Beruflich Dokumente
Kultur Dokumente
Interfernce
Hochgeladen von
Elvan Dwi WidyadiCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Interfernce
Hochgeladen von
Elvan Dwi WidyadiCopyright:
Verfügbare Formate
Evaluation of the interference of hemoglobin, bilirubin, and lipids on Roche Cobas
6000 assays
Jing Zhang Ji
a, b
, Qing H. Meng
a,
a
Department of Pathology and Laboratory Medicine, Royal University Hospital, University of Saskatchewan, Saskatoon, SK, Canada
b
Key Laboratory of Laboratory Medicine, Ministry of Education, Zhejiang Provincial Key Laboratory of Medical Genetics, School of Laboratory Medicine, Wenzhou Medical College,
Wenzhou, China
a b s t r a c t a r t i c l e i n f o
Article history:
Received 31 March 2011
Received in revised form 27 April 2011
Accepted 28 April 2011
Available online 6 May 2011
Keywords:
Analytical interference
Hemolysis
Icterus
Lipemia
Roche Cobas
Background: Pre-analytical error accounts for major total laboratory errors. We assessed the impacts of
hemolysis, icterus, and lipemia on laboratory tests on Roche Cobas 6000.
Methods: Various concentrations of hemoglobin, bilirubin, or Intralipid were added into the plasma to
simulate hemolytic, icteric, or lipemic samples. The analytes were then measured on Roche Cobas 6000 and
the change of the analyte concentrations was determined.
Results: For most of the chemistry assays, our data were in a good agreement with Roche package inserts.
However some assays had signicant interference at lower index values while others were affected at higher
index than the Roche package inserts indicated. In addition, we observed the positive interference by
hemolysis on ALT, lipase, total protein, potassium, and iron. Negative interference was noted on calcium and
CK. Most of the immunoassays were not affected by hemoglobin, bilirubin, and lipids although there were a
few exceptions. Several therapeutic drugs were affected either positively or negatively by hemolysis, icterus,
or lipemia to a certain extent.
Conclusions: We have demonstrated some test interferences which have not been reported previously on the
Cobas 6000. The implementation of the cut-off indices on Cobas 6000 would provide more accurate test result
reporting.
2011 Elsevier B.V. All rights reserved.
1. Introduction
Analytical interference caused by pre-analytical factors is a
signicant source of error in clinical laboratory measurements [1,2].
Analytical interference by hemolysis, bilirubin and lipids with
laboratory assays is the most common concern in laboratory
medicine. These altered results may lead to repeat tests, incorrect
interpretation, wrong diagnosis, and potentially inappropriate inter-
vention and unfavorable outcome for the patients [35]. Hemolysis,
icterus, and lipemia commonly interfere with spectrophotometric
methods with hemolysis being the most common [3,4,6,7]. This
interference is mainly caused by components released from the red
cells. Although direct spectral interference on chemistry analyzers has
been minimized with bichromatic and kinetic analysis, the contents of
red cells like potassium and lactate dehydrogenase falsely increase
these constituents in plasma or serum. There are other constituents
released from red cells that can interfere with test reactions. Finally,
analytes can also get diluted in hemolysis [4]. Hemolysis can occur in
vivo, but the major problem that clinical laboratories get is that it
occurs during and after collection of specimens. Bilirubin can interfere
with assays spectrally but also chemically in some reactions.
Normally, bilirubin concentrations N35 mmol/l are clinically dened
as hyperbilirubinemia, whereas icteric samples have bilirubin con-
centrations N100 mmol/l [3]. A lipemic sample is the result of high
concentrations of chylomicrons and very-low density lipoprotein
(VLDL). Lipemia may interfere in any assay that is based on the
detection of light transmission or scattering or by volume displace-
ment [8,9]. The presence of hemoglobin, bilirubin, and lipids in a
specimen can cause a positive or negative interference in the
measurement result of many analytes [1012]. Depending on the
magnitude of this interference, the results may lead to wrong
interpretation and inappropriate intervention [5,1315]. Interference
on the Beckman system has been well investigated [8,16]. Neverthe-
less, the degree of interference varies with the methodology and
individual instrumentation and comprehensive interference evalua-
tion and published data are not available on Roche Cobas system
(Indianapolis, IN).
2. Subjects and methods
To evaluate the impact of an interferent on analytes, the analyte
was measured in a sample spiked with increasing concentrations of
Clinica Chimica Acta 412 (2011) 15501553
Corresponding author at: Department of Pathology and Laboratory Medicine, Royal
University Hospital, University of Saskatchewan, 103 Hospital Drive, Saskatoon, SK,
Canada S7N 0W8. Fax: +1 306 655 2193.
E-mail address: qing.meng@usask.ca (Q.H. Meng).
0009-8981/$ see front matter 2011 Elsevier B.V. All rights reserved.
doi:10.1016/j.cca.2011.04.034
Contents lists available at ScienceDirect
Clinica Chimica Acta
j our nal homepage: www. el sevi er. com/ l ocat e/ cl i nchi m
the interferent, and the relative deviation of the result from the non-
spiked baseline value was then calculated.
2.1. Subjects
Pooled lithium heparinized plasma free of interferent was
prepared as test sample. Hemolysate was prepared by washing
packed red cells (0.9% saline and distilled water) and then freezing,
and thawing. Various concentrations of hemolysate were added into
the plasma to simulate different degrees of hemolysis. Icteric samples
were obtained with the addition of unconjugated bilirubin (Sigma-
Aldrich, St. Louis, MO) to specimens to achieve different concentra-
tions of bilirubin. Because of poor solubility of unconjugated bilirubin,
bilirubin was prepared in a stock solution in dimethyl sulfoxide [6,17].
The proportion of bilirubin in serum or plasma between conjugated
and unconjugated is not a signicant factor for interference [3].
Lipemic samples were prepared by adding Intralipid (20%) (Baxter,
Deereld, IL). The analytes were then measured on Roche Cobas 6000
(c501 and e601) and the change of the analyte concentrations was
compared with baseline samples.
2.2. Chemistry analyzer
The multi-channel Roche Cobas 6000 analyzer is a fully automated,
computer-controlled system designed for the analysis of routine
chemistry assays, immunoassays, and therapeutic drugs. The Cobas
6000 uses spectrophotometry to perform kinetic, end-point and non-
linear reactions. To a certain extent, the system, similar to most
modern analyzers, reduces spectral interference effects by application
of two-reagent procedures and bichromatic spectrophotometry. The
Cobas 6000 analyzer is able to detect hemolysis, icterus, and lipemia
in samples and can generate quantitative index values for the major
interfering substances of hemoglobin, bilirubin, and lipids expressed
as H-index (hemolysis), I-index (icterus), and L-index (lipemia). The
relationship of each HIL index with the SI unit or conventional unit is
listed in Table 1.
2.3. Analytes investigated
The following tests were investigated: 1-antitrypsin (A1AT),
acetaminophen, -fetoprotein (AFP), albumin (Alb), alcohol, alkaline
phosphatase (ALP), alanine aminotransferase (ALT), ammonia (Amm),
aspartate aminotransferase (AST), 2-microglobulin (2MI), human
chorionic gonadotropin (HCG), complement C
3
(C
3
), complement
C
4
(C
4
), calcium (CA), carbamazepine, carcinoembyronic antigen
(CEA), ceruloplasmin (Cerulo), cholesterol (Chol), creatine kinase
(CK), chloride (Cl), bicarbonate (CO
2
), cortisol (Cort), creatinine
(Creat), high-sensitivity C-reactive protein (hsCRP), direct bilirubin
(D. Bili), digoxin, ferrous iron (Fe
2+
), ferritin (FER), free triiodothy-
ronine(FRT3), free thyroxine (FRT4), follicle-stimulating hormone
(FSH), gentamicin, -glutamyltransferase (GGT), glucose, haptoglobin,
high density lipoprotein cholesterol (HDL-C), homocysteine, immu-
noglobin A (IgA), immunoglobin G (IgG), immunoglobin M (IgM),
potassium (K
+
), lactate dehydrogenase (LD), luteinizing hormone
(LH), lipase, magnesium (Mg
2+
), myoglobin (Myo), sodium (Na
+
),
pre-albumin (PreALB), phenobarbital, phosphorus (Phos), prolactin
(PRL), prostate-specic antigen (PSA), phenytoin, rheumatoid factor
(RHF), salicylate, total bilirubin (T.Bili), total bilirubin in cord blood
(TBICB), theophylline, tobramycin, total protein (TP), Troponin
T (TnT), thyroid stimulating hormone (TSH), unsaturated iron binding
capacity (UIBC), urea, uric acid, vancomycin, vitamin B12 (VB12), and
valproic acid.
2.4. Statistical analysis
Identify the measured hemolytic, icteric or lipemic index
that corresponds to the point where the test analyte differs by
N10% from the baseline. The percent change was calculated as:
Interference%=100(measured valuetrue value)/true value,
where the measured value is the apparent analyte concentration in
the presence of interferent, and the true value is the analyte
concentration in the baseline without any addition of interferent.
Signicant interference was dened when the change of the analyte
value exceeded 10% of the baseline value [13,17].
3. Results
3.1. Interferences of hemoglobin, bilirubin, and lipids on
chemistry assays
The overall interferences of hemoglobin, bilirubin, and lipids on
chemistry assays are listed in Table 2. For most of the chemistry
assays, our data were in a good agreement with what Roche reagent
package inserts indicated. However some assays were affected
Table 1
Relationship between interferent concentration and the corresponding index.
H=1 I =1 L=1
Conventional units 1 mg/dl 1 mg/dl 1
(without units)
SI units 0.621 mol/l 17.1 mol/l
Lipemic index is determined by the turbidity qualitatively with no units.
Table 2
List of interference on chemistry assays by HIL.
Test Hemolysis
interference
at index level
Icterous
interference
at index level
Lipemic
interference
at index level
Na
+
NS NS NS
K
+
Increased (H 200) NS NS
Cl
NS NS NS
CO
2
Decreased (H 130) Decreased (I 14) NS
A1AT NS NS Increased (L 1000)
Alb NS NS NS
Alcohol Decreased(H200) ND ND
ALP Decreased (H 235) NS NS
ALT Increased (H 235) NS Decreased (L 150)
Amm Increased (H 200) Increased (I 1) Decreased (L 50)
AST Increased (H 40) NS Decreased (L 179)
2MI NS NS NS
CA
2+
NS Decreased (I 7) NS
Cerulo NS NS Increased (L 50)
Chol Increased (H 600) NS NS
CK Increased (H 400) Decreased (I 14) Decreased (L 1200)
Creat NS Decreased (I 14) NS
hsCRP NS ND NS
D. Bili Decreased (H 40) NA Increased (L 233)
Fe
2+
Increased (H 40) ND NS
GGT Decreased (H 600) Decreased (I 14) Decreased (L 1200)
Glucose NS NS NS
Haptoglobin Decreased (H 118) NS Increased (L 1200)
HDL NS Decreased (I 25) Decreased (L 1200)
Homocysteine NS NS ND
LD Increased (H 41) NS Decreased (L 1200)
Lipase Increased (H 300) NS NS
Mg
2+
NS NS NS
PreALB Increased (H 400) NS Increased (L 50)
Phos Increased (H 200) NS NS
RHF Decreased (H 200) ND NS
T.Bili Increased (H 80) NA Increased (L 500)
TBICB ND ND Increased (L50)
TP Increased (H741) Decreased(I 14) NS
UIBC Increased (H 50) Decreased (I 3) Decreased (L 180)
Urea NS NS NS
Uric acid NS Decreased (I 25) NS
The index value listed indicates that signicant interference was observed at this level.
NA: not applicable, NS: not signicant, ND: not determined.
1551 J.Z. Ji, Q.H. Meng / Clinica Chimica Acta 412 (2011) 15501553
signicantly at lower indices in our study in comparison with those
listed in Roche package inserts. These assays included ceruloplasmin,
HDL-C, phosphorous, RHF, Total bilirubin, UIBC, and uric acid. Others
were affected by interferents at higher indices such as A1AT, CK, CO
2
,
GGT, and haptoglobin than those indicated by Roche. In addition, we
observed interferences on some assays which have not been reported
by the manufacturer or in the literature. For example, we observed
positive interference by hemoglobin on ALT, lipase, total protein,
potassium, ferrous iron and negative interference on calcium and CK.
3.2. Interferences of hemoglobin, bilirubin, and lipids on immunoassays
As expected, most of the immunoassays were not affected by
hemoglobin, bilirubin, and lipids with only a fewexceptions (Table 3).
Beta HCG and IgG were negatively interfered by bilirubin at I 13 and I
24, respectively. Free T3 level was falsely increased by bilirubin at I 13
while troponin T level was decreased by hemoglobin.
3.3. Interferences of hemoglobin, bilirubin, and lipids on therapeutic
drugs
Signicant interferences on drugs by hemoglobin, bilirubin, and
lipids are listed in Table 4. Acetaminophen levels were falsely
increased by hemoglobin and bilirubin. Digoxin, valproic acid, and
vancomycin levels were decreased by hemoglobin. Bilirubin falsely
increased levels of gentamicin, phenobarbital, and theophylline while
it decreased tobramycin levels. Blood levels of phenytoin, salicylate,
theophylline, and vancomycin were signicantly decreased by lipids.
We also observed that the degree of interference is dependent on the
initial drug concentration.
4. Discussion
The interferences of hemolysis, icterus, or lipemia with various
analytes on Roche Cobas 6000 analyzer were assessed. Our data
indicate that some index values are not fully concordant with those
provided by the manufacturer. Moreover, we demonstrate additional
assay interference ndings not reported by the manufacturer or
published in the literature.
The release of hemoglobin and other cellular components in
hemolysis can have impacts on laboratory results. We observed the
changes (positive or negative) of analytes such as potassium, LDH,
ammonia, AST, ALT, CK, Fe
2+
, acetaminophen, ethanol, either due to
interference by hemoglobin or increased release of the cellular
components from erythrocytes during hemolysis [7,1820]. Hemo-
globin begins to absorb around 340 nm and thus affects non-kinetic
methods based on the absorbance properties of NADH or NADPH
[21,22]. Similarly, bilirubin interference arises from its spectral
properties and its ability to react chemically with other reagents
resulting in decreased analyte values such as creatinine concentra-
tions. Although the enzymatic creatinine assay is reportedly to be less
interfered by bilirubin, negative interference of creatinine assay by
bilirubin was still observed on Roche Cobas 6000. This is consistent
with what was observed on Roche Modular system [17]. This
interference was presumed to be attributed to the consumption of
peroxide in the initial reaction mixture [23]. In lipemic samples,
chylomicrons and VLDL particles scatter light and produce turbidity.
Thus, lipemia may interfere in any assay that uses the transmission of
light as part of the detection scheme [9]. In this study, different
lipemia index had positive or negative effect on measured concen-
trations of various chemistry analytes such as A1AT, ALT, ammonia,
ceruloplasmin, prealbumin, and bilirubin. We did not observe
interference of albumin by lipemia as stated in the Roche package
insert.
For most of the immunoassays analyzed on Cobas, no signicant
interference by hemoglobin, bilirubin, or lipid was observed. This is
expected since the assay is two monoclonal antibodies directed
sandwich chemiluminescent assay with extensive washing and nal
measurement by electrical methods. In comparison with the electro-
immunoassays, interference was observed for some drugs measured
by turbidometric method but there is an improvement from the old
version of Cobas [13]. This is likely due to the similarity of the
methodology used for drugs and chemistry assays. We have also
observed that the degree of interference can be different depending
on the initial analyte concentrations in the specimen. It should be
apparent to the readers that a constant interference in any assay but
measured at either a low or high analyte concentration may result in
different percentage changes.
It should be noted that the in vitro results may not completely
support what would happen in vivo. For example, in vivo hemolysis
may be more complicated than the in vitro addition of hemoglobin. In
vivo hemolysis results in all the intracellular components released
into circulation. However, the incidence of samples with in vivo
hemolysis is extremely low compared with the samples that have
hemolysis introduced during and after the collection procedure. For
simplicity, we used unconjugated bilirubin to mimic icteric samples
instead of both conjugated and unconjugated bilirubin [3]. Future
studies should be conducted to determine if there is a difference in
terms of interference by different bilirubin species. Although
Table 3
List of interferences on immunoassays by HIL.
Test Hemolysis
interference
at index level
Icterus
interference
at Index level
Lipemic
interference
at index level
AFP NS ND ND
HCG NS Decreased (I 13) ND
B12 NS ND ND
C3 NS ND NS
C4 NS ND NS
CEA NS ND ND
Cort NS ND ND
FER NS NS ND
FRT3 NS Increased (I 13) ND
FRT4 NS ND ND
FSH NS ND ND
IgA NS NS NS
IgG NS Decreased (I 24) NS
IgM NS NS NS
LH NS ND ND
Myo NS ND ND
PRL NS ND ND
PSA NS ND ND
TnT Decreased (H 290) ND ND
TSH NS ND NS
The index value listed indicates that signicant interference was observed at this level.
NA: not applicable, NS: not signicant, ND: not determined.
Table 4
List of interferences on drugs by HIL.
Test Hemolysis
interference
at index level
Icterous
interference
at index level
Lipemic
interference
at index level
Acetaminophen Increased (H 40) Increased (I 40) NS
Carbamazepine Inconclusive ND NS
Digoxin Decreased (H 100) Inconclusive NS
Gentamicin NS Increased (I 2) NS
Phenobarbital NS Increased (I 5) NS
Phenytoin NS ND Decreased (L 112)
Salicylate NS NS Decreased (L 100)
Theophylline NS Increased (I 2) Decreased (L 50)
Tobramycin NS Decreased (I 3) NS
Valproic acid Decreased (H 80) ND NS
Vancomycin Decreased (H 600) NS Decreased (L 50)
The index value listed indicates that signicant interference was observed at this level.
NA: not applicable, NS: not signicant, ND: not determined.
1552 J.Z. Ji, Q.H. Meng / Clinica Chimica Acta 412 (2011) 15501553
Intralipid is the universally acceptable substance used for in vitro
interference study, it may not be completely comparable with in vivo
lipemic samples [24]. Intralipid is not the same as VLDL and
chylomicrons in terms of the spectrum of particle size. Lipemic
specimens are far more complex than Intralipid and there is a poor
relationship between triglyceride concentration and the lipemic
indices of absorbance. Thus, the heterogenous lipemic sample cannot
be easily simulated by addition of Intralipid[9]. Therefore the degree
of interference observed may not exactly represent what would occur
endogeneously [18]. Nevertheless, these data still provide useful
information as to the degree of analyte change inuenced by
hemolysis, icterus, and lipemia.
Generally, the visual judgment on these factors is variable and not
reliable which can affect the interpretation on the degree of
interference [3,25,26]. With implementation of automation and high
demand for fast turnaround time, it is not practical to inspect the
samples visually. We believe the implementation of HIL indices on the
Cobas 6000 will greatly improve the accuracy and the quality of the
test results. Comments based on our data will be added to results
when the interference is clinically signicant to help clinicians
interpret the results appropriately.
In conclusion, this study has produced useful information on
analyte interference by hemoglobin, bilirubin, and lipids on Roche
Cobas 6000. The use of the cut-off indices in the instrument instead of
visual inspection will provide more accurate information as to which
analyte is to be affected and to what degree. This automatic detection
system and corresponding comment on potential interference will
reduce error when reporting results.
Acknowledgments
We thank Ms. Lana Link, Ms. Cheryl Booth, and Ms. Cheryl Brenaut
for their technical assistance. We also thank Dr. John Krahn for
reviewing the manuscript.
References
[1] Carraro P, Plebani M. Errors in a stat laboratory: types and frequencies 10 years
later. Clin Chem 2007;53:133842.
[2] Lippi G. Governance of preanalytical variability: travelling the right path to the
bright side of the moon? Clin Chim Acta 2009;404:326.
[3] Kroll MH, Elin RJ. Interferences with clinical laboratory analyses. Clin Chem
1994;40:19962005.
[4] Dimeski G. Interference testing. Clin Biochem Rev 2008;29(Suppl 1):S438.
[5] Ryder KW, Glick MR. Erroneous laboratory results from hemolyzed, icteric, and
lipemic specimens. Clin Chem 1993;39:1756.
[6] Glick MR, Ryder KW, Jackson SA. Graphical comparisons of interferences in clinical
chemistry instrumentation. Clin Chem 1986;32:4704.
[7] Jay DW, Provasek D. Characterization and mathematical correction of hemolysis
interference in selected Hitachi 717 assays. Clin Chem 1993;39:180410.
[8] Steen G, Vermeer HJ, Naus AJ, Goevaerts B, Agricola PT, Schoenmakers CH.
Multicenter evaluation of the interference of hemoglobin, bilirubin and lipids on
Synchron LX-20 assays. Clin Chem Lab Med 2006;44:4139.
[9] Kroll MH. Evaluating interference caused by lipemia. Clin Chem 2004;50:19689.
[10] Vermeer HJ, Thomassen E, de Jonge N. Automated processing of serum indices
used for interference detection by the laboratory information system. Clin Chem
2005;51:2447.
[11] Vermeer HJ, Steen G, Naus AJ, Goevaerts B, Agricola PT, Schoenmakers CH.
Correction of patient results for Beckman Coulter LX-20 assays affected by
interference due to hemoglobin, bilirubin or lipids: a practical approach. Clin
Chem Lab Med 2007;45:1149.
[12] Simundic AM, Nikolac N, Ivankovic V, et al. Comparsion of visual vs. automated
detection of lipemic, icteric and hemolyzed specimens: can we rely on a human
eye? Clin Chem Lab Med 2009;47:13615.
[13] Ryder KW, Trandle DS, Bode MA, Cole RE, Moorehead WR, Glick MR. Effects of
hemolysis, icterus, and lipemia on automated immunoassays. Clin Chem 1991;37:
11345.
[14] Brady J, O'Leary N. Interference due to lipaemia in routine photometric analysis
survey of an underrated problem. Ann Clin Biochem 1994;31:2818.
[15] Bossuyt X, Blanckaert N. Evaluation of interferences in rate and xed-time
nephelometric assays of specic serum proteins. Clin Chem 1999;45:627.
[16] Randall AG, Garcia-Webb P, Beilby JP. Interference by haemolysis, icterus and
lipaemia in assays on the Beckman Synchron CX5 and methods for correction. Ann
Clin Biochem 1990;27:34552.
[17] Owen LJ, Keevil BG. Does bilirubin cause interference in Roche creatinine
methods? Clin Chem 2007;53:3701.
[18] Dimeski G. Effects of hemolysis on the Roche ammonia method for Hitachi
analyzers. Clin Chem 2004;50:9767.
[19] Lippi G, Salvagno GL, Montagnana M, Brocco G, Guidi GC. Inuence of hemolysis
on routine clinical chemistry testing. Clin Chem Lab Med 2006;44:3116.
[20] Hawkins RC. Correction and reporting of potassium results in haemolysed
samples. Ann Clin Biochem 2006;43:889.
[21] da Fonseca-Wollheim F. Haemoglobin interference in the bichromatic spectro-
photometry of NAD(P)H at 340/380 nm. Eur J Clin Chem Clin Biochem 1993;31:
595601.
[22] Sodi R, Darn SM, Davison AS, Stott A, Shenkin A. Mechanism of interference by
haemolysis in the cardiac troponin T immunoassay. Ann Clin Biochem 2006;43:
4956.
[23] van't Sant P, Kreutzer HJ. Articial icteric plasmas: unreliable indicators for
interference with creatinine assay on Beckman CX3. Clin Chem 1995;41:17734.
[24] Bornhorst JA, Roberts RF, Roberts WL. Assay-specic differences in lipemic
interference in native and intralipid-supplemented samples. Clin Chem 2004;50:
2197201.
[25] Hawkins R. Discrepancy between visual and spectrophotometric assessment of
sample haemolysis. Ann Clin Biochem 2002;39:5212.
[26] Glick MR, Ryder KW, Glick SJ, Woods JR. Unreliable visual estimation of the
incidence and amount of turbidity, hemolysis, and icterus in serum from
hospitalized patients. Clin Chem 1989;35:8379.
1553 J.Z. Ji, Q.H. Meng / Clinica Chimica Acta 412 (2011) 15501553
Das könnte Ihnen auch gefallen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- Genotype - Accupower Jak2Dokument34 SeitenGenotype - Accupower Jak2Elvan Dwi WidyadiNoch keine Bewertungen
- Menejeman LabDokument3 SeitenMenejeman LabElvan Dwi WidyadiNoch keine Bewertungen
- (Concentration: 30ng/ul Purity A260/280: 1.7) : Sample: Blood EDTA PCR PreparationDokument2 Seiten(Concentration: 30ng/ul Purity A260/280: 1.7) : Sample: Blood EDTA PCR PreparationElvan Dwi WidyadiNoch keine Bewertungen
- (Concentration: 30ng/ul Purity A260/280: 1.7) : Sample: Blood EDTA PCR PreparationDokument2 Seiten(Concentration: 30ng/ul Purity A260/280: 1.7) : Sample: Blood EDTA PCR PreparationElvan Dwi WidyadiNoch keine Bewertungen
- LaboratoryQualityStandardsandtheirImplementation 9C27 PDFDokument74 SeitenLaboratoryQualityStandardsandtheirImplementation 9C27 PDFElvan Dwi WidyadiNoch keine Bewertungen
- BV Blue TestDokument4 SeitenBV Blue TestElvan Dwi WidyadiNoch keine Bewertungen
- QC Levey-Jenning WestgurdDokument8 SeitenQC Levey-Jenning WestgurdElvan Dwi WidyadiNoch keine Bewertungen
- From The Sample To The Reading, Indirect ELISA Protocol: Buffer PreparationDokument2 SeitenFrom The Sample To The Reading, Indirect ELISA Protocol: Buffer PreparationElvan Dwi WidyadiNoch keine Bewertungen
- Rencana Pelaksanaan PembelajaranDokument5 SeitenRencana Pelaksanaan PembelajaranElvan Dwi WidyadiNoch keine Bewertungen
- Wesgardrules & MultirulesDokument18 SeitenWesgardrules & MultirulesElvan Dwi WidyadiNoch keine Bewertungen
- Glycatedalbumin (GA)Dokument22 SeitenGlycatedalbumin (GA)Elvan Dwi WidyadiNoch keine Bewertungen
- Performance of Bvblue Rapid Test in Detecting Bacterial Vaginosis Among Women in Mysore, IndiaDokument1 SeitePerformance of Bvblue Rapid Test in Detecting Bacterial Vaginosis Among Women in Mysore, IndiaElvan Dwi Widyadi100% (1)
- Pgl1 Penanda Lepra: Elvan, DR/ DR - Puspa Wardani,. Dr. SP - PKDokument8 SeitenPgl1 Penanda Lepra: Elvan, DR/ DR - Puspa Wardani,. Dr. SP - PKElvan Dwi WidyadiNoch keine Bewertungen
- Hem Lab6 RBC Morph Incl F12Dokument7 SeitenHem Lab6 RBC Morph Incl F12Elvan Dwi WidyadiNoch keine Bewertungen
- UrinalysisDokument10 SeitenUrinalysisElvan Dwi WidyadiNoch keine Bewertungen
- Phosphate BishopDokument3 SeitenPhosphate BishopElvan Dwi WidyadiNoch keine Bewertungen
- Sysmex Ca 600Dokument4 SeitenSysmex Ca 600Elvan Dwi WidyadiNoch keine Bewertungen
- Opini: Everybody in This World Has Dream. Whoever They Are, Wherever They Are FromDokument3 SeitenOpini: Everybody in This World Has Dream. Whoever They Are, Wherever They Are FromElvan Dwi WidyadiNoch keine Bewertungen
- Akut AbdomenDokument24 SeitenAkut AbdomenEka100% (1)
- Test Detail: Tes Tariff Registration Date Test Start Date Start Time Finish TimeDokument1 SeiteTest Detail: Tes Tariff Registration Date Test Start Date Start Time Finish TimeElvan Dwi WidyadiNoch keine Bewertungen
- Head & Brain Trauma: EMS Professions Temple CollegeDokument77 SeitenHead & Brain Trauma: EMS Professions Temple CollegeElvan Dwi WidyadiNoch keine Bewertungen
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- DM GestasionalDokument39 SeitenDM GestasionalAnggi CalapiNoch keine Bewertungen
- Obladen 2010Dokument7 SeitenObladen 2010Barna ÉvaNoch keine Bewertungen
- Dialog Interview A Foreigner at Sanur BeachDokument4 SeitenDialog Interview A Foreigner at Sanur BeachYugek AgungNoch keine Bewertungen
- Pathogenesis of Non-Hodgkin's Lymphoma: Ournal of Linical NcologyDokument9 SeitenPathogenesis of Non-Hodgkin's Lymphoma: Ournal of Linical NcologyZullymar CabreraNoch keine Bewertungen
- Journal ReadingDokument17 SeitenJournal ReadingDesty Friska KurniaNoch keine Bewertungen
- RN or BSRN or Case Manager or Manager Health Care or Dom or DireDokument4 SeitenRN or BSRN or Case Manager or Manager Health Care or Dom or Direapi-76957346Noch keine Bewertungen
- Management of Infiltration / Extravasation of Intravenous Iron TherapyDokument2 SeitenManagement of Infiltration / Extravasation of Intravenous Iron TherapyElaine BriososNoch keine Bewertungen
- Behavior Guidance For The Pediatric Dental Patient: Latest RevisionDokument19 SeitenBehavior Guidance For The Pediatric Dental Patient: Latest RevisionsinghsenoNoch keine Bewertungen
- Anti-Isthmus Autoimmunity in A Novel Feline Acquired Alopecia Resembling Pseudopelade of HumansDokument10 SeitenAnti-Isthmus Autoimmunity in A Novel Feline Acquired Alopecia Resembling Pseudopelade of HumansjenNoch keine Bewertungen
- Doctor of Physical Therapy (DPT) : Semester 1Dokument5 SeitenDoctor of Physical Therapy (DPT) : Semester 1M Irfan Bhutta100% (1)
- Registered NursingDokument11 SeitenRegistered Nursingapi-79193193Noch keine Bewertungen
- Craniopharyngioma-The Anesthetic, Critical Care and Syrgical Challenges in The Managemnet of CPNDokument5 SeitenCraniopharyngioma-The Anesthetic, Critical Care and Syrgical Challenges in The Managemnet of CPNpitriaNoch keine Bewertungen
- Buttonhole DislocationDokument13 SeitenButtonhole DislocationHanif Andhika WardhanaNoch keine Bewertungen
- 2008 Annual Report-From ILAEDokument113 Seiten2008 Annual Report-From ILAEarmythunder100% (2)
- International Dental and Medical Journal of Advanced ResearchDokument5 SeitenInternational Dental and Medical Journal of Advanced ResearchShirmayne TangNoch keine Bewertungen
- Chapter 14 Intravenous Solutions, Equipment, and CalculationsDokument48 SeitenChapter 14 Intravenous Solutions, Equipment, and CalculationsEn RasNoch keine Bewertungen
- Anaesthesia & Intensive Care Medicine Volume 11 Issue 4 2010 (Doi 10.1016/j.mpaic.2009.12.013) Ben Shelley Nick Sutcliffe - Total Intravenous AnaesthesiaDokument3 SeitenAnaesthesia & Intensive Care Medicine Volume 11 Issue 4 2010 (Doi 10.1016/j.mpaic.2009.12.013) Ben Shelley Nick Sutcliffe - Total Intravenous AnaesthesiamikhatiarNoch keine Bewertungen
- 50 Writing Task + Sample Letters PDFDokument173 Seiten50 Writing Task + Sample Letters PDFIrina Alexandra83% (86)
- ISBI Guidelines 1Dokument69 SeitenISBI Guidelines 1Mary Marlene López RamírezNoch keine Bewertungen
- Resume Joey Takemasa Russell RRT RCPDokument3 SeitenResume Joey Takemasa Russell RRT RCPapi-625837402Noch keine Bewertungen
- CP 1102 Assignment 1Dokument2 SeitenCP 1102 Assignment 1api-329210848Noch keine Bewertungen
- Elevated Risk of Blood Clots in Women Taking Birth Control Containing Drospirenone, Study ShowsDokument3 SeitenElevated Risk of Blood Clots in Women Taking Birth Control Containing Drospirenone, Study ShowsJomerlyn AbaricoNoch keine Bewertungen
- Intrathecal Antibiotic Administration Procedure (Paediatrics)Dokument9 SeitenIntrathecal Antibiotic Administration Procedure (Paediatrics)Abu Azzam Al-HadiNoch keine Bewertungen
- Benumof and Hagberg's Airway Management (2012, Saunders) PDFDokument1.314 SeitenBenumof and Hagberg's Airway Management (2012, Saunders) PDFmodreanu andreiNoch keine Bewertungen
- What Is Basic IV Therapy TrainingDokument6 SeitenWhat Is Basic IV Therapy TrainingJay Depakakibo GallardoNoch keine Bewertungen
- Coahman Galip Gurel Et Al 2012Dokument12 SeitenCoahman Galip Gurel Et Al 2012Jorge RojasNoch keine Bewertungen
- Nursing ResumeDokument1 SeiteNursing Resumeapi-545447839Noch keine Bewertungen
- Newborn Assesment in The Delivery RoomDokument9 SeitenNewborn Assesment in The Delivery RoomHaRoIsGaNoch keine Bewertungen
- Collet Gps DNP FNP Copy With Grades Blacked OutDokument4 SeitenCollet Gps DNP FNP Copy With Grades Blacked Outapi-251653314Noch keine Bewertungen
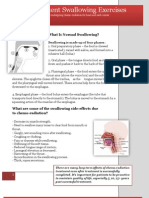
- Pre-Treatment Swallowing ExercisesDokument6 SeitenPre-Treatment Swallowing Exercisesshaghala38100% (4)