument15 SeitenPahang NEW STPM 2012 ChemistryMohd Iruan JanalNoch keine Bewertungen
- Soalan Ting 1.Dokument7 SeitenSoalan Ting 1.Dia Aizan HamdanNoch keine Bewertungen
- Test - Ch.18 ChemicalChanges - 2022Dokument4 SeitenTest - Ch.18 ChemicalChanges - 2022Kirstie KJSNoch keine Bewertungen
- Chemistry 1995 Paper 2Dokument14 SeitenChemistry 1995 Paper 2api-3824003Noch keine Bewertungen
- Graph Temp Change Solid Naphthalene HeatingDokument10 SeitenGraph Temp Change Solid Naphthalene HeatingFary SaidinNoch keine Bewertungen
- Chemistry - 3rd Form2001Dokument6 SeitenChemistry - 3rd Form2001Robert EdwardsNoch keine Bewertungen
- VIII CC Chemistry Home Work 5 On 9th April 2020Dokument4 SeitenVIII CC Chemistry Home Work 5 On 9th April 2020sultanaNoch keine Bewertungen
- Chemistry Mock Exam ss3Dokument3 SeitenChemistry Mock Exam ss3chrizyboyziNoch keine Bewertungen
- 16+ Chemistry PDFDokument12 Seiten16+ Chemistry PDFKhyzer HayyatNoch keine Bewertungen
- 25 Jan 23 Evening English 1Dokument18 Seiten25 Jan 23 Evening English 1DEPRESSED GAMERNoch keine Bewertungen
- 1 in Which Option Do The Three Particles Each Have The Same Number of ElectronsDokument12 Seiten1 in Which Option Do The Three Particles Each Have The Same Number of Electronssololexzib100% (1)
- Upper Sixth Organic ChemistryDokument6 SeitenUpper Sixth Organic ChemistryBaninla NerusNoch keine Bewertungen
- KCES2021Dokument13 SeitenKCES2021PremierLeagueNoch keine Bewertungen
- Chemistry s4 Theory and Pract.Dokument26 SeitenChemistry s4 Theory and Pract.kubwimanajeandamour359Noch keine Bewertungen
- 3 Oxidation and ReductionDokument25 Seiten3 Oxidation and ReductiondonutNoch keine Bewertungen
- Chem F4 Mid ExamDokument10 SeitenChem F4 Mid ExamYong SiewkuanNoch keine Bewertungen
- Aqueous Hydrogen Peroxide: Ó UCLES 2004 5070/01/M/J/04Dokument14 SeitenAqueous Hydrogen Peroxide: Ó UCLES 2004 5070/01/M/J/04thc8477Noch keine Bewertungen
- Pei Hwa Chem Prelim P1 2012 QNDokument16 SeitenPei Hwa Chem Prelim P1 2012 QNWonderfullyANoch keine Bewertungen
- Sample Chemistry Question Paper TitleDokument13 SeitenSample Chemistry Question Paper TitleShrikant KumarNoch keine Bewertungen
- Air and AtmosphereDokument12 SeitenAir and Atmospherebob leowNoch keine Bewertungen
- LW F6 Chem Mock Exam P1 1718 PDFDokument23 SeitenLW F6 Chem Mock Exam P1 1718 PDFVincent haNoch keine Bewertungen
- Stochiometry Classified QPDokument24 SeitenStochiometry Classified QPMang friesNoch keine Bewertungen
- 2011 H2 Chem SRJC Prelim Paper 1Dokument20 Seiten2011 H2 Chem SRJC Prelim Paper 1onnoezNoch keine Bewertungen
- 2010 SAJC H2 Chem Prelim P1Dokument19 Seiten2010 SAJC H2 Chem Prelim P1Giovanni AndersonNoch keine Bewertungen
- 2003 Csec Chem Paper 01Dokument10 Seiten2003 Csec Chem Paper 01Jesshaun Morris100% (6)
- Chem F3 QS2Dokument6 SeitenChem F3 QS2Okumu KevinsNoch keine Bewertungen
- Form Three Chemistry Set 1 QSDokument5 SeitenForm Three Chemistry Set 1 QSsadzivatNoch keine Bewertungen
- January 2014 - Question Paper - Chemistry U2Dokument20 SeitenJanuary 2014 - Question Paper - Chemistry U2lolomg90Noch keine Bewertungen
- 2004 MRSM Without AnswerDokument49 Seiten2004 MRSM Without AnswerChoong Wen JianNoch keine Bewertungen
- 2012 First Prelim Chemistry Paper 1 v2Dokument15 Seiten2012 First Prelim Chemistry Paper 1 v2jzhong_7Noch keine Bewertungen
- Manual Espctrofotômetro Jenway Mod GenovaDokument34 SeitenManual Espctrofotômetro Jenway Mod GenovaSheron CogoNoch keine Bewertungen
- Advantages and Disadvantages of Stainless Steel CookwareDokument15 SeitenAdvantages and Disadvantages of Stainless Steel CookwareFaIz FauziNoch keine Bewertungen
- 21 Pathogenesis of Carp Erythrodermatitis (CE)Dokument6 Seiten21 Pathogenesis of Carp Erythrodermatitis (CE)giuseppegnrNoch keine Bewertungen
- Mutu Manajemen PDFDokument238 SeitenMutu Manajemen PDFirdawanNoch keine Bewertungen
- np38 12b Product Data SheetDokument2 Seitennp38 12b Product Data Sheetapi-170472102Noch keine Bewertungen
- PhysRevD 99 052002 PDFDokument28 SeitenPhysRevD 99 052002 PDFBibhuprasad MahakudNoch keine Bewertungen
- RSBV Tech BulletinpdfDokument16 SeitenRSBV Tech BulletinpdfbalancedrumNoch keine Bewertungen
- Surface TextureDokument27 SeitenSurface Texturesohkimfai6340100% (5)
- A Facile Strategy To Simultaneously Improve The Mechanical and Fire SafetyDokument43 SeitenA Facile Strategy To Simultaneously Improve The Mechanical and Fire SafetyTia Nico Rossi RossiNoch keine Bewertungen
- Ultrasonic Testing Equipment Manufacturers - Canopus InstrumentsDokument1 SeiteUltrasonic Testing Equipment Manufacturers - Canopus InstrumentscanopusinstrumentsNoch keine Bewertungen
- 100-800 HP Boiler Control System PLC HMI Safety FunctionsDokument4 Seiten100-800 HP Boiler Control System PLC HMI Safety FunctionssebaversaNoch keine Bewertungen
- Re91001 01 X b2 - 2016 04Dokument30 SeitenRe91001 01 X b2 - 2016 04Bernardo Orozco LariosNoch keine Bewertungen
- Assignment 1Dokument4 SeitenAssignment 1Nur AfiqahNoch keine Bewertungen
- Thiabendazole RedDokument103 SeitenThiabendazole RedFandhi Adi100% (1)
- Material Data Sheet: Casting Material: Stainless Steel CF8MDokument9 SeitenMaterial Data Sheet: Casting Material: Stainless Steel CF8MakshayNoch keine Bewertungen
- Glassy Carbon - Wikipedia, The Free Encyclopedia PDFDokument5 SeitenGlassy Carbon - Wikipedia, The Free Encyclopedia PDFNaveen PalaNoch keine Bewertungen
- Glassfibre Reinforced Concrete ReviewDokument23 SeitenGlassfibre Reinforced Concrete ReviewmohamedNoch keine Bewertungen
- Process Modeling Using Aspen HYSYSDokument2 SeitenProcess Modeling Using Aspen HYSYSLinton WongNoch keine Bewertungen
- SS316 Butt WeldingDokument124 SeitenSS316 Butt Weldingdevinder_bamrahNoch keine Bewertungen
- TDS - RheoFIT 764Dokument2 SeitenTDS - RheoFIT 764aahtagoNoch keine Bewertungen
- 38CKC 8PDDokument36 Seiten38CKC 8PDjean_carlos_abreu_rNoch keine Bewertungen
- PublishedneemfullpaperDokument6 SeitenPublishedneemfullpaperClinton John Evangelista JuadiongNoch keine Bewertungen
- CP de VaporDokument6 SeitenCP de VaporNatalia MorenoNoch keine Bewertungen
- Standard Compressor R134a 115-127V 60Hz General Technical DataDokument2 SeitenStandard Compressor R134a 115-127V 60Hz General Technical Datawilmer cantilloNoch keine Bewertungen