Beruflich Dokumente
Kultur Dokumente
Ros PDF
Hochgeladen von
Aditya Dana Iswara0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
69 Ansichten7 SeitenLiver cell death induced by stresses such as ischemia-reperfusion, cholestasis and drug toxicity can trigger a sterile inflammatory response. Central to this inflammatory response is promotion of reactive oxygen species (ROS) ROS are the principal toxic mediators by which inflammatory cells kill their targets, e.g. Bacteria during host defense but also hepatocytes and other liver cells.
Originalbeschreibung:
Originaltitel
ROS (2).pdf
Copyright
© © All Rights Reserved
Verfügbare Formate
PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenLiver cell death induced by stresses such as ischemia-reperfusion, cholestasis and drug toxicity can trigger a sterile inflammatory response. Central to this inflammatory response is promotion of reactive oxygen species (ROS) ROS are the principal toxic mediators by which inflammatory cells kill their targets, e.g. Bacteria during host defense but also hepatocytes and other liver cells.
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
69 Ansichten7 SeitenRos PDF
Hochgeladen von
Aditya Dana IswaraLiver cell death induced by stresses such as ischemia-reperfusion, cholestasis and drug toxicity can trigger a sterile inflammatory response. Central to this inflammatory response is promotion of reactive oxygen species (ROS) ROS are the principal toxic mediators by which inflammatory cells kill their targets, e.g. Bacteria during host defense but also hepatocytes and other liver cells.
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 7
REVI EW
Reactive oxygen and mechanisms of inammatory liver
injury: Present concepts_6592 173..179
Hartmut Jaeschke
Department of Pharmacology, Toxicology and Therapeutics, University of Kansas Medical Center, Kansas City, USA
Abstract
Liver cell death induced by stresses such as ischemia-reperfusion, cholestasis and drug
toxicity can trigger a sterile inammatory response with activation of innate immune cells
through release of damage-associated molecular patterns (DAMPs). A similar inamma-
tory response can be induced by pathogen-associated molecular patterns (PAMPs) such as
endotoxin. Both DAMPs and PAMPs activate through toll-like receptors the resident
macrophages (Kupffer cells) and recruit activated neutrophils and monocytes into the liver.
Central to this inammatory response is promotion of reactive oxygen species (ROS)
formation by these phagocytes. ROS are the principal toxic mediators by which inam-
matory cells kill their targets, e.g. bacteria during host defense but also hepatocytes and
other liver cells. The mechanism of ROS-induced cell killing during inammation involves
the promotion of mitochondrial dysfunction through an intracellular oxidant stress in
hepatocytes leading mainly to oncotic necrosis and less apoptosis. The additional release of
cell contents amplies the inammatory injury. However, an inammatory oxidant stress
insufcient to directly cause cell damage can induce transcription of stress defence genes
including antioxidant genes. This preconditioning effect of ROS enhances the resistance
against future inammatory oxidant stress and promotes the initiation of tissue repair
processes. Despite the substantial progress in our understanding of mechanisms of inam-
matory liver injury during the last decade, more research is necessary to better understand
the role of ROS in acute liver inammation and to develop clinically applicable therapeutic
strategies that selectively target the detrimental effects of oxidant stress without compro-
mising the vital function of ROS in host defense.
Key words
alarmins, apoptosis, cholestasis, cytokines,
high mobility group box -1, hypochlorite,
ischemia-reperfusion, Kupffer cells, lipid
peroxidation, neutrophils.
Correspondence
Dr. Hartmut Jaeschke, Department of
Pharmacology, Toxicology and Therapeutics,
University of Kansas Medical Center, 3901
Rainbow Blvd, MS 1018, Kansas City, KS
66160, USA. Email: hjaeschke@kumc.edu
Conict of interest
The author does not have any conict of
interest to disclose.
Introduction
It is now generally accepted that innate immune cells, i.e. Kupffer
cells and inltrating monocytes/macrophages and neutrophils, are
activated after acute liver cell death and that under certain circum-
stances these inammatory cells can aggravate the initial liver
injury.
1
Experimental systems where direct cytotoxicity of neutro-
phils has been shown include hepatic ischemia-reperfusion
injury,
2,3
endotoxemia,
4
obstructive cholestasis,
5
alcoholic hepati-
tis,
6
halothane hepatitis,
7
and hemorrhagic shock.
8
Cytotoxicity of
Kupffer cells or inltrating macrophages has been implicated in
ischemia-reperfusion injury,
9,10
endotoxemia,
11
galactosamine
hepatotoxicity,
12
and corynebacterium parvum/endotoxin-induced
liver injury.
13
A common denominator of all inammatory models
is the involvement of various reactive oxygen species (ROS) in the
pathophysiology. ROS can have multiple effects in inammation
including direct cytotoxicity, a sensitizing or preconditioning
effect to a second insult, or involvement in the generation of
pro-inammatory mediators. However, understanding the sources
of ROS formation during liver injury processes is critical for
interpretation of experimental results and designing relevant thera-
peutic intervention strategies. Too often is evidence for general
ROS formation provided without evaluation of the cellular or
intracellular sources, the nature of the ROS involved and the iden-
tication of initiating events. This can lead to misinterpretation of
mechanisms and ultimately jeopardizes the translation of the nd-
ings to the human pathophysiology. This may be one of the main
reasons why despite that ROS appear to be important in every
experimental model of inammatory liver disease, no drug that
targets ROS is available for human therapy. This review will
provide an updated overview of the potential sources and the
impact of ROS in various inammatory liver injury models.
Alarmins, oxidant stress and initiation
of inammation
Although the detrimental role of neutrophils and Kupffer cells
is well established in various liver injury models and the self-
aggravating nature of the inammatory response to cell death
has been described,
14
the molecular basis for the initiation and
doi:10.1111/j.1440-1746.2010.06592.x
173 Journal of Gastroenterology and Hepatology 26 (2011) Suppl. 1; 173179
2011 Journal of Gastroenterology and Hepatology Foundation and Blackwell Publishing Asia Pty Ltd
propagation of inammation after tissue trauma was only more
recently recognized. In cases of tissue injury without involvement
of external pathogens, e.g. hepatic ischemia-reperfusion, the initial
ischemic insult causes limited necrosis within minutes of reperfu-
sion.
9,15
Necrotic cell death triggers release of cell contents with
some of the endogenous compounds (alarmins) being able to acti-
vate innate immune cells.
16
The nuclear protein high mobility
group box protein 1 (HMGB1) was one of the rst alarmins
identied that was related to endogenous tissue trauma
16
and
was therefore termed damage-associated molecular patterns
(DAMPs).
17
However, it is now obvious that a large number of
additional molecules can also function as DAMPs including heat
shock proteins, DNA, RNA and others.
17
Complexes between
HMGB1 and other DAMPs are particularly potent inamma-
gens.
18
The activation of innate immune cells by DAMPs occurs
through toll-like receptors, which recognize various molecular
patterns.
19
Thus, HMGB1 translocates from the nucleus to the
cytosol during ischemia and is subsequently released during rep-
erfusion.
20
HMGB1 is a critical mediator of inammation during
reperfusion by activation of the TLR4 receptor.
20
The mechanism
of HMGB1-mediated inammation include the direct activation of
neutrophils by activation of NADPH oxidase (NOX-2), the main
superoxide producing enzyme of phagocytes
21
and cytokine for-
mation through TLR4
22
signaling and in combination with DNA
fragments through TLR9 activation.
23
In addition to cytokine for-
mation, cell contents release also activates complement.
24
Both
cytokines and activated complement fragments (C5a) are potent
activators for Kupffer cells and neutrophils to produce ROS
and cause upregulation of cell adhesion molecules (CD11b/CD18)
on neutrophils and trigger their recruitment into the liver
vasculature.
2426
Because of the self-aggravating nature of this
sterile inammatory response (Fig. 1), any intervention at the level
of preventing the initial injury, neutralizing some of the DAMPs
released, or direct inactivation of innate immune cells can attenu-
ate the postischemic oxidant stress and liver injury.
In the case of sepsis or endotoxemia, lipopolysaccharide (LPS)
and other pathogen-associated molecular patterns (PAMPs) are
present. LPS is a potent trigger for cytokine formation particularly
in macrophages/Kupffer cells through the TLR4 receptor
(Fig. 1).
19
LPS can also prime Kupffer cells and neutrophils for
enhanced ROS formation in response to additional stimuli.
27
However, LPS does not directly trigger ROS generation by
Kupffer cells.
27
Instead, higher doses of LPS are sufcient to
activate complement induced Kupffer cell-mediated oxidant stress
through stimulation of the C5a receptor.
11
The oxidant stress by
Kupffer cells and the hepatic recruitment of primed neutrophils
during endotoxemia is initiated without tissue trauma and does not
lead to cell death.
11,27,28
One of the reasons is that enhanced release
of hepatocellular glutathione (GSH) into the vasculature during
inammation can detoxify oxidant stress derived from Kupffer
cells.
11
In addition, the neutrophils are only primed and require
additional stimulation during extravasation and adherence to target
cells to actually generate ROS.
27
The signal to transmigrate has to
come from the parenchyma in the form of a chemoattractant or
direct cell contact.
1
In the case of the galactosamine/LPS model,
the signal is apoptotic cell death.
29
Only the extravasated, full
activated adherent neutrophils generate ROS.
30,31
Another example of sterile inammation is obstructive cholesta-
sis.
5,32
It is generally believed that the intrahepatic accumulation of
bile acids after bile duct ligation is responsible for causing cell death
mainly through apoptosis.
33
However, recent data suggest that high
levels of the most common bile acids do neither cause apoptosis nor
necrosis in cultured mouse hepatocytes
34
In contrast, taurocholate,
deoxycholate and chenodeoxycholate directly induce the formation
of a large number of pro-inammatory cytokines and chemokines in
hepatocytes mostly dependent on the transcription factor Egr-1
(Fig. 1).
34
The formation of these mediators is responsible for the
recruitment of neutrophils and the substantial liver injury (bile
infarcts) caused by these inammatory cells.
5,32
The neutrophil-
induced oxidant stress and tissue injury requires extravasation.
32
Oxidant stress during inammatory liver injury
Extracellular versus intracellular oxidant stress
Sterile inammation is initiated by limited tissue trauma, which is
amplied through activation of innate immune cells (Fig. 1). In
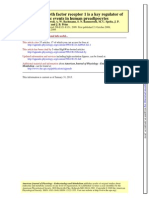
Figure 1 Mechanisms of an acute inammatory response after cellu-
lar stresses such as hepatic ischemia, drug toxicity, cholestasis and
endotoxemia (see text for details). Abbreviations: CXC, CXC chemok-
ines (IL-8); C5aR, complement fragment 5a receptor; CT adducts, chlo-
rotyrosine protein adducts; DAMPs, damage-associated molecular
patterns; DNA Frg, DNA fragments; HMGB1, high mobility group box 1
protein; HNE, hydroxynonenal (lipid peroxidation product); HSPs. Heat
shock proteins; HOCl, hypochlorous acid; ICAM-1, intercellular adhesion
molecule-1; IL-1, interleukin-1; LPS, lipopolysaccharide/endotoxin;
NF-kB, nuclear factor kB; Nox-2, NADPH oxidase; PAMPs, pathogen-
associated molecular patterns; ROS, reactive oxygen species; TLR, toll
like receptor; TNF-a, tumor necrosis factor-a.
Reactive oxygen and liver inammation H Jaeschke
174 Journal of Gastroenterology and Hepatology 26 (2011) Suppl. 1; 173179
2011 Journal of Gastroenterology and Hepatology Foundation and Blackwell Publishing Asia Pty Ltd
ischemia-reperfusion injury, this would include some cell death
during ischemia and during the initial reperfusion phase. Some
time ago, the assumption was that xanthine oxidase could be a
relevant intracellular source of ROS formation. However, there
was no direct evidence for an intracellular oxidant stress by xan-
thine oxidase during the early reperfusion phase.
35
In contrast,
most of the oxidant stress appears to occur in the vasculature with
Kupffer cells as the main source.
9,36
This was also conrmed by the
protective effect of vascular glutathione.
37,38
Neutrophils recruited
at this early phase are mainly primed for ROS formation but do not
actually contribute to the oxidant stress and reperfusion injury
until several hours later.
39,40
However, the long-lasting neutrophil-
induced ROS formation does not occur in the vasculature but
requires generally extravasation and adherence to the target.
41
Critical for adhesion and ROS formation is the b
2
integrin (CD11b/
CD18, Mac-1) on neutrophils.
42
Consequently, neutrophils gener-
ate ROS in close proximity to hepatocytes and thus, some ROS are
able to diffuse into hepatocytes. In support of this conclusion,
enhanced levels of intracellular glutathione disulde (GSSG) are
found during the neutrophil-mediated injury phase in the
galactosamine/LPS model
30,43
and during ischemia-reperfusion.
43
In addition, chlorotyrosine protein adducts are detected in hepato-
cytes during neutrophil cytotoxicity in both models
31,40
and after
bile duct ligation.
5,32
Based on these measurements, it can be
concluded that neutrophils generate hypochlorite, which can
diffuse into targets cells and form chlorotyrosine protein adducts.
In addition, neutrophil-derived hydrogen peroxide may also
diffuse across cell membranes or hypochlorite, causes mitochon-
drial dysfunction and mitochondrial ROS formation. In either
case, ROS generated by neutrophils get into the target, generate an
intracellular oxidant stress, which contributes to their rapid and
efcient cell killing.
ROS versus proteases during inammatory liver injury
Whereas there is little doubt that ROS are generated during all
inammatory liver disease conditions, there is not always consen-
sus on the pathophysiological relevance. The controversy origi-
nated from co-culture experiments showing that neutrophils kill
hepatocytes through release of proteases, not ROS.
44
However,
these experiments did not reect realistic inammatory conditions.
In particular, only control rat hepatocytes were exposed to
phorbol-ester stimulated human neutrophils. This resulted in cell
injury by slow proteolytic digestion in about 16 h.
44
In contrast,
neutrophils in vivo do not attack healthy hepatocytes.
1
Neutrophils
only attack stressed or injured cells exposed to inammatory
mediators generating chemokines, and cause the expression of
intercellular adhesion molecule-1 (ICAM-1), which is the main
receptor for b
2
integrins (CD11/CD18) on neutrophils.
1
Extrava-
sation in response to a distress signal and killing of the target
occurs within 1 h.
30
During a neutrophil attack, there is evidence
for an intracellular oxidant stress in the target hepatocytes.
30
The
enhanced formation of glutathione disulde suggest enhanced
levels of hydrogen peroxide
30
and the presence of chlorotyrosine
protein adducts
45
indicate diffusion of neutrophil-derived
hypochlorite into target cells.
5,31,32,40
Most importantly, deciency
of glutathione peroxidase, a defence mechanism against oxidant
stress, enhanced inammatory injury.
30
In addition, inhibition of
Nox-2, the superoxide generating enzyme in phagocytes, was
protective in models where neutrophils have been shown to play
a role.
31,46
Furthermore, strengthening intracellular antioxidant
defence mechanisms by ischemic or chemical preconditioning
inhibited Kupffer cell and neutrophil-induced liver injury.
47
Such
data are consistent with previous ndings that blocking CD11b or
the common b-chain (CD18) on neutrophils prevents ROS forma-
tion by these inammatory cells and prevents injury.
3,10
Thus,
extensive experimental evidence supports the conclusion that neu-
trophils kill target cells by ROS generation, not by protease
release. However, proteolytic enzymes of phagocytes are impor-
tant for digesting cell debris or foreign objects after phagocytosis;
they are also involved in the activation of pro-inammatory cytok-
ines and proteolytic cleavage of extracellular matrix proteins to
allow extravasation and facilitate movement of neutrophils into the
extracellular space.
48
This explains why various inhibitors against
proteases such as elastase
49
and others
1,41
can protect against an
inammatory liver injury. This, however, does not support a
protease-mediated cell injury mechanism.
Degranulation of neutrophils in the vasculature results in the
systemic release of proteolytic enzymes, which have the potential
to cause tissue injury. Anti-proteases in plasma can limit the nega-
tive impact in the vasculature.
50
However, the anti-proteases also
have the potential to interfere with the host-defence mission of
neutrophils. Interestingly, anti-proteases are more susceptible to
oxidative inactivation than their protease counterpart.
50
Thus, ROS
and especially hypochlorite protect neutrophil-derived proteases
from inactivation in the immediate vicinity of the neutrophil and
therefore enable extravasation, movement in the extracellular
space and the formation of chemoattractants.
50
This represents an
indirect mechanism by which ROS generated by inammatory
cells can promote tissue injury.
Mechanisms of ROS-mediated cell death
Lipid peroxidation
The most popular mechanism of cell death induced by extracellu-
lar reactive oxygen species is lipid peroxidation (LPO). This free
radical-mediated process leads to destruction of cell membranes
and as such can kill hepatocytes very rapidly. However, LPO is a
multi-step process that involves formation of various lipid radicals
from polyunsaturated fatty acids and the participation of metal
ions, mainly iron, as redox-catalysts. Because of the danger of this
process in an oxygen environment, cells developed very effective
defence mechanisms against ROS and LPO including enzymes
that detoxify ROS, chain-breaking antioxidants in cell membranes
(vitamin E) and iron chelators (ferritin and transferrin).
51
There-
fore, under realistic in vivo or in vitro conditions, LPO is not a very
effective cell death mechanism for hepatocytes. However, when
experimental conditions are used that do not necessarily reect
true pathophysiology, LPO can become more prominent both in
vivo
52
and in vitro.
53,54
If conditions are selected to induce cell
death by LPO in vivo, liver injury is accompanied by a 30- to
50-fold increase in all LPO parameters, e.g. malondialdhyde,
ethane and pentane exhalation, formation of hydroxyfatty acids,
hydroxynonenal, etc., and a profound protection by the lipid-
soluble antioxidant, vitamin E.
52,55,56
In contrast, the overwhelming
majority of in vivo experiments show at best a 2- to 3-fold increase
of LPO parameters, which implicates oxidant stress, but does not
H Jaeschke Reactive oxygen and liver inammation
175 Journal of Gastroenterology and Hepatology 26 (2011) Suppl. 1; 173179
2011 Journal of Gastroenterology and Hepatology Foundation and Blackwell Publishing Asia Pty Ltd
denitively prove that LPO is the actual injury mechanism.
55
In
addition, therapeutic intervention strategies that are assumed to act
as antioxidants may protect independently of LPO and reduce
LPO parameters as a secondary effect. Thus, effective endogenous
defense mechanisms limit the impact and relevance of LPO as an
injury mechanism in the liver.
Apoptosis
Superoxide formation induced by menadione or paraquat can
trigger apoptotic cell death in cultured hepatocytes.
57
This was
demonstrated in cell lines
58
and in primary rat hepatocytes.
59
The
oxidant stress can activate c-jun-N-terminal kinase (JNK) and
caspases to trigger apoptotic cell death.
58,59
Activation of protein
kinase C and extracellular signal-regulated kinase 1/2 (ERK1/2)
attenuate oxidant stress-induced cell death by down-regulating
JNK.
57
Interestingly, acute exposure of primary rat hepatocytes to
hydrogen peroxide or t-butyl hydroperoxide causes necrosis.
59,60
The reason for the discrepancy between the effect of superoxide
which triggers apoptosis (and also dismutates to hydrogen perox-
ide and oxygen) and the direct addition of hydrogen peroxide
might lie with the dose. Whereas superoxide is generated by
redox-cycling in a moderate amount over extended time periods,
hydrogen peroxide is generally added as a bolus dose.
59
Based on
these experiments, it is concluded that superoxide and oxidant
stress in general trigger apoptosis.
57,59
However, when a selective
oxidant stress was generated in rat hepatocytes in vivo using the
redox-cycling agent diquat, no apoptotic cell death was observed.
61
In contrast, the oxidant stress was effectively detoxied and only a
severe oxidant stress appeared to cause liver cell injury through
oncotic necrosis.
61,62
Although there was activation of JNK by
severe oxidant stress in vivo, JNK inhibitors did not protect.
61
In
addition, lower oxidant stress still caused moderate necrosis
without JNK activation.
61
Interestingly, menadione induces necro-
sis, not apoptosis in cultured mouse hepatocytes (Yan and Jae-
schke, unpublished observation). Thus, in contrast to studies with
cultured rat hepatocytes, intracellular superoxide generation does
not cause apoptosis in rat liver in vivo or cultured mouse hepato-
cytes; it is rst detoxied and only when the defense systems are
overwhelmed, the oxidant stress triggers necrosis.
61
Consistent
with this observation is the fact that oxidant stress-dependent
inammatory liver injury during hepatic ischemia-reperfusion
63
and obstructive cholestasis
64
involves almost exclusively oncotic
necrotic cell death.
Oncotic necrosis
Various ROS induce oncotic necrosis through mechanisms that
involve the mitochondrial permeability transition (MPT) pore
opening, breakdown of the membrane potential and ATP deple-
tion.
60
However, the extracellular ROS do not directly trigger the
MPT but act through oxidation of NAD(P)H, uptake of Ca
2+
and
mitochondrial ROS formation.
60
The MPT can also be induced by
Ca
2+
activated mitochondrial calpains during extracellular
oxidant stress.
65
In addition, translocation of lysosomal iron to
mitochondria can synergistically aggravate the mitochondrial dys-
function and mitochondrial oxidant stress induced by extracellular
ROS.
66
Nutrient-deprivation leads to enhanced ferritin protein
turnover with accumulation of iron in lysosomes.
67
Under these
conditions, hepatocytes are more susceptible to oxidant stress-
induced cell death due to lysosomal instability and iron release.
67
In addition to the release of lysosomal enzymes, the mitochondrial
iron translocation may be a critical event for the toxicity.
66
These
data are in agreement with in vivo ndings where diquat-induced
ROS formation triggers necrotic cell death,
61,62
which can be
enhanced by iron supplementation and reduced by iron chelation.
62
Although the original interpretation focused on the modulation of
the Fenton reaction and LPO,
62
the recent ndings of mitochon-
drial uptake of lysosomal iron, which is involved in the mitochon-
drial oxidant stress and MPT, under various oxidant stress
conditions may provide an alternative explanation.
66
As discussed, hepatocytes are not only exposed to hydrogen
peroxide during an inammatory response but also to more aggres-
sive oxidants such as hypochlorite and its derivatives including
chloramines.
68
In contrast to peroxides, hypochlorite oxidizes sulf-
hydryl groups, methionine and tryptophan residues, and triggers
formation of protein carbonyls and chlorotyrosine protein
adducts.
45,69
These events are closely followed by ATP depletion
and cell lysis.
69
Evidence for hypochlorite formation can be found
in all in vivo models where neutrophil cytotoxicity contributes to
liver injury including ischemia-reperfusion injury,
40
obstructive
cholestasis
5,32
and endotoxemia.
31
Preconditioning by subtoxic oxidant stress
Limited ROS formation, which may be insufcient to cause cell
death, can lead to a preconditioning effect with higher resistance to
subsequent oxidant stress.
47,70
Lower oxidant stress can activate the
Keap1-Nrf2-ARE (Kelch ECH associating protein 1nuclear
factor erythroid 2-related factor 2antioxidant response ele-
ments) signaling pathway, which regulates a large number of
defence genes against acute and chronic cell injury in response to
various stressors including oxidant stress.
71
Keap1 is associated
with Nrf2, which keeps Nrf2 in the cytosol to undergo proteolysis.
However, several cysteine residues in Keap1 serve as stress sensor.
Oxidation/conjugation of these cysteine residues leads to struc-
tural modications of Keap1, which causes the dissociation of the
Keap1-Nrf2 complex and the translocation of Nrf2 to the
nucleus.
71
Nrf2 and other associated proteins activate gene tran-
scription through binding to ARE. Nrf2-dependent genes that
enhance the antioxidant defence capacity include heme
oxygenase-1 (HO-1), superoxide dismutase-1, glutathione peroxi-
dase, glutathione reductase, peroxiredoxin, thioredoxin reductase
and both the catalytic subunit (Gclc) and the modier subunit
(Gclm) of glutamate cysteine ligase, the rate-limiting enzyme in
the glutathione biosynthesis.
71
Thus, any subtoxic oxidant stress
can lead to the induction of defence systems that increases the
resistance to subsequent more severe inammatory oxidant
stress.
47,70
One of the most sensitive oxidant stress-inducible genes
is HO-1, which has been shown to be protective in all warm and
cold hepatic ischemia models.
72
Inammation, regeneration and oxidant stress
Although innate immune cell activation after cell death can
severely aggravate the initial tissue injury, a sterile inammatory
response does not necessarily cause additional liver damage. In
fact, the main purpose of activating resident macrophages and
Reactive oxygen and liver inammation H Jaeschke
176 Journal of Gastroenterology and Hepatology 26 (2011) Suppl. 1; 173179
2011 Journal of Gastroenterology and Hepatology Foundation and Blackwell Publishing Asia Pty Ltd
recruiting monocytes and neutrophils into the liver after tissue
injury is to remove dead or dying cells. Removal of this cell
debris is essential for regeneration of lost tissue. A typical
example is acetaminophen hepatotoxicity, which triggers a pro-
found inammatory response with neutrophils appearing within
an hour after early liver injury with the recruitment of monocytes
within 24 h.
73,74
However, there is no reliable evidence that
this inammatory response contributes to tissue injury after
acetaminophen overdose.
26,73,74
In contrast, impairment of mac-
rophage recruitment substantially delays tissue repair after
acetaminophen-induced liver injury.
74
Interestingly, this repair is
not affected in mice with decient NADPH oxidase (Nox-2)
activity.
75
This suggests that in contrast to cell killing, removal of
cell debris by macrophages or neutrophils does not require ROS
formation. This may have important implications for therapeutic
intervention strategies because other mediators of sterile inam-
mation, such as cytokines or complement factors can have mul-
tiple roles in promoting tissue injury, enhancing defence
mechanisms and inducing tissue regeneration.
76,77
Thus, there is a
clear need for more studies to elucidate the mechanisms of tissue
injury and repair during an acute inammatory response and the
role of ROS in these processes.
Acknowledgments
Research in the authors laboratory is supported in part by
National Institutes of Health Grants R01 DK070195 and R01
AA12916 and by grants P20 RR016475 and P20 RR021940 from
the National Center for Research Resources (NCRR), a compo-
nent of the National Institutes of Health.
References
1 Jaeschke H. Mechanisms of Liver Injury. II. Mechanisms of
neutrophil-induced liver cell injury during hepatic
ischemia-reperfusion and other acute inammatory conditions.
Am. J. Physiol. Gastrointest. Liver Physiol. 2006; 290:
G1083G1088.
2 Jaeschke H, Farhood A, Smith CW. Neutrophils contribute to
ischemia/reperfusion injury in rat liver in vivo. FASEB J. 1990; 4:
33559.
3 Jaeschke H, Farhood A, Bautista AP, Spolarics Z, Spitzer JJ,
Smith CW. Functional inactivation of neutrophils with a Mac-1
(CD11b/CD18) monoclonal antibody protects against
ischemia-reperfusion injury in rat liver. Hepatology. 1993; 17:
91523.
4 Jaeschke H, Farhood A, Smith CW. Neutrophil-induced liver cell
injury in endotoxin shock is a CD11b/CD18-dependent mechanism.
Am. J. Physiol. 1991; 261: G1051G1056.
5 Gujral JS, Farhood A, Bajt ML, Jaeschke H. Neutrophils aggravate
acute liver injury during obstructive cholestasis in bile duct-ligated
mice. Hepatology. 2003; 38: 35563.
6 Bautista AP. Neutrophilic inltration in alcoholic hepatitis. Alcohol.
2002; 27: 1721.
7 You Q, Cheng L, Reilly TP, Wegmann D, Ju C. Role of neutrophils
in a mouse model of halothane-induced liver injury. Hepatology.
2006; 44: 142131.
8 Vedder NB, Fouty BW, Winn RK, Harlan JM, Rice CL. Role of
neutrophils in generalized reperfusion injury associated with
resuscitation from shock. Surgery. 1989; 106: 50916.
9 Jaeschke H, Farhood A. Neutrophil and Kupffer cell-induced oxidant
stress and ischemia-reperfusion injury in rat liver. Am. J. Physiol.
1991; 260: G355G362.
10 Liu P, McGuire GM, Fisher MA, Farhood A, Smith CW, Jaeschke
H. Activation of Kupffer cells and neutrophils for reactive oxygen
formation is responsible for endotoxin-enhanced liver injury after
hepatic ischemia. Shock. 1995; 3: 5662.
11 Jaeschke H. Enhanced sinusoidal glutathione efux during
endotoxin-induced oxidant stress in vivo. Am. J. Physiol. 1992; 263:
G60G68.
12 Shiratori Y, Kawase T, Shiina S et al. Modulation of hepatotoxicity
by macrophages in the liver. Hepatology. 1988; 8: 81521.
13 Arthur MJ, Bentley IS, Tanner AR, Saunders PK, Millward-Sadler
GH, Wright R. Oxygen-derived free radicals promote hepatic injury
in the rat. Gastroenterology. 1985; 89: 111422.
14 Jaeschke H. Molecular mechanisms of hepatic ischemia-reperfusion
injury and preconditioning. Am. J. Physiol. Gastrointest. Liver
Physiol. 2003; 284: G15G26.
15 Caldwell-Kenkel JC, Currin RT, Tanaka Y, Thurman RG, Lemasters
JJ. Reperfusion injury to endothelial cells following cold ischemic
storage of rat livers. Hepatology. 1989; 10: 2929.
16 Scafdi P, Misteli T, Bianchi ME. Release of chromatin protein
HMGB1 by necrotic cells triggers inammation. Nature. 2002; 418:
1915.
17 Bianchi ME. DAMPs, PAMPs and alarmins: all we need to know
about danger. J. Leukoc. Biol. 2007; 81: 15.
18 Bianchi ME. HMGB1 loves company. J. Leukoc. Biol. 2009; 86:
5736.
19 Schwabe RF, Seki E, Brenner DA. Toll-like receptor signaling in the
liver. Gastroenterology. 2006; 130: 1886900.
20 Tsung A, Sahai R, Tanaka H et al. The nuclear factor HMGB1
mediates hepatic injury after murine liver ischemia-reperfusion. J.
Exp. Med. 2005; 201: 113543.
21 Fan J, Li Y, Levy RM et al. Hemorrhagic shock induces NAD(P)H
oxidase activation in neutrophils: role of HMGB1-TLR4 signaling. J.
Immunol. 2007; 178: 657380.
22 Zhai Y, Shen XD, OConnell R et al. Cutting edge: TLR4 activation
mediates liver ischemia/reperfusion inammatory response via IFN
regulatory factor 3-dependent MyD88-independent pathway. J.
Immunol. 2004; 173: 71159.
23 Bamboat ZM, Balachandran VP, Ocuin LM, Obaid H, Plitas G,
DeMatteo RP. Toll-like receptor 9 inhibition confers protection from
liver ischemia-reperfusion injury. Hepatology. 2010; 51: 62132.
24 Jaeschke H, Farhood A, Bautista AP, Spolarics Z, Spitzer JJ.
Complement activates Kupffer cells and neutrophils during
reperfusion after hepatic ischemia. Am. J. Physiol. 1993; 264:
G801G809.
25 Bajt ML, Farhood A, Jaeschke H. Effects of CXC chemokines on
neutrophil activation and sequestration in hepatic vasculature. Am. J.
Physiol. Gastrointest. Liver Physiol. 2001; 281: G1188G1195.
26 Williams CD, Bajt ML, Farhood A, Jaeschke H.
Acetaminophen-induced hepatic neutrophil accumulation and
inammatory liver injury in CD18-decient mice. Liver Int. 2010;
30: 128092.
27 Bautista AP, Mszros K, Bojta J, Spitzer JJ. Superoxide anion
generation in the liver during the early stage of endotoxemia in rats.
J. Leukoc. Biol. 1990; 48: 1238.
28 Chosay JG, Essani NA, Dunn CJ, Jaeschke H. Neutrophil
margination and extravasation in sinusoids and venules of liver
during endotoxin-induced injury. Am. J. Physiol. 1997; 272:
G1195G1200.
29 Jaeschke H, Fisher MA, Lawson JA, Simmons CA, Farhood A,
Jones DA. Activation of caspase 3 (CPP32)-like proteases is
essential for TNF-alpha-induced hepatic parenchymal cell apoptosis
H Jaeschke Reactive oxygen and liver inammation
177 Journal of Gastroenterology and Hepatology 26 (2011) Suppl. 1; 173179
2011 Journal of Gastroenterology and Hepatology Foundation and Blackwell Publishing Asia Pty Ltd
and neutrophil-mediated necrosis in a murine endotoxin shock
model. J. Immunol. 1998; 160: 34806.
30 Jaeschke H, Ho YS, Fisher MA, Lawson JA, Farhood A.
Glutathione peroxidase-decient mice are more susceptible to
neutrophil-mediated hepatic parenchymal cell injury during
endotoxemia: importance of an intracellular oxidant stress.
Hepatology. 1999; 29: 44350.
31 Gujral JS, Hinson JA, Farhood A, Jaeschke H. NADPH
oxidase-derived oxidant stress is critical for neutrophil cytotoxicity
during endotoxemia. Am. J. Physiol. Gastrointest. Liver Physiol.
2004; 287: G243G252.
32 Gujral JS, Liu J, Farhood A, Hinson JA, Jaeschke H. Functional
importance of ICAM-1 in the mechanism of neutrophil-induced liver
injury in bile duct-ligated mice. Am. J. Physiol. Gastrointest. Liver
Physiol. 2004; 286: G499G507.
33 Miyoshi H, Rust C, Roberts PJ, Burgart LJ, Gores GJ. Hepatocyte
apoptosis after bile duct ligation in the mouse involves Fas.
Gastroenterology. 1999; 117: 66977.
34 Allen K, Jaeschke H, Copple BL. Bile acids induce inammatory
genes in hepatocytes: a novel mechanism of inammation during
obstructive cholestasis. Am. J. Pathol. 2011; DOI:
10.1066/j.ajpath.2010.11.026. in press.
35 Metzger J, Dore SP, Lauterburg BH. Oxidant stress during
reperfusion of ischemic liver: no evidence for a role of xanthine
oxidase. Hepatology. 1988; 8: 5804.
36 Jaeschke H, Bautista AP, Spolarics Z, Spitzer JJ. Superoxide
generation by Kupffer cells and priming of neutrophils during
reperfusion after hepatic ischemia. Free Radic. Res. Commun. 1991;
15: 27784.
37 Liu P, Fisher MA, Farhood A, Smith CW, Jaeschke H. Benecial
effects of extracellular glutathione against endotoxin-induced liver
injury during ischemia and reperfusion. Circ. Shock. 1994; 43:
6470.
38 Bilzer M, Baron A, Schauer R, Steib C, Ebensberger S, Gerbes AL.
Glutathione treatment protects the rat liver against injury after warm
ischemia and Kupffer cell activation. Digestion. 2002; 66: 4957.
39 Jaeschke H, Bautista AP, Spolarics Z, Spitzer JJ. Superoxide
generation by neutrophils and Kupffer cells during in vivo reperfusion
after hepatic ischemia in rats. J. Leukoc. Biol. 1992; 52: 37782.
40 Hasegawa T, Malle E, Farhood A, Jaeschke H. Generation of
hypochlorite-modied proteins by neutrophils during
ischemia-reperfusion injury in rat liver: attenuation by ischemic
preconditioning. Am. J. Physiol. Gastrointest. Liver Physiol. 2005;
289: G760G767.
41 Jaeschke H, Smith CW. Mechanisms of neutrophil-induced
parenchymal cell injury. J. Leukoc. Biol. 1997; 61: 64753.
42 Shappell SB, Toman C, Anderson DC, Taylor AA, Entman ML,
Smith CW. Mac-1 (CD11b/CD18) mediates adherence-dependent
hydrogen peroxide production by human and canine neutrophils. J.
Immunol. 1990; 144: 270211.
43 Jaeschke H. Reactive oxygen and mechanisms of inammatory liver
injury. J. Gastroenterol. Hepatol. 2000; 15: 71824.
44 Mavier P, Preaux AM, Guigui B, Lescs MC, Zafrani ES, Dhumeaux
D. In vitro toxicity of polymorphonuclear neutrophils to rat
hepatocytes: evidence for a proteinase-mediated mechanism.
Hepatology. 1988; 8: 2548.
45 Hazen SL, Crowley JR, Mueller DM, Heinecke JW. Mass
spectrometric quantication of 3-chlorotyrosine in human tissues
with attomole sensitivity: a sensitive and specic marker for
myeloperoxidase-catalyzed chlorination at sites of inammation.
Free Radic. Biol. Med. 1997; 23: 90916.
46 Lehnert M, Arteel GE, Smutney OM et al. Dependence of liver
injury after hemorrhage/resuscitation in mice on NADPH
oxidase-derived superoxide. Shock. 2003; 19: 34551.
47 Schauer RJ, Gerbes AL, Vonier D, op den Winkel M, Fraunberger P,
Bilzer M. Induction of cellular resistance against Kupffer
cell-derived oxidant stress: a novel concept of hepatoprotection by
ischemic preconditioning. Hepatology. 2003; 37: 28695.
48 Wiedow O, Meyer-Hoffert U. Neutrophil serine proteases: potential
key regulators of cell signalling during inammation. J. Intern. Med.
2005; 257: 31928.
49 Uchida Y, Freitas MC, Zhao D, Busuttil RW, Kupiec-Weglinski JW.
The protective function of neutrophil elastase inhibitor in liver
ischemia/reperfusion injury. Transplantation. 2010; 89: 10506.
50 Weiss SJ. Tissue destruction by neutrophils. N. Engl. J. Med. 1989;
320: 36576.
51 Jaeschke H. Antioxidant Defense Mechanisms. In: McQueen CA, ed.
Comprehensive Toxicology. Vol. 9, Oxford: Academic Press, 2010;
31937.
52 Wendel A, Feuerstein S. Drug-induced lipid peroxidation in miceI.
Modulation by monooxygenase activity, glutathione and selenium
status. Biochem. Pharmacol. 1981; 30: 251320.
53 Yan HM, Ramachandran A, Bajt ML, Lemasters JJ, Jaeschke H. The
oxygen tension modulates acetaminophen-induced mitochondrial
oxidant stress and cell injury in cultured hepatocytes. Toxicol. Sci.
2010; 117: 51523.
54 Halliwell B. Oxidative stress in cell culture: an under-appreciated
problem? FEBS Lett. 2003; 540: 36.
55 Mathews WR, Guido DM, Fisher MA, Jaeschke H. Lipid
peroxidation as molecular mechanism of liver cell injury during
reperfusion after ischemia. Free Radic. Biol. Med. 1994; 16: 76370.
56 Knight TR, Fariss MW, Farhood A, Jaeschke H. Role of lipid
peroxidation as a mechanism of liver injury after acetaminophen
overdose in mice. Toxicol. Sci. 2003; 76: 22936.
57 Singh R, Czaja MJ. Regulation of hepatocyte apoptosis by oxidative
stress. J. Gastroenterol. Hepatol. 2007; 22 (Suppl. 1): S45S48.
58 Czaja MJ, Liu H, Wang Y. Oxidant-induced hepatocyte injury from
menadione is regulated by ERK and AP-1 signaling. Hepatology.
2003; 37: 140513.
59 Conde de la Rosa L, Schoemaker MH, Vrenken TE et al. Superoxide
anions and hydrogen peroxide induce hepatocyte death by different
mechanisms: involvement of JNK and ERK MAP kinases. J.
Hepatol. 2006; 44: 91829.
60 Nieminen AL, Byrne AM, Herman B, Lemasters JJ. Mitochondrial
permeability transition in hepatocytes induced by t-BuOOH:
NAD(P)H and reactive oxygen species. Am. J. Physiol. 1997; 272:
C1286C1294.
61 Hong JY, Lebofsky M, Farhood A, Jaeschke H. Oxidant
stress-induced liver injury in vivo: role of apoptosis, oncotic
necrosis, and c-Jun NH2-terminal kinase activation. Am. J. Physiol.
Gastrointest. Liver Physiol. 2009; 296: G572G581.
62 Smith CV. Evidence for participation of lipid peroxidation and iron
in diquat-induced hepatic necrosis in vivo. Mol. Pharmacol. 1987;
32: 41722.
63 Gujral JS, Bucci TJ, Farhood A, Jaeschke H. Mechanism of cell
death during warm hepatic ischemia-reperfusion in rats: apoptosis or
necrosis? Hepatology. 2001; 33: 397405.
64 Gujral JS, Liu J, Farhood A, Jaeschke H. Reduced oncotic necrosis
in Fas receptor-decient C57BL/6J-lpr mice after bile duct ligation.
Hepatology. 2004; 40: 9981007.
65 Aguilar HI, Botla R, Arora AS, Bronk SF, Gores GJ. Induction of
the mitochondrial permeability transition by protease activity in rats:
a mechanism of hepatocyte necrosis. Gastroenterology. 1996; 110:
55866.
66 Uchiyama A, Kim JS, Kon K et al. Translocation of iron from
lysosomes into mitochondria is a key event during oxidative
stress-induced hepatocellular injury. Hepatology. 2008; 48:
164454.
Reactive oxygen and liver inammation H Jaeschke
178 Journal of Gastroenterology and Hepatology 26 (2011) Suppl. 1; 173179
2011 Journal of Gastroenterology and Hepatology Foundation and Blackwell Publishing Asia Pty Ltd
67 Ollinger K, Roberg K. Nutrient deprivation of cultured rat
hepatocytes increases the desferrioxamine-available iron pool and
augments the sensitivity to hydrogen peroxide. J. Biol. Chem. 1997;
272: 2370711.
68 Bilzer M, Lauterburg BH. Effects of hypochlorous acid and
chloramines on vascular resistance, cell integrity, and biliary
glutathione disulde in the perfused rat liver: modulation by
glutathione. J. Hepatol. 1991; 13: 849.
69 Schraufsttter IU, Browne K, Harris A et al. Mechanisms of
hypochlorite injury of target cells. J. Clin. Invest. 1990; 85: 55462.
70 Rdiger HA, Graf R, Clavien PA. Sub-lethal oxidative stress triggers
the protective effects of ischemic preconditioning in the mouse liver.
J. Hepatol. 2003; 39: 9727.
71 Kensler TW, Wakabayashi N, Biswal S. Cell survival responses to
environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev.
Pharmacol. Toxicol. 2007; 47: 89116.
72 Tsuchihashi S, Fondevila C, Kupiec-Weglinski JW. Heme oxygenase
system in ischemia and reperfusion injury. Ann. Transplant. 2004; 9:
847.
73 Cover C, Liu J, Farhood A et al. Pathophysiological role of the acute
inammatory response during acetaminophen hepatotoxicity. Toxicol.
Appl. Pharmacol. 2006; 216: 98107.
74 Holt MP, Cheng L, Ju C. Identication and characterization of
inltrating macrophages in acetaminophen-induced liver injury. J.
Leukoc. Biol. 2008; 84: 141021.
75 Smedsrd B, Le Couteur D, Ikejima K et al. Hepatic sinusoidal cells
in health and disease: update from the 14th International
Symposium. Liver Int. 2009; 29: 490501.
76 Teoh N, Field J, Sutton J, Farrell G. Dual role of tumor necrosis
factor-alpha in hepatic ischemia-reperfusion injury: studies in tumor
necrosis factor-alpha gene knockout mice. Hepatology. 2004; 39:
41221.
77 He S, Atkinson C, Qiao F, Cianone K, Chen X, Tomlinson S.
A complement-dependent balance between hepatic
ischemia/reperfusion injury and liver regeneration in mice. J. Clin.
Invest. 2009; 119: 230416.
H Jaeschke Reactive oxygen and liver inammation
179 Journal of Gastroenterology and Hepatology 26 (2011) Suppl. 1; 173179
2011 Journal of Gastroenterology and Hepatology Foundation and Blackwell Publishing Asia Pty Ltd
Das könnte Ihnen auch gefallen
- Clinical CaseDokument25 SeitenClinical CaseRussell Talan CilotNoch keine Bewertungen
- Systemic Lupus Erythematosus: Basic, Applied and Clinical AspectsVon EverandSystemic Lupus Erythematosus: Basic, Applied and Clinical AspectsGeorge C. TsokosNoch keine Bewertungen
- Biochemical and Cellular Mechanisms of Toxic Liver Injury: Neil Kaplowitz, M.DDokument8 SeitenBiochemical and Cellular Mechanisms of Toxic Liver Injury: Neil Kaplowitz, M.DAya AmerNoch keine Bewertungen
- Apoptosis and Therapy: Cold Spring Harbor Laboratory, Cold Spring Harbor, NY 11724, U.S.ADokument11 SeitenApoptosis and Therapy: Cold Spring Harbor Laboratory, Cold Spring Harbor, NY 11724, U.S.AAntonio RolonNoch keine Bewertungen
- 1-Advances in SepsisDokument10 Seiten1-Advances in SepsisGiovanna TardinNoch keine Bewertungen
- Leukocytes, Lymphocytes, Endothelial / Parenchymal Dan Dilanjutkan Dengan Humoral Respons Antara LainDokument11 SeitenLeukocytes, Lymphocytes, Endothelial / Parenchymal Dan Dilanjutkan Dengan Humoral Respons Antara Lainlalu irwan romadhanNoch keine Bewertungen
- SepsisDokument2 SeitenSepsisNannai02Noch keine Bewertungen
- Suspected Article Mos 2009 JMA XII 1Dokument6 SeitenSuspected Article Mos 2009 JMA XII 1Oana CraciunNoch keine Bewertungen
- 1 s2.0 S0085253815474071 MainDokument8 Seiten1 s2.0 S0085253815474071 MainBintiNoch keine Bewertungen
- Cell Death 2011Dokument15 SeitenCell Death 2011Paula Contreras MezaNoch keine Bewertungen
- NIH Public Access: Author ManuscriptDokument17 SeitenNIH Public Access: Author ManuscriptSanjeev SaxenaNoch keine Bewertungen
- Cellular and Molecular Mechanisms of Hepatic Fibrogenesis Leading To Liver CancerDokument9 SeitenCellular and Molecular Mechanisms of Hepatic Fibrogenesis Leading To Liver CancerMessia RaharjoNoch keine Bewertungen
- Stres Oxid.Dokument8 SeitenStres Oxid.Maria Luiza CraciunNoch keine Bewertungen
- Sepsis Si CoagulareDokument8 SeitenSepsis Si CoagulareCorina DavidNoch keine Bewertungen
- izab277Dokument10 Seitenizab277MartinaNoch keine Bewertungen
- Acetaminophen-Induced Liver Injury: From Animal Models To HumansDokument8 SeitenAcetaminophen-Induced Liver Injury: From Animal Models To HumansMiguel Adrian GaonaNoch keine Bewertungen
- Apoptotic Clearance RegulationDokument15 SeitenApoptotic Clearance Regulationare4erNoch keine Bewertungen
- Gabungan 3materi (Dipake)Dokument7 SeitenGabungan 3materi (Dipake)Indra SaputraNoch keine Bewertungen
- 1 Apoptosis DesDokument7 Seiten1 Apoptosis DessashiwiiNoch keine Bewertungen
- Activation of Adhesion Molecules in Septic ShockDokument8 SeitenActivation of Adhesion Molecules in Septic ShockIkhsan AliNoch keine Bewertungen
- Cellular Injury, Necrosis and ApoptosisDokument34 SeitenCellular Injury, Necrosis and ApoptosisSuraj UsmanNoch keine Bewertungen
- Does Ozone Therapy Normalize The Cellular Redox Balance?Dokument5 SeitenDoes Ozone Therapy Normalize The Cellular Redox Balance?Mariana MilesiNoch keine Bewertungen
- Recent Discoveries in the Immunopathogenesis of SepsisDokument11 SeitenRecent Discoveries in the Immunopathogenesis of SepsisjafralizNoch keine Bewertungen
- Role of ROS in metabolic diseases and chronic inflammationDokument9 SeitenRole of ROS in metabolic diseases and chronic inflammationFian AldyNoch keine Bewertungen
- Bilirubin 5Dokument8 SeitenBilirubin 5quattro2Noch keine Bewertungen
- Podocyte Injury and Its Consequences: Michio NagataDokument10 SeitenPodocyte Injury and Its Consequences: Michio Nagatablume diaNoch keine Bewertungen
- Possible Mechanisms of Disease Development in Tuberous SclerosisDokument7 SeitenPossible Mechanisms of Disease Development in Tuberous Sclerosisplastioid4079Noch keine Bewertungen
- Necrosis CPDDokument14 SeitenNecrosis CPDPŕìncëş Ğï ŘlNoch keine Bewertungen
- Apoptosis in Cancer: Carcinogenesis Vol.21 No.3 pp.485-495, 2000Dokument0 SeitenApoptosis in Cancer: Carcinogenesis Vol.21 No.3 pp.485-495, 2000dragoonlee75Noch keine Bewertungen
- Recent Findings On The Mechanism of Cisplatin-Induced Renal Cytotoxicity and Therapeutic Potential of Natural CompoundsDokument22 SeitenRecent Findings On The Mechanism of Cisplatin-Induced Renal Cytotoxicity and Therapeutic Potential of Natural CompoundsSamantha AdrianneNoch keine Bewertungen
- Autophagy in Vascular Endothelial Cells - 2016Dokument8 SeitenAutophagy in Vascular Endothelial Cells - 2016nazlibesirikNoch keine Bewertungen
- HHS Public Access: Unraveling The Complex Interplay Between T Cell Metabolism and FunctionDokument33 SeitenHHS Public Access: Unraveling The Complex Interplay Between T Cell Metabolism and Functionro111111Noch keine Bewertungen
- Coagulation and Innate Immune Responses: Can We View Them Separately?Dokument9 SeitenCoagulation and Innate Immune Responses: Can We View Them Separately?nuraswadNoch keine Bewertungen
- Necroptosis PDFDokument11 SeitenNecroptosis PDFdavid zambranoNoch keine Bewertungen
- Pharmacological Research: Nabila Bourebaba, Krzysztof MaryczDokument10 SeitenPharmacological Research: Nabila Bourebaba, Krzysztof Marycz2m96k96vp8Noch keine Bewertungen
- Guardian Del GenomaDokument7 SeitenGuardian Del GenomaNatalia NomesqueNoch keine Bewertungen
- Cancers 11 00Dokument17 SeitenCancers 11 00GeorgeNoch keine Bewertungen
- Macrophage Response Towards LPSDokument10 SeitenMacrophage Response Towards LPSajaewoongNoch keine Bewertungen
- F - 2178 JCD Role of Apoptosis in Amplifying Inflammatory Responses in Lung Disease - PDF - 2981 PDFDokument13 SeitenF - 2178 JCD Role of Apoptosis in Amplifying Inflammatory Responses in Lung Disease - PDF - 2981 PDFRanti WidiyantiNoch keine Bewertungen
- Neutrophils Induce Paracrine Telomere Dysfunction and Senescence in ROS-dependent MannerDokument19 SeitenNeutrophils Induce Paracrine Telomere Dysfunction and Senescence in ROS-dependent MannerValen EstevezNoch keine Bewertungen
- Role of Reactive Oxygen Species in p53 Activation During Cisplatin-Induced Apoptosis of Rat Mesangial CellsDokument7 SeitenRole of Reactive Oxygen Species in p53 Activation During Cisplatin-Induced Apoptosis of Rat Mesangial CellsPandji 'Utuh'Noch keine Bewertungen
- Novel Insights Into How Nitric Oxide Regulates Cell ProliferationDokument7 SeitenNovel Insights Into How Nitric Oxide Regulates Cell ProliferationArief NurmanNoch keine Bewertungen
- Polymer Drug Conjugates As Potent Against Cellular ApoptosisDokument8 SeitenPolymer Drug Conjugates As Potent Against Cellular ApoptosisGourav DasNoch keine Bewertungen
- Atherosclerosis: Evidence For Impairment of Resolution of Vascular Inflammation Governed by Specific Lipid MediatorsDokument12 SeitenAtherosclerosis: Evidence For Impairment of Resolution of Vascular Inflammation Governed by Specific Lipid MediatorsMaria AnghelacheNoch keine Bewertungen
- Review Article: Complement Diagnostics: Concepts, Indications, and Practical GuidelinesDokument12 SeitenReview Article: Complement Diagnostics: Concepts, Indications, and Practical GuidelinesGustavo NúñezNoch keine Bewertungen
- Microparticles in Vascular DiseaseDokument5 SeitenMicroparticles in Vascular DiseaseRichard ChandraNoch keine Bewertungen
- Cell Stress, Inflammatory Responses and Cell Death: SynopsisDokument29 SeitenCell Stress, Inflammatory Responses and Cell Death: SynopsisSasa PuiuNoch keine Bewertungen
- NETosis: Molecular Mechanisms, Role in Physiology and PathologyDokument22 SeitenNETosis: Molecular Mechanisms, Role in Physiology and PathologysebasgaravanoNoch keine Bewertungen
- Holmes 2013Dokument18 SeitenHolmes 2013Christina MountakiNoch keine Bewertungen
- Newsholme 2019 Oxidative Stress Pathways in Pancreatic Beta Cell and Insulin-Sensitive Cells and TissuesDokument14 SeitenNewsholme 2019 Oxidative Stress Pathways in Pancreatic Beta Cell and Insulin-Sensitive Cells and TissuesPaul SimononNoch keine Bewertungen
- Am J Clin Nutr 2006 Calder S1505 1519SDokument15 SeitenAm J Clin Nutr 2006 Calder S1505 1519SClever KaipNoch keine Bewertungen
- Complement and coagulation: partners in inflammationDokument9 SeitenComplement and coagulation: partners in inflammationMedranoReyesLuisinNoch keine Bewertungen
- Targeting Mitochondria For Cancer TherapyDokument18 SeitenTargeting Mitochondria For Cancer Therapysatya_chagantiNoch keine Bewertungen
- He Pa To BiliaryDokument14 SeitenHe Pa To BiliaryMayerly Camila González CórdobaNoch keine Bewertungen
- Pyroptosis: A Caspase-1-Dependent Programmed Cell Death and A Barrier To InfectionDokument21 SeitenPyroptosis: A Caspase-1-Dependent Programmed Cell Death and A Barrier To Infectionrovia achmad .Noch keine Bewertungen
- Eferocitosis en Salud y Enfermedad.Dokument14 SeitenEferocitosis en Salud y Enfermedad.Dana GonzalezNoch keine Bewertungen
- Pathobiology of Secondary Immune ThrombocytopeniaDokument14 SeitenPathobiology of Secondary Immune ThrombocytopeniateoNoch keine Bewertungen
- M7L7B1 The Endotoxin Hypothesis of NeurodegenerationDokument10 SeitenM7L7B1 The Endotoxin Hypothesis of NeurodegenerationLeocadio Alcalá MORILLASNoch keine Bewertungen
- Complement and T Cell RegulationDokument41 SeitenComplement and T Cell RegulationFrancelia Quiñonez RuvalcabaNoch keine Bewertungen
- Paper97 PDFDokument8 SeitenPaper97 PDFAditya Dana IswaraNoch keine Bewertungen
- Am J Physiol Endocrinol Metab 2009 Widberg E121 31Dokument12 SeitenAm J Physiol Endocrinol Metab 2009 Widberg E121 31Aditya Dana IswaraNoch keine Bewertungen
- Coffee Sport 2 Nutrients-13-00002Dokument17 SeitenCoffee Sport 2 Nutrients-13-00002Aditya Dana IswaraNoch keine Bewertungen
- F-3 Handle lever and switch assembly guide</TITLEDokument72 SeitenF-3 Handle lever and switch assembly guide</TITLEHesekiel Butar-butarNoch keine Bewertungen
- MODUL 5-Group JDokument44 SeitenMODUL 5-Group JAditya Dana IswaraNoch keine Bewertungen
- 1471 2210 2 7Dokument8 Seiten1471 2210 2 7Aditya Dana IswaraNoch keine Bewertungen
- Stewart and Costerton 2001Dokument4 SeitenStewart and Costerton 2001Aditya Dana IswaraNoch keine Bewertungen
- Cazclinic DLM EnglDokument8 SeitenCazclinic DLM EnglMaria-Lavinia HoinaruNoch keine Bewertungen
- WarfarinDokument19 SeitenWarfarinAditya Dana IswaraNoch keine Bewertungen
- KatsarosAmJOrthodDentofacialOrthop2006129352 357Dokument6 SeitenKatsarosAmJOrthodDentofacialOrthop2006129352 357Aditya Dana IswaraNoch keine Bewertungen
- 9 April 2013 PengecapanDokument63 Seiten9 April 2013 PengecapanAditya Dana IswaraNoch keine Bewertungen
- Jalt 04 I 3 P 94Dokument3 SeitenJalt 04 I 3 P 94Aditya Dana IswaraNoch keine Bewertungen
- Curriculum Resource GuideDokument113 SeitenCurriculum Resource GuideAditya Dana IswaraNoch keine Bewertungen
- Modul 7 CHAPTER 1 IntroductionDokument2 SeitenModul 7 CHAPTER 1 IntroductionAditya Dana IswaraNoch keine Bewertungen
- CONCLUSIONDokument1 SeiteCONCLUSIONAditya Dana IswaraNoch keine Bewertungen
- Trismus Aetiology Differentialdiagnosisandtreatment99616Dokument5 SeitenTrismus Aetiology Differentialdiagnosisandtreatment99616Ellizabeth LilantiNoch keine Bewertungen
- ChlorhexidineDokument1 SeiteChlorhexidineAditya Dana IswaraNoch keine Bewertungen
- KentutDokument6 SeitenKentutAditya Dana IswaraNoch keine Bewertungen
- SABiosciences Pathways Issue 7Dokument20 SeitenSABiosciences Pathways Issue 7triplegreenhand100% (5)
- Once The Barrier Is Breached How Are Pathogens Detected and Dealt With?Dokument64 SeitenOnce The Barrier Is Breached How Are Pathogens Detected and Dealt With?Seyeon Clare LeeNoch keine Bewertungen
- Shinjita Das M.D., Rachel Reynolds M.D. (Auth.), Joshua Zeichner (Eds.) - Acneiform Eruptions in Dermatology - A Differential Diagnosis-Springer-Verlag New York (2014) PDFDokument405 SeitenShinjita Das M.D., Rachel Reynolds M.D. (Auth.), Joshua Zeichner (Eds.) - Acneiform Eruptions in Dermatology - A Differential Diagnosis-Springer-Verlag New York (2014) PDFMaryNoch keine Bewertungen
- Russel J. Reiter Pedro Abreu-Gonzalez Alberto Dominguez-RodriguezDokument80 SeitenRussel J. Reiter Pedro Abreu-Gonzalez Alberto Dominguez-RodriguezFilipos ConstantinNoch keine Bewertungen
- PNAS 2013 Bellet 9897 902Dokument6 SeitenPNAS 2013 Bellet 9897 902Anca Maria GalisNoch keine Bewertungen
- Structure and Function of Joints PDFDokument53 SeitenStructure and Function of Joints PDFABHAY KUMARNoch keine Bewertungen
- From Pathogenesis of Acne Vulgaris To Anti-Acne AgentsDokument13 SeitenFrom Pathogenesis of Acne Vulgaris To Anti-Acne AgentsNazihan Safitri AlkatiriNoch keine Bewertungen
- Immunity All NotesDokument112 SeitenImmunity All NotesJonathan Carlisle CheungNoch keine Bewertungen
- Chapter 28 PROMDokument24 SeitenChapter 28 PROMTansya PurnaningrumNoch keine Bewertungen
- Stanojevic Exp. GerontologyDokument40 SeitenStanojevic Exp. GerontologyIvanaNoch keine Bewertungen
- Inmunidad InnataDokument7 SeitenInmunidad InnataWilfrido Jose Barrios AgamezNoch keine Bewertungen
- Fisiopatología de La Meningitis BacterianaDokument7 SeitenFisiopatología de La Meningitis BacterianaGiuliano AlbizziNoch keine Bewertungen
- Toll-Like Receptors Role in Periodontal Health and DiseaseDokument15 SeitenToll-Like Receptors Role in Periodontal Health and DiseaseAdyas AdrianaNoch keine Bewertungen
- In Ammation in Heart Failure: JACC State-of-the-Art ReviewDokument17 SeitenIn Ammation in Heart Failure: JACC State-of-the-Art ReviewAhmad FauzanNoch keine Bewertungen
- Toll Like Receptors and Their LigandsDokument21 SeitenToll Like Receptors and Their Ligandsakash_81087Noch keine Bewertungen
- Pseudomonas 2015Dokument319 SeitenPseudomonas 2015Julio Martinez RodriguezNoch keine Bewertungen
- Role of Progesterone in Tlr4-Myd88-Dependent Signaling Pathway in Pre-EclampsiaDokument5 SeitenRole of Progesterone in Tlr4-Myd88-Dependent Signaling Pathway in Pre-EclampsiaCarl Enrique PSNoch keine Bewertungen
- Pathophysiology Septic ShockDokument26 SeitenPathophysiology Septic ShockTinea Sycillia100% (1)
- RP105 and Its Role On PeriodontitisDokument4 SeitenRP105 and Its Role On PeriodontitisVijayalaxmi KokatnurNoch keine Bewertungen
- NF-KB Activation by Pamps and DampsDokument8 SeitenNF-KB Activation by Pamps and DampsShubham RastogiNoch keine Bewertungen
- Immunology KubyDokument24 SeitenImmunology KubySayanta Bera100% (1)
- Pgs 145-59, Neonatal Immunity by Constantin Bona (Humana Press) 2004 - $145.00Dokument401 SeitenPgs 145-59, Neonatal Immunity by Constantin Bona (Humana Press) 2004 - $145.00Riki TobingNoch keine Bewertungen
- Articulo Obesidad e InflamacionDokument19 SeitenArticulo Obesidad e InflamacionPaulo CardosoNoch keine Bewertungen
- Antiphospholipid Syndrome - From Pathogenesis To Novel Immunomodulatory TherapiesDokument6 SeitenAntiphospholipid Syndrome - From Pathogenesis To Novel Immunomodulatory TherapiesardirshajiNoch keine Bewertungen
- Naltrexone & Low-Dose NaltexoneDokument67 SeitenNaltrexone & Low-Dose NaltexoneShi_DMS100% (1)
- Damps, Pamps, and Lamps in Immunity and Sterile InflammationDokument26 SeitenDamps, Pamps, and Lamps in Immunity and Sterile InflammationCony G100% (1)
- Pathophysiology and Treatment Syok Septic in PediatricDokument41 SeitenPathophysiology and Treatment Syok Septic in PediatricGustomo PanantroNoch keine Bewertungen
- Circulating Heat Shock Protein 60 Levels Are Elevated in HIV Patients and Are Reduced by Anti-Retroviral TherapyDokument8 SeitenCirculating Heat Shock Protein 60 Levels Are Elevated in HIV Patients and Are Reduced by Anti-Retroviral TherapyAmalia WidowatiNoch keine Bewertungen
- Association Between Vasomotor Rhinitis and Irritable Bowel SyndromeDokument7 SeitenAssociation Between Vasomotor Rhinitis and Irritable Bowel SyndromedeaNoch keine Bewertungen