Beruflich Dokumente
Kultur Dokumente
Reactions
Hochgeladen von
Sofía I. Morales NavarroOriginalbeschreibung:
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Reactions
Hochgeladen von
Sofía I. Morales NavarroCopyright:
Verfügbare Formate
Reactions
Reaction 1. Electrophilic addition of hydrogen halides (HX) to alkenes.
Reagents and conditions
Mechanistic details
HCl, HBr in ether KI + H3PO4 room temperature
electrophilic addition carbocation intermediates Markovnikov's rule apply carbocation rearrangements possible no relative stereochemical preference
Reaction 2. Radical addition of hydrogen bromide (HBr) to alkenes. (NOT COVERED THIS TIME)
Reagents and conditions
Mechanistic details
radical initiators (usually peroxides) heat or light
chain reaction radical intermediates anti-Markovnikov's products no relative stereochemical preference
Reaction 3. Electrophilic addition of halogens (X2) to alkenes.
Reagents and conditions
Mechanistic details
Br2, Cl2 (or I2) in CH2Cl2 or CCl4 room temperature
electrophilic addition bromonium or chloronium ion intermediates anti addition
Reaction 4. Electrophilic addition of halogens to alkenes in the presence of water.
Reagents and conditions
Mechanistic details
Br2 or Cl2 in H2O or NBS in H2O/DMSO room temperature
electrophilic addition of X2 bromonium or chloronium ion intercepted by H2O Markovnikov's rule apply (with respect to H2O) anti addition
Reaction 5. Electrophilic addition of water to alkenes.
Reagents and conditions
Mechanistic details
acid catalyst needed with non-nucleophilic counter ion (H2SO4, HClO4, H3PO4) high temperature required often reversible (rather used to make alkenes from alcohols)
electrophilic addition carbocations intermediates Markovnikov's rule applies no relative stereochemical preference
Reaction 6. Oxymercuration of alkenes (formal addition of water).
Reagents and conditions
Mechanistic details
Hg(OAc)2 in H2O (or THF/H2O) reduction step required to replace mercury with hydrogen (NaBH4) room temperature
electrophilic addition of mercury compound mercurinium ion as the intermediate intercepted by H2O Markovnikov's rule applies with respect to H2O reduction step with a complicated mechanism the addition of H2O is anti, but reduction scrambles the stereochemistry (i.e. no relative stereochemical preference is observed)
Reaction 7. Hydroboration of alkenes (formal addition of water).
Reagents and conditions
Mechanistic details
BH3-THF complex in THF tri-fold addition (to borane) is common, giving trialkylboranes (R3B) oxidation step necessary (H2O2/OH) room temperature or heat
electrophilic addition of BH3 cyclic transition state, putting boron at the least substituted carbon of the double bond syn addition, preserved in the oxidation step (stereospecific replacement of BR2 with OH)) anti-Markovnikov alcohols
Reaction 8. Hydrogenation of alkenes.
Reagents and conditions
Mechanistic details
H2 gas over heterogeneous catalysts (Pd/C, Pt, PtO2) room temperature very facile reaction (many other functional groups remain untouched)
surface reaction syn addition from the less crowded face mechanism is complicated redox reaction
Reaction 9. Hydroxylation of alkenes.
Reagents and conditions
Mechanistic details
KMnO4/ OH (lower yield) OsO4/pyridine (higher yield but toxic and expensive) catalytic OsO4 with NMO
cyclic transition state and intermediate resulting in syn addition redox reaction
Reaction 10. Ozonolysis of alkenes.
Reagents and conditions
Mechanistic details
ozone at low temperature followed by reduction with Zn/AcOH
complicated mechanism with O3 oxidation followed by (partial) reduction
Reaction 11. Oxidation of diols.
Reagents and conditions
Mechanistic details
1,2-diol treated by HIO4 in H2O/THF equivalent to ozonolysis of the corresponding alkene
cyclic intermediate with HIO4
Reaction 12. Oxidation of alkenes with permanganate under acidic conditions.
Reagents and conditions
Mechanistic details
potassium permanganate under acidic or neutral conditions
redox reaction oxygen inserts into all former vinylic C-H bonds
Reaction 13. Electrophilic addition of hydrogen halides (HX) to alkynes.
Reagents and conditions
Mechanistic details
HCl, HBr in acetic acid
electrophilic addition vinyl carbocation as an intermediate Markovnikov's rule apply
first addition usually trans second addition often follows less reactive than alkenes
Reaction 14. Electrophilic addition of halogens (X2) to alkynes.
Reagents and conditions
Mechanistic details
Cl2, Br2 in CCl4
electrophilic addition Markovnikov's rule apply first addition usually trans (anti) as if the halonium ion was an intermediate (but most likely it is not) second addition often follows less reactive than alkenes
Reaction 15. Electrophilic addition of water to alkynes.
Reagents and conditions
Mechanistic details
HgSO4/H2O/H2SO4 no NaBH4 necessary to replace mercury (Hg) with hydrogen
electrophilic addition catalyzed by Hg2+ (mercurinium ion not involved) Markovnikov's rule apply the primary product is an enol, a less stable tautomer of a ketone
Reaction 16. Hydroboration of alkynes (formal addition of water).
Reagents and conditions
Mechanistic details
BH3/THF gives mixture of regioisomers for disubstituted alkynes, double addition with terminal alkynes R'2BH (R' = 1,2-dimethylpropyl) may be used for monoaddition to terminal alkynes
four - membered cyclic transition state for addition syn addition
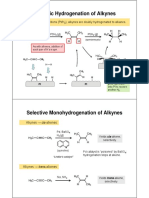
Reaction 17. Hydrogenation of alkynes.
Reagents and conditions
Mechanistic details
Lindlar catalyst used for cis product (Pd, CaCO3, Pb(OAc)2, quinoline) lithium metal in ammonia for trans product
hydrogenation is a heterogeneous reaction hydrogenation catalyst is poisoned (deactivated) to prevent further reduction of the double bond Li reduction involves electron - transfer process and proceeds via an intermediate vinylic carbanion
Reaction 18. Alkylation of acetylide anion.
Reagents and conditions
Mechanistic details
KNH2 or NaNH2 used as a base (in NH3 or THF)
the increased acidity of the sp hybridized carbon makes carbanion accessible (the lone electron pair in the
LDA in THF used as a base primary electrophiles (alkylating agents) work well
conjugate base, acetylide anion, has large s character) SN2 substitution mechanism followed (back-side attack on the electrophilic carbon)
Reaction 19. Oxidative cleavage of alkynes.
Reagents and conditions
Mechanistic details
KMnO4 or ozone often low yields
complicated oxidation mechanisms more difficult to oxidize than alkenes substituted "ends" yield the corresponding carboxylic acids, unsubstituted ones give CO2
Reaction 20. Electrophilic addition of HX to conjugated dienes.
Reagents and conditions
Mechanistic details
HCl or HBr in ether
electrophilic addition leading to allyl (resonance stabilized) carbocations the allyl cation can be attacked by the bromide anion at two positions the 1,2-adduct (A) is kinetically favored (predominates at low temperatures) the 1,4-adduct (B) is thermodynamically more stable and it predominates at higher temperatures
Reaction 21. Electrophilic addition of halogens to conjugated dienes.
Reagents and conditions
Mechanistic details
Br2 or Cl2 in CCl4 or CH2Cl2
electrophilic addition leading to halonium ion intermediate the 1,2-adduct is kinetically favored (predominates at low temperatures) the 1,4-adduct is thermodynamically more stable and predominates at higher temperatures
Reaction 22. Radical (chain) halogenation of alkanes.
Reagents and conditions
Mechanistic details
X2; the reaction is explosive for F2 and very sluggish for I2 (thermodynamic reasons) heat or light used to generate radicals in the initiation steps mixtures of products are obtained (mono- and polyhalogenated compounds, and different regioisomers) NBS in CCl4 (with light or initiators) used for allylic or benzylic brominations
radical chain reactions the initiation step generates X radical selectivity is established in the hydrogen-abstraction step by X the more reactive X, the less selective it is
Reaction 23. Conversion of alcohols into alkyl halides.
Reagents and conditions
Mechanistic details
HX in ether (works best for tertiary alcohols) PBr3 in ether or CH2Cl2 SOCl2 in pyridine TosCl/pyridine followed by X
SN1 mechanism for tertiary alcohols SN2 mechanism for primary alcohols hydroxyl group is converted to a better leaving group by reaction with the reagent of choice
Reaction 24. Nucleophilic substitution reaction on sp3 hybridized carbons.
Reagents and conditions
Mechanistic details
variety of conditions and solvents usually polar and protic solvents for SN1 reactions usually polar aprotic solvents for SN2 reactions
SN1 mechanism for tertiary substrates: the leaving group departs in a unimolecular rate-limiting step, generating the carbocation, which in the second step reacts with the nucleophile; ion pairs may be involved and carbocation rearrangements may compete SN2 mechanism for primary substrates: the nucleophile displaces the leaving group in one-step bimolecular back-side attack leading to inversion of configuration on stereogenic centers secondary, allylic or benzylic substrates may react by both mechanisms competition with elimination reactions (E1 and E2) often observed
Reaction 25. Elimination reaction to form carbon-carbon double bonds.
Reagents and conditions
Mechanistic details
variety of conditions and solvents usually strong bases favor E2 mechanism
E1 mechanism for tertiary or secondary allylic or benzylic substrates: the leaving group departs in a unimolecular ratelimiting step, generating the carbocation, which in the second step is deprotonated (with base) on the carbon adjacent to the cationc center, yielding the olefin; carbocation rearrangements may compete E2 mechanism favored by strong bases: the base removes a proton from the carbon adjacent to one bearing the leaving group in a one-step bimolecular reaction that requires periplanar orientation of the hydrogen and the leaving group (antiperiplanar preferred)
competition with substitution reactions (SN1and SN2) often observed Usually the most substituted olefin is the major product (Zaitsev's rule)
Reaction 26. Aromatic electrophilic substitution.
Reagents and conditions
Mechanistic details
Br2 and FeBr3 (or AlBr3) for bromination Cl2 and FeCl3 (or AlCl3) for chlorination I2 and H2O2 (or CuCl2) for iodination HNO3/H2SO4 for nitration SO3/H2SO4 for sulfonation RX and AlCl3 for alkylation RCOX and AlCl3 for acylation
positively charged electrophile adds to the aromatic ring in the rate-limiting step; the resulting carbocation reverts to aromaticity by the loss of proton the relative reactivity and regiochemistry of the reaction on substituted benzene derivatives is governed by the nature of the substituent: the substituents that are electron withdrawing by inductive and resonance effects are deactivating and meta-directing; the substituents that are electron withdrawing by inductive effects and electron donating by resonance are ortho- and para-directing and depending on the electron-density balance are deactivating (halides) or activating (O in ethers, N in amines or amides); the substituents that are electron donating by inductive and resonance (hyperconjugation) effects are activating and ortho- and para-directing
Reaction 27. Oxidation of side chains in aromatic compounds.
Reagents and conditions
Mechanistic details
KMnO4 or Na2Cr2O7
complex oxidation mechanism requires at least one benzylic hydrogen
Reaction 28. Hydrogenation of aromatic compounds.
Reagents and conditions
Mechanistic details
H2 (several hundred atm) over Pd H2 (1 atm) over Rh
heterogeneous catalysis with a complex mechanism no partial reduction possible
Reaction 29. Reduction of carbonyl compounds to alcohols.
Reagents and conditions
Mechanistic details
NaBH4 (or LiAlH4) for aldehydes and ketones LiAlH4 for carboxylic acids and esters BH3-THF for carboxylic acids
the hydrides deliver H to the carbonyl-group carbon (nucleophilic addition to C-O double bond) for carboxylic acids and their derivatives, the tetrahedral intermediate formed loses R'O group, and the newly formed carbonyl group is reduced again
Reaction 30. Addition of Grignard reagents to carbonyl compounds to yield alcohols.
Reagents and conditions
Mechanistic details
Grignard reagents are prepared by reacting organic halides with
nucleophilic addition of electron-rich (carbanion-like) carbon from the organometallic reagent to the
metallic magnesium in ether solvents usually carried out in ether solvents (ether, THF) organolithium compounds (RLi) can be used instead of Grignard reagents
electrophilic carbon of the carbonyl group the addition to esters takes place twice; the initially formed tetrahedral intermediate expels RO, regenerating the carbonyl group which reacts with the second molecule of the organometallic reagent
Reaction 31. Dehydration of alcohols.
Reagents and conditions
Mechanistic details
acid with a non-nucleophilic counterion (H2SO4) for tertiary substrates POCl3/pyridine for 2o and 1o alcohols
E1 mechanism for tertiary alcohols E2 mechanism for POCl3/pyridine (POCl3 converts -OH into a good leaving group: -OPOCl2) usually Zaitsev's rule followed (see Reaction 25)
Reaction 32. Oxidation of alcohols.
Reagents and conditions
Mechanistic details
PCC (pyridinium chlorochromate) or periodinane for oxidation of 1o alcohols to aldehydes (2o alcohols are oxidized to ketones) Jones' reagent (CrO3/H2SO4/H2O/acetone) or dichromate (Na2Cr2O7) for oxidation of 1o alcohols to carboxylic acids and 2o alcohols to ketones
E2-like elimination on chromate or periodinane intermediates
Reaction 33. The Williamson ether synthesis.
Reagents and conditions
Mechanistic details
alkoxides are prepared by reaction of alcohols with bases or alkali metals reaction of alkoxides with primary alkyl halides intramolecular reaction yields cyclic ethers
SN2 substitution reaction with oxygen serving as nucleophile
Reaction 34. Acidic cleavage of ethers.
Reagents and conditions
Mechanistic details
HI or HBr for 1o and 2o ethers HI, HBr and HCl for 3o ethers
SN2 for primary ethers (after protonation on oxygen, attack by X on the least substituted of the two carbons) SN1 for tertiary, benzylic or allylic ethers (after protonation on oxygen)
Reaction 35. Synthesis of epoxides with peroxyacids.
Reagents and conditions
Mechanistic details
peroxyacids (RCOOOH, for example m-chloroperoxybenzoic acid, MCPBA)
direct oxygen transfer from the peroxyacid to the alkene (syn stereochemistry)
Reaction 36. Ring-opening reactions of epoxides.
Reagents and conditions
Mechanistic details
acid catalysis (H2O, Cl, Br, I as nucleophiles) direct nucleophile addition (HO, RO, RNH2, R2N, RMgX)
under acid catalyzed conditions the protonated epoxide can be attacked by the nucleophile at the more (usually the major site of attack), or the less substituted site, depending on substitution patterns (anti stereochemistry results) direct nucleophilic attack (SN2) takes place at the least
substituted carbon (anti stereochemistry results)
Das könnte Ihnen auch gefallen
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Easter Island or Rapa NuiDokument6 SeitenEaster Island or Rapa NuiSofía I. Morales NavarroNoch keine Bewertungen
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- A Framework For The Integration of Ecosystem and Human HealthDokument14 SeitenA Framework For The Integration of Ecosystem and Human HealthSofía I. Morales NavarroNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- Brain Structure SummaryDokument2 SeitenBrain Structure SummarySofía I. Morales NavarroNoch keine Bewertungen
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (399)
- Urban Ecology and Urban Ecosystems, Understanding The Links To Human Health and Well BeingDokument8 SeitenUrban Ecology and Urban Ecosystems, Understanding The Links To Human Health and Well BeingSofía I. Morales NavarroNoch keine Bewertungen
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- Solubility Rules Made EasyDokument1 SeiteSolubility Rules Made EasySofía I. Morales NavarroNoch keine Bewertungen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- A Framework For The Integration of Ecosystem and Human HealthDokument14 SeitenA Framework For The Integration of Ecosystem and Human HealthSofía I. Morales NavarroNoch keine Bewertungen
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Interview QuestionsDokument1 SeiteInterview QuestionsSofía I. Morales NavarroNoch keine Bewertungen
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Interview QuestionsDokument1 SeiteInterview QuestionsSofía I. Morales NavarroNoch keine Bewertungen
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Med School Interview QuestionsDokument4 SeitenMed School Interview QuestionsSofía I. Morales NavarroNoch keine Bewertungen
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Amphetamines: ProfileDokument5 SeitenAmphetamines: ProfileSofía I. Morales NavarroNoch keine Bewertungen
- Med School Interview QuestionsDokument4 SeitenMed School Interview QuestionsSofía I. Morales NavarroNoch keine Bewertungen
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Table of Physical ConstantsDokument1 SeiteTable of Physical ConstantsSofía I. Morales NavarroNoch keine Bewertungen
- HFCS Everything You Wanted To Know But Were Afraid To AskDokument1 SeiteHFCS Everything You Wanted To Know But Were Afraid To AskSofía I. Morales NavarroNoch keine Bewertungen
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Effects of HFCSDokument8 SeitenThe Effects of HFCSSofía I. Morales NavarroNoch keine Bewertungen
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (73)
- Chapter 1 Introduction To Emergency Medical CareDokument38 SeitenChapter 1 Introduction To Emergency Medical CareSofía I. Morales NavarroNoch keine Bewertungen
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- Nmat Reviewer Orgchem PDFDokument15 SeitenNmat Reviewer Orgchem PDFAlice Katrina100% (1)
- Chapter 2 COE Alkenes and Alkynes II Addition ReactionsDokument54 SeitenChapter 2 COE Alkenes and Alkynes II Addition ReactionsRaven ShadeNoch keine Bewertungen
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Chapter 6: Reactions of Alkenes - Addition Reactions: Trans-1,2-Dimethylcyclopentane Cis-1,2-DimethylcyclopentaneDokument21 SeitenChapter 6: Reactions of Alkenes - Addition Reactions: Trans-1,2-Dimethylcyclopentane Cis-1,2-DimethylcyclopentaneRahma AshrafNoch keine Bewertungen
- @iitjeeadvancedmaterial Alcohols, Phenols & EthersDokument148 Seiten@iitjeeadvancedmaterial Alcohols, Phenols & EthersRonak kadamNoch keine Bewertungen
- Hydrocarbon-04 Solved ProblemsDokument14 SeitenHydrocarbon-04 Solved ProblemsRaju SinghNoch keine Bewertungen
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Haloalkanes and HaloarenesDokument28 SeitenHaloalkanes and HaloarenesDevansh TiwaryNoch keine Bewertungen
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- Alkynes: An Introduction To Organic Synthesis: (Ref: Mcmurry'S Organic Chemistry, 7 Edition)Dokument24 SeitenAlkynes: An Introduction To Organic Synthesis: (Ref: Mcmurry'S Organic Chemistry, 7 Edition)Tristan BadillaNoch keine Bewertungen
- 27 Alcohol Phenol Ether Formula Sheets Getmarks AppDokument15 Seiten27 Alcohol Phenol Ether Formula Sheets Getmarks AppFLASH FFNoch keine Bewertungen
- CHE 112 - Lecture 2Dokument103 SeitenCHE 112 - Lecture 2Martias WambiNoch keine Bewertungen
- Alkene Lecture - 3Dokument8 SeitenAlkene Lecture - 3Sukanya PhukonNoch keine Bewertungen
- CBSE Class 12 Chemistry Chapter 10 Haloalkanes and Haloarenes Revision NotesDokument99 SeitenCBSE Class 12 Chemistry Chapter 10 Haloalkanes and Haloarenes Revision NotesTECH STOVENoch keine Bewertungen
- CHM 2210 Practice Exam 3Dokument8 SeitenCHM 2210 Practice Exam 3Shaima MossamatNoch keine Bewertungen
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Organic Chemistry 2 Practice Exam 1Dokument15 SeitenOrganic Chemistry 2 Practice Exam 1KaybidoNoch keine Bewertungen
- Reaction of AlkenesDokument27 SeitenReaction of AlkenesHui JingNoch keine Bewertungen
- Mechanism of Organic ReactionsDokument45 SeitenMechanism of Organic ReactionsBapu ThoratNoch keine Bewertungen
- JPP-5 - (JLD 3.0) - Reaction Mechanisms - 05 SeptDokument50 SeitenJPP-5 - (JLD 3.0) - Reaction Mechanisms - 05 SeptHarsh ThakurNoch keine Bewertungen
- Chapter 9 Orgo Chem Test Bank PDFDokument77 SeitenChapter 9 Orgo Chem Test Bank PDFahmshiNoch keine Bewertungen
- Team Bootcamp - Reaction Summary Sheet PDFDokument30 SeitenTeam Bootcamp - Reaction Summary Sheet PDFDagne PovedaNoch keine Bewertungen
- Free Radical SubstitutionDokument22 SeitenFree Radical SubstitutionULFA TUFFAHATI100% (1)
- Reaction Road MapDokument2 SeitenReaction Road MapBenedick Jayson MartiNoch keine Bewertungen
- Haloalkanes And: HaloarenesDokument24 SeitenHaloalkanes And: HaloarenesWild SageNoch keine Bewertungen
- Chapter 3 AlkenesDokument63 SeitenChapter 3 AlkenesKonoli NuingNoch keine Bewertungen
- Overview of Alkene ReactionsDokument11 SeitenOverview of Alkene ReactionsVickyNoch keine Bewertungen
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- 5.2 Alkene 2Dokument6 Seiten5.2 Alkene 2Yusra IqbalNoch keine Bewertungen
- Reaction Mechanisms OverviewDokument19 SeitenReaction Mechanisms OverviewHanhHongDaoNoch keine Bewertungen
- Chapter 10 Alkenes - SmithDokument14 SeitenChapter 10 Alkenes - SmithRen Liew Jia Qing75% (4)
- Catalytic Hydrogenation of Alkynes: As With Alkenes, Addition of Each Pair of H's Is SynDokument6 SeitenCatalytic Hydrogenation of Alkynes: As With Alkenes, Addition of Each Pair of H's Is SynVishal SinghNoch keine Bewertungen
- Organic ChemistryDokument20 SeitenOrganic ChemistryGirish RaguvirNoch keine Bewertungen
- L14 - (JLD 3.0) - Reaction Mechanisms - 02 SeptDokument62 SeitenL14 - (JLD 3.0) - Reaction Mechanisms - 02 SeptkisuisoffbeatNoch keine Bewertungen
- Oc 1. Alkynes and Alkadienes Final RK Sir - 05.03.14 (01-16) PDFDokument16 SeitenOc 1. Alkynes and Alkadienes Final RK Sir - 05.03.14 (01-16) PDFAman9692100% (1)