Beruflich Dokumente
Kultur Dokumente
158 Full
Hochgeladen von
rantiadrianiOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
158 Full
Hochgeladen von
rantiadrianiCopyright:
Verfügbare Formate
158 Am J Clin Pathol 2007;128:158-163
158 DOI: 10.1309/69A5AV266W230AUA
American Society for Clinical Pathology
Clinical Chemistry / THYROID HORMONES IN AMNIOTIC FLUID
Establishment of Reference Intervals for Thyroid-
Stimulating Hormone and Free Thyroxine in Amniotic Fluid
Using the Bayer ADVIA Centaur
Nikola A. Baumann, PhD
*
, and Ann M. Gronowski, PhD
Key Words: Reference intervals; Thyroid hormone; Amniotic fluid
DOI: 10.1309/69A5AV266W230AUA
A b s t r a c t :
Thyroid hormone concentrations in amniotic fluid
(AF) can aid in the diagnosis of fetal hypothyroidism.
However, the availability of reference intervals for
thyroid hormones in AF using current methods is
limited. The purpose of this study was to validate the
Bayer ADVIA Centaur (Bayer Healthcare, Tarrytown,
NY) third-generation thyroid-stimulating hormone
(TSH), total thyroxine (T
4
), and free thyroxine (FT
4
)
assays for use with AF and establish reference intervals
for these thyroid hormones in AF.
Third-trimester AF samples were assayed for TSH,
T
4
, and FT
4
. Reference intervals were calculated using
nonparametric analyses. The reference intervals were
as follows: TSH (n = 131), 0.04 to 0.51 IU/mL (0.04-
0.51 mIU/L), with a median of 0.10 IU/mL (0.10
mIU/L); FT
4
(n = 133), less than 0.10 to 0.77 ng/dL
(1.29-9.93 pmol/L), with a median of 0.26 ng/dL (3.35
pmol/L). T
4
in AF was undetectable using the Centaur
assay, reinforcing the importance of validating different
manufacturers immunoassays for use with nonserum
specimens. These reference intervals represent the first
study performed to date using a third-generation TSH
assay and a sensitive FT
4
assay.
When thyroid disease arises during pregnancy, the moth-
er can be evaluated by using maternal serum. Evaluation of the
fetus for suspected thyroid abnormalities is far more difficult.
Proper fetal diagnosis is especially important because thyroid
disorders can lead to growth retardation and possible intrauter-
ine death. Approximately 3% of fetal thyroid disorders are
associated with goiter,
1
which can cause neck dystocia and
respiratory distress.
Evaluating thyroid abnormalities in the fetus is clinically
difficult; yet, because in utero treatment is available, proper
diagnosis is important. Although a fetal goiter can be distin-
guished during ultrasound examination, ultrasound does not
differentiate between hyperthyroidism and hypothyroidism.
For knowledge of specific thyroid derangements, direct sam-
pling of cord blood in utero can be performed, but percuta-
neous umbilical blood sampling is a technically demanding
procedure that poses risk of fetal bradycardia, hemorrhage,
and possible fetal death.
2
Although the risk of fetal death is
low, reported as 0.5% to 1% or less,
3
amniocentesis is a far
simpler and safer procedure, with less risk to the fetus.
Studies indicate that from the 12th week of gestation
onward, there are steady increases in the fetal serum concen-
trations of total thyroxine (T
4
), free thyroxine (FT
4
), thyroid-
stimulating hormone (TSH), and thyroxine-binding globu-
lin.
2,4
The increasing concentrations of fetal cord blood thy-
roid hormones correlate with increasing concentrations of
amniotic fluid TSH and FT
4
.
5
Furthermore, these increases are
independent of maternal concentrations of the same hor-
mones, indicating that there is no significant transplacental
passage of TSH and thyroxine.
2,5
A previous study from this laboratory established refer-
ence intervals for thyroid hormones in amniotic fluid using the
Am J Clin Pathol 2007;128:158-163 159
159 DOI: 10.1309/69A5AV266W230AUA 159
American Society for Clinical Pathology
Clinical Chemistry / ORIGINAL ARTICLE
Abbott AxSym method for TSH, total T
4
, and FT
4
.
6
In that
study, clinical case reports from the literature were also exam-
ined, and in all cases of fetal hypothyroidism, the amniotic
fluid TSH and FT
4
concentrations were outside the newly
established reference intervals. The data supported the use of
thyroid hormone concentrations in amniotic fluid as an aid in
the diagnosis of fetal hypothyroidism.
However, that study was limited because it used a second-
generation TSH assay. Today, more sensitive third-generation
TSH assays and more sensitive FT
4
assays are routinely used.
Furthermore, it is important to establish and/or validate refer-
ence intervals when changing assay methods or when assess-
ing different patient populations. Recent studies comparing
automated TSH methods have shown that TSH methods do
not provide comparable results across the analytic measure-
ment range.
7
The present study expands on previous studies,
establishing amniotic fluid reference intervals for TSH, T
4
,
and FT
4
using more sensitive assays on the ADVIA Centaur
automated immunoassay analyzer (Bayer Healthcare,
Tarrytown, NY).
Materials and Methods
Samples
Leftover amniotic fluid samples sent to the Barnes-
Jewish Hospital chemistry laboratory (St Louis, MO) for fetal
lung maturity testing from July 2002 through November 2005
and stored at 20C were used. All samples were visually
inspected for the presence of blood. Amniotic fluid specimens
with visible blood contamination were excluded from the
study.
The study was approved by the Washington University
Human Studies Committee (St Louis).
Validation of Assays for Use With Amniotic Fluid
The ADVIA Centaur automated immunoassay analyzer
was used for all thyroid hormone measurements. The ADVIA
TSH assay is a sandwich immunoassay with an analytic sen-
sitivity of 0.02 IU/mL (0.02 mIU/L). The ADVIA total T
4
assay is a competitive immunoassay with an analytic sensitiv-
ity of 0.3 g/dL (3.87 nmol/L). The ADVIA FT
4
assay is a
competitive immunoassay with an analytic sensitivity of 0.1
ng/dL (1.29 pmol/L). This assay cannot be diluted, and our
laboratory has established linearity in plasma to 4.5 ng/dL
(58.1 pmol/L).
Validation of the ADVIA Centaur thyroid hormone
assays using amniotic fluid as a matrix was performed. Serum
(20% vol/vol) with a known concentration of TSH, total T
4
, or
FT
4
was added to amniotic fluid and then serially diluted
using amniotic fluid with a low concentration of TSH, total T
4
,
or FT
4
as the diluent. Samples were analyzed in duplicate, and
recovery was calculated. As expected, because of binding pro-
tein equilibrium, FT
4
cannot be accurately recovered from
diluted specimens.
Precision Studies
Intra-assay precision for the TSH and FT
4
assays was
determined by measuring amniotic fluid samples 10 consecu-
tive times. Precision was established at 2 concentrations of
TSH and FT
4
.
Analyte Stability Studies
The effect of the freeze-thaw process on the stability of
TSH and FT
4
was determined by measuring the hormones in
9 specimens before (within 24 hours of collection) and after
freezing (24-48 hours) at 20C. Results obtained before and
after freezing were analyzed by using the t test. A P value of
less than .05 was considered statistically significant.
Calculation of Reference Intervals
Thorough chart review for all cases was performed, and
samples were excluded if there was a patient history of fetal
anomalies, multiple gestations, or any evidence of thyroid dis-
ease. Gestational ages ranged from 31.3 to 39.4 weeks (medi-
an, 36 weeks). The level of TSH was measured in 131 speci-
mens and of FT
4
in 133 specimens.
Central 95% reference intervals for TSH and FT
4
were
calculated using nonparametric analysis.
8
This process ranks
data according to numeric value and calculates percentiles as
a function of these ranked numbers. The concentrations at the
2.5 and 97.5 percentiles provide an estimate of the central
95% reference interval for the population based on the given
set of data.
Results
The ADVIA Centaur TSH assay was validated for use
with amniotic fluid. Recovery of serum TSH diluted in amni-
otic fluid was measured at 12 concentrations in the range
between 0.08 IU/mL (0.08 mIU/L) and 37 IU/mL (37
mIU/L) Figure 1A. The TSH concentrations (IU/mL; to
convert to mIU/L, multiply by 1.0) and percentages of recov-
ery were as follows, respectively: 0.08 and 103%; 0.10 and
111%; 0.13 and 127%; 0.21 and 110%; 0.35 and 109%; 0.64
and 108%; 1.22 and 88%; 2.37 and 86%; 4.68 and 78%; 9.30
and 84%; 18.50 and 94%; and 37.00 and 89%. The mean SD
recovery of serum TSH was 99% 15%.
Recovery of serum T
4
when diluted in amniotic fluid is
shown in Figure 1B. The T
4
concentrations (g/dL; to con-
vert to nmol/L, multiply by 12.9) and percentages of recovery
were as follows, respectively: 0.9 and 182%; 1.8 and 164%,
160 Am J Clin Pathol 2007;128:158-163
160 DOI: 10.1309/69A5AV266W230AUA
American Society for Clinical Pathology
Baumann and Gronowski / THYROID HORMONES IN AMNIOTIC FLUID
2.7 and 158%; 3.5 and 159%; and 4.3 and 158%. The mean
SD recovery of serum T
4
was 164% 10%. The recovery data
demonstrated overrecovery of T
4
, suggesting that the amniot-
ic fluid matrix may affect the measurement of serum T
4
with
this assay.
FT
4
in serum (20% vol/vol) was diluted into amniotic
fluid, and FT
4
recovery was measured using the ADVIA
Centaur assay. FT
4
recovery was 195% in this sample. This
indicates that serum FT
4
can be recovered and measured in
amniotic fluid using the ADVIA Centaur assay. However, as
expected, detailed recovery studies could not be performed
because FT
4
cannot be diluted and accurately measured owing
to the effect of dilution on binding protein equilibrium.
9
Intra-assay precision studies were performed on patient
amniotic fluid samples. The coefficients of variation (CVs) for
the TSH and FT
4
assays were less than 10%. The intra-assay
CVs for the TSH assay in 10 samples were 9.0% and 4.1% at
TSH concentrations of 0.06 IU/mL (0.06 mIU/L) and 0.11
IU/mL (0.11 mIU/L), respectively. The FT
4
assay had intra-
assay CVs of 9.6% and 9.8% at FT
4
concentrations of 0.34
ng/dL (4.39 pmol/L) and 0.52 ng/dL (6.71 pmol/L), respec-
tively (n = 10).
It is interesting that we found that T
4
could not be detect-
ed in any patient amniotic fluid samples using the ADVIA
Centaur method. This did not seem to be simply a matrix
effect because serum T
4
could be diluted into amniotic fluid
and measured (Figure 1B). This inability to measure amniotic
fluid T
4
was further confirmed with 10 amniotic fluid samples
that were measured using the ADVIA Centaur and the Abbott
AxSym (Abbott Laboratories, Abbott Park, IL) instruments.
Amniotic fluid samples analyzed using the Abbott AxSym
total T
4
assay gave results between 1.8 and 2.9 g/dL (23.22-
37.41 nmol/L), whereas all samples had undetectable T
4
(<1.0
g/dL [12.9 nmol/L]) by the ADVIA Centaur assay.
Amniotic fluid specimens were stored frozen (20C)
before analysis. Thus, it was necessary to determine the stabil-
ity of TSH and FT
4
after specimens were subjected to a
freeze-thaw cycle. We analyzed 9 amniotic fluid specimens
before (within 24 hours of collection) and after freezing (24-
48 hours) at 20C. A single freeze-thaw cycle did not have a
significant effect on thyroid hormone concentrations in amni-
otic fluid. Postfreeze TSH and FT
4
results were within a mean
SD of 21% 27% (P = .22) and 11% 21% (P = .08) of
the original result, respectively Figure 2.
Samples from 133 patients met selection criteria (see the
Materials and Methods section) and were subsequently
thawed and assayed immediately for TSH and FT
4
(not all
samples could have both assays performed owing to insuffi-
cient sample volume). The frequencies of amniotic fluid TSH
and FT
4
results are shown in Figure 3. Central 95% refer-
ence intervals, median values, and ranges for TSH and FT
4
were calculated and are given in Table 1.
0
10
20
30
40
0 10 20
Expected TSH (IU/mL)
O
b
s
e
r
v
e
d
T
S
H
(
I
U
/
m
L
)
30 40
3
2
1
0
0 1 2 3
0
1
2
3
4
5
6
7
0 1 2 3 4 5 6 7
Expected T
4
(g/dL)
O
b
s
e
r
v
e
d
T
4
(
g
/
d
L
)
A B
Figure 1 Recovery of serum thyroid-stimulating hormone (TSH; A) and total thyroxine (T
4
; B) in amniotic fluid. Serum (20%
vol/vol) with a known concentration of TSH or T
4
was added to amniotic fluid and then serially diluted using amniotic fluid as the
diluent. The dashed line represents a slope of 1. A, Slope = 0.909; y-intercept = 0.012. Inset graph shows recovery in the low
TSH concentration range. Samples were analyzed in duplicate. B, Slope = 1.518; y-intercept = 0.22. Values are given in
conventional units; to convert to Systme International units, for TSH (mIU/L), multiply by 1.0; for T
4
(nmol/L), multiply by 12.9.
Am J Clin Pathol 2007;128:158-163 161
161 DOI: 10.1309/69A5AV266W230AUA 161
American Society for Clinical Pathology
Clinical Chemistry / ORIGINAL ARTICLE
Discussion
We established normal amniotic fluid reference intervals
for TSH and FT
4
. To our knowledge, this is the first study that
has established these reference intervals using the ADVIA
Centaur automated immunoassay analyzer. Our reference
intervals for TSH and FT
4
compare favorably with those in
previous third-trimester studies.
5,6,10-12
An interesting and unexpected result from this study was
the observation that amniotic fluid T
4
cannot be measured
using the ADVIA Centaur. This does not seem to be simply a
0.0
0.1
0.2
0.3
0.4
0.5
TSH Prefreeze TSH Postfreeze
T
S
H
(
I
U
/
m
L
)
0.00
0.25
0.50
0.75
1.00
1.25
FT
4
Prefreeze FT
4
Postfreeze
F
T
4
(
n
g
/
d
L
)
0
.
0
2
0
.
0
6
0
.
1
0
0
.
1
4
0
.
1
8
0
.
2
2
0
.
2
6
0
.
3
0
0
.
3
4
0
.
3
8
0
.
4
2
0
.
4
6
0
.
5
0
0
.
5
4
0
.
5
8
0
.
6
2
0
.
6
6
0
.
7
0
0
.
7
4
0
.
7
8
0
.
8
2
0
.
8
6
0
.
9
0
0
.
9
4
0
.
9
8
TSH (IU/mL)
0
10
20
30
40
F
r
e
q
u
e
n
c
y
0
.
1
0
0
.
1
5
0
.
2
0
0
.
2
5
0
.
3
0
0
.
3
5
0
.
4
0
0
.
4
5
0
.
5
0
0
.
5
5
0
.
6
0
0
.
6
5
0
.
7
0
0
.
7
5
0
.
8
0
0
.
8
5
0
.
9
0
0
.
9
5
1
.
0
0
1
.
0
5
1
.
1
0
1
.
1
5
1
.
2
0
1
.
2
5
1
.
3
0
FT
4
(ng/dL)
0
5
10
15
20
25
30
F
r
e
q
u
e
n
c
y
A B
A B
Figure 2 Stability of thyroid-stimulating hormone (TSH; A) and free thyroxine (FT
4
; B) in amniotic fluid. Amniotic fluid
specimens were assayed for TSH (A) and FT
4
(B) before (within 24 hours of collection) and after freezing (24-48 hours) at 20C.
Values are given in conventional units; to convert to Systme International units, for TSH (mIU/L), multiply by 1.0; for free T
4
(pmol/L), multiply by 12.9.
Figure 3 Frequencies of amniotic fluid thyroid hormone results. A, Thyroid-stimulating hormone (TSH). B, Free thyroxine (FT
4
).
Values are given in conventional units; to convert to Systme International units, for TSH (mIU/L), multiply by 1.0; for free T
4
(pmol/L), multiply by 12.9.
Table 1
Reference Intervals for TSH and Free T
4
Calculated Using Nonparametric Analysis
*
No. of Samples Median Range Central 95% Reference Interval
TSH (IU/mL) 131 0.10 0.05-0.99 0.04-0.51
Free T
4
(ng/dL) 133 0.26 <0.1-1.31 <0.10-0.77
TSH, thyroid-stimulating hormone; T
4
, thyroxine.
*
Values are given in conventional units; to convert to Systme International units, for TSH (mIU/L), multiply by 1.0; for free T
4
(pmol/L), multiply by 12.9.
162 Am J Clin Pathol 2007;128:158-163
162 DOI: 10.1309/69A5AV266W230AUA
American Society for Clinical Pathology
Baumann and Gronowski / THYROID HORMONES IN AMNIOTIC FLUID
matrix effect of the fluid because serum with a known concen-
tration of total T
4
can be recovered when diluted into amniot-
ic fluid (recovery approximately 160%). Furthermore, 10
samples with undetectable total T
4
concentrations using the
ADVIA Centaur had detectable T
4
concentrations using the
Abbott AxSym analyzer. This observation could be the result
of failure of the protein binding inhibitor used in the Centaur
assay to release T
4
bound to proteins in amniotic fluid. This
observation highlights the potential obstacles in adapting
assays for use with a novel matrix such as amniotic fluid. It
also emphasizes the importance of validating reference inter-
vals before transferring them between platforms and methods.
In addition, because of the inability to dilute FT
4
specimens
and perform in-depth validation experiments and the lack of
information available on T
4
binding capacity in amniotic fluid
or available information on amniotic fluid binding proteins,
we believe that TSH is the preferred analyte for assessment of
thyroid status in amniotic fluid when using the ADVIA
Centaur immunoassay platform. Total T
4
has been shown to
be a useful analyte using other manufacturers assays; howev-
er, all assays must be validated before specimens are analyzed
for diagnostic purposes. It is also possible that the ADVIA
Centaur T
4
assay does not recognize sulfated T
4
, which is a
metabolite of T
4
in neonates and amniotic fluid.
13,14
This find-
ing reinforces the importance of validating different manufac-
turers immunoassays for use with nonserum specimens.
The samples used in this study were obtained from a pop-
ulation of women undergoing amniocentesis for fetal lung
maturity analysis and do not necessarily represent a normal
healthy population. Although this is important, we believe that
the reference intervals are still useful as supported by a previ-
ous study.
6
Six specimens from women with known thyroid
anomalies were excluded. All 6 of these women had amniotic
fluid TSH and FT
4
concentrations within our established refer-
ence intervals and, to the best of our knowledge, delivered nor-
mal infants. This supports the idea that amniotic fluid thyroid
hormone concentrations reflect the thyroid status of the fetus
and are independent of the thyroid status of the mother.
A potential limitation of the present study is that amniot-
ic fluid specimens were stored frozen for up to 3 years before
analysis. However, we demonstrated that TSH and FT
4
were
not significantly affected by a freeze-thaw cycle (Figure 2).
Previous studies
15
have also reported that TSH and thyroxine
are stable in serum for up to 5 years at 20C, lending support
to the idea that these analytes are stable in specimens stored
frozen for extended periods.
The usefulness of thyroid hormone measurement in
amniotic fluid lies in its use not only for diagnosis but also for
management. Many authors
16-19
have treated cases of fetal
hypothyroid goiter with intrauterine thyroxine, using ultra-
sound examination to show regression of fetal goiter and
amniotic fluid to show decreases in amniotic fluid TSH and/or
increases in amniotic fluid T
4
levels. Application of our amni-
otic fluid reference intervals may provide a more relevant end
point for therapy than ultrasound monitoring because the
regression of goiter does not necessarily confer a euthyroid
state. In addition, amniotic fluid is a simple sample to obtain
in this setting because it is readily accessible when intrauter-
ine thyroxine injections are performed.
Fetal hyperthyroidism is currently diagnosed through
findings of fetal goiter, tachycardia, advanced bone matura-
tion, and abnormal cord serum testing. The usefulness of the
amniotic fluid thyroid hormone reference ranges for diagnos-
ing hyperthyroidism is unclear. We know of no published case
reports of fetal hyperthyroidism in which amniotic fluid thy-
roid tests were performed. It is not possible to know how use-
ful amniotic fluid TSH and FT
4
measurements are in diagnos-
ing and monitoring fetal hyperthyroidism until several cases
of fetal hyperthyroidism are reported.
We established reference intervals for TSH and FT
4
in
third-trimester amniotic fluid by using the ADVIA Centaur.
This work expands on previous studies demonstrating that ref-
erence intervals for thyroid hormones (especially TSH) in
amniotic fluid can be of significant use in the diagnosis and
management of fetal hypothyroidism, reducing the need for
umbilical blood sampling.
From the Department of Pathology and Immunology, Division of
Laboratory Medicine, Washington University School of Medicine
St Louis, MO,
Supported in part by Bayer Healthcare by supplying reagents.
Presented in part at the American Association for Clinical
Chemistry Annual Meeting; July 2005; Orlando, FL.
Address reprint requests to Dr Baumann: Dept of Pathology,
University of Illinois Medical Center at Chicago, 840 S Wood St,
Room 201-G, Chicago, IL 60612.
Acknowledgment: We thank Mark H. Wener, MD, University
of Washington, Seattle, for testing samples using the Abbott AxSym
instrument.
*
Dr Baumann is currently with the Department of Pathology,
University of Illinois Medical Center at Chicago.
References
1. Volumenie JL, Polak M, Guibourdenche J, et al. Management
of fetal thyroid goiters: a report of 11 cases in a single perinatal
unit. Prenat Diagn. 2000;20:799-806.
2. Thorpe-Beeston JG, Nicolaides KH, McGregor AM. Fetal
thyroid function. Thyroid. 1992;2:207-217.
3. Daffos F. Fetal blood sampling. Annu Rev Med. 1989;40:319-
329.
4. Ballabio M, Nicolini U, Jowett T, et al. Maturation of thyroid
function in normal human foetuses. Clin Endocrinol (Oxf).
1989;31:565-571.
5. Yoshida K, Sakurada T, Takahashi T, et al. Measurement of
TSH in human amniotic fluid: diagnosis of fetal thyroid
abnormality in utero. Clin Endocrinol (Oxf). 1986;25:313-318.
Am J Clin Pathol 2007;128:158-163 163
163 DOI: 10.1309/69A5AV266W230AUA 163
American Society for Clinical Pathology
Clinical Chemistry / ORIGINAL ARTICLE
6. Singh PK, Parvin CA, Gronowski AM. Establishment of
reference intervals for markers of fetal thyroid status in
amniotic fluid. J Clin Endocrinol Metab. 2003;88:4175-4179.
7. Rawlins ML, Roberts WL. Performance characteristics of six
third-generation assays for thyroid-stimulating hormone.
Clin Chem. 2004;50:2338-2344.
8. Solberg HE. Establishment and use of reference values. In:
Burtis CA, Ashwood ER, eds. Tietz Textbook of Clinical
Chemistry. Philadelphia, PA: Saunders; 1999.
9. ADVIA Centaur Assay Manual. Tarrytown, NY: Bayer
Healthcare; 2001.
10. Kourides IA, Heath CV, Ginsberg-Fellner F. Measurement of
thyroid-stimulating hormone in human amniotic fluid. J Clin
Endocrinol Metab. 1982;54:635-637.
11. Kourides IA, Berkowitz RL, Pang S, et al. Antepartum
diagnosis of goitrous hypothyroidism by fetal ultrasonography
and amniotic fluid thyrotropin concentration. J Clin Endocrinol
Metab. 1984;59:1016-1018.
12. Hollingsworth DR, Alexander NM. Amniotic fluid
concentrations of iodothyronines and thyrotropin do not
reliably predict fetal thyroid status in pregnancies complicated
by maternal thyroid disorders or anencephaly. J Clin Endocrinol
Metab. 1983;57:349-355.
13. Chopra IJ, Santini F, Hurd RE, et al. A radioimmunoassay for
measurement of thyroxine sulfate. J Clin Endocrinol Metab.
1993;76:145-150.
14. Polk DH. Thyroid hormone metabolism during development.
Reprod Fertil Dev. 1995;7:469-477.
15. Vasiliades J, Hilgers T, Gentrup B. Long term stability of
hormones in serum [abstract]. Clin Chem. 1995;41(suppl):S59.
16. Perelman AH, Johnson RL, Clemons RD, et al. Intrauterine
diagnosis and treatment of fetal goitrous hypothyroidism.
J Clin Endocrinol Metab. 1990;71:618-621.
17. Bruner JP, Dellinger EH. Antenatal diagnosis and treatment
of fetal hypothyroidism: a report of two cases. Fetal Diagn Ther.
1997;12:200-204.
18. Perrotin F, Sembely-Taveau C, Haddad G, et al. Prenatal
diagnosis and early in utero management of fetal
dyshormonogenic goiter. Eur J Obstet Gynecol Reprod Biol.
2001;94:309-314.
19. Abuhamad AZ, Fisher DA, Warsof SF, et al. Antenatal
diagnosis and treatment of fetal goitrous hypothyroidism: case
report and review of the literature. Ultrasound Obstet Gynecol.
1995;6:368-371.
Das könnte Ihnen auch gefallen
- Pang Imunopathogenesis Dss PDFDokument3 SeitenPang Imunopathogenesis Dss PDFrantiadrianiNoch keine Bewertungen
- Aslmualaikum WR WBDokument6 SeitenAslmualaikum WR WBrantiadrianiNoch keine Bewertungen
- Rajapakse Dengue ShockDokument9 SeitenRajapakse Dengue ShockrantiadrianiNoch keine Bewertungen
- GipsDokument13 SeitenGipsrantiadrianiNoch keine Bewertungen
- Handbook: For Clinical Management of DengueDokument124 SeitenHandbook: For Clinical Management of DengueraattaiNoch keine Bewertungen
- AbstractDokument10 SeitenAbstractrantiadrianiNoch keine Bewertungen
- MRI in MTDokument13 SeitenMRI in MTrantiadrianiNoch keine Bewertungen
- Practice Tracing B Prek PDFDokument1 SeitePractice Tracing B Prek PDFrantiadrianiNoch keine Bewertungen
- Practice Tracing D PrekDokument1 SeitePractice Tracing D PrekrantiadrianiNoch keine Bewertungen
- Practice Tracing B Prek PDFDokument1 SeitePractice Tracing B Prek PDFrantiadrianiNoch keine Bewertungen
- Practice Tracing e PrekDokument1 SeitePractice Tracing e PrekrantiadrianiNoch keine Bewertungen
- Devarajan, Dss DGN ArdsDokument4 SeitenDevarajan, Dss DGN ArdsrantiadrianiNoch keine Bewertungen
- Practice Tracing C PrekDokument1 SeitePractice Tracing C PrekrantiadrianiNoch keine Bewertungen
- Management of Immune Thrombocytopenic PurpuraDokument7 SeitenManagement of Immune Thrombocytopenic PurpuraApriliza RalasatiNoch keine Bewertungen
- YUVARAJAN-DeNGUE Pulmonal ComplicationDokument6 SeitenYUVARAJAN-DeNGUE Pulmonal ComplicationrantiadrianiNoch keine Bewertungen
- Daftar Pustaka Sepsis Tugas UjianDokument2 SeitenDaftar Pustaka Sepsis Tugas UjianrantiadrianiNoch keine Bewertungen
- Daftar Pustaka RadiologiDokument1 SeiteDaftar Pustaka RadiologirantiadrianiNoch keine Bewertungen
- Eficacy Surfactant 2016Dokument13 SeitenEficacy Surfactant 2016rantiadrianiNoch keine Bewertungen
- European ConsensusDokument19 SeitenEuropean ConsensusEdgar Quelex VicenteNoch keine Bewertungen
- 6 PDFDokument5 Seiten6 PDFUgaugaaNoch keine Bewertungen
- Shock Risk FactorDokument4 SeitenShock Risk FactorrantiadrianiNoch keine Bewertungen
- Handbook: For Clinical Management of DengueDokument124 SeitenHandbook: For Clinical Management of DengueraattaiNoch keine Bewertungen
- Pang Imunopathogenesis Dss PDFDokument3 SeitenPang Imunopathogenesis Dss PDFrantiadrianiNoch keine Bewertungen
- Preterm Birth - WHO RecommendationDokument108 SeitenPreterm Birth - WHO RecommendationBonifacius Bayu Erlangga KusumaNoch keine Bewertungen
- Cardiac Anomali ResearchDokument5 SeitenCardiac Anomali ResearchrantiadrianiNoch keine Bewertungen
- Clinical Presenyation Dengue in ChildrenDokument4 SeitenClinical Presenyation Dengue in ChildrenrantiadrianiNoch keine Bewertungen
- GipsDokument13 SeitenGipsrantiadrianiNoch keine Bewertungen
- Role of Hematologic Scoring System in Early Diagnosis of Neonatal SepticemiaDokument6 SeitenRole of Hematologic Scoring System in Early Diagnosis of Neonatal SepticemiarantiadrianiNoch keine Bewertungen
- A Dede Ji 422017 Aj Mah 32627Dokument9 SeitenA Dede Ji 422017 Aj Mah 32627rantiadrianiNoch keine Bewertungen
- JClinNeonatol2125-1530533 041505Dokument5 SeitenJClinNeonatol2125-1530533 041505rantiadrianiNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (399)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (73)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (120)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Maternal and Child NursingDokument30 SeitenMaternal and Child NursingJack Bisarra Sanchez0% (1)
- Episiotomy and RepairDokument20 SeitenEpisiotomy and RepairwendeliciousNoch keine Bewertungen
- Menstrual HygieneDokument8 SeitenMenstrual HygieneManvendra Pratap SinghNoch keine Bewertungen
- Incompetent Cervix As One of The Antenatal ComplicationsDokument14 SeitenIncompetent Cervix As One of The Antenatal ComplicationsKenje Kate Agripo100% (2)
- OccipitoposteriorDokument11 SeitenOccipitoposteriorVijith.V.kumar100% (1)
- Safety and Effectiveness of Oral Misoprostol For Induction of Labour in A Resource-Limited Setting: A Dose Escalation StudyDokument8 SeitenSafety and Effectiveness of Oral Misoprostol For Induction of Labour in A Resource-Limited Setting: A Dose Escalation StudyAfria Tika AvicennaNoch keine Bewertungen
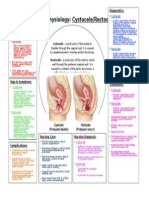
- Cystocele-Rectocele Patho MapDokument1 SeiteCystocele-Rectocele Patho MapAnn-Marie MorrisonNoch keine Bewertungen
- Masturbation TipsDokument4 SeitenMasturbation Tipsjoan_padilla200025% (4)
- Rajiv Gandhi University of Health Sciences, Bangalore: Proforma For Registration of Subject For SynopsisDokument20 SeitenRajiv Gandhi University of Health Sciences, Bangalore: Proforma For Registration of Subject For SynopsisRevathi DadamNoch keine Bewertungen
- CV MikhailDokument2 SeitenCV MikhailMikhail NurhariNoch keine Bewertungen
- Skor Bishop, Profil Biofisik Janin, Dan Tanda Kehamilan Post-TermDokument9 SeitenSkor Bishop, Profil Biofisik Janin, Dan Tanda Kehamilan Post-TermMuhammad KhoiruddinNoch keine Bewertungen
- Aqa Byb2 W MS Jan05Dokument6 SeitenAqa Byb2 W MS Jan05Mike YauNoch keine Bewertungen
- Form 2D Health Plan Implementation Pregnancy Prenatal CareDokument2 SeitenForm 2D Health Plan Implementation Pregnancy Prenatal CareLora CarpioNoch keine Bewertungen
- Contractedpelvis 100515015729 Phpapp01Dokument45 SeitenContractedpelvis 100515015729 Phpapp01Kevyn TanNoch keine Bewertungen
- Chapter 10: Reaching The Age of AdolescenceDokument3 SeitenChapter 10: Reaching The Age of AdolescenceAarti RajanikantNoch keine Bewertungen
- Form No.1 Birth Report PDFDokument4 SeitenForm No.1 Birth Report PDFRiyas TkNoch keine Bewertungen
- Get A Girl You LikeDokument9 SeitenGet A Girl You LikeKollin2150% (2)
- PlantreproductionlabDokument4 SeitenPlantreproductionlabapi-264004169Noch keine Bewertungen
- Safe MotherhoodDokument16 SeitenSafe MotherhoodOswald DachagaNoch keine Bewertungen
- High Risk Pregnancy FinaleDokument149 SeitenHigh Risk Pregnancy FinaleQuolette Constante100% (1)
- APES ExcelDokument3 SeitenAPES ExcelNathan SherNoch keine Bewertungen
- IcdDokument21 SeitenIcdVicky AprizanoNoch keine Bewertungen
- Female Genitile Mutilation RPDokument8 SeitenFemale Genitile Mutilation RPPalak GroverNoch keine Bewertungen
- Daftar Barang/Alat Laboratorium Jurusan Kebidanan Panthom JumlahDokument3 SeitenDaftar Barang/Alat Laboratorium Jurusan Kebidanan Panthom JumlahEva SavitriNoch keine Bewertungen
- Benign Gynecologic TumorsDokument57 SeitenBenign Gynecologic TumorsDexter IanNoch keine Bewertungen
- Julie A Greenberg - Intersexuality and The Law Why Sex MattersDokument180 SeitenJulie A Greenberg - Intersexuality and The Law Why Sex MattersLaura Belli100% (1)
- 3rd Preliminary Examination 2023 - 2024 Grade 10 ScienceDokument4 Seiten3rd Preliminary Examination 2023 - 2024 Grade 10 ScienceJerome Tala-oc100% (1)
- 6th Grade Structure Processes and Responses To Plants 6bDokument9 Seiten6th Grade Structure Processes and Responses To Plants 6bapi-232424041Noch keine Bewertungen
- B Kerala PSC Staff Nurse Grade 2 Model Questions 2017 Part 2 - Kerala PSC QuestionsDokument6 SeitenB Kerala PSC Staff Nurse Grade 2 Model Questions 2017 Part 2 - Kerala PSC QuestionsMrudula SureshNoch keine Bewertungen
- Worksheet-4C - TEAM ALPHADokument4 SeitenWorksheet-4C - TEAM ALPHARina RomanoNoch keine Bewertungen