Beruflich Dokumente
Kultur Dokumente
Jurnal Baso4 Anor
Hochgeladen von
Hahahihi_D_flu_9405Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Jurnal Baso4 Anor
Hochgeladen von
Hahahihi_D_flu_9405Copyright:
Verfügbare Formate
Journal of Crystal Growth 234 (2002) 533538
Flux growth of baryte-type BaSO4 from chloridic alkaline metal solvents
D. Ehrentraut*, M. Pollnau
Department of Microtechnique, Institute of Applied Optics, Swiss Federal Institute of Technology, CH-1015 Lausanne, Switzerland Received 14 May 2001; accepted 29 August 2001 Communicated by T. Hibiya
Abstract The growth of BaSO4 from high-temperature uxes of both chloridic alkaline metal and alkaline-earth metal solvents has been investigated. Two binary alkaline-metal solvent systems containing LiCl and the additive ternary system with CsClKClNaCl where optimized with respect to solute concentration and growth temperature. It is found that the solution of BaSO4 in these solvents increases with decreasing average radius of the solvent cation. In addition, the solubility of BaSO4 is increased in the LiCl-containing uxes by a chemical reaction between solute and solvent. Binary solvents of LiClKCl and LiClCsCl delivered prismatic crystals of 2 cm size, whereas the CsClKClNaCl ux prefers the growth of short-prismatic crystals. The crystals grown under optimized conditions were crack free, colorless, and transparent. r 2002 Elsevier Science B.V. All rights reserved.
PACS: 61.72.Qq; 81.05.Je; 81.10.Dn Keywords: A1. Solubility; A1. Solvents; A2. Growth from solution; B1. Barium compounds; B1. Lithium compounds
1. Introduction Transition-metal (TM) ions are widely used as activator ions in laser crystals. Prominent examples are Al2O3 : Cr3+, Ca2+co-doped Y3Al5O12 : Cr4+, and Al2O3 : Ti3+, and the valence states of TM ions vary signicantly (for example, the valence states of Cr vary theoretically from Cr2+ to Cr6+). An interesting but so far rather poorly investigated material is the baryte type of BaSO4. First,
*Corresponding author. Fax: +41-21-6933701. E-mail address: dirk.ehrentraut@ep.ch (D. Ehrentraut).
this host oers the possibility to substitute the sulphur ion in the SO2 4 -tetrahedron by fourfoldcoordinated TM ions in their high valence states (typically TM5+ or TM6+). Second, BaSO4 forms solid solutions with SrSO4 [1] and CaSO4 [2]. In this series, the refractive index n decreases with decreasing size of the cation. For example, in the solid solution BaxSr1xSO4 n ranges from n 1:636 [3] (x 1) to n 1:631 [4] (x 0:78). This feature oers the possibility to create waveguide structures by adjusting a lower n for the substrate compared to the active layer. The disadvantage of BaSO4 is that it cannot be grown from its melt because of thermal decomposition [5]. In addition,
0022-0248/02/$ - see front matter r 2002 Elsevier Science B.V. All rights reserved. PII: S 0 0 2 2 - 0 2 4 8 ( 0 1 ) 0 1 6 9 3 - 1
534
D. Ehrentraut, M. Pollnau / Journal of Crystal Growth 234 (2002) 533538
BaSO4 undergoes a phase transition [6] at 10901C to a high-temperature a-form. Therefore, only growth techniques from solutions are applicable. Group I (alkaline metal) and group II (alkalineearth metal) chlorides are suitable solvents for alkaline tungstates, molybdates, and sulphates [7]. These chlorides are non-toxic and available in large quantities of adequate purity. We investigated the growth of BaSO4 crystals from dierent uxes of chloridic alkaline metals and alkaline-earth metals. Our main emphasis when searching for a suitable solvent was put on the solubility of BaSO4 in the solvent. Dierences of the valence states between solute and solvent [8] and a lower ionic radius of the solvent compared to the solute are necessary to achieve a high solubility. Furthermore, the solubility should be suciently high at lower temperatures, because TM ions have the tendency to reduce their valence states with increasing growth temperatures. In this study, we report on ux optimization for the growth of large BaSO4 crystals. Mixtures of LiCl, NaCl, KCl, RbCl, CsCl, CaCl2, and MgCl2 were investigated systematically as suitable solvents for BaSO4 at temperatures lower than 6001C. Large BaSO4 crystals will be used as substrates in a follow-up layer-growth process by means of liquid-phase epitaxy (LPE) to fabricate optical thin lms activated with TM ions in their high oxidation states (TM5+ or TM6+) at temperatures below 6001C.
2. Experimental procedure We used chemicals with purities from 3 to 4.5 N. The chemicals were dried at 1501C for 24 h. After weighing and mixing, they were lled into Al2O3 crucibles and immediately heated for melting. The ux experiments were carried out under ambient atmosphere in a resistance heated furnace which provided a precision in temperature control of 0.3 K. Start temperatures Tstart between 5101C and 6001C for the LiClKCl system, 4501C and 6001C for the LiClCsCl system, and 5701C and 6001C for the CsClKClNaCl system were chosen. The cooling rate from Tstart down to the solidication temperature Ts was 1.0 K min1 for all experiments. Table 1 shows the quantitative variation of the compositions of the solvents and the concentrations of BaSO4. The water-insoluble BaSO4 crystals can be extracted easily from the ux after the experiment by solving with distilled water. The crystals were checked for phase purity using X-ray powder diraction (Guinier-de Wol method) with silicon as the internal standard. Optical characterization was carried out by means of microscopy.
3. Results and discussion The crystal symmetry of baryte-type BaSO4 is orthorhombic (Pnma) with lattice constants ( b0 5:450 A, ( and c0 7:152 A ( [3]. a0 8:878 A,
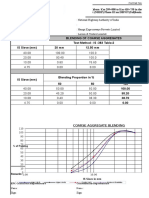
Table 1 Solidication point (Ts ), growth time (tG ), cooling rate, and the concentrations of BaSO4 cBaSO4 for dierent compositions of solvents for the ux growth of baryte-type BaSO4 Composition (mol%) LiCl : KCl 65 : 35 60 : 40 35 : 65 LiCl : CsCl 57 : 43 50 : 50 CsCl : KCl: NaCl 45.2 : 24.4 : 30.4 Ts (1C) 405 348 590 326 333 480 Ref. in [10] Fig. 1777 Fig. 1777 Fig. 1777 Fig. 1227 Fig. 1227 Fig. 3241 tG (h) 165 112252 10 105255 215245 120 Cooling rate (K h1) 1 1 1 1 1 1 cBaSO4 (wt%) 15.0 9.030.0 12.015.0 3.011.0 4.09.0 2.56.0
D. Ehrentraut, M. Pollnau / Journal of Crystal Growth 234 (2002) 533538
535
We investigated the following ux systems in more detail because of their high yield of grown BaSO4 crystals in our preliminary experiments: the binary LiClKCl and LiClCsCl, and the additive ternary CsCLKClNaCl solvents. The latter was reported [9] to be successful in ux-growth experiments of TM-ion doped BaSO4. Combinations of the group II chlorides SrCl2 and BaCl2 with group I and/or group II chlorides are not of interest for our purposes due to melting points above 6001C [10]. The parameters in our ux experiments were: (a) concentrations c of the constituents of the solvent, (b) concentration cBaSO4 of BaSO4 in the solvent, and (c) the start temperature Tstart of the experiment. The variation of parameter (a) will inevitably change the temperature of solidication of the solvent. 3.1. Solubility The solubility of BaSO4 in chloridic solvents was estimated under growth conditions. The solubility sBaSO4 is the value of cBaSO4 ; the concentration of BaSO4, where nucleation of BaSO4 is initiated. In each system we added dierent amounts of the solute BaSO4 in order to determine the solubility of BaSO4. High supersaturation s; s cBaSO4 ce =ce ; with ce ; the equilibrium concentration of the solute, may result in growth of dendrites (instable growth), whereas a solute concentration cBaSO4 below the critical supersaturation will not initiate crystal growth. The region between the curves of ce and sBaSO4 in a typical concentration-versus-temperature diagram is the Ostwald-Miers-region, a metastable region of s: The results of our experiments are shown in Figs. 1ac. The LiClKCl (Fig. 1a) and LiClCsCl (Fig. 1b) uxes show a dierent behavior in the solution of BaSO4 compared to the CsClKCl NaCl ux (Fig. 1c). Each straight line in Figs. 1ac corresponds to one ux experiment. The dierences in cBaSO4 between Tstart (open squares) and Ts (solid squares) represent the amounts of BaSO4 deposit obtained in each experiment. Complete dissolution of BaSO4 in the solvent gives a line without slope, and no deposit is obtained, see line 6 in Fig. 1a and lines 4, 5, and 7 in Fig. 1b. A
similar behavior, complete dissolution of BaSO4 and no deposit at the end of the experiment, was not found in the additive ternary system and we obtained BaSO4 deposits for all concentrations of BaSO4 between 2.5 and 6.0 wt%, Fig. 1c. The two binary solvents are distinguished by the solubility of BaSO4, which diers by a factor of almost 2 to the favor of LiClKCl. The results of Figs. 1a and b show an increase of the solubility sBaSO4 of BaSO4 with increasing LiCl concentration. This increase can partly be explained by the decreasing average radius of the solvent cations r xA rA xB rB ; with x; the mole fractions of the salts A, B, and r; the radii of the cations A+, B+. The values of the ionic radii are taken from [11]. Fig. 2 displays the dependence of sBaSO4 on r : The average cation radius of the ( for the 65 : 35 solvent increases from 0.91 A ( LiCl : KCl system to 1.55 A for the 67 : 33 CsCl : NaCl system. The increase of sBaSO4 with decreasing average cation radius is very pronounced. The dierence in sBaSO4 between the 65 : 35 LiCl : KCl solvent (symbol 1 in Fig. 2), and the CsCl : KCl : NaCl solvent (symbol 6 in Fig. 2), is almost one order of magnitude. In addition, the solubility depends on the chemical nature of the solvent. In certain cases, a chemical reaction between solvent and solute may take place, which changes the solubility of the solute. Alkaline-earth metal chlorides are completely dissociated in the molten state [12], and are, therefore, comparable which each other in their solubility of a common solute. As we will see in the following, this is not the case for all the alkalinemetal solvents. The following results conrm our assumption of the existence of chemical reaction during the process of BaSO4 dissolution, and the formation of new phases. The minimum amounts of BaSO4 to initiate crystal growth are: 9 wt% for LiClKCl, 5.5 wt% for LiClCsCl, and o2.5 wt% for CsClKClNaCl. However, the highest yield of crystallized BaSO4 of 83.3% was achieved with the CsClKClNaCl system, the system with the smallest sBaSO4 : The LiCl KCl and the LiClCsCl systems delivered highest yields of only 55.6% and 42.8%, respectively. By use of the values [13] for the enthalpy of formation DHf ; we calculated the following reac-
536
D. Ehrentraut, M. Pollnau / Journal of Crystal Growth 234 (2002) 533538
Fig. 1. Experimental determination of the solubility of BaSO4 in group I chlorides in dependence on temperature and concentration: (a) LiClKCl solvent (values for 100 : 0 are derived from [10], value for the eutectic temperature Teutectic after [LAG], (b) LiClCsCl solvent (value for Teutectic after [16], and (c) CsClKClNaCl solvent (value for Teutectic after [10]. The compositions of the solvents are given. The dierences in the solidication temperatures Ts for the LiCl-containing systems in (a) and (b) are due to the dierent compositions of the solvents. The values for Tstart (&) and Ts () are given for each curve. The dierence in cBaSO4 between Tstart and Ts indicates the mass in wt% of the crystalline BaSO4 deposit of each experiment.
tion to be energetically preferred in the LiClcontaining systems: 2LiCl BaSO4 - Li2 SO4 BaCl2 : At temperatures of 600 K and above, DHf increases to the favor of the products, DHf (600 K)=
T
6.731 kJ mol1, DHf (800 K)=131.258 kJ mol1. In contrast, in the additive ternary system no sulfate other than BaSO4 is energetically stable, because it does not contain LiCl. The shift of the chemical equilibrium toward the formation of Li2SO4 and BaCl2 is probably initiated during dissociation of LiCl and BaSO4
D. Ehrentraut, M. Pollnau / Journal of Crystal Growth 234 (2002) 533538
537
Fig. 2. Solubiltity of BaSO4 (sBaSO4 ) as function of the average solvent cation radius r : The strong increase of sBaSO4 with decreasing r is due to the increased LiCl concentration in the ux, which, rstly, decreases r ; and, secondly, introduces a chemical reaction between solute and solvent. Fig. 3. Comparison of the habits of BaSO4 crystals grown from: (a) the LiClKCl solvent and (b) the CsClKClNaCl solvent. The aspect ratios for the specimen of (a) and (b) are 8 and 2, respectively.
while heating up the ux. The concentrations of Li2SO4 and BaCl2 increase with increasing temperature due to the increasing gain in DHf : While cooling down, the chemical reaction is probably only partly reversed and both formatted salts remain in the liquid ux. Part of the formed Li2SO4 and BaCl2 remain in the ux even after its solidication. Due to the high solubility of Li2SO4 and BaCl2 in water, 3.11 and 1.78 mol l1 at 251C [3], respectively, these components are lost during extraction of the insoluble BaSO4 crystals (solubility 105 mol l1 at 251C [3]). The phase diagram of the Li2SO4BaCl2 system [10] describes the coexistence of solid BaSO4 and a liquid solution of Li2SO4BaCl2 below the liquidus line at temperatures much lower than 9001C. This is yet another indication for the hypothesis of the formation of Li2SO4 and BaCl2 during the ux growth of BaSO4 from LiCl-containing solvents. 3.2. Crystal habit, quality, and size In general, the habit of a crystal is in close relation to its growth mechanism and the incor-
poration of impurities and inclusions, which both depend on the growth conditions [14]. The ability to control the habit oers the chance to grow tailor-made crystals with respect to follow-up applications. Figs. 3a and b show typical ux-grown BaSO4 crystals. We observed prismatic habits for all the systems. The fastest growth direction is the a-direction, 1 0 0; and therefore the crystals are elongated along 1 0 0: The b=c-ratio is almost unity for the solvents investigated. The largest a=c-ratio of 15 was found for the LiClCsCl solvent whereas the additive ternary CsClKCl NaCl system delivered crystals with a more compact, short prismatic habit (Fig. 3b). The a=c-ratio was determined to be almost 2 in the latter case. Both binary solvents seem to favor the growth of large b- or c-oriented substrates. Typical growth rates of the ux-grown crystals are 1050 mm h1 for 1 0 0; and 510 mm h1 for
538
D. Ehrentraut, M. Pollnau / Journal of Crystal Growth 234 (2002) 533538
0 1 0 and 0 0 1: The higher values are for the crystals grown from LiCl-containing uxes. Needle-like crystals with high optical quality could be grown at high growth rates of 5001000 mm h1 for 1 0 0 by use of the 35 : 65 wt% LiClKCl solvent. The crystal size reached up to 2 cm in 1 0 0 and 0.3 cm in 0 0 1 and 0 1 0: The crystals grown under optimized growth conditions are clear, transparent, and colorless. The best optical quality was obtained for the crystals grown from the additive ternary CsCl KClNaCl system. A few cracks observed are possibly due to mechanical stresses during ux removal and handling. BaSO4 exhibits a cleavage from good for f0 1 0g to perfect for f0 0 1g [15].
! -Paul Salathe ! for his support. equipment and Rene D.E. thanks Christine Klemenz for fruitful discus. mer and Thomas sions. We also thank Karl Kra Brunold for experimental support and discussions. This work was partially supported by the Swiss National Science Foundation.
References
. [1] K. Bostrom, J. Frazer, J. Blankenburg, Arkiv Mineral. Geol. 4 (1968) 477. ! , J. Inorg. Nucl. [2] O. Vojtch, J. Moravec, F. Volf, M. Dlouha Chem. 32 (1970) 3725. [3] D.R. Lide (Ed.), CRC Handbook of Chemistry and Physics, 81st Edition, CRC Press, Boca Raton, FL, 2000. [4] S.V. Nechaev, Zap. Vses. Mineralogy. Obshchestva 92 (1963) 363. [5] P. Mohazzabi, A.W. Searcy, J. Chem. Soc. Farad. Trans. 72 (1976) 290. [6] H. Sawada, Y. Takeuchi, Z. Krist. 191 (1990) 161. [7] B.N. Roy, Crystal Growth from Melts. Applications to Growth of Groups 1 and 2 Crystals, Wiley, Chichester, 1992. [8] C. Klemenz, H.J. Scheel, Mater. Res. Forum 276277 (1998) 175. [9] T.C. Brunold, H.U. Gudel, . Inorg. Chem. 36 (9) (1997) 1946. [10] Dierent (Eds.), Phase Diagrams for Ceramists, The American Ceramic Society Inc., Academic Press, New York, 1966. [11] I.S. Grigoriev, E.Z. Meilikhov (Eds.), Handbook of Physical Quantities, CRC Press, Boca Raton, FL, 1997. [12] Y. Marcus, Introduction to Liquid State Chemistry, Wiley, London, 1977. [13] I. Barin, Thermodynamical Data of Pure Substances, Part II, 1989, and Vol. 3, 3rd Edition, VCH, Weinheim, 1995. [14] D. Elwell, H.J. Scheel, Crystal Growth from HighTemperature Solutions, Academic Press, London, 1975. [15] L.L.Y. Chang, R.A. Howie, J. Zussman, Rock-Forming Minerals, Non-silicates: Sulphates, Carbonates, Phosphates, Halides, Vol. 5B, 2nd Edition, Longman, Harlow, Essex, 1996. [16] J.J. Lagowski (Ed.), The Chemistry of Non-Aqueous Solvents, Vol. 1.
4. Conclusions The solubility of BaSO4 in pure chloridic solvents at start temperatures between 4501C and 6001C was investigated. Binary LiClKCl solvents are favored for the growth of large 1 0 0elongated crystals with high optical quality. We observed a high solubility of BaSO4 in LiClcontaining solvents. This eect was attributed to the decrease in average cation radius with increasing LiCl contents and the formation of Li2SO4 and BaCl2 with increasing temperature. The highest growth rate is along 1 0 0 for all solvent systems investigated. The use of CsClKClNaCl as solvent favors the growth of short prismatic crystals. Crystals with the best optical quality achieved, without cracks, and without inclusions were grown from the CsClKClNaCl ux. Furthermore, this system enables us to work at constant crystal-growth conditions because of its chemical stability to the solute BaSO4, a fact that is of high importance for the following LPE growth to achieve uniform layers of highest optical quality.
Acknowledgements The authors wish to thank Benoit Deveaud! dran for the outstanding help with laboratory Ple
Das könnte Ihnen auch gefallen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Configuration A: Unloaded BJT Transistor AmplifiersDokument3 SeitenConfiguration A: Unloaded BJT Transistor AmplifiersdasdNoch keine Bewertungen
- Research and Development at The BENSON Test RigDokument20 SeitenResearch and Development at The BENSON Test RigsbktceNoch keine Bewertungen
- Numbers (0, 1, 2, 3,... ) Are Defined To Be Natural Numbers, Including Zero, That Does Not ContainDokument9 SeitenNumbers (0, 1, 2, 3,... ) Are Defined To Be Natural Numbers, Including Zero, That Does Not ContainRomela EspedidoNoch keine Bewertungen
- Start Up and Commissioning of Chilled Water PumpsDokument6 SeitenStart Up and Commissioning of Chilled Water PumpsAlaa AnwerNoch keine Bewertungen
- HST TrainingDokument11 SeitenHST TrainingRamesh BabuNoch keine Bewertungen
- FP - ES - 28 - Rindu Grahabhakti Intani - PERMEABLE ENTRY CHARACTERIZATION AT DARAJAT FIELD, WEST JAVA PDFDokument4 SeitenFP - ES - 28 - Rindu Grahabhakti Intani - PERMEABLE ENTRY CHARACTERIZATION AT DARAJAT FIELD, WEST JAVA PDFrindu_intaniNoch keine Bewertungen
- Semi Conductors: The Start of Information AgeDokument15 SeitenSemi Conductors: The Start of Information AgeMarvin LabajoNoch keine Bewertungen
- PC - Section 1.3 - Worksheet PDFDokument2 SeitenPC - Section 1.3 - Worksheet PDFAnabbNoch keine Bewertungen
- Ball Charge ManagementDokument14 SeitenBall Charge ManagementSalud Y SucesosNoch keine Bewertungen
- 00.concrete Mix Design-RailwayDokument38 Seiten00.concrete Mix Design-RailwaySoundar PachiappanNoch keine Bewertungen
- WhiteLED1 8Dokument12 SeitenWhiteLED1 8Smyle KatariaNoch keine Bewertungen
- EC SyllDokument4 SeitenEC SyllDDIYNoch keine Bewertungen
- Form in MusicDokument8 SeitenForm in MusicAndri KurniawanNoch keine Bewertungen
- Iec 60243-1-2013Dokument58 SeitenIec 60243-1-2013FernandoCrespoMon100% (1)
- Arithmetic Unit: Dr. Sowmya BJDokument146 SeitenArithmetic Unit: Dr. Sowmya BJtinni09112003Noch keine Bewertungen
- Linux NFS and Automounter Administration (Craig Hunt Linux Library)Dokument0 SeitenLinux NFS and Automounter Administration (Craig Hunt Linux Library)Bryan BowmanNoch keine Bewertungen
- Multiple Choice Questions (The Answers Are Provided After The Last Question.)Dokument6 SeitenMultiple Choice Questions (The Answers Are Provided After The Last Question.)VijendraNoch keine Bewertungen
- Yang Learning Face Age CVPR 2018 PaperDokument9 SeitenYang Learning Face Age CVPR 2018 Paperandr234ibateraNoch keine Bewertungen
- App NandDokument30 SeitenApp NandRajesh MedampudiNoch keine Bewertungen
- Basic Use of Datum Plane in Creo ParametricDokument8 SeitenBasic Use of Datum Plane in Creo ParametricKartik BhararaNoch keine Bewertungen
- Physics Gcse Coursework Resistance of A WireDokument8 SeitenPhysics Gcse Coursework Resistance of A Wiref5dq3ch5100% (2)
- Italy CasestudyDokument21 SeitenItaly CasestudylozaNoch keine Bewertungen
- Rexx Programmers ReferenceDokument723 SeitenRexx Programmers ReferenceAnonymous ET7GttT7Noch keine Bewertungen
- Data Cable Containment SizingDokument21 SeitenData Cable Containment SizingAngelo Franklin100% (1)
- Beam Design: BackgroundDokument2 SeitenBeam Design: BackgroundolomizanaNoch keine Bewertungen
- W.R. Klemm (Auth.) - Atoms of Mind - The - Ghost in The Machine - Materializes-Springer Netherlands (2011)Dokument371 SeitenW.R. Klemm (Auth.) - Atoms of Mind - The - Ghost in The Machine - Materializes-Springer Netherlands (2011)El equipo de Genesis ProjectNoch keine Bewertungen
- Rekonfigurasi Jaringan Distribusi Radial Untuk Mengurangi Rugi Daya Pada Penyulang Jatirejo Rayon Mojoagung Menggunakan Metode BinaryDokument4 SeitenRekonfigurasi Jaringan Distribusi Radial Untuk Mengurangi Rugi Daya Pada Penyulang Jatirejo Rayon Mojoagung Menggunakan Metode BinaryNurindra CahyantaNoch keine Bewertungen
- Form 1 ExercisesDokument160 SeitenForm 1 Exerciseskays MNoch keine Bewertungen
- The Power of Human Connection Review 2Dokument81 SeitenThe Power of Human Connection Review 2Tajam SoftNoch keine Bewertungen
- Chem Cheat Sheet MasterDokument6 SeitenChem Cheat Sheet MasteradamhamelehNoch keine Bewertungen