Beruflich Dokumente
Kultur Dokumente
Schwann Cells
Hochgeladen von
Rho Vince Caño MalagueñoCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Schwann Cells
Hochgeladen von
Rho Vince Caño MalagueñoCopyright:
Verfügbare Formate
SCHWANN CELLS
Schwann cells (named after physiologist Theodor Schwann) or neurolemmocytes are the
principal glia of the peripheral nervous system (PNS). Glial cells function to support neurons
and, in the PNS, also include satellite cells, olfactory ensheathing cells, enteric glia and glia that
reside at sensory nerve endings, such as the Pacinian corpuscle. There are two types of Schwann
cell, myelinating and nonmyelinating. Myelinating Schwann cells wrap around axons of motor
and sensory neurons to form the myelin sheath.
Schwann cells are involved in many important aspects of peripheral nerve biologythe
conduction of nervous impulses along axons, nerve development and regeneration, trophic
support for neurons, production of the nerve extracellular matrix, modulation of neuromuscular
synaptic activity, and presentation of antigens to T-lymphocytes.
Charcot-Marie-Tooth disease (CMT), Guillain-Barr syndrome (GBS), schwannomatosis, acute
inflammatory demyelinating polyradiculopathy, and chronic inflammatory demyelinating
polyneuropathy (CIDP), and leprosy are all neuropathies involving Schwann cells.
Named after the German physiologist Theodor Schwann, Schwann cells are a variety of glial
cell that keep peripheral nerve fibres (both myelinated and unmyelinated) alive. In myelinated
axons, Schwann cells form the myelin sheath (see above). The sheath is not continuous.
Individual myelinating Schwann cells cover about 100 micrometres of an axonequating to
approximately 10,000 Schwann cells along a 1 metre length of the axonwhich can be up to a
metre or more in length. The gaps between adjacent Schwann cells are called nodes of Ranvier
(see above). The vertebrate nervous system relies on the myelin sheath for insulation and as a
method of decreasing membrane capacitance in the axon. The action potential jumps from node
to node, in a process called saltatory conduction, which can increase conduction velocity up to
ten times, without an increase in axonal diameter. In this sense, Schwann cells are the peripheral
nervous system's analogues of the central nervous system's oligodendrocytes. However, unlike
oligodendrocytes, each myelinating Schwann cell provides insulation to only one axon (see
image). This arrangement permits saltatory conduction of action potentials with repropagation at
the nodes of Ranvier. In this way, myelination greatly increases speed of conduction and saves
energy.
[1]
Non-myelinating Schwann cells are involved in maintenance of axons and are crucial for
neuronal survival. Some group around smaller axons (External image here) and form Remak
bundles.
[2]
Myelinating Schwann cells begin to form the myelin sheath in mammals during fetal
development and work by spiraling around the axon, sometimes with as many as 100
revolutions. A well-developed Schwann cell is shaped like a rolled-up sheet of paper, with layers
of myelin in between each coil. The inner layers of the wrapping, which are predominantly
membrane material, form the myelin sheath while the outermost layer of nucleated cytoplasm
forms the neurolemma. Only a small volume of residual cytoplasm communicates the inner from
the outer layers. This is seen histologically as the Schmidt-Lantermann incisure.
Schwann cell transplantation and regeneration
A number of experimental studies since 2001 have implanted Schwann cells in an attempt to
induce remyelination in multiple sclerosis-afflicted patients.
[3]
In the past two decades, many
studies have demonstrated positive results and potential for Schwann cell transplantation as a
therapy for spinal cord injury, both in aiding regrowth and myelination of damaged CNS
axons.
[4]
Schwann cell transplants in combination with other therapies such as Chondroitinase
ABC have also been shown to be effective in functional recovery from spinal cord injury.
[5]
Indeed, Schwann cells are known for their roles in supporting nerve regeneration.
[6]
Nerves in the
PNS consist of many axons myelinated by Schwann cells. If damage occurs to a nerve, the
Schwann cells will aid in digestion of its axons (phagocytosis). Following this process, the
Schwann cells can guide regeneration by forming a type of tunnel that leads toward the target
neurons. The stump of the damaged axon is able to sprout, and those sprouts that grow through
the Schwann-cell tunnel do so at the rate of approximately 1mm/day in good conditions. The
rate of regeneration decreases with time. Successful axons can therefore reconnect with the
muscles or organs they previously controlled with the help of Schwann cells, however,
specificity is not maintained and errors are frequent, especially when long distances are
involved.
[7]
Because of their ability to impact regeneration of axons, Schwann cells have been
connected to preferential motor reinnervation as well. If Schwann cells are prevented from
associating with axons, the axons die. Regenerating axons will not reach any target unless
Schwann cells are there to support them and guide them. They have been shown to be in advance
of the growth cones. Schwann cells are essential for the maintenance of healthy axons. They
produce a variety of factors, including neurotrophins, and also transfer essential molecules across
to axons.
Schwann cell lineage
Schwann cells are of neural crest origin. During mouse embryonic development, neural crest
cells first differentiate into Schwann cell precursors (SCPs) at around embryonic day (E) 1213.
These precursor cells subsequently differentiate into immature Schwann cells at approximately
E1516, persisting until birth. The postnatal fate of the immature Schwann cell depends on its
random association with axons. In a process called radial sorting, whereby Schwann cells
segregate axons by extending processes into axon bundles, the Schwann cells that happen to
associate with a large diameter axon (>1 m) will develop into myelinating Schwann cells. Small
diameter axons become entrenched in the invaginations of non-myelinating Schwann cells, also
called Remak bundles. A key regulator of this process is the axonally-derived signal Neuregulin-
1, which binds to cell surface receptors on the Schwann cell and promotes myelination of large
diameter axons and sorting of small diameter axons in Remak bundles, dependent on the activity
of the -secretase BACE1 (
[8][9][10][11][12]
). A further class of non-myelinating Schwann cell, the
terminal (or perisynaptic) Schwann cell, exists at the neuromuscular junction, in close proximity
to the neuron-muscle synapse. The transition from immature Schwann cell to myelinating/non-
myelinating Schwann cell is reversible. When the nerve is injured, Schwann cells can
dedifferentiate to form a cell type resembling the immature Schwann cell, often referred to as a
denervated or dedifferentiated Schwann cell. This allows them to re-enter the cell cycle in order
to proliferate and aid nerve regeneration.
[13]
Immunoreactivity
The different classes of Schwann cells express characteristic antigenic markers that can be
targeted with antibodies. Myelinating Schwann cells can be visualised by immunohistochemistry
using antibodies against the proteins S-100, Myelin protein zero (P-Zero) and Myelin basic
protein (MBP). Non-myelinating Schwann cells such as those that form Remak bundles and
terminal Schwann cells are positive for S-100 and Glial fibrillary acidic protein (GFAP).
Ganglioside
9-O-acetyl GD3 ganglioside is an acetylated glycolipid which is found in the cell membranes of
many types of vertebrate cells. During peripheral nerve regeneration, 9-O-acetyl GD3 is
expressed by schwann cells.
[14]
Das könnte Ihnen auch gefallen
- QuestDokument1 SeiteQuestRho Vince Caño MalagueñoNoch keine Bewertungen
- Cranial Epidural AbscessDokument31 SeitenCranial Epidural AbscessRho Vince Caño MalagueñoNoch keine Bewertungen
- ASTHMA TreatmentDokument27 SeitenASTHMA TreatmentRho Vince Caño Malagueño0% (1)
- TonsillectomyDokument27 SeitenTonsillectomyRho Vince Caño MalagueñoNoch keine Bewertungen
- The Bradyarrhythmias: Disorders of The Atrioventricular NodeDokument34 SeitenThe Bradyarrhythmias: Disorders of The Atrioventricular NodeRho Vince Caño MalagueñoNoch keine Bewertungen
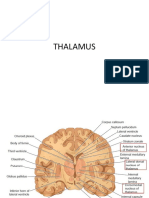
- ThalamusDokument30 SeitenThalamusRho Vince Caño Malagueño100% (1)
- Operating Room StandardsDokument28 SeitenOperating Room StandardsRho Vince Caño MalagueñoNoch keine Bewertungen
- Pediatric Sample ExamDokument5 SeitenPediatric Sample ExamRho Vince Caño MalagueñoNoch keine Bewertungen
- Cabinet: Title NameDokument5 SeitenCabinet: Title NameRho Vince Caño MalagueñoNoch keine Bewertungen
- LABOR and Delivery: Cristina Cleofe, RM BSN - IiiDokument17 SeitenLABOR and Delivery: Cristina Cleofe, RM BSN - IiiRho Vince Caño MalagueñoNoch keine Bewertungen
- Reading As A Physiological ProcessDokument2 SeitenReading As A Physiological ProcessRho Vince Caño Malagueño89% (9)
- CD-NTG BupcDokument9 SeitenCD-NTG BupcRho Vince Caño MalagueñoNoch keine Bewertungen
- Arrow of GodDokument24 SeitenArrow of GodRho Vince Caño Malagueño100% (1)
- Arrowroot Powder vs. Cornstarch: Why Arrowroot Powder Is A Better ChoiceDokument53 SeitenArrowroot Powder vs. Cornstarch: Why Arrowroot Powder Is A Better ChoiceRho Vince Caño MalagueñoNoch keine Bewertungen
- Henry Sy Success StoryDokument6 SeitenHenry Sy Success StoryRho Vince Caño MalagueñoNoch keine Bewertungen
- QuestionnaireDokument4 SeitenQuestionnaireRho Vince Caño MalagueñoNoch keine Bewertungen
- A Typology of Nursing Problems in Family Nursing ProblemDokument5 SeitenA Typology of Nursing Problems in Family Nursing ProblemRho Vince Caño MalagueñoNoch keine Bewertungen
- MYASTHENIA GRAVIS Case PresentationDokument78 SeitenMYASTHENIA GRAVIS Case PresentationRho Vince Caño Malagueño50% (4)
- Transmittal Letter or Cover Letter-1Dokument3 SeitenTransmittal Letter or Cover Letter-1Rho Vince Caño MalagueñoNoch keine Bewertungen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Syllogisms in EducationDokument6 SeitenSyllogisms in EducationJonathan AcuñaNoch keine Bewertungen
- Electromyography & It's Application in OrthodonticsDokument22 SeitenElectromyography & It's Application in Orthodonticsprasadgayake100% (1)
- 3 Marr's Model of PerceptionDokument3 Seiten3 Marr's Model of PerceptionronnyNoch keine Bewertungen
- Teaching and Learning: Philosophies, Principles and PracticesDokument10 SeitenTeaching and Learning: Philosophies, Principles and PracticesGilang NugrahaNoch keine Bewertungen
- Learning Style ReflectionDokument3 SeitenLearning Style Reflectionapi-404093704Noch keine Bewertungen
- Vanessa Murillo Ramírez - Cognitiva 03Dokument7 SeitenVanessa Murillo Ramírez - Cognitiva 03Vanessa Murillo RamírezNoch keine Bewertungen
- Final SHS-Perdev-Q1-Module 5Dokument18 SeitenFinal SHS-Perdev-Q1-Module 5Ira Jane CaballeroNoch keine Bewertungen
- Chapter 4 QuizDokument10 SeitenChapter 4 QuizlcjenleeNoch keine Bewertungen
- Using CuriosityDokument53 SeitenUsing CuriosityGissel GuardadoNoch keine Bewertungen
- (Oxford Library of Psychology) Richard M. Ryan-The Oxford Handbook of Human Motivation-Oxford University Press (2012) PDFDokument598 Seiten(Oxford Library of Psychology) Richard M. Ryan-The Oxford Handbook of Human Motivation-Oxford University Press (2012) PDFPrueba95% (19)
- Assignment 1. Mind MapDokument1 SeiteAssignment 1. Mind MapMaestros Plantel BayitoNoch keine Bewertungen
- Como Hacer dx-1Dokument6 SeitenComo Hacer dx-1Krys Vargas DazaNoch keine Bewertungen
- Fatima Memorial Medical & Dental College & AHS, LahoreDokument8 SeitenFatima Memorial Medical & Dental College & AHS, LahoreAqsa MohsinNoch keine Bewertungen
- Who Am I?: An Introduction To Self-AwarenessDokument23 SeitenWho Am I?: An Introduction To Self-AwarenessStar LuchavezNoch keine Bewertungen
- Glasgocoma ScaleDokument35 SeitenGlasgocoma Scalepop lopNoch keine Bewertungen
- FINAL ACTIVITY 10 - (Sto. Nino)Dokument2 SeitenFINAL ACTIVITY 10 - (Sto. Nino)Wena Mae LavideNoch keine Bewertungen
- Pain in Children With Autism Spectrum Disorder: Experience, Expression, and AssessmentDokument6 SeitenPain in Children With Autism Spectrum Disorder: Experience, Expression, and Assessmentflower21Noch keine Bewertungen
- Chapter IDokument7 SeitenChapter IAshley PanganNoch keine Bewertungen
- Sudn 11Dokument280 SeitenSudn 11triddle1969100% (2)
- Possibilities: Limitations andDokument12 SeitenPossibilities: Limitations andHazel Kinilog88% (8)
- Guide To Spiritual AwakeningDokument12 SeitenGuide To Spiritual Awakeningconnect2rahul4204100% (4)
- Lab Report Group 6 Nerves and ReflexesDokument5 SeitenLab Report Group 6 Nerves and ReflexesKristina Esta50% (2)
- Poster: Sensory Garden Design For Adolescents With Dual Sensory ImpairmentDokument1 SeitePoster: Sensory Garden Design For Adolescents With Dual Sensory ImpairmentNeroulidi854Noch keine Bewertungen
- DS Case StudyDokument2 SeitenDS Case StudyKevin PascuaNoch keine Bewertungen
- Behavioural Aspects of Language Attitude Mean DescriptionDokument6 SeitenBehavioural Aspects of Language Attitude Mean Descriptionjhehan galmanNoch keine Bewertungen
- Chapter 1Dokument11 SeitenChapter 1Jasie Joy GilloNoch keine Bewertungen
- For Interacting With Individuals With Autism: 9 TipsDokument5 SeitenFor Interacting With Individuals With Autism: 9 TipsSweet'nSpicy LabNoch keine Bewertungen
- Creative Engineering - John E. ArnoldDokument244 SeitenCreative Engineering - John E. ArnoldMuhammad Choirul AzisNoch keine Bewertungen
- Describing LearnersDokument5 SeitenDescribing LearnersFlorenciaCarrizo030% (1)
- Educ2181 PP WeeblyDokument4 SeitenEduc2181 PP Weeblyapi-363014935Noch keine Bewertungen