Beruflich Dokumente
Kultur Dokumente
The Chemical Composition and Nature of A Cell
Hochgeladen von
LencesiOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
The Chemical Composition and Nature of A Cell
Hochgeladen von
LencesiCopyright:
Verfügbare Formate
THE CHEMICAL COMPOSITION/NATURE OF A CELL:
BIOLOGICAL MACROMOLECULE Nucleic Acids GENERAL STRUCTURE Nucleic acids are made of a sugar, a phosphate group and a nitrogenous base. The sugar determines whether it is DNA or RNA (which includes tRNA: transfer RNA, mRNA: messenger RNA and rRNA: Ribosomal RNA). DNA has deoxyribose while RNA has ribose. The phosphate group remains generally constant. The nitrogenous base determines which nucleic acid it is. The nucleobases are: adenine (A), thymine (T), cytosine (C) and guanine (G). Below is the structure of DNA, the genetic information in your body. FUNCTION tRNA: Serves as a carrier molecule for amino acids to be used in protein synthesis mRNA: Carries genetic sequence information between DNA and ribosomes, directing protein synthesis rRNA: Major component of the ribosome, catalyses peptide bond formation DNA: Contains genetic instructions used for the development and functioning of all known living organisms
Proteins
The structure of a protein is described in four different levels of organisation. The first level is the primary structure; this is just the order of amino acids in the molecule. The secondary structure is the local 3D folding which is formed by hydrogen bonds. There are three main types of secondary structure, theres the alpha helix, the beta plated sheets and a random coil. One long polypeptide chain could have some alpha helix, some beta plated sheets and some random coil along the one chain. The tertiary structure is the total folding of the protein that is held together by hydrogen bonds or ionic bonds. Finally there is the quaternary structure. This is a structure containing two or more polypeptide chain. Below is a nice diagram of all the protein structures.
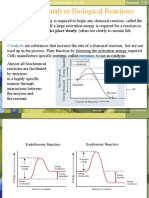
There are thousands of different proteins in every single cell of your body and they all have a special function. Here are some: Antibodies: Defends the body from antigens (foreign invaders) Contractile Proteins: Responsible for movement Enzymes: Act as catalysts to speed up chemical reactions without being used up in the reaction itself Hormones: Messenger proteins that help coordinate certain bodily activities Structural Proteins: Fibrous and stringy, they help give support Storage proteins: Store amino acids Transport proteins: Carrier proteins which move molecules from one place to another around the body
Carbohydrates
Carbohydrates are organic compounds made up of carbon, hydrogen and oxygen. Carbohydrates containing one sugar unit are called monosaccharides (like glucose, the energy source for plants and animals). Two sugar units are called disaccharides (like sucrose, thats table sugar). These two carbohydrates are called simple carbohydrates. Many sugar units joined together are called polysaccharides (like starch, the stuff in potatoes, or cellulose, the component in plant cell walls), also known as, complex carbohydrates. The picture here shows the structure of lactose, a disaccharide sugar found most notably in milk.
Carbohydrates form a major part of your diet and help in building body strength, by generating energy.
Lipids
Lipids are the general term for fats oils and waxes. They consist of carbon, hydrogen and oxygen but contain relatively little water. This lack of water allows them to carry much more energy per molecule than any other compound found in either plants or animals. Fats have little or no attraction to water and are insoluble. This makes them hydrophobic molecules. A phospholipid has two fatty acids attached to a glycerol molecule
A lipid primary purpose is for longterm energy storage in comparison to carbohydrates, which are for short-term usage. A very small amount of energy, which is excellent if you dont happen to have a ready supply around. In adipose tissue, lipids can also serve as cushioning.
WATER
GENERAL STRUCTURE
FUNCTION/PROPERTIES
3
A water molecule has two hydrogen atoms joined onto one oxygen atom by bonds called covalent bonds. So the hydrogen atoms share electrons with the larger oxygen atom. Overall, the water molecule has a neutral charge. However, when looked at closely, the oxygen end of the molecule has a slight negative charge and the hydrogen ends are slightly positive, like a polarized magnet. Thus, water molecules are attracted to other water molecules. This attraction is called hydrogen bonding, it is not very strong but it is the reason why water stays together.
Water is a very common compound on Earth but also one with very uncommon properties. It exists naturally as a liquid, solid and gas. It is a good solvent due to its polarity and is essential to all living creatures. Water is also called dihydrogen monoxide (two hydrogens, one oxygen) that is a very cool long name and makes you sound like a genius just by saying it. It is white solid or almost colourless and transparent, with a slight hint of blue. It boils at 100 C (actually, Wikipedia says 99.98C) and freezes at 0C. Pure water will also have a neutral pH of 7.
PROVIDE DEFINITIONS FOR KEY WORDS: ORGANIC: Organic describes forms, methods and patterns found in living systems such as the organisation of cells, to populations, communities, and ecosystems INCORGANIC: Part of or derived from non-biological material MONOMERS: A single unit from which a polymer is built POLYMERS: Naturally occurring or synthetic compound consisting of large molecules made up of a linked series of repeated simply monomers HYDROPHILIC: Having a strong affinity for water, tending to dissolve in, mix with, or be wetted by water HYDROPHOBIC: Lacking affinity for water, tending to repel and not absorb water, not mixing, or be wetted by water. It could also describe a person who has an abnormal fear of water.
MATERIAL TRANSPORT:
4
TRANSPORT MODE
DEFINITION OR FUNCTION
PASSIVE OR ACTIVE
ENERGY SOURCE
Osmosis
The movement of water molecules through a selectively permeable membrane down a water potential gradient. Basically, water moving from an area of high water potential to an area of low, crossing a membrane as it moves.
Passive
Osmosis releases energy, and can be made to do work. The energy is from the cells own kinesis.
Diffusion
This is the spread of particles through random motion of higher concentration to regions of lower concentration.
Passive
From the cells own energy.
Active Transport
The movement of a substance against its concentration gradient (so, from low to high concentration). Specialized proteins recognize the substance and allow it to access, or exit, the cell via the membrane.
Active
This process uses chemical energy, such as from adenosie triphosphate (ATP).
Carrier Proteins
These are proteins involved in the movement of ions, small molecules, or macromolecules, such as another protein, across a biological membrane. The proteins can assist in the movement of substance by facilitated diffusion or active transport.
Active/Passive
Uses the kinetic energy of cells or, if helping with active transport, it uses chemical energy. Cells own kinetic energy. Protein channels are just holes basically, they only allow some molecules that can fit in. The cells own energy. It s only used when the substance need cannot pass through the hydrophobic plasma or the cell membrane. The cells own energy, used when the substance cannot pass
6
Protein Channels
A watery pathway through the interstices of a protein molecule by which ions and small molecules can cross a membrane into or out of the cell by diffusion or active transport.
Active/Passive
Endocytosis
This is the process in which cells absorb molecules (such as proteins) by engulfing them. The amoeba eats like that.
Active
Exocytosis
This is the process in which cells rids itself of unwanted molecules by directing the contents out of the cell membrane. I guess the
Active
amoeba vomits like that.
through the hydrophobic plasma or the cell membrane. Active Cells cellular energy
Phagocytosis
The cellular process of engulfing solid particles by the cell membrane. Imagine small arms reaching out from the cell membrane and engulfing something.
Pinocytosis
A form of exocytosis in which small particles are bought into the cell suspended within small vesicles that subsequently fuse with lysosomes to hydrolyse, or to break down, the particles. It is like pinching the substance through the membrane.
Active
The cells cellular energy.
Facilitated Diffusion
Also known as facilitated transport or passive-mediated transport. Polar molecules and charged ions are dissolved in water but they cannot diffuse freely across the membrane due to the hydrophobic nature of fatty acid tails of the phospholipid bilayer. So the carrier proteins have to open and allow them access into the cell.
Passive
The cells own kinetic energy and the permeability of its membrane
HYPERTONIC SOLUTIONS: A solution with a higher salt concentration than in normal cells of the body and the blood
HYPOTONIC SOLUTIONS: A solution with a lower salt concentration than in normal cells of the body and the blood
ISOTONIC SOLUTIONS: A solution with the same salt concentration than in normal cells of the body and the blood
Das könnte Ihnen auch gefallen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (73)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- 3:5 BioDokument4 Seiten3:5 BioMaya FinkleNoch keine Bewertungen
- CHON Lecture PDFDokument25 SeitenCHON Lecture PDF2d HoehoeNoch keine Bewertungen
- Instalasi Laboratorium: Klinik Cahaya HusadaDokument9 SeitenInstalasi Laboratorium: Klinik Cahaya HusadaEnjuff OscarNoch keine Bewertungen
- Prestigi0us BiochemistryDokument30 SeitenPrestigi0us Biochemistrybovey69015Noch keine Bewertungen
- Amino Acids and Proteins ReviewerDokument11 SeitenAmino Acids and Proteins ReviewerJohn-Karl JimenezNoch keine Bewertungen
- Essay On ProteinDokument2 SeitenEssay On ProteinLester MendigoriaNoch keine Bewertungen
- BIO156 Enzyme Lab-Effect of Temperature On Enzyme Activity-F21Dokument8 SeitenBIO156 Enzyme Lab-Effect of Temperature On Enzyme Activity-F21Shiloh FrederickNoch keine Bewertungen
- CHAPTER 19 Tricarboxylic Acid CycleDokument11 SeitenCHAPTER 19 Tricarboxylic Acid Cycle楊畯凱Noch keine Bewertungen
- Histidine Operon LeaderDokument2 SeitenHistidine Operon LeaderVijayant Kumar PandeyNoch keine Bewertungen
- GFP TrapDokument4 SeitenGFP TrapAllele BiotechnologyNoch keine Bewertungen
- მიტოქონდრიის ფუნქცია და კიბოDokument1 Seiteმიტოქონდრიის ფუნქცია და კიბოEMD GROUPNoch keine Bewertungen
- Chapter 21 Lipid MetabolismDokument45 SeitenChapter 21 Lipid MetabolismWaad MajidNoch keine Bewertungen
- Molecular Basis of Inheritance Practice TestDokument13 SeitenMolecular Basis of Inheritance Practice TestPartha Shee100% (1)
- Unit 1: Structure Determination: Protein Structure Database PDB PDB File Format Ramachandran PlotDokument33 SeitenUnit 1: Structure Determination: Protein Structure Database PDB PDB File Format Ramachandran PlotSaran.S.MenonNoch keine Bewertungen
- Elements Found in Living Things2Dokument6 SeitenElements Found in Living Things2Maevrick HaleyNoch keine Bewertungen
- Homework Marking Scheme 16.1: Inherited ChangeDokument2 SeitenHomework Marking Scheme 16.1: Inherited ChangeMikeNoch keine Bewertungen
- Research QuestionDokument2 SeitenResearch QuestionSiegNoch keine Bewertungen
- Protein Synthesis Worksheet PART A. Read The FollowingDokument5 SeitenProtein Synthesis Worksheet PART A. Read The Followingapi-39708077950% (2)
- Enzymes WorksheetDokument5 SeitenEnzymes WorksheetgyunimNoch keine Bewertungen
- Flashcards - Topic 5 Enzymes - CAIE Biology IGCSEDokument25 SeitenFlashcards - Topic 5 Enzymes - CAIE Biology IGCSEJane LeaNoch keine Bewertungen
- Bio MapDokument2 SeitenBio Mapdqnxfdw6bjNoch keine Bewertungen
- Protein Science - 2021 - Rai - Liquid Liquid Phase Separation of Tau From Molecular Biophysics To Physiology and DiseaseDokument21 SeitenProtein Science - 2021 - Rai - Liquid Liquid Phase Separation of Tau From Molecular Biophysics To Physiology and DiseaseKeshav JhalaniNoch keine Bewertungen
- 1.3b Enzymes EditedDokument10 Seiten1.3b Enzymes EditedSoleil R.Noch keine Bewertungen
- Molecules of Life PDFDokument113 SeitenMolecules of Life PDFArpit Pradhan100% (1)
- Asam Amino Dan ProteinDokument13 SeitenAsam Amino Dan ProteinSitti hanafiahNoch keine Bewertungen
- BANING Activity No. 5.2 DNA Base PairingDokument3 SeitenBANING Activity No. 5.2 DNA Base PairingFatima Nur Faiza HandaNoch keine Bewertungen
- Learning Activity 2.3: Proteins: BiomoleculesDokument5 SeitenLearning Activity 2.3: Proteins: BiomoleculesaikeeNoch keine Bewertungen
- Membran Sel, Matriks (DR Karismananda)Dokument84 SeitenMembran Sel, Matriks (DR Karismananda)An iNoch keine Bewertungen
- Short Course On Basic Biology IDokument47 SeitenShort Course On Basic Biology ICed HernandezNoch keine Bewertungen
- 4TH Q Physical ScienceDokument14 Seiten4TH Q Physical Sciencemarvin agubanNoch keine Bewertungen