Beruflich Dokumente
Kultur Dokumente
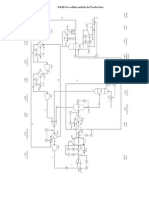
Disc Bowl Flow Chart
Hochgeladen von
Saints Burner Christopher0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
54 Ansichten3 SeitenFlow chart for disc bowl centrifuge experiment
Copyright
© © All Rights Reserved
Verfügbare Formate
PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenFlow chart for disc bowl centrifuge experiment
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
54 Ansichten3 SeitenDisc Bowl Flow Chart
Hochgeladen von
Saints Burner ChristopherFlow chart for disc bowl centrifuge experiment
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 3
EXPERIMENT 4 : DISC BOWL CENTRIFUGE
Experiment 1A: Investigate the Effect of Frequency on Centrifuge Separation
Prepare 1 L of Solution A (100% water) and determine its density.
Prepare 1 L of Solution B (100% oil) and determine its density. Record down all the
readings.
Mix both solutions into a 2 L beaker and determine the density
Place two 1 L beakers to the liquid collecting outlets. Label the beaker as top
sample and bottom sample
Switch ON the apparatus. Press the 'Run' green button on the frequency inverter.
Set the frequency to roughly 15 Hz.
Allow the apparatus to run for 3 minutes. Record down the speed of the bowl.
Pour the mixed solution into the mixing tank
Keep an deye on the liquid collecting outlets. Stop the appratus when there is no
liquid coming out from the outlets.
Collect sample from each of the 1L beaker. Determine the density of the collected
solution
Compare the density with initial solution density.
Repeat with 20 Hz, 25 Hz and 30 Hz.
Experiment 1B: Investigate the Effect of Density on Centrifuge Separation
Prepare 1 L of Solution A (100% water) and determine its density.
Prepare 0.5 L of Solution B (50% oil) and determine its density. Record down all the
readings.
Mix both solutions into a 2 L beaker and determine the density
Place two 1 L beakers to the liquid collecting outlets. Label the beaker as top
sample and bottom sample
Switch ON the apparatus. Press the 'Run' green button on the frequency inverter.
Set the frequency to roughly 15 Hz.
Allow the apparatus to run for 3 minutes. Record down the speed of the bowl.
Pour the mixed solution into the mixing tank
Keep an deye on the liquid collecting outlets. Stop the appratus when there is no
liquid coming out from the outlets.
Collect sample from each of the 1L beaker. Determine the density of the collected
solution
Compare the density with initial solution density.
Repeat with using 1.2 L of A with 0.4 L of B and repeat again for 0.5 L of A with 1 L
of B
Clean and wash mixing tank, disc bowl and collecting tray.
Experiment 2: Density Determination
Take the measuring cylinder and measure its mass, in grams as
accurately as possible.
Take the measuring cylinder off the balance and add the water
carefully, either by careful pouring or with a pipette until the level
is as close to the 10 ml mark as possible. Put the measuring
cylinder back on the balance. Measure and record the new mass
(cylinder plus water), in grams.
Repeat the procedure, adding 10 ml at a time as accurately as possible and
recording the volume and total mass, until the measuring cylinder is full. Then,
for each volume calculate the mass of the liquid alone.
Repeat steps for the oil
Draw a graph of mass of liquid (y-axis) against volume (x-axis).
For each set of data try and draw a straight best fit line
passing through the origin. Calculate the density of each liquid
from the gradient of its graph line.
Das könnte Ihnen auch gefallen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Visio Stream Table Part3Dokument1 SeiteVisio Stream Table Part3Saints Burner ChristopherNoch keine Bewertungen
- Garbage Enzyme University SarawakDokument6 SeitenGarbage Enzyme University SarawakSaints Burner ChristopherNoch keine Bewertungen
- UEMK4353 Assignment - Mech DesignDokument2 SeitenUEMK4353 Assignment - Mech DesignSaints Burner ChristopherNoch keine Bewertungen
- Visio IndexDokument1 SeiteVisio IndexSaints Burner ChristopherNoch keine Bewertungen
- Industrial Training Report UtarDokument29 SeitenIndustrial Training Report UtarSaints Burner Christopher100% (1)
- HAZOP TemplateDokument12 SeitenHAZOP TemplateSaints Burner ChristopherNoch keine Bewertungen
- Tutorial 5Dokument7 SeitenTutorial 5Saints Burner ChristopherNoch keine Bewertungen
- Foodchapter 1 PDFDokument1 SeiteFoodchapter 1 PDFSaints Burner ChristopherNoch keine Bewertungen
- Part 1 (A)Dokument7 SeitenPart 1 (A)Saints Burner ChristopherNoch keine Bewertungen
- P&IDDokument1 SeiteP&IDSaints Burner ChristopherNoch keine Bewertungen
- Introduction For Batch Reactor ExperimentDokument5 SeitenIntroduction For Batch Reactor ExperimentSaints Burner Christopher25% (4)
- Experiment 1: Batch Reactor: Experiment 1: 6.1. Calibration Curve - Conductivity Vs ConversionDokument2 SeitenExperiment 1: Batch Reactor: Experiment 1: 6.1. Calibration Curve - Conductivity Vs ConversionSaints Burner ChristopherNoch keine Bewertungen
- Chemical Reaction Engineering Catalyst ResearchDokument4 SeitenChemical Reaction Engineering Catalyst ResearchSaints Burner ChristopherNoch keine Bewertungen
- Fluidiesed BedDokument14 SeitenFluidiesed BedSaints Burner ChristopherNoch keine Bewertungen
- Assignment 1 (CRE 2)Dokument1 SeiteAssignment 1 (CRE 2)Saints Burner ChristopherNoch keine Bewertungen
- Gas Absorption Report PDFDokument13 SeitenGas Absorption Report PDFSaints Burner Christopher100% (1)
- Microsoft Word - Cpci Assignment NewDokument25 SeitenMicrosoft Word - Cpci Assignment NewSaints Burner ChristopherNoch keine Bewertungen
- Table 10.4 Food Packaging ConsiderationsDokument1 SeiteTable 10.4 Food Packaging ConsiderationsSaints Burner ChristopherNoch keine Bewertungen
- Cre 1 Solution PDFDokument21 SeitenCre 1 Solution PDFSaints Burner Christopher100% (1)
- For Temperature Changes Per TimeDokument1 SeiteFor Temperature Changes Per TimeSaints Burner ChristopherNoch keine Bewertungen
- Part 1 (A)Dokument7 SeitenPart 1 (A)Saints Burner ChristopherNoch keine Bewertungen
- Chapter 3 - Tutorial SolutionDokument8 SeitenChapter 3 - Tutorial SolutionSaints Burner ChristopherNoch keine Bewertungen
- For Temperature Changes Per TimeDokument1 SeiteFor Temperature Changes Per TimeSaints Burner ChristopherNoch keine Bewertungen
- Eis AssignmentDokument4 SeitenEis AssignmentSaints Burner ChristopherNoch keine Bewertungen
- French Flash Card 18Dokument1 SeiteFrench Flash Card 18Saints Burner ChristopherNoch keine Bewertungen
- For Temperature Changes Per TimeDokument1 SeiteFor Temperature Changes Per TimeSaints Burner ChristopherNoch keine Bewertungen
- Chemical Engineering Compulsory SubjectDokument1 SeiteChemical Engineering Compulsory SubjectSaints Burner ChristopherNoch keine Bewertungen
- Ni Hao Hi Chi Le Ma? Have You Eaten? Zai Jian Good ByeDokument1 SeiteNi Hao Hi Chi Le Ma? Have You Eaten? Zai Jian Good ByeSaints Burner ChristopherNoch keine Bewertungen
- Flash Card FrenchDokument6 SeitenFlash Card FrenchSaints Burner ChristopherNoch keine Bewertungen
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- S861 & S866 Alfa LavalDokument176 SeitenS861 & S866 Alfa LavalarmaganNoch keine Bewertungen
- Ramachandran S., Mechanics of Fluids, 2016Dokument600 SeitenRamachandran S., Mechanics of Fluids, 2016Safa Eng90Noch keine Bewertungen
- Mrs Lawrence Manuscript 1 PDFDokument11 SeitenMrs Lawrence Manuscript 1 PDFOyebanji Oluwayinka MathewNoch keine Bewertungen
- Feed Study Co2 Transport PipelineDokument143 SeitenFeed Study Co2 Transport PipelineSalvatore MonacoNoch keine Bewertungen
- Percent Air Voids in Compacted Dense and Open Asphalt MixturesDokument4 SeitenPercent Air Voids in Compacted Dense and Open Asphalt MixturesAhmad KhreisatNoch keine Bewertungen
- Basic Calibration of Master Viscometers and Viscosity Oil StandardsDokument7 SeitenBasic Calibration of Master Viscometers and Viscosity Oil StandardsIntegridad de Equipos y Gestión de Activos NelcamNoch keine Bewertungen
- Draught Survey Code by UNECEDokument19 SeitenDraught Survey Code by UNECEHobbyLogicNoch keine Bewertungen
- Iso 10294-3-1999Dokument16 SeitenIso 10294-3-1999Nguyen Trung KienNoch keine Bewertungen
- Airdata Multimeter Adm 870c Operating InstructionsDokument86 SeitenAirdata Multimeter Adm 870c Operating InstructionsALEJANDRO IPATZINoch keine Bewertungen
- Fan Foundation DesignDokument47 SeitenFan Foundation DesignsomumallidiNoch keine Bewertungen
- ALPS 2208 Physics AssignmentDokument14 SeitenALPS 2208 Physics AssignmentHemang JainNoch keine Bewertungen
- Energies 14 00930 v2Dokument16 SeitenEnergies 14 00930 v2CloudyshaNoch keine Bewertungen
- Brochure ClausDokument7 SeitenBrochure ClausJUAN VICTOR SULVARANNoch keine Bewertungen
- Module 1 - Introduction To Fluid MechanicsDokument94 SeitenModule 1 - Introduction To Fluid MechanicsFarhan Shajahan100% (1)
- Lab Note 2 Sand Equivalent Test and Specific Gravity of Fine and Coarse AggregateDokument5 SeitenLab Note 2 Sand Equivalent Test and Specific Gravity of Fine and Coarse Aggregatemarlon lumanogNoch keine Bewertungen
- General Physics I: Physical Quantities & Vectors Quarter 1, Module 1Dokument9 SeitenGeneral Physics I: Physical Quantities & Vectors Quarter 1, Module 1Denies VargasNoch keine Bewertungen
- Review 3 SearchableDokument16 SeitenReview 3 SearchablehquynhNoch keine Bewertungen
- Thermophysical PropertiesDokument14 SeitenThermophysical PropertiesKalinga BalNoch keine Bewertungen
- ASTM D5453-16e11Dokument11 SeitenASTM D5453-16e11Andres Muñoz Aguirre100% (2)
- Chapter 29 Pressure in Fluids: 367 © John Bird Published by Taylor and FrancisDokument8 SeitenChapter 29 Pressure in Fluids: 367 © John Bird Published by Taylor and FrancisRmbj AntNoch keine Bewertungen
- FVT Fundamentals of Vacuum Technology EN58774555441f3Dokument200 SeitenFVT Fundamentals of Vacuum Technology EN58774555441f3velmuruganNoch keine Bewertungen
- Klein On Evaluating Foam PDFDokument2 SeitenKlein On Evaluating Foam PDFFarhana DiyanaNoch keine Bewertungen
- C1162Dokument2 SeitenC1162reefreefNoch keine Bewertungen
- Higher Unit 11 Topic Test: NameDokument17 SeitenHigher Unit 11 Topic Test: NamesadiyaNoch keine Bewertungen
- Soil Detailed NotesDokument175 SeitenSoil Detailed Notesrishi choudhary100% (1)
- Petroserv Limited: Section 06: Roadworks 04: Unbound Pavement MaterialsDokument4 SeitenPetroserv Limited: Section 06: Roadworks 04: Unbound Pavement MaterialsJabin100% (1)
- Agar50series MPFM SpecDokument4 SeitenAgar50series MPFM SpecJADNoch keine Bewertungen
- Autodyn Verberne Pieter 201411 MAS Thesis PDFDokument115 SeitenAutodyn Verberne Pieter 201411 MAS Thesis PDFYousaf SaidalaviNoch keine Bewertungen
- Sample Reviewer GEODokument10 SeitenSample Reviewer GEOZherrinore Rasay50% (2)
- The Fleet Type Submarine HydraulicsDokument155 SeitenThe Fleet Type Submarine HydraulicsSan Francisco Maritime National Park Association100% (1)