Beruflich Dokumente
Kultur Dokumente
Sintesis de Urea Con Aspen
Hochgeladen von
Olfer ClarosCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Sintesis de Urea Con Aspen
Hochgeladen von
Olfer ClarosCopyright:
Verfügbare Formate
Aspen Plus Urea Synthesis Loop Model
Contents
1Introduction...............................................................................1
2Components and Units of Measurement.......................................1
3Process Description....................................................................2
4Physical Properties.....................................................................3
5Chemical Reactions inetics........................................................5
!"imulation #pproach...................................................................$
$"imulation Results......................................................................$
%Conclusions................................................................................&
References.................................................................................1'
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
i
1 Introduction
This document describes the steady-state Aspen Plus
model of the high-pressure synthesis loop of a
urea plant, with a capacity of about 1,100 metric tons of prilled urea per year.
This simulation is based on the Stamicarbon !" Stripping Process, which is a popular and fast growing
process for manufacturing urea.
The wor# demonstrates the capability of Aspen Plus to rigorously model the urea synthesis process. The
modeling is complicated due to the formation of ammonium carbamate, an intermediate product for
which a special property pac#age has to be de$eloped. This type of model is useful to analy%e the plant
performance and to impro$e plant operation, including&
'nergy sa$ing studies to impro$e economics of the plant.
Studies of indi$idual pieces of e(uipment with a $iew of increasing their throughput and)or
impro$ing their performance.
*lowsheet modification for better plant operation.
+dentifying bottlenec#s.
As a basis for optimi%ation study and for de$eloping on-line control system of the plant.
,hile this document describes the simulation of the Stamicarbon !" stripping process, the accurate
results obtained support the applicability of Aspen Plus and the data pac#age to other urea processes.
2 Components and Units of Measurement
The table below lists the components modeled in the simulation.
Components
Component Name Component ID Type ormula
,ater -"! !./ -"!
Ammonia .-0 !./ -0.
arbon 1io2ide !" !./ !"
3rea 34'A !./ -5."!
Ammonium arbamate A46 !./ -7."!"
.itrogen ." !./ ."
!2ygen !" !./ !"
A small amount of biuret 8"-9.0!": is produced during the synthesis. +n this wor#, the biuret production
is not considered, but the component can easily be added if re(uired.
The pure component properties of all the components e2cept ammonium carbamate can be retrie$ed
from the Aspen Plus databan#s. Special efforts were made to incorporate pure component properties of
ammonium carbamate in the simulation.
A special contribution of this wor# is the de$elopment of a physical property model to describe the
simultaneous physical-chemical e(uilibrium occurring in the urea-synthesis process.
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
1
;etric units are used in this wor#, e2cept that temperature unit is
o
, pressure unit is #g)cm
"
8abs:,
enthalpy flow unit is mmcal)hr, and mole flow unit is #mol)day.
! Process Description
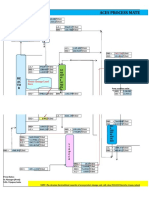
A simulation flowsheet of the synthesis loop is shown in *igure 1.
S05
S0<
S1"
S10
S19
S01
'0"
401
FLA!"
*01
'01
#T$I%
A0"
EP
SP1
#&I''
A01
'00
(I)E#
;01
%$"
Feed
%$"
tripper
*!+
Feed
#eactor
%arbamate
%ondenser
crubber
,ent
&as
Urea
olution
i"ure 1# Simulation lo$sheet of Urea Synthesis Loop
The high-pressure loop is operated at around 151 #g)cm
"
8abs: and consists of the following #ey pieces
of e(uipment&
%&uipment Purpose
401 3rea 4eactor, where ammonium carbamate is dehydrated to urea
'01 -.P. !" Stripper, where the bul# of the unreacted carbamate from the reactor
effluent is decomposed by stripping with !" gas and with heat input.
'0" -.P. ondenser, where the gaseous !" and .-0 condense and react to form
ammonium carbamate.
'00 -.P Scrubber, where the recycled carbamate solution from the downstream low-
pressure section is used to absorb unreacted gases from the reactor.
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
"
S05
S0< S1" S1 0 S19S01'0" 401FLA!"*01'01 #T$I%A0"EPSP1#&I' ' A0 1 '00(I)E#;01
%$"
Feed
%$"
tripper
*!+
Feed
#eactor %arbamate
%ondenser
crubber
,ent
&as
Urea
olution
The process is described as follows&
*eed !" gas 8S0<: is fed to the !" stripper, '01, to strip the urea solution coming from the
reactor. +n the stripper, ammonium carbamate decomposes, liberating more .-0 and !" to be
stripped out. -eat is supplied on the shell side of tubes by condensing "=9 psig steam while the urea
solution falls inside the tubes countercurrently down past the rising !" stripping gas. The outlet
li(uid solution from the stripper is rich in urea and goes to the downstream section for urea
purification.
+n the adiabatic urea reactor, 401, an a(ueous solution of .-0 and !" 8much in the form of
ammonium carbamate: and $apors flow upward through = stages of reactor $olume to minimi%e
bac#-mi2ing and pro$ide enough residence time for urea formation. 4emaining gases condense and
carbamate decomposes in the reactor to pro$ide heat for the slightly endothermic reaction of
carbamate to urea. The urea solution 8S07: o$erflows from the top of the reactor bac# to the
Stripper '01, while the unreacted gases 8S09: pass out the top of the reactor.
These unreacted gases pass to the Scrubber, '00, where recycled carbamate solution from the
'$aporation)4ecirculation section 8S10: is passed o$er the top of a pac#ed bed and fills the tube
side section of $ertical tubes. The gases rise up through the tubes and pass up through the pac#ed
section before lea$ing the top of the $essel. The contact with the carbamate solution absorbs the
unreacted .-0 and !", while the inert gases of !", .", and others 8S19: $ent out from the top. !n
the bottom, recirculated cooling water flowing inside tubes remo$es the heat of absorption from the
carbamate solution. arbamate solution 8S1<: o$erflows out of the $essel.
The solution 8S1<: together with the top $apor stream from the Stripper 8S0=: is fed to the
arbamate ondenser, '0", through the use of an e>ector, where the ammonia feed 8S01: ser$es as
the pumping fluid. Ammonium carbamate forms in this condenser. The $apor-li(uid mi2ture 8S00:
falls through tubes and the heat of reaction is remo$ed by generation of 90 psig steam on the shell
side. The mi2ture goes to the bottom of the reactor for urea production.
The bottom stream 8S0?: from the Stripper '01 is sent to the downstream section to reco$er urea.
The recycled stream after reco$ering the urea 8S10: is bac# to the -.P. Scrubber to complete the
loop.
' Physical Properties
The model for the thermodynamic properties of the .-0-!"--"!-34'A-A46-."-!" system is based
upon the S4-P!@A4 model within Aspen Plus 8Soa$e, 1?<"A Penelou2 and 4au%y, 1?="A
Schwart%entruber and 4enon, 1?=?:. The model uses an e(uation of state and is thus suitable for the
high-pressure, high-temperature conditions of urea synthesis. *urther, the model contains e2tensions
that enable an accurate description of the phase and chemical e(uilibria, the density and the other
thermodynamic properties 8e.g., enthalpy: of this system.
,e chose our approach to the modeling of the thermodynamic properties after studying the pre$ious
modeling attempts in the literature and analy%ing the a$ailable data. *rB>ac(ues 81?5=:, Cawasumi
81?9", 1?90 and 1?95: and @em#owit% 81?=0: de$eloped chemical and thermodynamic models by
postulating reactions for urea formation and $arious simplified assumptions for the phase non-ideality.
The simplifying assumptions do not permit an accurate and general model for the chemical-
thermodynamic properties. 6ernadis et al. 81?=?: and +sla et al. 81??0: de$eloped impro$ed theoretical
models by including ionic species and describing the nonideality of the li(uid phase by a modified
3.+D3A model. ,e belie$e that under the high temperatures 8170 to "00: and the relati$ely low
water concentrations of urea synthesis, the e2tent of ioni%ation will be small. *urther, modern e(uations
of state such as the S4-P!@A4 model are well suited to the description of the thermodynamic properties
of nonideal systems at high pressures and temperatures. Thus we ha$e chosen to use the S4-P!@A4
model as the physical-property option.
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
+
1e$elopment of a data pac#age for this system is difficult since most of the data are only a$ailable as
combined physical and chemical e(uilibria. 1isassociation pressure data are a$ailable for ammonium
carbamate 8Eanac#e, 1?00:, which is the pressure at a specified temperature where ammonia and
carbon dio2ide are in e(uilibrium with the condensed-phase ammonium carbamate. 1ata are also
a$ailable for the e(uilibrium con$ersion of defined mi2tures of .-0-!"--"! to urea 8Cawasumi, 1?9",
1?90 and 1?95A +noue, 1?<":. *inally, bubble pressures ha$e been measured for defined mi2tures of
.-0-!"--"! at chemical e(uilibrium. The Aspen Plus data analysis capabilities 814S and 1ATA-*+T:
ha$e been used to obtain a simultaneous good fit of these $aried and comple2 sets of data.
Forlo$s#ii and Cucherya$yi 81?<?: ha$e analy%ed the urea con$ersion data reported by many authors
and ha$e produced an empirical correlation that is recogni%ed to be accurate. *igure " compares the
e(uilibrium con$ersions predicted by the present AspenTech model with the Forlo$s#ii-Cucherya$yi
correlation. The present correlation clearly pro$ides an accurate fit of the data.
i"ure 2# %&uili(rium Con)ersion of C*2 to Urea at 1+,C Comparison of
Aspen Plus- %*S to Correlation of .orlo)s/ii and 0ucherya)yi 1123,4
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
-
*igure 0 presents a comparison between the present AspenTech model and the bubble pressure data
measured by @em#owit% 81?<1, 1?<" and 1?<<:. The model pro$ides an accurate description of the data
and, in particular, pro$ides an accurate description of the minimum in bubble pressure, which is
necessary for an effecti$e description of the urea synthesis process.
;easured and alculated 6ubble Pressures in the .-0-!" System
omparison of ASP'. P@3S '!S to 1ata of @em#owit% et al. 81?<2:
150G
170G
1=0G
"00G
0
90
100
190
"00
"90
000
0.79 0.<0 0.<9 0.=0 0.=9 0.?0 0.?9
;ole *raction .-0 in *eed
6
u
b
b
l
e
P
r
e
s
s
u
r
e
8
b
a
r
:
i"ure !# Measured and Calculated 5u((le Pressures in the N6!C*2 System
Comparison of Aspen Plus- %*S to Data of Lem/o$it7 et al8 112+94
The AspenTech model pro$ides an accurate description of the phase and chemical e(uilibria related to
urea synthesis. +t also accurately describes the other properties needed for reliable simulations, namely
enthalpies and densities.
: Chemical ;eactions 0inetics
There are two main reactions that ta#e place in the urea synthesis process&
81: ".-0 H !" A46
8": A46 34'A H -"!
The first reaction, which ta#es place in the li(uid phase, con$erts ammonia and carbon dio2ide into
ammonium carbamate. This reaction is highly e2othermic and fast. hemical e(uilibrium is readily
reached under the operating conditions in the reactor. The second reaction also ta#es place in the li(uid
phase and is endothermic. +ts rate is slow and e(uilibrium is usually not reached in the reactor.
A user subroutine, 3S34A.*, was de$eloped to include the reaction #inetics of both reactions.
3S34A.* is used in the reactor simulations. 6oth forward and re$erse reactions were considered. The
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
.
#inetics of 4eaction 1 has been set to be rapid so that e(uilibrium is effecti$ely reached. @imited
literature data were used for the #inetics of 4eaction ".
The reaction #inetics has been formulated to approach the e(uilibrium composition for large residence
times. The e(uilibrium has been described in terms of the fugacity coefficients since an e(uation of
state is used as the thermodynamic model. The e(uilibrium constant for 4eaction 1, in terms of mole
fractions, is written as follows&
( )
1
]
1
,
_
'
CARB
CO NH CO NH CARB
P
P
RT
G G G
K
"
"
0
"
0
0
"
0
0
0
1
"
e2p
,here,
T - Temperature
P - Pressure
2 - ;ole fraction $ector
4 - Fas constant
P
0
- 4eference pressure 8I 1 atmosphere:
0
i
G - +deal-gas Fibbs *ree energy of component i at T, P
0
i - *ugacity coefficient of component i at T, P, 2
The e(uilibrium constant for 4eaction 1 in terms of mole fractions is as follows&
"
"
0
1
CO NH
CARB
x x
x
K
Similar e(uilibrium e(uations can be written for 4eaction ".
The rates for 4eactions 1 and ", in units of #mol)s)m
0
, are as follows&
'
1
"
"
+ 1 1
K
x
x x k Rate
CARB
CO NH
'
"
"
" "
K
x x
x k Rate
O H UREA
CARB
The two rate e2pressions ha$e been formulated so that they will necessarily reach e(uilibrium at large
residence times. The rate constant for 4eaction 1 8C1: is set to a large $alue so that this reaction is
essentially at e(uilibrium. The rate constant for 4eaction " determines the urea con$ersion in the
reactor. !nly scant information is a$ailable to determine C" and it is usually best to ad>ust its $alue to fit
plant data. A reasonable appro2imation for C" is the following&
L
RT
v e k ) 10 J . 19
7
10 J . 100
=
"
,
_
,here 4I=015.0 and /
@
is the molar $olume of the li(uid.
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
/
< Simulation Approach
The !" Stripper, '01, is of the falling film type, which was appro2imated by a 4A1*4A 8multistage
distillation: bloc# with 10 stages. -eat is supplied to the stages "-? to simulate the heat transfer from the
tubes. The urea solution, falling down on tube walls, is stripped off $olatile .-0 by the entering !" gas
8S0<:. !n each stage the model considers the e(uilibrium of carbamate in the li(uid as well as the /@'
of the mi2ture. .ote that the #inetics of carbamate formation is large enough to ensure that chemical
e(uilibrium for the carbamate reaction is reached in each stage of the 4A1*4A bloc#.
The 3rea 4eactor 401, <.9" ft in diameter and ?9 ft in length, is modeled with an 4P@3F bloc#. The
#inetics is pro$ided by the user subroutine 3S34A in the 4P@3F bloc#.
+n the reactor, the e2othermic carbamate reaction and the endothermic urea formation reaction are
ta#ing place. The reactor is designed such that its $olume is big enough for the desired urea production.
The -.P. Scrubber is modeled using a 9-stage 4A1*4A bloc#. -eat is ta#en out from the bottom
stage. Similar to the Stripper, '01, on each stage the model considers the e(uilibrium of carbamate in
the li(uid as well as the /@' of the mi2ture.
The '0" -.P. !" ondenser is modeled with an 4ST!+ bloc#. A 1esign Specification paragraph is
included to monitor the specified reactor outlet temperature of 1=0
o
by ad>usting the e2tent of the
carbamate reaction in '0".
This simulation is based on a closed-loop flowsheet. The downstream section is appro2imated by using
a S'P bloc# to lin# the Stripper bottom urea solution 8S0?: to the recycled carbamate solution 8S10:.
+ Simulation ;esults
The Aspen Plus run was made using /ersion "07.9. Some of the results are shown below, and a
simulation flowsheet with stream data is shown in *igure 5. The simulation results of this generic model
are reasonable compared with similar plant operations.
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
0
i"ure '# Simulation lo$sheet of Urea Synthesis Loop
Cey Process Simulation results&
%&uipment =aria(le =alue Unit
;,1 > ;eactor
-eat duty 0 mm#cal)hr
Top temperature 1=0.0 8spec.:
o
3rea in e2it stream 5",<9" #g)hr
%,1 > C*2 Stripper
-eat duty 17.0 8spec.: mm#cal)hr
Top temperature 1=9.0
o
6ottom temperature 179.7
o
Top stream 8S0=:, =0,"0? #g)hr
6ottom stream 8S0?:, <7,<71 #g)hr
3rea production 5",707 #g)hr
%,2 ? C*2 Condenser
-eat duty -1<.?" mm#cal)hr
'2it temperature 17< 8spec.:
o
%,! ? Scru((er
-eat duty -0." 8spec.: mm#cal)hr
Top temperature =7.1
o
6ottom temperature 171.<
o
Top stream 8S19: 1707.1 #g)hr
6ottom stream 8S1<: 00,?=7 #g)hr
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
1
3 Conclusions
1. This urea process model has been de$eloped using Aspen Plus /ersion "007.9. This is a rigorous
closed-loop model for the plant while the reco$ery section is appro2imated by using a S'P model.
The carbon dio2ide compression section is not included. *rom the results, it is shown that the S4-
P!@A4 property pac#age used for simulation is appropriate.
". *or further refinement of the model, the following upgrades should be made&
a: The cooling water circuit for '00 and the low pressure steam circuit for '0" should be
implemented. This implementation is useful for energy sa$ing studies.
b: '0" is simulated in this wor# by a 4ST!+ model. -owe$er, a 4P@3F model is more suitable
for the simulation of this e(uipment. -owe$er, to do so, detailed e(uipment data for '0" are
needed.
c: The stripper '01 is a falling-film type e(uipment. +t embodies /@' e(uilibrium, mass transfer,
reaction, and heat transfer. To rigorously simulate this e(uipment, rate-based calculations
84ateSep: should be used. A special subroutine will be needed to incorporate the mass transfer
limitations of the falling-film. +n this simulation, the 4A1*4A model was used with component
efficiencies for .-0, !" and -"! as a wor# around.
0. The accurate results obtained in the present simulation indicate that Aspen Plus and the physical-
property data pac#age will pro$ide accurate simulations of other urea processes.
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
2
;eferences
K1L 6ernadis, ;.A ar$oli, F.A Santini, ;., M3rea-.-0-!"--"! /@' alculations 3sing an '2tended
3.+D3A '(uation,N *luid Phase '(uilibria, 90, "0<-"1= 81?=?:.
K"L *rB>ac(ues, ;., MTheoretical 6asis of the +ndustrial Synthesis of 3rea,N him. +nd., 70, ""-09 81?5=:.
K0L Forlo$s#ii, 1.;.A Cucherya$yi, /.+., M'(uation for 1etermination of the '(uilibrium 1egree of !"
on$ersion 1uring Synthesis of 3rea,N Oh. Pri#l. Chim., 90, "95=-"991 81?<?:.
K5L +noue, S.A Canai, C.A !tsu#a, '., M'(uilibrium of 3rea Synthesis. +.N 6ull. hem. Soc. Eapan, 59,
1000?-1059 81?<":.
K9L +sla, ;.A.A +ra%o(ui, -.A.A Fenoud, .;., MSimulation of a 3rea Synthesis 4eactor. 1.
Thermodynamic *ramewor#,N +P' 4esearch, 0", "77"-"7<0 81??0:.
K7L Eanec#e, '., O. 'lectrochem., 07, 759 81?00:.
K<L Cawasumi, S., M'(uilibrium of the !"-.-0-3rea--"! System 3nder -igh Temperature,N 6ull.
hem. Soc. Eapan, "9, ""<-"0= 81?9":.
K=L Cawasumi, S., M'(uilibrium of the !"-.-0-3rea--"! System 3nder -igh Temperature and
Pressure. ++. @i(uid-/apor '(uilibrium in the @oading ;ole 4atio of ".-0 to !",N 6ull. hem. Soc.
Eapan, "7, "1=-""< 81?90:.
K?L Cawasumi, S., M'(uilibrium of the !"-.-0-3rea--"! System 3nder -igh Temperature and
Pressure. +/. @i(uid-/apor '(uilibrium in the Presence of '2cess Ammonia or arbon 1io2ide,N
6ull. hem. Soc. Eapan, "<, "95-"9? 81?95:.
K10L Cotula, '.A., M/apor-@i(uid '(uilibrium of the .-0-!"--"!-3rea System at 'le$ated Pressure,N
E. hem. Technol. 6iotechnol., 01, 100-110 81?=1:
K11L @em#owit%, S.;.A Foedegebuur, E.A $an den 6erg, P.E., M6ubble-Point ;easurements in the
Ammonia-arbon 1io2ide System,N E. Appl. hem. 6iotechnol., "1, ""?-"0" 81?<1:.
K1"L @em#owit%, S.;.A $an 'rp E..A 4e#ers, 1.;.A $an den 6erg, P.E., MPhae '(uilibria in the
Ammonia-arbon 1io2ide Systems at and Abo$e 3rea Synthesis onditions,N E. Appl. hem.
6iotechnol., 00, =9-101 81?=0:.
K10L @em#owit%, S.;.A /et, '.A $an den 6erg, P.E., MA Phase ;odel for the Fas-@i(uid '(uilibria in the
Ammonia-arbon 1io2ide ,ater-3rea System in hemical '(uilibrium at 3rea Synthesis
onditions. ++. '2perimental /erification,N E. Appl. hem. 6iotechnol., "<, 009-05= 81?<<:.
K15L @em#owit%, S.;.A /et, '.A $an den 6erg, P.E., MPhase 6eha$ior in the Ammonia-arbon 1io2ide
System at and Abo$e 3rea Synthesis onditions,N E. Appl. hem. 6iotechnol., "", <"<-<0< 81?<":.
K19L Penelou2, A.A 4au%y, '., MA onsistent orrection for 4edlich-Cwong-Soa$e /olumes,N *luid
Phase '(uilibria, =, <-"0 81?=":.
K17L Schwart%entruber, E.A 4enon, -., M'2tension of 3.+*A to -igh Pressures and Temperatures by
the 3se of a ubic '(uation of State,N +P' 4esearch, "=, 105?-1099 81?=?:.
K1<L Soa$e, F., M'(uilibrium onstants from a ;odified 4edlich-Cwong '(uation of State,N hem. 'ng.
Sci., "<, 11?7-1"00 81?<":.
Aspen Plus Example Library
Proprietary Information of AspenTech
Unauthorized duplication or distribution strictly prohibited without prior written permission
13
Das könnte Ihnen auch gefallen
- Urea ModelingDokument20 SeitenUrea ModelingekmagisNoch keine Bewertungen
- Urea Toyo ACES21 - BrochureDokument8 SeitenUrea Toyo ACES21 - BrochureSanti SevenfoldNoch keine Bewertungen
- Urea - Aspen TutrialDokument19 SeitenUrea - Aspen TutrialAmanda BittencourtNoch keine Bewertungen
- AmmoniaDokument24 SeitenAmmonianghiemta18100% (2)
- Simulation and ParametricDokument12 SeitenSimulation and ParametricAmiruddin KubikNoch keine Bewertungen
- Urea PDFDokument11 SeitenUrea PDFSteve WanNoch keine Bewertungen
- Ammonia D PDFDokument10 SeitenAmmonia D PDFrkm_rkmNoch keine Bewertungen
- REACTOR DESIGN FOR AMMONIA OXIDATIONDokument5 SeitenREACTOR DESIGN FOR AMMONIA OXIDATIONabdul rehmanNoch keine Bewertungen
- Simulation of a Urea Synthesis Reactor Thermodynamic FrameworkDokument9 SeitenSimulation of a Urea Synthesis Reactor Thermodynamic FrameworktreyzzztylerNoch keine Bewertungen
- Simulation of A Urea Synthesis Reactor. 1. ThermodynamicDokument10 SeitenSimulation of A Urea Synthesis Reactor. 1. ThermodynamicYaraKanawatiNoch keine Bewertungen
- Improved ACES Urea Technology Case Studies China IndonesiaDokument17 SeitenImproved ACES Urea Technology Case Studies China IndonesiaZeeshan Khan100% (1)
- Aces Process Material Balance: RE AC TO RDokument4 SeitenAces Process Material Balance: RE AC TO Rwaheed ahmadNoch keine Bewertungen
- 2015 01 Baboo NFL Ammonia and Urea Plant Energy Consumption CalculationsDokument4 Seiten2015 01 Baboo NFL Ammonia and Urea Plant Energy Consumption CalculationsjunaidNoch keine Bewertungen
- Designing Urea ReactorDokument20 SeitenDesigning Urea ReactordcobasbNoch keine Bewertungen
- P Urea Smcarb 2018 Ok Ok OkDokument143 SeitenP Urea Smcarb 2018 Ok Ok Okهشام حدودNoch keine Bewertungen
- Stamicarbon Urea Process Data PDFDokument1 SeiteStamicarbon Urea Process Data PDFtreyzzztylerNoch keine Bewertungen
- Case Study 109 Ammonia OxidationDokument9 SeitenCase Study 109 Ammonia OxidationkaalkankNoch keine Bewertungen
- Reactor Modeling and Simulations in Synthesis Gas ProductionDokument39 SeitenReactor Modeling and Simulations in Synthesis Gas ProductionSteven Sandoval100% (1)
- Rate Based MDEA ModelDokument24 SeitenRate Based MDEA ModelDebrudra MitraNoch keine Bewertungen
- Manufacturing DAP using the cross processDokument9 SeitenManufacturing DAP using the cross processNabeel SiddiqueNoch keine Bewertungen
- Ammonia Synthesis Loops Variables Investigated by Steady-State SimulationDokument14 SeitenAmmonia Synthesis Loops Variables Investigated by Steady-State Simulationpolonium2310Noch keine Bewertungen
- UREA PROCESSDokument8 SeitenUREA PROCESSIshan HaiderNoch keine Bewertungen
- Urea - Kirk Othmer PDFDokument15 SeitenUrea - Kirk Othmer PDFusman_hafeez86Noch keine Bewertungen
- Major Ammonia Leak From HP Ammonia Feed Pump: P.Hari Narayana Reddy, R. Raghavan and Ramashray SinghDokument10 SeitenMajor Ammonia Leak From HP Ammonia Feed Pump: P.Hari Narayana Reddy, R. Raghavan and Ramashray Singhvaratharajan g rNoch keine Bewertungen
- Commissioning of The First Ammonia-Urea Complex: A Challenging Venture For The Vietnamese EconomyDokument13 SeitenCommissioning of The First Ammonia-Urea Complex: A Challenging Venture For The Vietnamese Economyvaratharajan g rNoch keine Bewertungen
- Draft Report For Urea ProductionDokument59 SeitenDraft Report For Urea ProductionBryan Jesher Dela CruzNoch keine Bewertungen
- Urea Plant Material Balance (ACES Process)Dokument7 SeitenUrea Plant Material Balance (ACES Process)muks19950% (2)
- Equilibria in The Ammonium Carbamate Urea-Water System: K. G. Clark, L. C. DDokument5 SeitenEquilibria in The Ammonium Carbamate Urea-Water System: K. G. Clark, L. C. DjcortezcNoch keine Bewertungen
- 2008 Morikawa TEC IFA ACES21 Advanced Urea Production Technology - 2Dokument15 Seiten2008 Morikawa TEC IFA ACES21 Advanced Urea Production Technology - 2MubasharNoch keine Bewertungen
- Reaction Kinetics-Reactor DesignDokument17 SeitenReaction Kinetics-Reactor Designzorro21072107Noch keine Bewertungen
- Process Simulation of Ammonia PlantDokument9 SeitenProcess Simulation of Ammonia Planthamidrezaee008Noch keine Bewertungen
- Chemical Ammonia Report PDFDokument72 SeitenChemical Ammonia Report PDFAli J. Hojeij100% (1)
- Methanol Reactor ModelDokument11 SeitenMethanol Reactor ModeljoeNoch keine Bewertungen
- KRIBHCO Urea Plant OverviewDokument25 SeitenKRIBHCO Urea Plant OverviewHarsh DholakiyaNoch keine Bewertungen
- Nirbhay Urea Final PDFDokument99 SeitenNirbhay Urea Final PDFHimanshu vikram100% (1)
- Ammonia and Urea Production PDFDokument10 SeitenAmmonia and Urea Production PDFئارام ناصح محمد حسێنNoch keine Bewertungen
- (MEA) CO2 Capture PFDsDokument95 Seiten(MEA) CO2 Capture PFDsfNoch keine Bewertungen
- Ammonia AspenDokument24 SeitenAmmonia AspenMohammadAlAmeenNoch keine Bewertungen
- 1255 Chemical Process Simulation Using Ms ExcelDokument11 Seiten1255 Chemical Process Simulation Using Ms ExcelLex ERafael100% (1)
- New SynCOR Ammonia™ ProcessDokument12 SeitenNew SynCOR Ammonia™ Processrukam18Noch keine Bewertungen
- CDB 4313 Heat Integration - Basic Heat IntegrationDokument25 SeitenCDB 4313 Heat Integration - Basic Heat Integrationharvin raoNoch keine Bewertungen
- K Factor or Watson FactorDokument6 SeitenK Factor or Watson FactorZakyAlFatonyNoch keine Bewertungen
- Chapter 3 Separation of Oil and Gas - 1987 - Developments in Petroleum ScienceDokument50 SeitenChapter 3 Separation of Oil and Gas - 1987 - Developments in Petroleum ScienceFalokid RaboNoch keine Bewertungen
- 05 PDFDokument491 Seiten05 PDFNatasha Mgt JoharNoch keine Bewertungen
- Bubble Column ReactorDokument21 SeitenBubble Column ReactorMuhammad Hamzah SyahrirNoch keine Bewertungen
- Urea Production Process OverviewDokument38 SeitenUrea Production Process OverviewVishal SinghNoch keine Bewertungen
- Meyer Coal 2014Dokument23 SeitenMeyer Coal 2014Molote Erwin MalieheNoch keine Bewertungen
- Final ReportDokument46 SeitenFinal ReportVarun Gupta100% (1)
- Basic Design of Methanol Production Process 2019Dokument112 SeitenBasic Design of Methanol Production Process 2019Santosh Bhandari100% (1)
- Adiabatic Fixed-Bed Reactors: Practical Guides in Chemical EngineeringVon EverandAdiabatic Fixed-Bed Reactors: Practical Guides in Chemical EngineeringNoch keine Bewertungen
- Vapor-Liquid Equilibria Using Unifac: A Group-Contribution MethodVon EverandVapor-Liquid Equilibria Using Unifac: A Group-Contribution MethodNoch keine Bewertungen
- Principles and Case Studies of Simultaneous DesignVon EverandPrinciples and Case Studies of Simultaneous DesignNoch keine Bewertungen
- Dynamic Programming in Chemical Engineering and Process Control by Sanford M RobertsVon EverandDynamic Programming in Chemical Engineering and Process Control by Sanford M RobertsNoch keine Bewertungen
- Chemical Process Design and Simulation: Aspen Plus and Aspen Hysys ApplicationsVon EverandChemical Process Design and Simulation: Aspen Plus and Aspen Hysys ApplicationsBewertung: 2 von 5 Sternen2/5 (1)
- Distillation Design and Control Using Aspen SimulationVon EverandDistillation Design and Control Using Aspen SimulationBewertung: 5 von 5 Sternen5/5 (2)
- Biogas PlantDokument37 SeitenBiogas PlantMahaManthra100% (1)
- Configure Dynamic NAT and PATDokument20 SeitenConfigure Dynamic NAT and PATMary Amirtha Sagayee. GNoch keine Bewertungen
- Contacts From MotorolaDokument26 SeitenContacts From MotorolamvayangcoNoch keine Bewertungen
- Dernord Dn40 220v380v 3kw 4.5kw 6kw 9kw Sus304 He... - VicedealDokument1 SeiteDernord Dn40 220v380v 3kw 4.5kw 6kw 9kw Sus304 He... - Vicedealadrianchelaru21Noch keine Bewertungen
- Summarized ResumeDokument2 SeitenSummarized Resumeapi-310320755Noch keine Bewertungen
- Wind Load Calculations For PV Arrays: Solar America Board For Codes and StandardsDokument24 SeitenWind Load Calculations For PV Arrays: Solar America Board For Codes and StandardsSalman KhanNoch keine Bewertungen
- Catalog de Aparatura Si Instrumentar Veterinar Eikemeyer-GermaniaDokument336 SeitenCatalog de Aparatura Si Instrumentar Veterinar Eikemeyer-GermaniaDr. Dragos CobzariuNoch keine Bewertungen
- 2012 Product CatalogDokument407 Seiten2012 Product CatalogSe SamnangNoch keine Bewertungen
- An Instrument and It's Repertoire No 4Dokument6 SeitenAn Instrument and It's Repertoire No 4Michael BellemoreNoch keine Bewertungen
- TAM Acquisition and Analysis ManualDokument92 SeitenTAM Acquisition and Analysis ManualJulian UribeNoch keine Bewertungen
- BS WaterDokument0 SeitenBS WaterAfrica OdaraNoch keine Bewertungen
- SST CrossguideDokument39 SeitenSST Crossguideardacho1968Noch keine Bewertungen
- Web Based Attendance Management SystemDokument19 SeitenWeb Based Attendance Management SystemAnonymous zenL7LNoch keine Bewertungen
- E - 9039 - Sika - Per - 3326 EG-HDokument4 SeitenE - 9039 - Sika - Per - 3326 EG-HBogdan DavidescuNoch keine Bewertungen
- Effisus Vapour FR System Technical Data SheetDokument4 SeitenEffisus Vapour FR System Technical Data SheetPaula MoçoNoch keine Bewertungen
- Wheel Loader Links, Bellcranks, Pins & BushingsDokument7 SeitenWheel Loader Links, Bellcranks, Pins & BushingsMax SashikhinNoch keine Bewertungen
- Mitsubishi Electric Heat Pump Parts Outdoor MUZ-GE25-80VA (2) - A1, A2 - OBB532E PDFDokument10 SeitenMitsubishi Electric Heat Pump Parts Outdoor MUZ-GE25-80VA (2) - A1, A2 - OBB532E PDFTony SavageNoch keine Bewertungen
- Tools For Pipe Welding: W E L D T E C HDokument24 SeitenTools For Pipe Welding: W E L D T E C HChano HanokNoch keine Bewertungen
- Centrifugal Vs Reciprocating Compressor - Turbomachinery Magazine PDFDokument2 SeitenCentrifugal Vs Reciprocating Compressor - Turbomachinery Magazine PDFReyes SanchezNoch keine Bewertungen
- GAC-12-VX-RMH-LAH-008 - Attachments - 1 To 7Dokument14 SeitenGAC-12-VX-RMH-LAH-008 - Attachments - 1 To 7Dipayan DasNoch keine Bewertungen
- 1.overview Construction N DevelopmentDokument27 Seiten1.overview Construction N Development_ain_Noch keine Bewertungen
- CCR Process GuideDokument55 SeitenCCR Process GuideJean Claude EidNoch keine Bewertungen
- Performance of Fiber ReinforcedDokument16 SeitenPerformance of Fiber ReinforcedBang BangNoch keine Bewertungen
- ANSI E1.50-1 2017 Entertainment Technology - Requirements For The Structural Support of Temporary LED, Video & Display SystemsDokument19 SeitenANSI E1.50-1 2017 Entertainment Technology - Requirements For The Structural Support of Temporary LED, Video & Display SystemsGabriel neagaNoch keine Bewertungen
- Electronic Patient Record Use During Ward Rounds: A Qualitative Study of Interaction Between Medical StaffDokument8 SeitenElectronic Patient Record Use During Ward Rounds: A Qualitative Study of Interaction Between Medical StaffpsikubNoch keine Bewertungen
- VA CarparkManagementSysDokument22 SeitenVA CarparkManagementSysmerkermanNoch keine Bewertungen
- Design and Detailing of RCC BeamsDokument4 SeitenDesign and Detailing of RCC BeamsPhilip JohnNoch keine Bewertungen
- Engineering Tilt UpDokument334 SeitenEngineering Tilt UpMTCLSNoch keine Bewertungen
- Terrible Old Games You Have Probably Never Heard ofDokument274 SeitenTerrible Old Games You Have Probably Never Heard ofNagyKristóf100% (2)