Beruflich Dokumente
Kultur Dokumente
Gastrointestinal Tract (Partial Edit)
Hochgeladen von
applesncoreCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Gastrointestinal Tract (Partial Edit)
Hochgeladen von
applesncoreCopyright:
Verfügbare Formate
PATHOLOGY OF
GASTROINTESTINAL TRACT
Dr Dexter MD FRC Path
Undercover Professor
Department of pathology SOM,SGU
Grenada (W.I.)
Overview of clinical features
Symptoms
Dysphagia
Epigastric pain
Heart burn, dyspepsia, flatulence
Loss of appetite
Nausea, Vomiting
Hematemesis
Overview of clinical features
Colicky abdominal pain
Abdominal distension
Diarrhea
Dysentery
Constipation
Alternate diarrhea and constipation
Blood in stools
Occult- Guaiac test
Hematochezia
Melena
Anemia
Overview of clinical features
Signs
Epigastric tenderness
Epigastric mass
Hepatomegaly
Splenomegaly
Ascites
Visible peristalsis
Abdominal rigidity or guarding
Abdominal mass
Common investigations
Stool for occult blood
Plain X ray
Barium meal
Barium follow through
Barium enema
Upper GI endoscopy
Colonoscopy, Sigmoidoscopy
Ultrasonography
Arteriography
Fine Needle Aspiration Cytology (FNAC)
Biopsy
ORAL CAVITY
Leukoplakia
Refers to well defined ZKLWH SODTXHV
caused by epidermal proliferations
White plaque on the oral mucosa that can
not be removed with scraping and can not
be classified clinically and microscopically
as any other disease
Benign epithelial lesions to highly
dysplastic lesions
3-7% undergo malignant transformation
Precancerous until proven otherwise
Buccal mucosa, tongue, floor of mouth
Most commonly at vermillion border
Predisposing factors:
tobacco use
ill fitting dentures,
persistent irritants
HPV infection
Erythroplakia
Aka Erythroplasia
Less common but more ominous
Red velvety eroded area
Poorly circumscribed
Much more atypical epithelial changes
(dysplasia)
Malignant transformation in more than 50%
Oral Hairy Leukoplakia(OHL)
Seen in patients with HIV
White confluent fluffy or hairy
hyperkeratotic thickenings
Epstein Barr Virus in majority
Layers of keratotic squames on underlying
mucosal acanthosis (hyperkeratotic)
No malignant potential
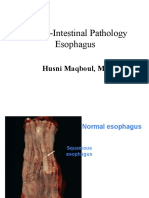
ESOPHAGUS
Esophagus-Normal
Wall has 4 layers:
Mucosa
Sub mucosa (rich network of lymphatics)
Muscularis propria
Adventitia
In sharp contrast to rest of GIT mostly
devoid of serosa
Lesions -overview
Benign Benign Malignant Malignant
Congenital
malformations
Non neoplastic
conditions
Neoplasms
Atresia
Stenosis
Heterotopia
Cysts
Diverticula
Hiatal hernia
Achalasia
Mallory Weiss tear
Esophagitis
Barrets esophagus
Varices
Esophageal Atresia
Disruption of elongation & seperation
of esophogus and trachea during
embryogenesis
Commonly associated with tracheo-
esophageal fistulas
Maternal polyhydroamnios, single umblical
artery
Excessive drooling of saliva in new born
Choking and cyanosis with first feed
Hiatal Hernia
Herniation of stomach through enlarged
esophageal hiatus in diaphragm
Sliding type (95%)
Paraesophageal type
Leads to incompetence of lower esophageal
sphincter (LES) especially in sliding type
Reflux of gastric contents leads to epigastric pain,
heart burn (sliding type)
Respiratory distress, volvulus, strangulation in
paraesophageal type
Achalasia / cardiospasm
Incomplete relaxation of LES in response to
swallowing
Functional esophageal obstruction
Three main features
Aperistalsis
Partial or incomplete relaxation of LES
Increased resting tone of LES
Primary
Loss of intrinsic inhibitory innervation of LES and
and smooth muscle
Loss or absence of ganglion cells in myenteric
plexus
Secondary (aka pseudoachalasia)
Impaired function from a variety of causes e.g.
Chagas, polio, sarcoidosis
Clinical features
Gradual onset of dysphagia
Substernal discomfort
Odynophagia
Reflux of contents
vomiting
Aspiration pneumonia
Progressive dilatation of esophagus above LES
Manometry is diagnostic
Risk of developing squamous cell
carcinoma (in about 5%)
Mallory Weiss Syndrome
Longitudinal mucosal tears at esophageal
gastric junction
In alcoholics after bout of severe retching
In bulimics following vomiting
Inadequate relaxation of LES during
vomiting
Hematemesis
Usually heal but some times fatal
Esophagitis
IRRITANTS - alcohol, acids, alkalis etc.
INFECTIONS - Herpes simplex and
Cytomegalovirus virus, Candidiasis
UREMIA
ALLERGIC - Eosinophilic esophagitis
ANTICANCER THERAPY
HIATAL HERNIA - Sliding type
LES
tone
Delayed
esophageal
clearance
Increased
gastric volume
Reparative
capacity of
esophagus
CNS depressants,
hypothyroidism, pregnancy,
alcohol, tobacco,
nasogastric intubation.
REFLUX
ESOPHAGITIS
Hyperemia
Presence of inflammatory cells
eosinophils
Elongation of lamina propria papillae
Basal zone hyperplasia
Clinically
Heart burn
Water brash
Symptoms increase after lying down or after a
large meal
Nocturnal cough, hoarseness of voice
Complications
Bleeding
Stricture formation
Barrets esophagus, Adenocarcinoma
Aspiration pneumonitis
Barrets Esophagus
The esophageal mucosa is replaced by
metaplastic columnar epithelium
because of prolonged injury i.e.
chronic esophageal reflux.
Usually in patients with long standing
reflux esophagitis
After injury there is proliferation of stem
cells which differentiate into columnar
cells (more resistant to acid peptic injury)
Salmon pink patch, tongue or large area
above EG, surface becomes granular
Clinical features
Long history of reflux
Features of GERD
Asymptomatic in one third patients
30-40 fold increased risk of
adenocarcinoma
Monitored by repeated endoscopic
biopsies to look for dysplasia
Esophageal Varices
Dilated tortuous vessels (collaterals) in the
lower end of esophagus
Associated with portal hypertension
90% in cirrhotic patients
Dilated tortuous veins in mucosa
and submucosa
Overlying mucosa may be normal or inflamed or
ulcerated
Usually no symptoms till they rupture
Responsible for 50% deaths in patients
with advanced cirrhosis
Neoplasms
BENIGN
Rare
MALIGNANT
(More common)
Squamous cell
carcinoma
Adenocarcinoma
(More common in US)
Squamous Cell Carcinoma
Adults over the age of 50
More common in males
High incidence in central Asia and
Northern China
In United states incidence is 2-8
per 100,000 yearly
Blacks are at higher risk than whites
Etiologies
Clinically
Dysphagia to solid food initially
Weight loss
Chest pain, cough, hoarseness due to
direct local extension of tumor
Lymph node involvement- cervical,
mediastinal, paratracheal, tracheobronchial,
gastric and celiac nodes
SCC
GROSS
20% upper third of esophagus
50% middle third of esophagus
30% lower third of esophagus
PATTERNS
Exophytic
Diffusely Infiltrative
Ulcerated/ Excavated
Growth Patterns of tumors
Adenocarcinoma
Lower third of esophagus
Barrets esophagus is precursor lesion
Multistep process through dysplasia
Median age 50 years
Poor prognosis
Appear as flat, raised patches which
develop into large, nodular masses
Signs and symptoms similar to squamous
cell carcinoma
Most are mucin producing
adenocarcinomas
Prognosis poor
Surveillance in Barrets esophagus is
mandatory
STOMACH
Lesions- overview
NON
NEOPLASTIC
Pyloric stenosis
Gastritis
- Acute
-Chronic
Peptic ulcer disease
NEOPLASTIC
BENIGN
Gastric polyps
Adenomas
Leiomyomas
MALIGNANT
Carcinomas
Carcinoids
Lymphomas
(MALTOMAS)
Pyloric Stenosis
CONGENITAL
Has genetic basis
ACQUIRED
Chronic antral
gastritis
Peptic ulcers
Malignancy
Congenital Pyloric Stenosis
More common in first male child
Concentric hypertrophy of circular muscle
Clinically - Manifests 2-3 weeks after birth:
Regurgitation Regurgitation
Projectile vomiting Projectile vomiting
Palpable epigastric mass Palpable epigastric mass
Visible peristalsis
Visible peristalsis
Treated by surgery (myotomy)
Acute Gastritis
Is acute inflammatory process of the
mucosa usually transient in nature
Etiological factors
NSAIDS
Excessive alcohol consumption
Heavy smoking
Ischemia and shock
Severe stress (burns, surgery)
Cancer chemotherapy
Systemic infections
Uremia
Clinical Features
May be asymptomatic
Epigastric pain, nausea and vomiting
Hematemesis and melena
Bleeding can be fatal
Pathogenesis is related to
Increased acid secretion with back diffusion
Decreased bicarbonate secretion
Decreased mucosal blood flow
Direct damage to epithelium
Loss of surface epithelium = erosions
Erosions along with hemorrhage (acute
erosive gastritis)
Hyperemia, punctate areas of hemorrhage
Edema and congestion of lamina propria
Neutrophils in the surface epithelium and
lumina of the glands
Chronic Gastritis
Definition presence of chronic mucosal
inflammatory changes leading eventually to
mucosal atrophy and epithelial metaplasia
Etiology
Chronic infection with H. pylori
Immunologic causes (autoimmune)
Alcohol and smoking
Post surgical e.g. after antrectomy
Radiation
Granulomatous conditions
Common
causes
Helicobacter pylori
Gram negative, curvilinear, motile
(flagella), non invasive and urease positive
bacillus
Associated with two
types of gastritis
- Antral type
- Multifocal atrophic type
OTHER DISEASES
CAUSED BY H.PYLORI
-Peptic ulcers
-Gastric carcinoma
-Gastric lymphoma
H. pylori
Attracts PMNs
& other inflammatory
cells
Enzymes
Proteases
Ureases
Phosopholipases
Damage to mucosa
Chronic gastritis, Peptic ulcers
Uncontrolled
proliferation of B cells
lymphoma
Autoimmune
Auto antibodies against parietal cells
Gland destruction
ATROPHY
Loss of Loss of
Acid production intrinsic factor
(Achlorhydria) VitaminB12 deficiency
(Pernicious anemia)
Mainly involves body
and fundus
Hypergastrinemia
Clinical Features of chronic
gastritis
Asymptomatic
Nausea, vomiting
Epigastric discomfort
Dyspepsia
Indigestion
Endoscopy-Boggy appearing mucosa
with thick mucosal folds
MICROSCOPY
Inflammatory infiltrate (lymphocytes,
plasma cells) in the lamina propria
PMNs in the surface epithelium and
glandular lumen (Activity)
Lymphoid aggregates ( marker for H.pylori
infection)
Intestinal metaplasia
Glandular atrophy especially severe in
autoimmune type
Parietal cell absence in pernicious anemia
Look for DYSPLASIA especially in long
standing cases
Rapid urease test
Histopathology (special stains)
Culture
polymerase chain reaction
Urea breath test
Serology (lgG, lgA)
PCR in saliva and feces
DIAGNOSIS OF H.PYLORI
ENDOSCOPY BASED
INVASIVE TESTS
NON INVASIVE TESTS
Lab investigations depend upon cause
1. Achlorhydria
2. Increased gastrin levels
3. Auto antibodies to parietal cell antigens
4. Urease breath test
5. Endoscopic biopsy
Long term risk of gastric carcinoma esp. in
autoimmune type
Autoimmune
type
Acute Gastric Ulceration
Aka stress ulcers
Small mucosal erosions to deep lesions
involving the entire mucosal thickness
Can present with massive upper GIT bleed
Usually multiple and asymptomatic
5-10% of patients admitted in Intensive
Care Units
Severe trauma, major surgeries
Extensive burns (Curling ulcers)
Head injuries and other intracranial lesions
(Cushing Ulcers)
Pathogenesis is related to
Systemic acidosis and hypoxia (severe
trauma and burns)
Vagal stimulation (intra cranial lesions)
multiple small and circular
gastric rugae are normal
base is not indurated
adjacent gastric mucosa normal
Peptic Ulcer Disease
Ulcer is defined as breach in the mucosa
of the alimentary tract that extends
through the muscularis mucosae into the
submucosa or deeper
Are chronic, often solitary lesions that
occur in any part of the GIT exposed to
aggressive action of the gastric acid peptic
juices
Sites (Descending order)
Duodenum
Stomach
Gastro esophageal junction
Margins of gastrojejunostomy
Meckels diverticulum
Stomach, duodenum and jejunum in
Zollinger Ellison syndrome
Clinical Features
Burning epigastric pain 1-3 hours after
meals
Relieved by food and alkali esp. duodenal
ulcers
Worse at night
Associated weight loss
Gastric outlet obstruction
Etiopathogenesis
DAMAGING
FORCES
GASTRO DUODENAL
MUCOSAL DEFENSES
1) H.PYLORI- 70% of gastric and 90% of
duodenal ulcers
2) NSAIDS- inhibit prostaglandin synthesis
3) ZOLLINGER ELLISON SYNDROME-
multiple ulcers
4) SMOKING- impairs mucosal blood flow
5) ALCOHOL
6) PSYCHOLOGICAL STRESS
50% < 2cms
Round to oval, punched out with relatively
straight walls
Sharp and raised margins but not everted
Depth varies but may penetrate entire wall
Smooth and clean base
Radiating surrounding mucosal folds
Active ulcer has four zones (From inwards )
1) Necrotic fibrinoid debris
2) Non specific inflammatory infiltrate
(Predominantly neutrophilic)
3) Granulation tissue
4) Fibrosis and collagenous scar
Features of chronic gastritis in adjacent
mucosa
COMPLICATIONS
Bleeding
Perforation
Gastric outlet obstruction
MALIGNANT TRANSFORMATION -
Unknown in duodenal ulcer and
exceedingly rare in gastric ulcers
Gastric Carcinoma
ENVIRONMENTAL
GENETIC
HOST FACTORS
Blood Gp A, Family History, HNPCC
H. pylori, Chronic atrophic
Gastritis, Intestinal metaplasia
Partial gastrectomy,
Gastric adenoma
Diet,Smoking,Low SES
SITES
Pylorus, Antrum - 50-60%
Cardia 25%
Body and Fundus
GROWTH PATTERN
Exophytic
Flat
Exacavated
Classification
BASED ON DEPTH
EARLY GASTRIC
CARCINOMA
ADVANCED GASTRIC
CARCINOMA
Confined to mucosa and
submucosa
Regardless of
involvement of
regional lymph nodes
Classification
Neoplastic glands
resemble colon
Associated with
H. pylori
Intestinal
metaplasia is
precursor lesion
Don't form glands
Infiltrate as single cells
or small clusters
Intestinal metaplasia is not
precursor lesion
Linitis plastica
Signet ring cells
Role of E cadherin
HISTOLOGICAL
(LAURENS CLASSIFICATION)
INTESTINAL DIFFUSE
Spread
Regional
Transcelomic
Lymphatic
Hematogenous
The Virchows lymph node
Mainly asymptomatic
Weight loss, anorexia, abdominal pain
Pyloric obstruction
Krukenberg tumor
Left supraclavicular lymphadenopathy
(Virchows lymph node)
Umbilical nodule (sister Mary Joseph nodule)
Prognosis depends upon depth of invasion and
nodal status
Clinically
Gastro Intestinal Stromal Tumors
(GIST)
Mesenchymal tumors
Derived from cells of Cajal the pace maker
cells
Many of these previously k. a. Leiomyomas
Stomach, small intestine, large intestine
Can be benign or malignant
Usually submucosal
Whorls and bundles of spindle shaped cells
C-kit (CD 117) is the tumor marker
SMALL INTESTINE
Small Intestine-Normal histology
Lining epithelium has four types of cells
Columnar absorptive cells with microvilli
Goblet cells
Endocrine cells
Paneth cells (contain antimicrobial enzymes)
Pathological Lesions-overview
CONGENITAL
ANOMALIES
MALABSORPTION
SYNDROMES
INFLAMMATORY
BOWEL DISEASE
NEOPLASMS
Duplication
Malrotation
Heterotopia
Omphalocoele
Duodenal atresia
MECKELS
DIVERTICULUM
Celiac sprue
Tropical sprue
Whipples disease
Pancreatic insufficiency
Crohns disease
Ulcerative colitis
Adenomas
Adenocarcinomas
Carcinoid tumor
Lymphomas
Meckels Diverticulum
Incomplete of involution of vitelline duct
Rule of 2 - 2% of normal population
- with in 2 feet of ileocecal valve
- average 2 cm in length
On anti-mesenteric border
All layers of GIT (true diverticulum)
Some times lined by gastric mucosa or
pancreatic tissue
Commonly asymptomatic
COMPLICATIONS
Hemorrhage and Peptic ulceration
Intestinal obstruction
Diverticulitis
Perforation
Fistula
Malabsorption Syndromes
Suboptimal absorption of fats, fat soluble vitamins and other vitamins,
proteins, carbohydrates, electrolytes and water.
BASIC DEFECT CAN BE IN
Intra luminal
digestion
Terminal
digestion
Transepithelial
transport
CAUSES
-Celiac disease
-Pancreatic insuffiency
-Crohns disease
-Cholestatic liver disease
-Tropical sprue
-Whipples disease
-Giardiasis
(Giardia)
Clinical Features
Steatorrhea, Flatus, abdominal distension
Weight loss, failure to thrive (children)
Anemia
Muscle wasting, weakness
Osteopenia,Tetany
Amenorrhea
Infertility
CELIAC DISEASE (Gluten Sensitive
Enteropathy)
Common in whites (1 in 3000)
Age of presentation 1-10 years
Pathogenesis Sensitivity to gluten
Mucosa exposed to gluten
Accumulation of T & B lymphocytes
Damage to enterocytes
MALABSORPTION
In Predisposed
Persons (HLA
DQ2
and DQ8)
About 10% cases have dermatitis herpetiformis
Diffuse enteritis
Changes more marked in proximal part of
intestine
Marked atrophy and loss of villi (reduced area
for absorption)
Increased intraepithelial lymphocytes
Elongated and hyperplastic crypts
Increased number of lymphocytes,
macrophages and plasma cells in lamina propria
Reversal of changes after gluten free diet
Diagnosis
-Documentation of malabsorption
-Small intestine biopsy
-Reversal of changes and signs and
symptoms after gluten free diet
-Anti tissue transglutaminase
and anti endomysial antibodies
Small percentage associated with IgA deficiency
Long term risk of intestinal lymphomas (T
cell type)
Tropical Sprue (Post Infectious
Sprue)
In people living in or visiting tropics
Symptoms appear after months or even
years after visit
Pathogenesis is related to bacterial
infection superimposed on pre existing
small intestine injury
All parts of small intestine are involved
equally
Near normal to diffuse enteritis
Blunting of villi to complete flattening of villi
Difficult to differentiate from celiac disease
Responds to antibiotics
WhippleS Disease
Systemic disease
Mainly involves intestine, joints and CNS
Caused by a gram positive sickle shaped
bacilli Trepophyrema whippelii
More common in males (10:1)
Mucosa laden with distended
macrophages in lamina propria
Contain PAS positive granules
Rod shaped bacilli can be seen on EM
Villi are broad and expanded
Mesenteric lymphadenopathy can be seen
Similar bacilli laden macrophages in joints,
CNS
BUT No associated lymphocytic or
neutrophilic infiltration
Treated with Antibiotics
Clinically
Arthralgias, GIT sysmptoms
Weight loss, generalized
lymphadenopathy, low grade fever
CNS symptoms (dementia, seizures)
Hyperpigmentation in sun exposed areas
Neoplasms
3-6 % of all GIT tumors
Benign tumors are slightly more common
than malignant ones
BENIGN
MALIGNANT
Adenomas
Leiomyoma
Lipoma
Hemangioma
Neuroma
Adenocarcinoma
Carcinoid tumor
Lymphomas
Sarcomas
Carcinoid Tumor
Arise from endocrine cells
Tumors with low malignant potential
Secrete a variety of bioactive products &
hormones
Commonly secreted products are 5HT,
5-HIAA, histamine, bradykinin and
kallikrein
Metastasis to liver produces carcinoid
syndrome
Common sites-(descending order)
Small intestine (ileum)-usually multiple
Rectum
Stomach
Appendix (tip)
Colon
Usually located in submucosa
Cut Surface- solid & yellow tan
Fibrosis of mesentery so leads to kinking and
obstruction
Those less than 1 cms and rectal and
appendiceal carcinoids do not metastasize
Islands, trabeculae, nests and sheets of
monomorphic cells
Have round to oval stippled nuclei (salt and
pepper appearance)
Minimal pleomorphism
EM dense core granules
IHC - chromogranin A
Many are asymptomatic
Kinking and obstruction of bowel due to
fibroblastic reaction
Carcinoid syndrome-
Vasomotor disturbances
wheezing, dyspnea,
Intestinal hypermotility
Systemic fibrosis, may lead to right sided valvular
lesions
Overall 5 year survival rate is 50%
GIT Lymphomas
1-4% of all GIT malignancies
Sites
Stomach (50%)> small intestine (37%)>
colon and rectum
By definition no bone marrow, liver or
spleen involvement at the time of diagnosis
(However regional lymphadenopathy can
be present)
Risk factors
patients with helicobacter gastritis
people living in mediterranean,
Immunodeficient persons
celiac disease
Types of Lymphoma
Sporadic
Mediterranean
Sprue associated (celiac disease)
Sporadic: (most common)
Arise from B cells of MALT tissue
MC site Stomach, SI and Colon
Appendix and esophagus RARE
Remain localized for long time
t(11:18) is characteristic
Morphology
Plaque like expansions of mucosa and
submucosa
Some times diffuse mural thickening
Diffuse sheets of small cleaved lymphoid
cells
Infiltrate and destroy epithelial glands
(Lymphoepithelial lesions)
APPENDIX
Appendix-Normal
Average length 7 cm
Four layers as rest of GIT
Colonic type of epithelium
But mucosa and submucosa rich in
lymphoid tissue
Acute Appendicitis
Inflammation of the appendix
Underlying obstruction of the lumen in 50-
80% cases
Pathogenesis unclear in non obstructive
cases
Pathogenesis
Obstruction
Continued secretion of mucinous fluid
Increased intraluminal pressure
Collapse of draining veins
Ischemic injury
Bacterial proliferation
Inflammation and edema
Edematous and congested appendix
Serosa dull and granular
Fibropurulent exudate
Severe cases gangrene
Neutrophilic exudate in mucosa, sub
mucosa and muscularis propria
Edema and congestion
Neutrophilic infiltration of the
muscularis propria is the criteria for
diagnosis
Clinically
Adolescents and young adults
Pain is the most common symptom,
initially periumblical then localizes to right
quadrant
Nausea, vomiting
Tenderness (McBurneys point)
Mild fever, Leucocytosis (Neutrophilia )
Complications
Perforation
Peritonitis
Periappendiceal abscess
Liver abscess
Bacteremia
Tumors of the Appendix
Rare
Carcinoid tumors (usually incidental
finding)
Adenomas
Adeno carcinomas (mucin producing)
Mucocele
Swelling of the appendix because of accumulation
of inspissated mucus
Obstruction
Mucinous tumors
Mucinous
cystadenocarcinoma
Mucinous
cystadenoma
Rupture
Intraperitoneal
spread
PSEUDOMYXOMA PERITONEI
Fecolith
Stricture
Other cause
-Mucinous tumors
of the ovary
LARGE INTESTINE
Large Intestine-Normal histology
Flat mucosa (no villi)
Straight tubular crypts
Epithelium composed of
Columnar absorptive cells
Goblet cells
Endocrine cells
Pathological lesions-overview
Congenital
malformations
Inflammatory
lesions
Tumors & tumor
like lesions
Hirschsprungs
Disease
Colitis
IBD
Non neoplastic polyps
Adenomas
Adenocarcinomas
Carcinoid
Lymphomas
Most common cause of congenital intestinal
obstruction
Incidence is 1 in 5000 to 1 in 8000
M:F is 4:1
In 10% patients of Down Syndrome
Rectum is always affected
Absence of ganglion cells in Meissner and
Auerbachs plexus
Dilatation and hypertrophy proximal to
aganglionic segment (congenital megacolon)
Hirschsprungs Disease
Defect in migration and survival of neuroblasts
Congenital absence of ganglion cells
Functional obstruction
DILATATION PROXIMAL TO OBSTRUCTION
(megacolon)
Clinical features
Delayed passage of meconium
Constipation
Abdominal distension
Diagnosed by rectal biopsy
Complications -Enterocolitis
-Perforation and peritonitis
Diverticular disease
Includes diverticulosis and diverticulitis
Common in western world
50% in older than 60 years
Flask like structures ( 95% sigmoid colon)
extending from lumen through muscular layer
Pathogenesis
Lack of dietary fiber leads to sustained bowel
contractions and increased intraluminal
pressure
Herniation of colonic wall at sites of focal
defects
Not true diverticula
Clinically
Divertculosis is usually asymptomatic but
some times painless bleeding
Diverticulitis
Lower abdominal pain
Constipation, diarrhea, flatulence
Bleeding
fever
Diverticulitis can lead to perforation
Colitis
common causes
1) Infections bacterial
viral
protozoal (Amebic colitis)
2) Necrotizing enterocolitis
3) Antibiotic associated colitis
(Pseudo membranous colitis)
2) Ischemic colitis
3) Idiopathic-Inflammatory bowel disease
Clinical features of colitis
Diarrhea
Mucoid
Bloody
Abdominal pain (Usually below umbilicus)
Abdominal cramps
Tenesmus (painful defecation)
Amebic Colitis
Common in developing countries
Caused by Entamoeba histolytica
Cecum and ascending colon
Invades the crypts and burrows into
submucosa
Flask shaped ulcers
Little inflammatory infiltrate in the ulcer
Can produce Amebic liver abscess
Pseudo Membranous Colitis
Associated with broad spectrum antibiotic
use
Caused by Clostridium difficile
Toxin mediated damage
Colon, particularly rectosigmoid, exhibits
raised yellow plaques
Fibrinopurulent-necrotic debris
(PSEUDOMEMBRANES)
Surface epithelium denuded
Superficially damaged crypts distended by
mucopurulent exudate which erupts to
form a mushrooming cloud
Coalescence of these clouds leads to
pseudo membrane formation
Transmural (all layers)
Mural (mucosa and submucosa)
Mucosal (not deep than mucosa)
Ischemic bowel disease
1.Arterial thrombosis- systemic atherosclerosis,
systemic vasculitis, hypercoagulable states,
angio- graphic procedures
2.Arterial embolism- cardiac vegetations, athero-
thromboembolism and angiographic procedures
3.Venous thrombosis- hypercoagulable states,
Oral contraceptives, intraperitoneal, sepsis, post
operative etc.
4.Non occlusive ischemia- cardiac failure,
shock, dehydration
5.Miscellaneous- radiation, volvulous,
herniation
PREDISPOSING CONDITIONS
Clinical Features
Common in elderly
Severe abdominal pain and tenderness
Nausea, vomiting and bloody stools
Shock and vascular collapse
Mortality is very high (50-75%)
Mild features in mucosal and mural
infarction
Gangrene, perforation, peritonitis
Hemorrhagic infarction
Intestine is edematous, congested and red
purple in color
Lumen may contain frank blood
Demarcation from normal mucosa is
sharply defined in arterial causes (Not in
venous)
Chronic Ischemic colitis
Develops insidiously
Chronic inflammation and fibrosis
Stricture formation ( Splenic flexure-the
water shed area)
Intermittent attacks of pain- Intestinal
angina
Can mimic Inflammatory bowel disease
Idiopathic Inflammatory Bowel
Disease
Includes Crohns disease (CD) and
Ulcerative colitis (UC)
Are chronic relapsing, inflammatory
disorders of obscure origin
CD- Granulomatous inflammation which
can affect any part of GIT from mouth to
anus
UC- Non granulomatous inflammation
limited to colon
Etiopathogenesis
GENETICS
? INFECTIONS
ABNORMAL
HOST
REACTIVITY
Activation of inflammatory
cells (PMNs)
cytokines & mediators
tissue injury
Net result
Crohns Disease
Synonyms- Terminal ileitis, regional ileitis and
granulomatous colitis
characterized by
Sharply delimited & transmural involvement by an
inflammatory process
Presence of non caseating granulomas
Mucosal fissuring with formation of fistulas
Common disease in U.S.(3 in 100,000)
Adolescents and young adults
More common in Jews
Crohns disease involves
Small intestine alone 30%
Small intestine and colon 40%
Colon alone 30%
Serosa dull and granular
Creeping up of mesenteric fat because of
fibrosis
Mesenteric lymphadenopathy
Mesentery thickened and fibrotic
Long narrow thickened segments of small
intestine (unlike tubercular strictures which
are short in length)
Wall is thickened (because of edema,
hypertrophy, fibrosis and inflammation)
Lumen is narrow (radiographically- String
sign)
Morphology
Focal mucosal ulcers which may coalesce to
form linear ulcers
Intervening mucosa is relatively spared but
coarsely textured- Cobble stone appearance
Mucosal fissures , if penetrate deeply
Fistulas Sinus tract Perforation Localized abscess
1.Neutrophilic infiltration of the epithelium and
crypt abscesses
2.Architectural distortion- Villous blunting,
irregular branching crypts and crypt atrophy
3.Ulcerations (deep mucosal fissures)
4.Transmural inflammation- lymphoid
aggregates scattered through out the wall
(mucosa,submucosa,mucularis propria and
serosa)
5.Non caseating granulomas (50% cases) In all
layers even in regional lymph nodes BUT
absence of granulomas does not preclude
diagnosis
6.Fibrosis of mucosa ,sub mucosa and muscularis
propria
Clinical features
Low grade fever, malise, weight loss
Intermittent attacks of nonbloody diarrhea,
Steady or cramping right lower quadrant pain
Palpable tender mass in right lower quadrant
Features of malabsorption
Small intestine- features of subacute intestinal
obstruction
Large intestine- features of colitis
Perianal disease- fissures, fistulas and
abscesses
Extra intestinal Manifestations
Seen in both CD and UC (though more
common with Ulcerative colitis)
Can develop even before the onset of GI sign
symptoms
Migratory polyarthritis, Sacroileitis , Ankylosing
spondylitis
Erythema nodosum
Clubbing of finger tips
Sclerosing cholangitis
Aphthous ulcers, gall stones
5-6 times increased risk of developing
malignancy (Less than UC)
Ulcerative Colitis
Ulcero-inflammatory disease limited to
colon and affecting mucosa and sub
mucosa
Extends in continuous fashion
More common in females
Also more common in whites
Peak between 20 and 25 years
Starts in rectum and then extends
proximally to involve whole of the colon
Clinical features
Relapsing disorder
Attacks of bloody mucoid diarrhea
Lower abdominal pain, abdominal cramps
Tenesmus
Cramping abdominal pain relieved by
defecation
Flare ups with physical and mental stress
Fever and weight loss in severe cases
10% cases back
wash ileitis
Serosa is usually
normal
Mucosa red granular
and friable
Broad based ulcers
Isolated islands of
regenerating mucosa
bulge in between to
create pseudopolyps
INTESTINAL ULCERS
Duodenal ulcers
Typhoid
Tuberculosis
Crohns disease
Ischemic colitis
Amebic colitis
Infective causes
Carcinomas
Severe cases toxic damage to muscularis
propria and neural plexus shut down of
neuromuscular function- TOXIC
MEGACOLON
Mucosal inflammation, cryptitis, crypt
abscesses, crypt distortion
Long standing cases nuclear atypia and
features of dysplasia
When carcinoma arises, usually multicentric
Risk of carcinoma increases with pancolitis
and duration of disease ( > 10 years)
GIT Polyps
Non neoplastic
Neoplastic
Inflammatory Non inflammatory Benign Malignant
A polyp is a mass that protrudes into the lumen of
the gut
Hyperplastic
Hamartomatous
Lymphoid
Tumors of the Colon And
Rectum
EPITHELIAL
MESENCHYMAL
LYMPHOID
Lymphomas
BENIGN
BENIGN
MALIGNANT
MALIGNANT
Adenomas
Tubular
Tubulovillous
Villous
Adeno-
carcinoma
carcinoid
GIST
Lipoma
Neuroma
Angioma
GIST
Kaposi
sarcoma
GIST-Gastrointestinal Stromal Tumors
Hyperplastic Polyp
Small nipple like protrusions
Common in elderly
Well differentiated glands and crypts lined
by non neoplastic epithelium
Because of delayed shedding there is
crowding, infoldings and serrated epithelial
profile
MOST LIKELY NO MALIGNANT
POTENTIAL
Juvenile Polyp
Common in children < 5 years of age
Rectum is most common site
In adults aka retention polyp
Rarely juvenile polyposis syndrome (AD)
Usually 1-3 cms, lobulated with stalk
Lamina propria forms the bulk and
encloses abundant cystically dilated
glands
Inflammatory cells can be present
NO MALIGNANT POTENTIAL
Peutz Jeghers Polyp
Hamartomatous polyp
P.J. syndrome is autosomal dominant
Multiple polyps in whole GIT
Melanotic pigmentation in mucocutaneous
areas, lips, perioral areas, face, genitalia
and palms
Are large pedunculated lesions
Arborizing network of connective tissue
and smooth muscle extending into the
polyp and surrounds glands
Glands are lined by an epithelium rich in
goblet cells
No malignant potential per se
BUT such patients are at risk of
developing carcinomas of pancreas,
breast, lung, ovary and uterus
Normal mucosa
Hyperplastic
polyp
Juvenile
Polyp or retention poly
Peutz Jeghar
polyp
Lymphoid
polyp
Saw tooth
or
Summary of non neoplastic polyps
Adenomas
All arise as a result of epithelial
proliferative dysplasia
Three types-tubular, villous and
tubulovillous
Are precursor lesions of carcinoma
Risk increases with
Polyp size (most important criteria)
Histologic architecture
Severity of dysplasia
Tubular Adenomas
Usually solitary
90% colon
Stalked and coarsely lobulated
Adenomatous epithelium lines the glands,
seen as tall, hyperchromatic and
disordered epithelium
Invasion of the submucosal stalk
constitutes invasive carcinoma
Villous Adenomas
Larger and more ominous
Sessile, upto10 cm, velvety and
cauliflower like
Frond like villiform extensions of the
mucosa
No stalk so no buffer zone
Direct invasion of the sub mucosa
Tubulovillous Adenomas
Contain both tubule
like and villi like
structures
Clinical Features
Clinically most tubular and tubulovillous
adenomas are usually asymptomatic
May present with features of anemia
because of occult blood loss
In villous adenoma loss of fluid, proteins
and electrolytes-hypoproteinemia,
hypokalemia
Rarely intussception
All adenomatous polyps are considered
potentially malignant
Familial Polyposis Syndromes
FAP-genetic defect in APC gene on
Chromosome 5
Usually tubular type
Some times in small intestine and stomach
Typically 500-2500 mucosal adenomas
(minimum number for diagnosis is 100)
Attenuated FAP
Carcinoma occurs in young individuals
Prophylactic colectomy is done
Gardner Syndrome- Tubular adenomas with
multiple osteomas and epidermal cysts
Turcot Syndrome- adenomas and CNS gliomas
Colorectal Carcinoma
Occurs in elderly individuals
Occurs in young individuals in setting of
ulcerative colitis and polyposis syndromes
Risk factors:
Age ( 90% in older than 50 years)
dietary habits, diet low in indigestible fiber and rich in
animal fat
obesity, physical inactivity
Family history
Antioxidants protective
80-90% cases arise in an adenomatous polyp
(adenoma-carcinoma sequence)
10-15% cases defect in DNA mismatch repair
genes e.g. MLH1 ( HNPCC)
Adenoma-carcinoma sequence
Usually solitary
More than half occur proximal to splenic
flexure
Multiple sites-ulcerative colitis, polyposis
syndromes and HNPCC
Proximal colon- expohytic polypoidal
lesions and obstruction is uncommon
Distal colon- annular, encircling napkin
ring constrictions
Neoplastic glands invading submucosa
and muscularis propria
10-15 % can be mucin producing
Spread to regional lymph nodes is
frequent
Systemic liver, lung and bones
Prognosis -most important factor is extent
of tumor at the time of diagnosis
Clinical Features
Asymptomatic
RIGHT SIDED- fatigue, weakness & iron
deficiency anemia (Bulky & bleed easily)
LEFT SIDED- altered bowel habits
Tumor marker - CEA
Iron deficiency anemia in elderly man is
due to GIT malignancy unless proved
otherwise
Hereditary Non Polyposis Colorectal
Cancer (HNPCC)
Aka Warthin-Lynch Syndrome
Defective DNA repair genes (AD)-MLH1 gene
Fewer number of polyps as compared to FAP
More common on right side (proximal colon)
Multiple sites
Mucinous histology , increased number of
intratumoral lymphocytes
Associated with carcinomas of other sites
endometrium, ovaries, stomach, small intestine,
biliary tract
Guppal GIT Path SGUSOM
LIVER,GALL BLADDER AND LIVER,GALL BLADDER AND LIVER,GALL BLADDER AND LIVER,GALL BLADDER AND
PANCREAS PANCREAS PANCREAS PANCREAS
Liver-normal
Normal weight 1400-1600 gm
Anastamosing cords of hepatocytes (2 cell
thick)
Sinusoids lined by fenestrated endothelial
cells and Kupffer cells
Space of Disse -contains Ito cells
Clinical features-overview
Symptoms:
Right upper quadrant pain
Anorexia
Yellowish discoloration of skin
Pruritus
Fever
Mental confusion
Easy bruising
Signs:
Icterus
Hepatomegaly
Splenomegaly
Palmar erythema
Spider angiomas
Gynecomastia
Testicular atrophy
Purpura
Skin pigmentation
Tenderness in right hyopchondrium
Chronic liver disease
Laboratory investigations
Serum bilirubin Direct and Indirect
Transaminases Alanine amino transferase
(ALT) and Aspartate amino transferase (AST)
ALT is more specific for liver injury than AST.
ALT (Cytosolic enzyme) and AST (Primarily
Mitochondrial)
In Alcoholic Liver Disease AST/ALT >2
Viral hepatitis AST/ALT <1
Alkaline phosphatase- Levels are increased in
obstructive liver disease
-glutamyl transferase (GGT) Alcoholic liver
disease
Serum albumin- Decreased levels in chronic
liver diseases
Prothrombin time- Prolonged in acute and
chronic liver diseases
Serum ammonia levels-raised in hepatic failure
Immunological markers -Anti smooth muscle
antibodies, anti mitochondrial antibodies
Viral markers and antibodies to various viral
proteins
Ultrasonography
Endoscopic Retrograde Cholangio
Pancreatography (ERCP)
FNAC
Percutaneous needle biopsy
Jaundice
Yellowish discoloration of skin
Yellowish discoloration of sclera-Icterus
Because of accumulation of bilirubin
When bilirubin levels > 2.0 mg/dl it is
clinically evident
Both conjugated & unconjugated bilirubin
can accumulate
CHOLESTASIS -in addition to bilirubin
retention of bile salts and cholesterol
163
Bilirubin & Bile Formation
Aged RBCs
Hepatic hemoproteins Minor
Premature destruction of RBC component
precursors in bone marrow
Heme Biliverdin
Heme oxygenase
(MPS)
Reductase
(MPS)
Released in circulation
Bound to albumin
Bilirubin
Carrier mediated uptake
At sinusoidal membrane
Conjugation in ER
Bilrubin glucouronides
(water soluble) in bile
Bacterial deconjugases
In intestine
Urobilinogens
Feces
Reabsorbed
Small amount in urine
1
2
3 4
5
6
1- 4 unconjugated hyperbilirubinemia
5,6 conjugated hyperbilirubinemia
Jaundice
Conjugated bilirubin
Water soluble
Excreted in urine
Loosely bound to
albumin
Aka direct bilirubin
Unconjugated bilirubin
Water insoluble
Lipid and alcohol
soluble
Can not be excreted in
urine
Tightly bound to
albumin
Aka indirect bilirubin
Present Absent
Absent Present in excess
Present Absent
Urine Bilirubin
Urine
urobilinogen
Urine bile salts
Obstructive
jaundice
Hemolytic
jaundice
Predominantly Unconjugated
Hyperbilirubinemia
PREHEPATIC:
Excess Production Of Bilirubin Hemolytic
anemias, resorption of blood from internal
hemorrhages, ineffective erythropoiesis
HEPATIC:
a) Reduced Hepatic Uptake- Drugs interfering
with carrier system & few cases of Gilbert
syndrome
b) Impaired Bilirubin Conjugation- Diffuse
hepatocellular damage e.g. viral hepatitis,
cirrhosis etc., Physiologic jaundice of new
born, breast milk jaundice, Crigler-Najjar
syndrome I & II
Predominantly Conjugated
Hyperbilirubinemia
c) Decreased Hepatic Secretion Of
Bilirubin Glucuronides- deficiency in
membrane transporters (Dubin Johnson
and Rotor syndromes), hepatocellular
damage (Viral hepatitis), Drugs,
POSTHEPATIC:
Extrahepatic Biliary Obstruction
Gall stones, Ca head of pancreas, Ca of
extrahepatic bile ducts, Biliary atresia
Cholestatic liver disease
Caused by hepatocellular dysfunction/intra
or extra hepatic biliary obstruction
Pruritus
Skin xanthomas
Features of fat malabsorption
Elevated alkaline phosphatase
Extra hepatic biliary obstruction is
amenable to surgery
Cirrhosis
Defined by three characteristics
a) Bridging fibrous septae
b) Parenchymal nodules created by
regeneration
c) Architectural disruption
Changes should be diffuse not focal
Micronodular < 3mm nodules
Macronodular > 3 mm nodules
170
Pathogenesis
Hepatocellular
Injury
Regeneration
Progressive
fibrosis
CIRRH
OSIS
Causes of Cirrhosis
Alcoholic liver Disease (Most common)
Viral hepatitis
Biliary tract diseases
Hereditary hemochromatosis
Wilsons disease
Alpha 1 anti trypsin deficiency
Cryptogenic cirrhosis
Rare
Pathogenesis
Chronic inflammation
(TNF-,TGF- & IL-10)
Cytokine production by endogenous
Cells (Kupffer cells, hepatocytes)
Disruption of ECM
Direct stimulation of
Ito cells by toxins
Stimulate Ito cells
DEPOSITION OF COLLAGEN I & III
IN SPACE OF DISSE & HEPATIC LOBULES
Loss of fenestrations
In endothelial cells
Impaired hepatocellular
Secretion of proteins (Albumin,
Clotting factors)
New vascular channels
In fibrous septae
Shunting of blood
Obstruction of
Biliary channels
Jaundice
Clinically
May be clinically silent
Non specific signs and symptoms
Signs and Symptoms are usually because
of
Portal hypertension
Ascites
Splenomegaly
Features of Liver failure
Portal Hypertension
Simply means increased resistance to
portal blood flow
Can also occur in many disorders other
than cirrhosis
Normal= 5-10 mm Hg
Consequences of Portal
Hypertension
Ascites Portosystemic shunts
Sites-
Cardioesophageal junction
Rectum
Retroperitoneum
Abdominal wall
Congestive
Splenomegaly
Hepatic
Encephalo-
pathy
Clinically as
Esophageal varices
Hemorrhoids
Caput medusae
Viral Hepatitis
Most common infective disease of the liver
Many systemic viral infections also involve liver
e.g. Infectious mononucleosis, CMV, yellow
fever etc.
However the term viral hepatitis is used for
group of diseases caused by a group of
hepatotropic viruses
They are hepatitis viruses A to E
Virological aspects will be taught in microbiology
course
Hepatitis A
Caused by single stranded RNA virus
Incubation period is 2-6 weeks
Does not cause chronic hepatitis
No carrier state
Common in children
Spread is by feco-oral route
Diagnosis- IgM appear at the onset of sign
and symptoms
After few months IgG
Hepatitis B (Serum Hepatitis)
Caused by enveloped DNA virus
Long incubation period 4-26 weeks
Present in all pathological and
physiological fluids
Transmission occurs by
Transfusion of blood and blood products
Sexual intercourse
I/V drug abuse
Homosexuals
Needle stick injuries
Structure of HBV
Nucleocapsid core-
Has core antigen
(HBcAg) and HBeAg
Envelope glycoprotein
is HBeAg
DNA polymerase
HBX role in
hepatocellular
carcinoma
Serologic Diagnosis
Pre symptomatic stage HBsAg, HBcAg
and DNA polymerase
With onset of Symptoms-Anti HBc IgM
Later on- Anti HBe and Anti HBs
Window period- Anti HBc IgM
Replication markers- HBeAg, DNA
polymerase and HBsAg
Hepatitis C
Most important cause of transfusion
associated hepatitis and chronic liver
disease
Incubation period is 1-3 weeks
Much more propensity for chronicity
Single stranded RNA virus
Inherently unstable virus so no vaccine yet
PERSISTENT INFECTION and CHRONICITY ARE
HALL MARK
Hepatitis D
Is replication defective RNA virus
Infective only when encapsulated by
HBsAg
Either co-infection or established HBV
infection before (super infection)
Super infection is more dangerous
Hepatitis E
Enterically transmitted, as endemics
Unenveloped single stranded RNA virus
NO CHRONICITY
Usually a self limited illness
But high mortality in pregnant females
Severe cholestasis which persists for
longer period
Clinico pathologic Syndromes
(viral hepatitis)
Asymptomatic infection
Carrier state
Acute hepatitis
Chronic hepatitis
Fulminant hepatitis
Asymptomatic Infection
Identified incidentally
Elevated transaminases
Carrier State
Healthy carriers
Chronic carriers
Harbour virus
Can transmit Infection
Usually have no signs & symptoms
Normal liver biopsy
Ground glass hepatocytes
Features of chronic hepatitis in hepatitis C
Acute Viral Hepatitis
Incubation period
Pre-icteric or symptomatic phase
Icteric phase
Convalescence
Signs and symptoms usually abate with onset of
jaundice
Predominantly conjugated type
Enlarged reddened liver some times greenish
because of cholestasis
Morphology of Acute Viral
Hepatitis
ARCHITECTURE
HEPATOCYTES SINUSOIDS PORTAL TRACTS
Ballooning
degeneration
Necrosis (Lytic/
bridging / confluent
/ massive)
Regeneration
(disarray)
Reactive changes
Kupffer cell
hyperplasia
Contain cell
debris
Inflammation
Mononuclear
May spill into
adjacent
hepatocytes (k.a.
Interface
hepatitis)
Lobular disarray
190
Acute Viral Hepatitis
Chronic Hepatitis
Symptomatic, biochemical or clinical evidence of
continuing or relapsing hepatic disease of > 6
months duration in addition to histological
documentation of inflammation and necrosis
Mainly by HBV and HCV
Clinically non specific Signs and symptoms
On examination- Mild hepatomeagly, spider
angiomas, palmar erythema and mild
splenomeagly
Prothrombin time prolonged
Other causes of chronic hepatitis
Alcoholism
Wilsons disease
Alpha 1 anti trypsin deficiency
Drugs
Autoimmune
Chronic Viral Hepatitis
Shares some features with acute viral
hepatitis
Hepatocyte injury, necrosis, regeneration
Portal tracts show mononuclear
inflammation
Fibrosis- can be portal, periportal or
bridging fibrous septae
In HCV - a few special features
Bile ductular proliferation in portal tracts
Presence of lymphoid aggregates
Fatty change of the hepatocytes
Fulminant Hepatitis
Denotes clinical hepatic insufficiency that
progresses rapidly to hepatic
encephalopathy with in 2-3 weeks
Viral hepatitis is the most important cause
(60-70% cases)
Remaining cases - drug and chemical
toxicity
Rare causes include ischemic necrosis,
acute fatty liver of pregnancy, Wilsons
disease
Fulminant Hepatitis
Morphology is same regardless of
causative agent
Because of massive loss of liver
parenchyma liver is shrunken
Acute yellow atrophy
Massive destruction of hepatocytes
involving adjacent lobules
Collapsed reticulin framework
Little inflammation in first week
Autoimmune hepatitis
Common in females
Morphological features of chronic hepatitis
Young female with jaundice, spider nevi, acne , hirsutism
Amenorrhea can occur
Extrahepatic autoimmune disease can be seen e.g.
Sjogren syndrome, arthritis, ulcerative colitis
INVESTIGATIONS:
Hyperbilirubinemia, elevations in aminotransferases
hypergammaglobulinemia
Anti nuclear and anti smooth muscle antibodies
Anti LKM (Liver kidney microsomal type) antibodies
Respond to immunosuppressive therapy
Alpha-1 Antitrypsin Deficiency
Autosomal recessive disorder
Markedly low levels of alpha antitrypsin (is
protease inhibitor)
Is a glycoprotein encoded by PiMM gene on
chromosome 14
Synthesized by liver
Most allelic variants have slightly reduced levels
PiZZ is homozygote form (markedly levels)
Alpha-1 Antitrypsin Deficiency
Associated hepatic syndromes are variable
Neonatal hepatitis with or without cholestasis
Smoldering chronic inflammation
Cirrhosis at early age
characterized by round to oval globular
cytoplasmic inclusions
These are acidophilic and indistinct on H & E
strongly PAS positive
Risk of hepatocellular carcinoma
Liver transplantation is the only treatment
Alcoholic Liver Disease
3 patterns of liver disease
Hepatic steatosis
Alcoholic hepatitis
Cirrhosis
201
ALCOHOL
Shunting of normal
substrates towards
lipid synthesis,
Defective
assembly of lip-
oproteins
Catabolism of fat
Induction of P-450
Toxic metabolites
Free radicals
In MES
Acetaldehyde
Lipid
peroxidation
Hepatocyte-acetal
dehyde adducts
Immunologically altered
hepatocytes
Susceptible to
Immunological attack
By alcohol
oxidation React with memb
ranes & proteins
Hepatic injury
Steatosis
Necrosis
Inflammation Fibrosis
ALD
Morphology Of Alcoholic
Steatosis
Occurs even with moderate intake
Liver is grossly enlarged, is soft and
greasy
Microvesicular and Macrovesicular
Fat granulomas or lipogranulomas
Alcoholic Hepatitis
1) Hepatocyte swelling (water and lipids)
and necrosis
2) Mallory bodies-Tangled skeins of
intermediate filaments
Are seen as eosinophilic cytoplasmic
inclusions
Other causes of Mallorys
hyaline-
Primary biliary cirrhosis
Wilsons disease
Chronic cholestasis
NASH (Non Alcoholic
Steato Hepatitis)
Neutrophilic reaction- neutrophils
permeate lobules and accumulate around
degenerating hepatocytes (especially
those having Mallorys hyaline)
Admixed macrophages and lymphocytes
are also present
Fibrosis- Perivenular initially
Later on perisinusoidal aka Chicken wire
fibrosis
Steatosis
Steatonecrosis
Mallorys hyaline
Highly suggestive of
Alcoholic hepatitis
Alcoholic Cirrhosis
Initially enlarged yellow fatty liver later
over the span of years brown, non fatty
and shrunken
Is micronodular
Macronodules may be formed at a later
stage
Jaundice, ascites
Wasted extremities, palmar erythemas
Esophageal varices
Splenomeagly
Spider angioma, palmar erythema,
testicular atrophy
Elevated levels of alkaline phosphatase,
gamma glutamyl transferase
Elevated serum transaminases AST/ALT
ratio >2
Prolonged prothrombin time
Hypoproteinemias reversal of albumin
globulin ratio
Long term outlook varies
Important causes of death include
Hepatic coma
Upper GIT bleed
Intercurrent infections
Hepatorenal syndrome
Rarely hepatocellular carcinoma
Non alcoholic fatty liver disease
(NAFLD or NASH)
Changes like ALD in non drinkers
Steatosis with or without hepatitis
Risk factors
Obesity
Insulin resistance
Hyperlipidemias
Usually asymptomatic
Mild elevation of serum transferases
May lead to cirrhosis
Hemochromatosis
Excessive accumulation of iron
Characterized by
Micronodular cirrhosis
Diabetes mellitus
Skin pigmentation
Can be genetic (primary) or acquired
(secondary)
Primary- AR (HFE gene on chromosome 6)
-increased Fe absorption
- Common in males
Causes of Secondary
Hemochromatosis
Parentral iron overload
-Repeated blood transfusions
-Iron dextran injections
Ineffective erythropoiesis
-beta thalassemias
-other chronic hemolytic anemias
Increased oral intake
- Bantus disease
Chronic liver disease
Pathogenesis
EXCESSIVE IRON ACCUMULATION
Iron catalyzes free
Radical formation
Lipid peroxidation
Stimulation of
collagen
synthesis
Direct damage
to DNA
HEAPTIC INJURY
AND FIBROSIS
Deposition of iron in
Liver
Pancreas
Myocardium
Pituitary
Adrenals
Thyroid and Parathyroid
Joints and skin
In liver:
Initially golden yellow pigment in cytoplasm of
periportal hepatocytes
Later on whole of the lobule, bile duct
epithelium and even in Kupffer cells
NO INFLAMMATION
Ultimately cirrhosis
Hepatic iron concentration increased
(normal < 1000gm/gm dry weight of liver)
Iron is stained with Prussian blue
Hepatomegaly, abdominal pain
PANCREAS
Intensely pigmented
Diffuse interstitial fibrosis
Hemosiderin in both acinar and Islet cells
Diabetes mellitus
HEART
Hemosiderin in myocardial fibres
Delicate interstitial fibrosis
SKIN
Slate gray coloration
Iron in dermal melanophages
Increased melanin production
Clinical Features
Genetic variety rarely manifests before 40
More common in males
Hepatomeagly
Abdominal pain
Skin pigmentation (Sun exposed areas)
Diabetes mellitus
Cardiac dysfunction
Clinical Features
Death because of cirrhosis or cardiac
disease
200 fold increased risk of hepatocellular
carcinoma
Screening is done by serum iron and
ferritin levels
Genetic studies (In patients with family
history)
Wilsons Disease
Autosomal recessive disorder
Gene on chromosome 13
Characterized by accumulation of toxic
levels of Cu in Liver, Brain and Eyes
WILSONS DISEASE
Dietary copper
After absorption
goes to liver
In hepatocytes
Cu+2 globulin
Plasma
Senescent
ceruloplasmin
Defect in ATP7B gene
(chromosome 13)
Cu transporting ATPase
On canalicular memebrane
Ceruloplasmin
Excreted in bile
Cu
Excessive cu in hepatocytes
TOXIC INJURY TO LIVER
Spills in blood and damages
Brain, eyes and other organs
Morphology
In liver variable morphology
Fatty change
Acute hepatitis
Chronic hepatitis
Ultimately cirrhosis
Rarely massive necrosis
BRAIN
Deposited in basal ganglia,
may even produce cavitation
EYES
Deposits of copper in Descemets membrane
of corneal limbus k.a. Kayser Fleischer ring
In some sun flower cataracts
Clinically
Presentation is rare before 6 years of age
Features of acute or chronic liver disease
Neuropsychiatriac manifestations, chorea
In some patients -hemolytic anemia
Investigations
Decreased serum ceruloplasmin levels
Increased hepatic Cu (>250gm/gm dry
weight of liver is diagnostic)
Increased urinary copper excretion
222
Secondary
Biliary Cirrhosis
Caused by extra hepatic obstruction
Cholestasis
Secondary
inflammation
Fibrosis
Secondary
bacterial infection
Ascending cholangitis
Yellow green pigmented liver
Cut surface- Hard and granular with
coarse fibrous septae
Embedded bile ducts in septae (May
contain inspissated bile)
Extensive proliferation of small bile ducts
and edema
Marked feathery degeneration of
hepatocytes and bile lakes
Primary Biliary Cirrhosis
Chronic, progressive and fatal liver
disease
Possibly autoimmune
Non suppurative, granulomatous
destruction of medium sized bile ducts
Disease of middle aged women(6:1)
225
Pathogenesis
Autoimmune etiology
Aberrantly expressed
mitochondrial Ag on surface
of biliary epithelium
Auto reactive T cells
ANTI MITOCHONDRIAL
ANTIBODIES
DESTRUCTION OF
BILE DUCTS
Other associated autoimmune
Disorders may be present
bile ducts destroyed by granulomas (Florid
duct lesion)
Bile duct proliferation in portal tracts
upstream the obstruction
Dense infiltrate of lymphocytes, plasma
cells, macrophages and occasional
eosinophils
Ultimately cirrhosis (indistinguishable from
secondary biliary cirrhosis)
Clinically
Pruritus
Jaundice develops late
Hepatomeagly
Raised alkaline phosphatase levels
Raise cholesterol levels, xanthomas
Anti mitochondrial antibodies in 90%
Death usually occurs because of liver
failure
228
Primary sclerosing cholangitis
Inflammation, fibrosis and dilatation of intra
and extra hepatic ducts
M>F
Two third have chronic ulcerative colitis
? Gut derived toxins
? Immunological mediated
Onion skin fibrosis of the bile ducts
Lymphocytic infiltrate of the portal tracts
Strictures, ERCP- beading of the biliary tree,
Cholestasis
P-ANCA in about 80% cases
Neoplasms of The Liver
Malignant
Hepatoblastoma
Hepatocellular
carcinoma
Cholangiocarcinoma
Metastasis
Benign
Hepatic adenomas
Long term oral
contraceptive
use
Can be picked
incidently
May produce
hemorrhage
Hepatocellular Carcinoma
Aka Hepatoma
Global distribution is strongly related to
prevalence of HBV
Highest frequencies in Taiwan,
Mozambique and China
Male preponderance
Usually in adult life
Etiopathogenesis
HBV
Chronic liver disease (HCV & Alcoholism)
Aflatotoxins
But many factors interact age, sex,
chemicals, nutrition, alcohol, viruses etc.
Exact pathogenesis varies between HBV
prevalent population Vs low incidence
population
Morphology
Unifocal
Multifocal
Diffuse infiltrative
Are usually paler than surrounding hepatic
parenchyma
Strong propensity for invading vascular
channels (portal vein, IVC)
Well differentiated to poorly differentiated
type
Trabecular,sinusoidal or pseudoacinar
pattern
Bile production by tumor cells
Cirrhosis in adjacent liver parenchyma
Fibrolamellar Variant
M=F
No association with HBV or cirrhosis
Usually single hard tumor with fibrous
bands traversing through it
Well differentiated polygonal cells in cords
or nests, separated by fibrous septae
Clinical Features of HCC
Not characteristic
Usually masked by underlying liver disease
Ill defined upper abdominal pain, fatigue,
cachexia
Raised alpha fetoprotein levels in 60-75%
(markedly increased)
Diagnosis- FNAC, biopsy
Fibrolamellar variant has better
prognosis
FALSE
POSITIVE AFP
Yolk sac
tumors,
Pregnancy,
Fetal distress,
Cirrhosis,
Massive liver
necrosis
Cholangiocarcinoma
Is carcinoma of bile duct origin
No well known risk factors
Only known influences are
primary sclerosing cholangitis,
Parasites- Clonorchis sinesis
exposure to Thorotrast
Cut surface - Firm and Gritty
M/E- well differentiated tumors
Well defined glandular and tubular
structures
Abundant stroma (Desmoplasia)
Hematogenous and lymphatic metastasis
usually present (More common than HCC)
Death usually occurs with in six months
PANCREAS
Pancreas-Normal
About 15 cm in length
Weight 60-140 gm
Histologically two separate components
Exocrine & Endocrine pancreas
Exocrine - 80-85%, composed of acini
Endocrine - Islets of Langerhans
Pathological lesions
Inflammatory
lesions
Neoplasms
Acute pancreatitis
Chronic pancreatitis
Pancreatic Ca
Endocrine tumors
-Insulinomas
-Gastrinomas
Acute Pancreatitis
Characterized by acute onset of
abdominal pain resulting from enzymatic
necrosis and inflammation of the
pancreas
80% cases are associated with GALL
STONES and ALCOHOLISM
Other important CAUSES include
Infections Mumps, Coxsackie and
Mycoplasma
Acute ischemia Shock, trauma, vascular
thrombosis, embolism, vasculitis etc.
Hyperlipoproteinemias (uncommon)
Drugs Diuretics, Azothioprine,
Estrogens, sulfonamides etc
Pathogenesis
Basic mechanism is release of
inappropriately activated pancreatic
enzymes
Cause auto digestion of the pancreas
Hence subsequent inflammation
Three proposed mechanisms for release
of these enzymes
All above listed causes lead to one of
these three mechanisms
244
PANCREATIC DUCT
OBSTRUCTION
Gall stones
Ductal concretions
PRIMARY ACINAR
CELL INJURY
Drugs
Trauma
Ischemia
viruses
DEFECTIVE INTRA-
CELLULAR
TRANSPORT OF
PROENZYMES
Alcohol
Metabolic injury
Interstitial edema
Impaired blood flow
ischemia
Delivery of proenzymes
to lysosomes
ACINAR CELL INJURY
1 2 3
Morphology
Interstitial edema
Focal areas of fat necrosis in pancreas and
peripancreatic tissue
Also in fatty tissue of the abdominal cavity
Appear radiopaque on radiographs
Severe cases necrosis of pancreatic tissue
(acini, ducts and islets)
If sufficient damage to vessels then hemorrhage
in parenchyma (Hemorrhagic pancreatitis)
Blue black areas of hemorrhage admixed
with chalky areas of fat necrosis
vacuolated adipocytes are converted to
shadowy outlines of cell membranes
Calcium salts are seen as basophilic
masses on microscopy
247
Clinical Features
Pain can vary from mild
to very severe
Usually severe epigastric
pain with nausea and
vomiting
Constant, intense and is
referred to upper back
Release of enzymes,
toxins and cytokines in
circulation leads to
activation of systemic
inflammatory response
OTHER CAUSES OF
EPIGASTRIC PAIN (GIT)
Esophagitis (reflux)
Gastritis
Peptic ulcers
Cholecystitis
Leucocytosis
DIC
Hemolysis
Peripheral vascular collapse
Shock with ATN, ARDS
Hypocalcemia, tetany
Raised amylase levels in first 24 hours
Followed by lipase with in 72-96 hours
Complications-
ARDS
ATN
Pancreatic abscess
Pancreatic pseudocyst-
Localized collections of pancreatic
secretions
There occurs drainage of secretions
from ducts into interstitium
no true epithelial lining
Can get infected
Chronic Pancreatitis
Repeated bouts of mild to moderate
pancreatic inflammation with loss of
pancreatic parenchyma and its
replacement by fibrous tissue
Common in middle aged alcoholics
Pancreatic divisum in about 12%
Other cause very rare
40% no obvious cause
Pathogenesis
Associated :
Ductal obstruction by concretions
Oxidative stress
Decreased secretion of lithostatin (a protein
that inhibits preipitation of calcium carbonate)
Once interstial fibrosis, further affects
ductal secretions
Densely fibrotic, small sized organ
Hard, ducts may be dilated & may contain
concretions
Irregularly distributed areas of fibrosis
Sparing of Islets (diabetes is not a
common feature)
Chronic mononuclear infiltrate around
lobules and ducts
Some time squamous metaplasia of duct
epithelium
Clinical Features
Repeated attacks of moderately severe pain
Or persistent abdominal and back pain
Later on pancreatic insufficiency and diabetes
may develop
Features of malabsorption, corrected by
pancreatic enzyme supplements
Diagnosed by high degree of suspicion
X ray and CT- calcifications
Pseudocysts in 10%
Moderately increased risk of carcinoma
(Alcohol)
Carcinoma of The Pancreas
Fifth most common cause of death in U.S.
Only convincing association is with
smoking
Other proposed risk factors like alcohol,
diet rich in fats are not consistent
Familial relapsing pancreatitis (very rare
itself) is strongly associated with
Carcinoma pancreas
Head 60-70%
Body 5-10%
Tail 10-15%
Diffuse involvement in 20%
All are adenocarcinomas arising from duct
epithelium
Carcinoma of the head of pancreas
causes obstruction to bile flow
Ca of Head early symptoms, jaundice
Ca of body and tail- detected late
Invade adjacent retroperitoneal structures
Usually moderately to poorly differentiated
adenocarcinomas
Well differentiated tumors are very rare
Dense stromal fibrosis (desmoplasia)
Propensity for perineural invasion
Clinical Features
Remain silent till late
Pain is usually first symptom (because of
invasion posterior abdominal wall and nerves
Obstructive jaundice
Trousseaus sign (migratory thrombophlebitis) in
10%(because of release of platelet activating factors
and procoagulants from tumor and its necrotic products)
No single specific marker
Raised levels of CA 19-9
Very bad prognosis
Islet Cell Tumors
Neuroendocrine origin
Resemble carcinoids tumors
Functional (elaborate pancreatic enzymes)
or non functional
Insulinomas
Gastrinomas (Zollinger Ellison Syndrome)
Insulinomas
Usually benign, solitary
Arise from cells
1) Signs and symptoms because of hypoglycemia
2) Hypoglycemia accentuated by fasting and
relieved with intake of glucose
3) Low blood glucose levels
Aka Whipples triad
Insulin levels are increased
Histologically appear as giant islets
Gastrinomas ( Zollinger- Ellison Syndrome)
Can also arise in duodenum and
peripancreatic tissues
Hypergastrenemia
Multiple ulcers- esophagus, stomach,
duodenum and jejunum
Refractory to conventional treatment
GALL BLADDER
Lesions-Overview
Cholelithiasis
Acute cholecystitis
Chronic cholecystitis
Carcinoma
Cholelithiasis (Gall Stones)
10% population of northern hemisphere of
western countries
More common in Latin American countries
Two main types- Cholesterol and Pigment
stones
Cholesterol stones are more common
(80%)
264
Risk Factors
CHOLESTEROL
STONES
PIGMENT
STONES
Common in western
Advancing age
Female sex hormones
Rapid weight reduction
Gall bladder stasis
Hyperlipidemia syndromes
Common in Asians
Chronic hemolytic
syndromes
Biliary infections
Ileal disease e.g.
Crohnsdisease, ileal
resection and cystic
fibrosis of pancreas
Pathogenesis
Cholesterol stones
Pigment stones
Biliary lipid
Supersaturation
Gall bladder hypomotility
& cholesterol nucleation
Cholesterol monohytrate crystals
Accretion
Cholesterol stones
Promoters are
mucus
hypersecretion,
hypomoility & ca
salts
Hemolysis Infection of
the biliary
tract
Release of microbial
glucuronidases
Hydrolysis of bilirubin
glucuronides
Unconjugate
d bilirubin
Calcium salts
PIGMENT STONES
+
Morphology
CHOLESTEROL STONES
Pure cholesterol stones are pale yellow
With increasing proportion of calcium carbonate,
phosphates and bilirubin exhibit discoloration
Multiple and faceted
Granular and hard external surface
Glistening radiating crystalline palisade
Mostly radiolucent
Cholesterolosis-excess cholesterol esters
accumulate in lamina propria and look like
yellow flecks
PIGMENT STONES
Black usually in sterile bile
Brown in infected biliary tract
Mainly composed of ca salts of bilirubin
Black stones are multiple and crumble on
touch
Brown stones are few in number & are
soapy in consistency (retain fatty acids
because of bacterial phospholipases)
Are radio opaque (Ca content)
Clinically
70-80% asymptomatic
May present with pain constant or colicky
Complications include
Empyema
Perforation
Fistulas
Cholangitis
Pancreatitis
Gall stone ileus
Increased risk of carcinoma
Cholecystitis
ACUTE
CHRONIC ACUTE ON
CHRONIC
Calculous Acalculous Calculous Acalculous
(Common)
(Rare, occurs in
severly ill
patients)
Acute Cholecystitis (Pathogenesis)
Chemical irritation and inflammation in setting of
obstruction to flow
Mucosal phospholipase convert lecithin to lysolecithin
Damage to glycoprotein layer of mucosa
Further release of prostaglandins from mucosa
All these events lead to mucosal & mural inflammation
Gall bladder dysmotility and increased intraluminal
pressure
Later on bacterial contamination
Enlarged tense gall bladder
Reddish, blotchy or greenish black
external surface
Lumen contains stones
some times pus (Empyema gall bladder)
Wall is edematous & thickened
Severe cases gangrene
Clinically
Pain right hypochondrium or epigastrium
May appear like surgical emergency
Associated fever, nausea and vomiting
Rarely jaundice if obstruction of CBD
Most patients recover
Chronic Cholecystitis
Can be sequel of repeated bouts of acute
cholecystitis
BUT more commonly no antecedent
attacks of acute cholecystitis
Role of gall stones is not clear
Microorganisms can be cultured from bile
in one third patients
Morphology
Serosa usually smooth and glistening but
some times granular because of fibrosis
Wall is thickened
Lumen usually contain stones or clear
secretions (Hydrops gall bladder)
Mucosa is normal
Sub epithelial and sub serosal fibrosis
Mononuclear infiltrate
Rarely extensive dystrophic calcification
k. a. PORCELAIN GALL BLADDER
(increased association with cancer)
Carcinoma Gall Bladder
Occurs in seventh decade of life
Slightly more common in females
Stones in 60-90% patients
Other risk factors include pyogenic and
parasitic infections of the biliary tract
Carcinogenic derivatives of bile may play a
role
Infiltrating or exophytic type
Some times poorly defined seen as diffuse
thickening and induration of gall bladder
Fundus is the most common site
Some have papillary architecture
Most invade liver by the time they are
discovered
Das könnte Ihnen auch gefallen
- MCQ BTSDokument41 SeitenMCQ BTSZonera IqraNoch keine Bewertungen
- SURG - Hepatobiliary, Pancreas, SpleenDokument230 SeitenSURG - Hepatobiliary, Pancreas, SpleenJoan Timbol100% (1)
- CardiopathophysiologyDokument63 SeitenCardiopathophysiologyapplesncoreNoch keine Bewertungen
- CardiopathophysiologyDokument63 SeitenCardiopathophysiologyapplesncoreNoch keine Bewertungen
- Hematopoietic SystemDokument39 SeitenHematopoietic SystemapplesncoreNoch keine Bewertungen
- ImmunopathologyDokument21 SeitenImmunopathologyapplesncoreNoch keine Bewertungen
- Bowel ObstructionDokument48 SeitenBowel ObstructionPatrick John100% (1)
- SURGPATH - 2.1 The Gastrointestinal Tract (Robbins) - TableDokument8 SeitenSURGPATH - 2.1 The Gastrointestinal Tract (Robbins) - TableAngela Caguitla100% (1)
- Special Pathology - Topical Past Papers (2005-2020)Dokument70 SeitenSpecial Pathology - Topical Past Papers (2005-2020)Oga bogaNoch keine Bewertungen
- CIRRHOSIS OF LIVER (Practice of Medicine)Dokument84 SeitenCIRRHOSIS OF LIVER (Practice of Medicine)simran kaurNoch keine Bewertungen
- Survival Guide To Gastrointestinal Mucosal BiopsiesDokument247 SeitenSurvival Guide To Gastrointestinal Mucosal BiopsiesAlex Constantin100% (2)
- Oral Medicine and Radiology MCQDokument7 SeitenOral Medicine and Radiology MCQsamhita100% (1)
- 006 Large IntestineDokument26 Seiten006 Large IntestineDeepthi SreenivasNoch keine Bewertungen
- Biliary Tract Disease - Emmet AndrewsDokument52 SeitenBiliary Tract Disease - Emmet AndrewsBoneyJalgarNoch keine Bewertungen
- Biliary Tract DiseaseDokument52 SeitenBiliary Tract DiseaseAna Cotoman100% (1)
- Rectal CancerDokument71 SeitenRectal CancerGio Balisi100% (2)
- eMRCS Pathology 326 MCQsDokument101 SeiteneMRCS Pathology 326 MCQsSaad KhanNoch keine Bewertungen
- 2011 - Essentials of Oral Pathology, 3rd EditionDokument647 Seiten2011 - Essentials of Oral Pathology, 3rd EditionAdrian Chivu89% (28)
- Colorectal Cancer 1Dokument71 SeitenColorectal Cancer 1Anupam SisodiaNoch keine Bewertungen
- 07a. Non Neoplastic Disease of OesophagusDokument35 Seiten07a. Non Neoplastic Disease of Oesophagus21701101016 - Juliana Ayu NugrahaNoch keine Bewertungen
- PA GI Tract Skuad Tandingan Elnineno!Dokument281 SeitenPA GI Tract Skuad Tandingan Elnineno!Arsya AliNoch keine Bewertungen
- Esophagus: Diseases of The EsophagusDokument60 SeitenEsophagus: Diseases of The EsophagusSalma NajjarNoch keine Bewertungen
- Esophageal ConditionsDokument43 SeitenEsophageal ConditionsMICHAEL SAKALANoch keine Bewertungen
- Esophagous Stomach Small Intestine PathologyDokument58 SeitenEsophagous Stomach Small Intestine PathologytahaNoch keine Bewertungen
- Diseases of OesophagusDokument46 SeitenDiseases of OesophagusBrother GeorgeNoch keine Bewertungen
- Large Intestine, PancreasDokument30 SeitenLarge Intestine, PancreasElijah WoodNoch keine Bewertungen
- Gastrointestinal and Liver PathologyDokument99 SeitenGastrointestinal and Liver PathologyMuhammad Rifki El-MuammaryNoch keine Bewertungen
- Pancreatic Diseases: Dr. Norizal Mohd NoorDokument53 SeitenPancreatic Diseases: Dr. Norizal Mohd NoorTower AlangNoch keine Bewertungen
- GIT PathologyDokument27 SeitenGIT PathologyIshali NuwanjiniNoch keine Bewertungen
- Chapter 17 - Gastrointestinal TractDokument29 SeitenChapter 17 - Gastrointestinal TractAgnieszka WisniewskaNoch keine Bewertungen
- Large Bowel ObstructionDokument36 SeitenLarge Bowel Obstructionpuleng matshabaNoch keine Bewertungen
- GI Esophagus - UPDATED PDFDokument46 SeitenGI Esophagus - UPDATED PDFBatool SherbiniNoch keine Bewertungen
- Overview of Gastric Pathology: Non-Neoplastic DiseasesDokument23 SeitenOverview of Gastric Pathology: Non-Neoplastic DiseasesErnest Jerome MalamionNoch keine Bewertungen
- Disorders of The Upper-GI Tract: Touro College of Osteopathic MedicineDokument107 SeitenDisorders of The Upper-GI Tract: Touro College of Osteopathic Medicinebahadar94Noch keine Bewertungen
- Dysphagia: Dr. Sangeeta Aggarwal Assistant Professor, E.N.T Deptt GMCH, PatialaDokument29 SeitenDysphagia: Dr. Sangeeta Aggarwal Assistant Professor, E.N.T Deptt GMCH, PatialaVishalNoch keine Bewertungen
- Lecture 9 GitDokument42 SeitenLecture 9 GitWaqar Ul HaqNoch keine Bewertungen
- Inflammatory Bowel DiseaseDokument52 SeitenInflammatory Bowel DiseaseWilliams Emmanuel AdeyeyeNoch keine Bewertungen
- Diseases of The StomachDokument72 SeitenDiseases of The StomachAMANUEL HABTEWOLDNoch keine Bewertungen
- Management of Malignant AscitesDokument34 SeitenManagement of Malignant Ascitesadaiah Soibi-HarryNoch keine Bewertungen
- Upper GI Bleeding & It's ManagementDokument40 SeitenUpper GI Bleeding & It's ManagementManeesha KhalseNoch keine Bewertungen
- Liver AbscessDokument34 SeitenLiver Abscessdebdeepbhattacharya411Noch keine Bewertungen
- Dr. Arif Gunawan Teguh WDokument64 SeitenDr. Arif Gunawan Teguh WarifgteguhNoch keine Bewertungen
- Abdominal Emergencies Presentation (Recovered)Dokument126 SeitenAbdominal Emergencies Presentation (Recovered)kityamuwesiNoch keine Bewertungen
- I Am Sharing 'Class - 4 - GIT' With You - 240122 - 164403Dokument21 SeitenI Am Sharing 'Class - 4 - GIT' With You - 240122 - 164403phuyalaryan666Noch keine Bewertungen
- Intestinal Obstruction in ChildrenDokument63 SeitenIntestinal Obstruction in Childreng1381821Noch keine Bewertungen
- Small and Large IntestineDokument124 SeitenSmall and Large IntestinemariahemnksNoch keine Bewertungen
- Oesophagus StomachDokument28 SeitenOesophagus Stomachwanja91Noch keine Bewertungen
- Inflammatory Bowel Disease: 7 Year DR Felix MicheloDokument55 SeitenInflammatory Bowel Disease: 7 Year DR Felix MicheloFreeburn SimunchembuNoch keine Bewertungen
- Disorders of Small IntestineDokument41 SeitenDisorders of Small IntestineSamuel kuriaNoch keine Bewertungen
- 07b.NON NEOPLASTIC DISEASE OF STOMACHDokument45 Seiten07b.NON NEOPLASTIC DISEASE OF STOMACHINDRINoch keine Bewertungen
- Bowel ObstructionDokument36 SeitenBowel ObstructionyoanNoch keine Bewertungen
- Esophageal DisordersDokument37 SeitenEsophageal DisordersDanielle FosterNoch keine Bewertungen
- Ulcerative ColitisDokument63 SeitenUlcerative ColitismahalakshmiNoch keine Bewertungen
- Jaundice: Checklist For HistoryDokument17 SeitenJaundice: Checklist For HistoryMustafa H. KadhimNoch keine Bewertungen
- Gastro Week 4Dokument39 SeitenGastro Week 4Reynaldi FernandesNoch keine Bewertungen
- GIT Pathology Lecture 2005Dokument161 SeitenGIT Pathology Lecture 2005api-3700579100% (5)
- AmebiasisDokument34 SeitenAmebiasisashuNoch keine Bewertungen
- Benign Tumors of The StomachDokument55 SeitenBenign Tumors of The Stomachapi-19641337Noch keine Bewertungen
- Wa0001Dokument41 SeitenWa0001Riya ShindeNoch keine Bewertungen
- GastroenterologySlides2020 Copy TG6Dokument33 SeitenGastroenterologySlides2020 Copy TG6yasmeen.houshiehNoch keine Bewertungen
- Common XraysDokument56 SeitenCommon XraysToluwani Lade-igeNoch keine Bewertungen
- Upper Gastrointestinal BleedingDokument49 SeitenUpper Gastrointestinal BleedingUmar AzlanNoch keine Bewertungen
- Disorders of Esophagus and StomachDokument29 SeitenDisorders of Esophagus and StomachSamuel kuriaNoch keine Bewertungen
- Acute Appendic ItsDokument24 SeitenAcute Appendic ItsDr-Wisam Mhmd AliNoch keine Bewertungen
- Gambaran Patologi Anatomik Kelainan Sistem Pencernaan: Fajar L Gultom Departemen Patologi Anatomik FK Uki Oktober 2016Dokument57 SeitenGambaran Patologi Anatomik Kelainan Sistem Pencernaan: Fajar L Gultom Departemen Patologi Anatomik FK Uki Oktober 2016ajengdmrNoch keine Bewertungen
- Polyps of Large IntestineDokument20 SeitenPolyps of Large IntestineAroosha IbrahimNoch keine Bewertungen
- Neoplasms of The Gastrointestinal Tract: Laura W. Lamps, M.DDokument39 SeitenNeoplasms of The Gastrointestinal Tract: Laura W. Lamps, M.DINDRINoch keine Bewertungen
- Sharpy's Surgical Series PresentationDokument244 SeitenSharpy's Surgical Series PresentationhappyhappylandNoch keine Bewertungen
- Benign Colorectal Disease: Ahmed AlzahraniDokument47 SeitenBenign Colorectal Disease: Ahmed Alzahraniahmad alzahraniNoch keine Bewertungen
- The Stomach and Duodenum: Dr. Elian EliasDokument48 SeitenThe Stomach and Duodenum: Dr. Elian EliasmodarNoch keine Bewertungen
- Powerpoint: Chronic Inflammatory DisordesDokument55 SeitenPowerpoint: Chronic Inflammatory Disordesj.doe.hex_87Noch keine Bewertungen
- Esophageal Conditions: Kibrom Gebreselassie, MD, FCS-ECSA Cardiovascular and Thoracic SurgeonDokument54 SeitenEsophageal Conditions: Kibrom Gebreselassie, MD, FCS-ECSA Cardiovascular and Thoracic SurgeonVincent SerNoch keine Bewertungen
- Inflammatory Bowel DiseaseDokument40 SeitenInflammatory Bowel DiseaseSK TalkNoch keine Bewertungen
- Pediatric PathologyDokument27 SeitenPediatric PathologyapplesncoreNoch keine Bewertungen
- Cardiovascular + Clinical ScenariosDokument36 SeitenCardiovascular + Clinical ScenariosapplesncoreNoch keine Bewertungen
- Renal Pathology: Kidney and The Urinary Collecting SystemDokument37 SeitenRenal Pathology: Kidney and The Urinary Collecting Systemapplesncore100% (1)
- Pathology of The Lung Objectives: Define and Use in Proper Context The Following TermsDokument45 SeitenPathology of The Lung Objectives: Define and Use in Proper Context The Following TermsapplesncoreNoch keine Bewertungen
- Pathology of Infections Objectives: ©bharti B-Pathologyof infections-SGUSOM 2Dokument25 SeitenPathology of Infections Objectives: ©bharti B-Pathologyof infections-SGUSOM 2applesncoreNoch keine Bewertungen
- Fluid and Hemodynamic DisordersDokument19 SeitenFluid and Hemodynamic DisordersapplesncoreNoch keine Bewertungen
- Neoplasia Path NotesDokument13 SeitenNeoplasia Path NotesapplesncoreNoch keine Bewertungen
- Gluteraldehyde, Peracetic Acid Alcohol, Iodophors QAC (Quarternary Ammonium Compounds)Dokument3 SeitenGluteraldehyde, Peracetic Acid Alcohol, Iodophors QAC (Quarternary Ammonium Compounds)applesncoreNoch keine Bewertungen
- Anil Degaonkar, Nikhil Bhamare, Mandar Tilak Arterio-Enteric Fistula A Case ReportDokument6 SeitenAnil Degaonkar, Nikhil Bhamare, Mandar Tilak Arterio-Enteric Fistula A Case ReportDr. Krishna N. SharmaNoch keine Bewertungen
- Jurnal Double Kontras FIXDokument33 SeitenJurnal Double Kontras FIXWisnuHeriPurwantoNoch keine Bewertungen
- Biological Special Programs - Overview: Organs Controlled From The BrainstemDokument19 SeitenBiological Special Programs - Overview: Organs Controlled From The BrainstemYogesh Kharche100% (1)
- GI Polyps DR OdzeDokument30 SeitenGI Polyps DR OdzeJuliana Do CoutoNoch keine Bewertungen
- Clinical Presentation, Diagnosis, and Staging of Colorectal CancerDokument20 SeitenClinical Presentation, Diagnosis, and Staging of Colorectal CancerJose Luis Marin CatacoraNoch keine Bewertungen
- Molecular Genetics of Colorectal Cancer - UpToDateDokument41 SeitenMolecular Genetics of Colorectal Cancer - UpToDateToweran ToweraneNoch keine Bewertungen
- Academy of Life Underwriting - ALU 201 - Intermediate Medical Life Insurance Underwriting (2022)Dokument478 SeitenAcademy of Life Underwriting - ALU 201 - Intermediate Medical Life Insurance Underwriting (2022)KALPESH SHAHNoch keine Bewertungen
- Appendix and ColonDokument59 SeitenAppendix and ColonEzekiel ArtetaNoch keine Bewertungen
- # Davidson - Review # GastroenterologyDokument30 Seiten# Davidson - Review # Gastroenterologyemtiaz zamanNoch keine Bewertungen
- Pathoma Study OutlineDokument19 SeitenPathoma Study OutlineJustine Val RevalidaNoch keine Bewertungen
- Danial McCraeDokument3 SeitenDanial McCraeSi Thi0% (1)
- Peutz-Jeghers Syndrome Clinical Manifestations, Diagnosis, and Management - UpToDateDokument19 SeitenPeutz-Jeghers Syndrome Clinical Manifestations, Diagnosis, and Management - UpToDateHartemes RosarioNoch keine Bewertungen
- CPSP Demo Mcqs - 2022Dokument24 SeitenCPSP Demo Mcqs - 2022Farhan AfzalNoch keine Bewertungen
- Colorectal Cancer Genetic SyndromesDokument1 SeiteColorectal Cancer Genetic SyndromesRyan GosserNoch keine Bewertungen
- Colonic Polyps and Polyposis SyndromesDokument30 SeitenColonic Polyps and Polyposis SyndromesIndhumathiNoch keine Bewertungen
- Causes, Risk Factors, and PreventionDokument19 SeitenCauses, Risk Factors, and PreventionvictorNoch keine Bewertungen
- Pathology For The Health Professions 4Th Edition Damjanov Test Bank Full Chapter PDFDokument20 SeitenPathology For The Health Professions 4Th Edition Damjanov Test Bank Full Chapter PDFcrastzfeiej100% (9)
- Kanker Kolorektral: Gastrointestinal SistemDokument19 SeitenKanker Kolorektral: Gastrointestinal Sistem11Muhammad Agung RozaqNoch keine Bewertungen
- Transanal Protocol PDFDokument4 SeitenTransanal Protocol PDFDesiree FlorendoNoch keine Bewertungen
- 19 Gastrointestinal Tract Polyps - Libre PathologyDokument20 Seiten19 Gastrointestinal Tract Polyps - Libre PathologyfadoNoch keine Bewertungen