Beruflich Dokumente
Kultur Dokumente
Thermal Physics Lecture 16
Hochgeladen von
OmegaUserCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Thermal Physics Lecture 16
Hochgeladen von
OmegaUserCopyright:
Verfügbare Formate
Physics 301 18-Oct-2002 16-1
The Sackur-Tetrode Entropy and Experiment
In this section well be quoting some numbers found in K&K which are quoted from
the literature.
You may recall that Ive several times asked how one would measure absolute entropy?
I suspect that I pretty much gave it away (if you hadnt gured it out already) in the last
section. The answer is you have to measure heat transfers from a state of known absolute
entropy to the desired state so that you can calculate
_
dQ/. What is a state of known
entropy? Answer, at absolute 0, one expects the entropy to be very small and we can take
it to be 0. Actually, there is the third law of thermodynamics (not as famous as the rst
two!) which says that the entropy should go to a constant as 0.
At absolute 0, a reasonable system will be in its ground state. In fact the ground state
might not be a single state. For example if we consider a perfect crystal, its ground
state is clearly unique. But real crystals have imperfections. Suppose a crystal is missing
a single atom from its lattice. If there are N atoms in the crystal there are presumably N
dierent sites from which the atom could be missing so the entropy is log N. Also, theres
presumably an energy cost for having a missing atom, so the crystal is not really in its
ground state. But this might be as close as we can get with a real crystal. The point is
that the energy and the entropy are both very small in this situation and very little error
is made by assuming that (0) = 0. (Compare log N with N log(n
Q
/n) when N 10
23
!)
In fact, a bigger problem is getting to very low temperatures. In practice, one gets as
low as one can and then extrapolates to = 0 using a Debye law (assuming an insulating
solid). So to measure the entropy of a monatomic ideal gas such as neon, one makes heat
capacity measurements and does the integral
_
C() d/. The heat capacity measure-
ments go to as low a as needed to get a reliable extrapolation to 0 with the Debye law.
According to K&K, the calculation goes like this: solid neon melts at 24.55 K. At the
melting point, its entropy (by extrapolation and numerical integration) is
S
melting
S
0
= 14.29
J
mol K
.
To melt the solid (which occurs at a constant temperature) requires 335 J mol
1
so the
entropy required to melt is
S
melt
= 13.65
J
mol K
.
Again, a numerical integration is required to nd the entropy change as the liquid neon is
taken from the freezing point to the boiling point at 27.2 K. This is
S
boiling
S
freezing
= 3.85
J
mol K
.
Finally, 1761 J mol
1
is required to boil the neon at the boiling point, and
S
boil
= 64.74
J
mol K
.
Copyright c 2002, Princeton University Physics Department, Edward J. Groth
Physics 301 18-Oct-2002 16-2
Now we have a gas to which we can apply the Sackur-Tetrode expression. Assuming
S
0
= 0, the total is
S
vapor
= S
boil
+ (S
boiling
S
freezing
) +S
melt
+ (S
melting
S
0
) = 96.40
J
mol K
,
= 6.98 10
24
/mol ,
where I have quoted the sum from K&K which diers slightly from the sum you get by
adding up the four numbers presumably because there is some round-o in the input
numbers. (For example, using a periodic table on the web, I nd the melting and boiling
points of neon are 24.56 K and 27.07 K.) According to K&K, the Sackur-Tetrode value
for neon at the boiling point is
S
SackurTetrode
= 96.45
J
mol K
,
which is in very good agreement with the observed value.
When I plug into the Sackur-Tetrode expression I actually get,
S
SackurTetrode
= 96.47
J
mol K
,
still in very good agreement with the observed value. Why did I get a slightly dierent
value than that quoted in K&K? I used
S = R
_
log
_
_
mkT
2h
2
_
3/2
_
p
kT
_
+
5
2
_
,
Everything can be looked up, but Im using p/kT instead of N
0
/V , which assumes the
ideal gas law is valid. However, this expression is being applied right at the boiling point,
so its not clear that the ideal gas law should work all that well.
Some other things to note. (1) If we had left out the N! over counting correction, We
would have to add
R(log N
0
1) = 447
J
mol K
,
to the above. This swamps any slight problems with deviations from the ideal gas law or
inaccuracies in the numerical integrations! (2) The Sackur-Tetrode expression includes h
which means it depends on quantummechanics. So this is an example where measurements
of the entropy pointed towards quantum mechanics. Of course, h occurs inside a logarithm,
so it might not have been so easy to spot!
Copyright c 2002, Princeton University Physics Department, Edward J. Groth
Physics 301 18-Oct-2002 16-3
The Ideal Fermi Gas
Consider a metal like sodium or copper (or the other metals in the same columns in the
periodic table). These metals have one valence electronan electron which can be easily
removed from the atom, so these atoms often form chemical bonds as positively charged
ions. In the solid metal, the valence electrons arent bound to the atoms. How do we know
this? Because the metals are good conductors of electricity. If the electrons were bound to
the atoms they would be insulators. Of course, there are interactions between the electrons
and the ions and between the electrons and other electrons. But, as a rst approximation
we can treat all the valence electrons as forming a gas of free (non-interacting) particles
conned to the metal.
Lets do a little numerology. First, lets calculate the quantum concentration for an
electron at room temperature,
n
Q
=
_
m
e
kT
2h
2
_
3/2
,
=
_
(9.108 10
28
g) (1.380 10
16
erg K
1
) (300 K)
2 (1.054 10
27
erg s)
2
_
3/2
,
= 1.26 10
19
cm
3
,
=
_
1
43
A
_
3
.
In other words, the density of electrons is equal to the room temperature quantum con-
centration if there is one electron every 43
A. Now consider copper. It has a density of
8.90 g cm
3
and an atomic mass of 63.54 amu. So the number density of copper atoms is
n
Cu
= 8.44 10
22
cm
3
=
_
1
2.3
A
_
3
.
The number of electrons in the electron gas (assuming one per copper atom) exceeds the
quantum concentration by a factor of 6700. For copper the actual concentration and the
quantum concentration are equal at a temperature of about 100,000 K (assuming we could
get solid copper that hot!).
The upshot of all this is that we are denitely not in the classical domain when dealing
with an electron gas in metals under normal conditions. We will have to use the Fermi-
Dirac distribution function. Low energy states are almost certain to be lled. When this
is true, the system is said to be degenerate. Furthermore, the electron gas is cold in the
sense that thermal energies are small compared to the energies required to conne them
to high densities. (This is most easily seen from an uncertainty principle argument.)
Copyright c 2002, Princeton University Physics Department, Edward J. Groth
Physics 301 18-Oct-2002 16-4
So as a rst approximation, we can use the Fermi-Dirac distribution at zero temper-
ature. This means we will ignore thermal energies altogether. At zero temperature, the
Fermi-Dirac distribution becomes,
f() =
1
e
( )/
+ 1
_
1, < ;
0, > .
Imagine a chunk of copper in which all the valence electrons have been removed (it would
have a rather large electric charge . . .). Add back one valence electron remembering that
the temperature is 0. This electron goes into the lowest available state. Add another
electron, it goes into the state with the next lowest energy. Actually its the same center
of mass state and the same energy, but the second electron has its spin pointing in the
opposite direction from the rst. The third electron goes in the state with the next lowest
energy. And so on. What we are doing is lling up states (with no gaps) until we run out
of valence electrons. Since we have the lowest possible energy, this conguration must be
the ground state (which is the state the system should be in at 0 temperature!).
We must choose the chemical potential so that our metal has the correct number
of valence electrons. To do this, we need to know the number of states. Since we are
considering free electrons, we are dealing with single particle states in a box. This is the
same calculation weve done before. The number of states with position vector in the
element d
3
x and momentum vector in the element d
3
p is
dn(x, p) = 2
d
3
xd
3
p
8
3
h
3
,
where the factor of 2 arises because there are two spin states for each center of mass state.
When the number of states is used in an integral, the integral over d
3
x just leads to the
volume of the box, V . The element d
3
p = p
2
dp d and the solid angle may be integrated
over to give 4p
2
dp. Finally, the independent variable may be converted from momentum
to energy with p
2
/2m = , and we have
dn() =
V
2
2
_
2m
h
2
_
3/2
d .
Its customary to write this as the density of states per unit energy
D() d =
V
2
2
_
2m
h
2
_
3/2
d .
Now were ready to calculate . At zero temperature, the occupancy is 1 up to and
0 above , so the total number of electrons is
N =
_
0
D() d .
Copyright c 2002, Princeton University Physics Department, Edward J. Groth
Physics 301 18-Oct-2002 16-5
Before we do this integral, a bit of jargon. The energy of the highest lled state at zero
temperature is called the Fermi energy,
F
. So ( = 0) =
F
. Then
N =
_
F
0
V
2
2
_
2m
h
2
_
3/2
d =
V
3
2
_
2m
h
2
_
3/2
3/2
F
,
or
F
= (3
2
n)
2/3
h
2
2m
,
where n = N/V is the concentration. One also speaks of the Fermi temperature dened by
F
= kT
F
=
F
. This is not actually the temperature of anything, but is a measure of the
energy that separates degenerate from non-degenerate behavior. The Fermi temperature
is a few tens of thousands of Kelvins for most metals, so the electron gas in typical metals
is cold. Having determined the Fermi energy, we can determine the total energy of the
gas. We just add up the energies of all the occupied states
U
0
=
_
F
0
V
2
2
_
2m
h
2
_
3/2
d =
V
5
2
_
2m
h
2
_
3/2
5/2
F
=
V
5
2
(3
2
n)
5/3
h
2
2m
=
3
5
N
F
,
where the subscript on U indicates the ground state energy.
In the ground state, the average energy of an electron is 3/5 the Fermi energy. Also
note that the concentration contains V
1
which means U
0
V
2/3
which means that as
the system expands, the energy goes down which means it must be exerting a pressure on
its container. This is called degeneracy pressure. In fact,
p =
_
U
0
V
_
,N
=
2
3
U
0
V
,
so
pV =
2
3
U
0
,
just as for an ideal gas. Note that the derivative is to be taken at constant entropy. We
are dealing with the ground state, so the entropy is constant at 0.
As a point of interest, using the concentration for copper that we calculated earlier,
we nd
F
= 7.02 eV and T
F
= 81, 500 K,
and the electron gas in copper really is cold all the way up to the point where copper
melts! (1358 K)
Copyright c 2002, Princeton University Physics Department, Edward J. Groth
Physics 301 18-Oct-2002 16-6
Heat Capacity of a Cold Fermi Gas
In the preceding section we considered the Fermi gas in its ground state. This does
not allow us to consider adding heat to the gas because the gas would no longer be in the
ground state. To calculate the heat capacity, we need to expand our treatment a bit.
What we need to do is calculate the energy of the gas as a function of temperature. We
will calculate the dierence between the ground state energy and the energy at temperature
and we will make use of the fact that the gas is cold.
With a cold gas, all the action occurs within of . That is the occupancy goes from
1 to 0 over a range of a few centered at . Since we have a cold gas, this is a relatively
narrow range.
The dierence in energy between the gas at temperature and the gas in the ground
state is
U() =
_
0
D()f() d U
0
,
=
_
0
D()f()(
F
) d +
_
0
D()f()
F
d U
0
,
=
_
0
D()f()(
F
) d +N
F
U
0
.
Now, we dierentiate with respect to to get the heat capacity.
C
V
=
U
,
=
_
0
D()
df()
d
(
F
) d .
So far, everything is exact, now we start making approximations. At = 0, the distribu-
tion is a step function, so its derivative is a delta function (a very sharply peaked function
in the neighborhood of the step). This means that the main contribution to the integral
occurs (even if is not 0) when is very close to . So we will ignore the variation in the
density of states, evaluate it at and take it out of the integral. What about ? At = 0,
=
F
. As increases, decreases, but when
F
, the change in is negligibly small
(plot some curves!), so we will take =
F
. Then we have
C
V
= D(
F
)
_
0
df()
d
(
F
) d .
Now,
df
d
=
d
d
_
1
e
(
F
)/
+ 1
_
=
F
2
e
(
F
)/
_
e
(
F
)/
+ 1
_
2
.
Copyright c 2002, Princeton University Physics Department, Edward J. Groth
Physics 301 18-Oct-2002 16-7
At this point, we change variables to x = (
F
)/ and we have
C
V
= D(
F
)
_
+
x
2
e
x
dx
(e
x
+ 1)
2
,
where the actual lower limit of integration,
F
/, has been replaced by since all the
contribution to the integral is in the neighborhood of x = 0. It turns out that the integral
is
2
/3, so
C
V
=
2
3
D(
F
) ,
a surprisingly simple result! If we plug in the expression for the density of states, we have
D(
F
) =
3N
2
F
,
and
C
V
=
2
2
N
F
=
2
2
N
F
.
In conventional units,
C
V
=
2
2
Nk
T
T
F
.
Some comments. This is proportional to T which means that the energy of the electron
gas is U
0
+ constant
2
. Can we see how this happens? In going from 0 to , we are
exciting electrons in the energy range from
F
F
by giving them a thermal energy of
roughly . The number of such electrons is roughly N/
F
, so the added energy is roughly
N
2
/
F
. The Fermi gas heat capacity is quite a bit smaller than that of a classical ideal gas
with the same energy and pressure. This is because only the fraction /
F
of the electrons
are excited out of the ground state. At low temperatures, heat capacities of metals have
a linear term due to the electrons and a cubic term due to the lattice. Some experimental
data may be found in K&K.
Copyright c 2002, Princeton University Physics Department, Edward J. Groth
Das könnte Ihnen auch gefallen
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Tensor AnalysisDokument29 SeitenTensor AnalysisPugazhenthi Thananjayan100% (2)
- Part 3 Lattice Quantum Ising RGDokument28 SeitenPart 3 Lattice Quantum Ising RGOmegaUserNoch keine Bewertungen
- Fundamentals of Statistical Physics: Leo P. Kadanoff University of Chicago, USADokument17 SeitenFundamentals of Statistical Physics: Leo P. Kadanoff University of Chicago, USAOmegaUserNoch keine Bewertungen
- Part 7 Mean Field TheoryDokument40 SeitenPart 7 Mean Field TheoryOmegaUserNoch keine Bewertungen
- Math 404 Lecture NotesDokument31 SeitenMath 404 Lecture NotesOmegaUserNoch keine Bewertungen
- Part 5 Momentum HopsDokument27 SeitenPart 5 Momentum HopsOmegaUserNoch keine Bewertungen
- Outline For Fundamentals of Statistical PhysicsDokument13 SeitenOutline For Fundamentals of Statistical PhysicsOmegaUserNoch keine Bewertungen
- Part 2 Basics of Statistical Physics1Dokument24 SeitenPart 2 Basics of Statistical Physics1OmegaUserNoch keine Bewertungen
- Part 8 Beyond Mean FieldDokument42 SeitenPart 8 Beyond Mean FieldOmegaUserNoch keine Bewertungen
- Part 4 Diffusion and HopsDokument24 SeitenPart 4 Diffusion and HopsOmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L19Dokument3 SeitenStatistical Mechanics Lecture Notes (2006), L19OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L20Dokument4 SeitenStatistical Mechanics Lecture Notes (2006), L20OmegaUserNoch keine Bewertungen
- Part 6 Boson-FermionDokument16 SeitenPart 6 Boson-FermionOmegaUserNoch keine Bewertungen
- Chapter 1 ChemDokument4 SeitenChapter 1 ChemOmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L14Dokument4 SeitenStatistical Mechanics Lecture Notes (2006), L14OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L30Dokument4 SeitenStatistical Mechanics Lecture Notes (2006), L30OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L28Dokument5 SeitenStatistical Mechanics Lecture Notes (2006), L28OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L21Dokument4 SeitenStatistical Mechanics Lecture Notes (2006), L21OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L13Dokument7 SeitenStatistical Mechanics Lecture Notes (2006), L13OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L15Dokument4 SeitenStatistical Mechanics Lecture Notes (2006), L15OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L16Dokument4 SeitenStatistical Mechanics Lecture Notes (2006), L16OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L26Dokument5 SeitenStatistical Mechanics Lecture Notes (2006), L26OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L3Dokument5 SeitenStatistical Mechanics Lecture Notes (2006), L3OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L18Dokument6 SeitenStatistical Mechanics Lecture Notes (2006), L18OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L7Dokument8 SeitenStatistical Mechanics Lecture Notes (2006), L7OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L12Dokument7 SeitenStatistical Mechanics Lecture Notes (2006), L12OmegaUserNoch keine Bewertungen
- Statistical Mechanics Lecture Notes (2006), L5Dokument11 SeitenStatistical Mechanics Lecture Notes (2006), L5OmegaUserNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5795)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1091)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Spring Return Dosing PumpsDokument12 SeitenSpring Return Dosing PumpsheleonmNoch keine Bewertungen
- Steel BS 5950Dokument26 SeitenSteel BS 5950Rodain NajjarNoch keine Bewertungen
- P22 23 CRD PDFDokument2 SeitenP22 23 CRD PDFMikka IdanoNoch keine Bewertungen
- Design properties of HEA profiles for S235 steel class (γ = 1.00, units = mm)Dokument18 SeitenDesign properties of HEA profiles for S235 steel class (γ = 1.00, units = mm)scarto08Noch keine Bewertungen
- Rodera GarciaDokument120 SeitenRodera GarciaBathula BujjiNoch keine Bewertungen
- Experiment To Observe Temporary Mount of A Leaf Peel To Show StomataDokument3 SeitenExperiment To Observe Temporary Mount of A Leaf Peel To Show StomataEliseo Pamandanan0% (1)
- Pressure Vessel, Asme CodeDokument10 SeitenPressure Vessel, Asme Codeabhi_mohit2005100% (1)
- Lessons For The Young Economist Robert P MurphyDokument422 SeitenLessons For The Young Economist Robert P Murphysleepyninjitsu100% (1)
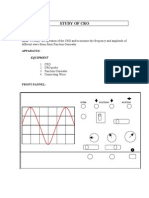
- CRODokument4 SeitenCROboopathy566Noch keine Bewertungen
- Conductivity TesterDokument4 SeitenConductivity TesterFelix PintoNoch keine Bewertungen
- Thermodynamics (TKJ3302) Lecture Notes - 2 Energy, Energy Transfer, and General Energy AnalysisDokument37 SeitenThermodynamics (TKJ3302) Lecture Notes - 2 Energy, Energy Transfer, and General Energy Analysismamat88100% (4)
- Final End Sem Solution-2018Dokument9 SeitenFinal End Sem Solution-2018Sriram MudunuriNoch keine Bewertungen
- (ASTM - D.1586) Diskripsi Tanah Standard Penetration TestDokument6 Seiten(ASTM - D.1586) Diskripsi Tanah Standard Penetration TestAndi KasmaNiaNoch keine Bewertungen
- Part A1 Chapter 2 - ASME Code Calculations Stayed Surfaces Safety Valves FurnacesDokument25 SeitenPart A1 Chapter 2 - ASME Code Calculations Stayed Surfaces Safety Valves Furnacesfujiman35100% (1)
- SG 2Dokument5 SeitenSG 2Hakdog SheinNoch keine Bewertungen
- Parastesh (2019) - Shape Optimization of Cold-Formed Steel Beam-Columns With Practical and Manufacturing ConstraintsDokument11 SeitenParastesh (2019) - Shape Optimization of Cold-Formed Steel Beam-Columns With Practical and Manufacturing ConstraintsGogyNoch keine Bewertungen
- Gunclean Toftejorg I65 DDokument55 SeitenGunclean Toftejorg I65 Dpetrakou aglaiaNoch keine Bewertungen
- Pumping MachinesDokument2 SeitenPumping MachinesKerwinPerezNoch keine Bewertungen
- Metal Film Precision Resistors: Resistive Metal Films and A New Resistor ConceptDokument19 SeitenMetal Film Precision Resistors: Resistive Metal Films and A New Resistor ConceptfbaldnerNoch keine Bewertungen
- Weekly Home Learning Plan (Science) : Week 1, Quarter 1, October 5 - 9, 2020Dokument4 SeitenWeekly Home Learning Plan (Science) : Week 1, Quarter 1, October 5 - 9, 2020Iris AriesNoch keine Bewertungen
- Practice Test 4Dokument14 SeitenPractice Test 4shreyshreyNoch keine Bewertungen
- The Touchstone of Life (Molecular Information, Cell Communication and The Foundations of Life) by Werner R. Loewenstein (1999) RDokument376 SeitenThe Touchstone of Life (Molecular Information, Cell Communication and The Foundations of Life) by Werner R. Loewenstein (1999) RAnonymous yu09qxYCMNoch keine Bewertungen
- Scia Engineer 2014 - Advanced Concept Training - FEM PDFDokument102 SeitenScia Engineer 2014 - Advanced Concept Training - FEM PDFericfgNoch keine Bewertungen
- Steam TrapsDokument10 SeitenSteam Trapsmaninder_khasria100% (1)
- Juan Dela Cruz, RRT: Personal InformationDokument3 SeitenJuan Dela Cruz, RRT: Personal InformationShiela Leomo-VillaramaNoch keine Bewertungen
- 3850 Certificate in Mathematics - Sample Paper Stage 3: Candidate Name (First, Middle, Last)Dokument20 Seiten3850 Certificate in Mathematics - Sample Paper Stage 3: Candidate Name (First, Middle, Last)shan100% (1)
- S-18 20Dokument2 SeitenS-18 20Raghavendra MurthyNoch keine Bewertungen
- Vector NotationDokument2 SeitenVector NotationAmmar BegenNoch keine Bewertungen
- IPSF Code of Points 2017-18 Final EnglishDokument135 SeitenIPSF Code of Points 2017-18 Final EnglishIsabela BiazottoNoch keine Bewertungen
- Elevator Traction Machine 20131018 Elevator Traction MachineDokument8 SeitenElevator Traction Machine 20131018 Elevator Traction MachineTh NattapongNoch keine Bewertungen