Beruflich Dokumente
Kultur Dokumente
What Is A Buffer Solution?: Analticaltechniques Assignment COURSE#509
Hochgeladen von
Altaf Ur RehmanOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
What Is A Buffer Solution?: Analticaltechniques Assignment COURSE#509
Hochgeladen von
Altaf Ur RehmanCopyright:
Verfügbare Formate
ANALTICALTECHNIQUES ASSIGNMENT
COURSE#509
WHAT IS A BUFFER SOLUTION?
DEFINITION
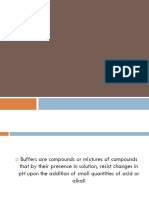
A buffer solution is one which resists changes in pH when small quantities of an acid or
an alkali are added to it.
ACIDIC BUFFER SOLUTIONS
An acidic buffer solution is simply one which has a pH less than 7. Acidic buffer solutions
are commonly made from a weak acid and one of its salts - often a sodium salt.
A common example would be a mixture of ethanoic acid and sodium
ethanoate in solution. In this case, if the solution contained equal molar concentrations
of both the acid and the salt, it would hae a pH of !.7". It wouldn#t matter what the
concentrations were, as long as they were the same.
$ou can change the pH of the buffer solution by changing the ratio of acid to salt, or by
choosing a different acid and one of its salts.
ALKALINE BUFFER SOLUTIONS
An alkaline buffer solution has a pH greater than 7. Alkaline buffer solutions are
commonly made from a weak base and one of its salts.
A frequently used example is a mixture of ammonia solution and ammonium
chloride solution. If these were mixed in equal molar proportions, the solution would
hae a pH of %.&'. Again, it doesn#t matter what concentrations you choose as long as
they are the same.
BUFFER SOLUTION
A buffer solution is one in which the pH of the solution is (resistant( to small additions of
either a strong acid or strong base. )uffers usually consist of a weak acid and its
con*ugate base, in relatiely equal and (large( quantities. +alculations are based on the
equation for the ioni,ation of the weak acid in water forming the hydronium ion and the
con*ugate base of the acid. (HA( represents any weak acid and (A-( represents the
con*ugate base.
HA(aq) H!O(l) ""# H$O(aq) A"(aq)
Ka % &H$O'&A"'
&HA'
WHAT IS A BUFFER AND HOW DOES IT WORK? " THE CONCE(T
A buffer must contain an acid component and a basic component. -ypically a buffer may
either consist of an acid and its con*ugate base or of a base and its con*ugate acid. In
either case it contains a (common( ion, i.e. an ion that is common to the acid.base
equilibrium/
Example)
CH$COOH H!O CH$COO" H$O
MUHAMMAD DANISH 1 17-03-
07
ANALTICALTECHNIQUES ASSIGNMENT
COURSE#509
Acid con*ugated base
0ikewise/
NH$ H!O NH* OH"
)ase con*ugated acid
In the discussion that follows below, we will discuss the acetic acid.acetate buffer
system.
+ontains a large reseroir of acid 1CH$COOH)and its con*ugate base (CH$COO")+
+oncentrations of acid and con*ugate base are about equal 1typically about 2 molar
solutions3
&H$O' or &OH"' concentrations are generally much smaller than acid or con*ugated base
of the buffer solution
CH$COOH H!O CH$COO" H$O
4hen a small amount of strong acid is added to the buffer solution, it will react with the
con*ugated base (CH$COO")+ and the equilibrium will shift to the left, increasing
concentration of CH$COOH, +onsequently, the pH of the buffer solution will decrease,
but only slightly.
0ikewise, if a small amount of a strong base is added, it will shift the equilibrium to the
right, and the pH of the buffer solution will slightly increase.
HOW TO (RE(ARE A BUFFER
A compound can buffer the pH of a solution only when its concentration is sufficient and
when the pH of the solution is close 1within about one pH unit3 to its p5a. -o make a
buffer you must first pick a compound whose p5a is close to the pH you want for the
solution, and then decide what the buffer concentration should be. -ypically, buffer
concentrations are between 2 m6 and &77 m6, depending on the desired ionic strength
and the buffering capacity required. If the pH is expected to decrease during the
experiment, choose a buffer with a p5a slightly below the working pH. +onersely, if the
pH is expected to increase during the experiment, select a buffer with a p5a slightly
aboe the working pH. Haing decided on the total buffer concentration, you must
ad*ust the ratio of the protonated and unprotonated forms of the buffer in your solution so
MUHAMMAD DANISH 2 17-03-
07
ANALTICALTECHNIQUES ASSIGNMENT
COURSE#509
as to gie the desired pH. -ypically, buffers are composed of weak acids and their salts,
or weak bases and their salts. If the protonated form is uncharged, it is an acid 1like
acetic acid3, and its unprotonated form is a salt 1e.g., sodium acetate3. +onersely, if the
unprotonated form is uncharged it is a base 1like -ris base3, and its protonated form is a
salt 1e.g., -risH+l3.
BUFFER S-STE.
A buffer system can be made by mixing a soluble compound that contains the con*ugate
base with a solution of the acid such as sodium acetate with acetic acid or ammonia with
ammonium chloride. -he aboe equation for 5a can be rearranged to sole for the
hydronium ion concentration. )y knowing the 5a of the acid, the amount of acid, and the
amount of con*ugate base, the pH of the buffer system can be calculated.
&H$O' % Ka&HA'
&A"'
pH % "lo/&H$O'
CALCULATION OF THE (H OF A BUFFER SOLUTION
In order to calculate the pH of the buffer solution you need to know the amount of acid
and the amount of the con*ugate base combined to make the solution. -hese amounts
should be either in moles or in molarities. -he 5a of the acid also needs to be known.
Example/ A buffer solution was made by dissoling 27.7 grams of sodium acetate in
&77.7 m0 of 2.77 6 acetic acid. Assuming the change in olume when the sodium
acetate is not significant, estimate the pH of the acetic acid.sodium acetate buffer
solution. -he 5a for acetic acid is 2.7 x 27-'.
8irst, write the equation for the ioni,ation of acetic acid and the 5a expression.
9earrange the expression to sole for the hydronium 0on concen12a10on,
CH$COOH(aq) H!O(l) ""# H$O(aq) CH$COO"(aq)
&H$O' % Ka&CH$COOH'
&CH$COO"'
:econd, determine the number of moles of acid and of the con*ugate base.
12.77 6 +H;+<<H 3 1&77.7 m03 12 0.2777 m03 = 7.&77 mol +H;+<<H
127.7 g >a+H;+<<3 12 mol.?&.7; g3 = 7.2&& mol >a+H;+<<
:ubstitute these alues, along with the 5a alue, into the aboe equation and
sole for the hydronium ion concentration. +onert the hydronium ion concentration
into pH.
@H;<AB = 12.7 x 27-'3 17.&77.7.2&&3 = &.7% x 27
-'
pH = !.'"
BUFFER CA(ACIT- AND BUFFER RAN3E)
-he buffer capacity is the ability to resist change in pH. -he more concentrated the
components of a buffer, the greater the buffer capacity
MUHAMMAD DANISH 3 17-03-
07
ANALTICALTECHNIQUES ASSIGNMENT
COURSE#509
REFERENCES
http/..www.thomasregister.com.olc.";%7777?.crane.htm
http/..www.school-for-champions.com.science.chembases.htm
http/..www.chemguide.co.uk.physical.acidbaseeqia.buffers.html
MUHAMMAD DANISH 4 17-03-
07
Das könnte Ihnen auch gefallen
- Biochemistry : A Practical ManualVon EverandBiochemistry : A Practical ManualBewertung: 5 von 5 Sternen5/5 (1)
- 09 Exp 11 Buffer SolutionsDokument8 Seiten09 Exp 11 Buffer SolutionsShainmaugne AdvientoNoch keine Bewertungen
- Experiment 11 - Buffor SolutionsDokument7 SeitenExperiment 11 - Buffor SolutionsBridget BurnsNoch keine Bewertungen
- A Further Investigation of the Symmetrical Chloride of Paranitroorthosulphobenzoic AcidVon EverandA Further Investigation of the Symmetrical Chloride of Paranitroorthosulphobenzoic AcidNoch keine Bewertungen
- Assignment 2Dokument3 SeitenAssignment 2edelyn telewikNoch keine Bewertungen
- Buffers: Analytical TechniquesDokument17 SeitenBuffers: Analytical TechniquesAbdul FarooqNoch keine Bewertungen
- Buffers CompleteDokument46 SeitenBuffers CompleteSunshine_Bacla_4275100% (2)
- Buffer Solution (1.2)Dokument5 SeitenBuffer Solution (1.2)Kuldipsinh ZalaNoch keine Bewertungen
- Buffer AssignmentDokument4 SeitenBuffer AssignmentHannahnel Anasco QuidatoNoch keine Bewertungen
- BioChem34: Exp1Dokument2 SeitenBioChem34: Exp1Nonee Quesada CornebyNoch keine Bewertungen
- Buffer SolutionsDokument3 SeitenBuffer Solutionsemad_abdelaal57Noch keine Bewertungen
- Buffer Solution - Acidic and Basic Buffers, Preparations, Examples PDFDokument5 SeitenBuffer Solution - Acidic and Basic Buffers, Preparations, Examples PDFSakshi DevalekarNoch keine Bewertungen
- Analytical Techniques: Topic: BuffersDokument18 SeitenAnalytical Techniques: Topic: BuffersAbdul FarooqNoch keine Bewertungen
- CHEM 102 Instructional Objectives: - Additional Aqueous EquilibriaDokument29 SeitenCHEM 102 Instructional Objectives: - Additional Aqueous EquilibriarajNoch keine Bewertungen
- BuffersDokument20 SeitenBuffersgopimpharmNoch keine Bewertungen
- How Do We Choose A BufferDokument2 SeitenHow Do We Choose A BufferShahir AshuNoch keine Bewertungen
- Buffer (Larutan Penyangga)Dokument6 SeitenBuffer (Larutan Penyangga)Budiman ApriyossaNoch keine Bewertungen
- Unit-V - PH, Buffer, Buffer Equation, Isotonicity-1Dokument27 SeitenUnit-V - PH, Buffer, Buffer Equation, Isotonicity-1IKHLASH MOHDNoch keine Bewertungen
- PH Is A Measure of The Acidity or Basicity of A Solution.: Glass ElectrodeDokument9 SeitenPH Is A Measure of The Acidity or Basicity of A Solution.: Glass Electrodehelion45Noch keine Bewertungen
- 2.pH, Buffers and IsotonicDokument48 Seiten2.pH, Buffers and Isotonicrajender91% (11)
- Buffer ChemistryDokument9 SeitenBuffer ChemistrySidra chaudharyNoch keine Bewertungen
- Buffer Solutions.: Ass. Prof. I. R. BekusDokument27 SeitenBuffer Solutions.: Ass. Prof. I. R. BekusNanda ThyarezaNoch keine Bewertungen
- Mechanism of Buffering SystemDokument8 SeitenMechanism of Buffering SystemRezaul Karim TutulNoch keine Bewertungen
- 6.0 Conceitos Sobre PH e Soluções TampãoDokument13 Seiten6.0 Conceitos Sobre PH e Soluções TampãoFernando SperandioNoch keine Bewertungen
- Lec 7 Analytical (Buffer Solution)Dokument13 SeitenLec 7 Analytical (Buffer Solution)alhyaly857Noch keine Bewertungen
- Acid-Base Equilibria and Salt EquilibriaDokument14 SeitenAcid-Base Equilibria and Salt EquilibriaRaven LampaNoch keine Bewertungen
- The Synopsis of Buffering System-Its Chemical ReviewDokument6 SeitenThe Synopsis of Buffering System-Its Chemical ReviewRezaul Karim TutulNoch keine Bewertungen
- Acid-Base Equilibria in Aqueous SolutionsDokument48 SeitenAcid-Base Equilibria in Aqueous SolutionsAdrian ChombaNoch keine Bewertungen
- General Chemistry 2: Buffer SolutionsDokument15 SeitenGeneral Chemistry 2: Buffer SolutionsSteiner100% (1)
- Buffer SolutionsDokument19 SeitenBuffer SolutionsMuskaan BindalNoch keine Bewertungen
- BUFFERSDokument21 SeitenBUFFERSRose-ann Gallema PauloNoch keine Bewertungen
- BUFFERDokument10 SeitenBUFFERakinolaboluwatife83Noch keine Bewertungen
- Lecture 6 Buffer SolutionDokument5 SeitenLecture 6 Buffer SolutionhusseinNoch keine Bewertungen
- BufferDokument82 SeitenBufferDeepa DevanathanNoch keine Bewertungen
- 6.0 Acid-Base and 7.0 Solubility EquilibriaDokument29 Seiten6.0 Acid-Base and 7.0 Solubility Equilibriaparkinsondilys7Noch keine Bewertungen
- Buffered and Isotonic Solutions-DikonversiDokument35 SeitenBuffered and Isotonic Solutions-DikonversiKoming SukawatiNoch keine Bewertungen
- Buffer Solutions FinalDokument62 SeitenBuffer Solutions Finalshripathyd1100% (1)
- Buffer Solutions: September 2020Dokument22 SeitenBuffer Solutions: September 2020Nitin DapkeNoch keine Bewertungen
- BufferDokument25 SeitenBuffernaghma KhanNoch keine Bewertungen
- Chem 102 Class Notes: Prof. Upali Siriwardane, Chemistry Program, Louisiana Tech University, Ruston, LA 71272Dokument28 SeitenChem 102 Class Notes: Prof. Upali Siriwardane, Chemistry Program, Louisiana Tech University, Ruston, LA 71272Jape GarridoNoch keine Bewertungen
- 6 - IonizationDokument48 Seiten6 - IonizationYashfa YasinNoch keine Bewertungen
- Scientific Method: Jayson Nota Maria Angelica BalicocoDokument21 SeitenScientific Method: Jayson Nota Maria Angelica BalicocoGrace Ann ArimadoNoch keine Bewertungen
- Lecture 1Dokument24 SeitenLecture 1mvps9248Noch keine Bewertungen
- Vdocument - in Buffer Solutions 568b0b28b560dDokument14 SeitenVdocument - in Buffer Solutions 568b0b28b560dAmiraNoch keine Bewertungen
- 2023 BCH 211 Masondo - BL 202126087 Prac5 WriteupDokument6 Seiten2023 BCH 211 Masondo - BL 202126087 Prac5 WriteupBenson MasondoNoch keine Bewertungen
- Lesson 3.2. PH and Buffer SystemsDokument13 SeitenLesson 3.2. PH and Buffer SystemsGemma CabañasNoch keine Bewertungen
- Lecture5 Pharmaceutics (Buffer Partition)Dokument6 SeitenLecture5 Pharmaceutics (Buffer Partition)haroon41Noch keine Bewertungen
- Chapter 17: Additional Aspects of Aqueous Equilibria: Common-Ion EffectDokument28 SeitenChapter 17: Additional Aspects of Aqueous Equilibria: Common-Ion EffectrajNoch keine Bewertungen
- Chemistry, Tenth Edition (PDFDrive) - 100 - 100Dokument10 SeitenChemistry, Tenth Edition (PDFDrive) - 100 - 100Ezat JrNoch keine Bewertungen
- Acid-Base Equilibria and Solubility EquilibriaDokument24 SeitenAcid-Base Equilibria and Solubility EquilibriaAndrew John CellonaNoch keine Bewertungen
- Unit 4 Notes Part 7 Buffers Student Notes WlykqtDokument5 SeitenUnit 4 Notes Part 7 Buffers Student Notes WlykqtIsiwat KazeemNoch keine Bewertungen
- CH 15 More Equilibrium: Buffers, K, KDokument20 SeitenCH 15 More Equilibrium: Buffers, K, KDamris MuhamadNoch keine Bewertungen
- General Chemistry Q4 M4Dokument16 SeitenGeneral Chemistry Q4 M4roxan clabria100% (1)
- Biochemistry LN03Dokument16 SeitenBiochemistry LN03Rahaf Al-muhtasebNoch keine Bewertungen
- Acid Base BuffersDokument8 SeitenAcid Base BuffersAmiraAtiyaNoch keine Bewertungen
- Chem 40.1 - Midterms Reviewer Discussion: Buffers: - (Aq) 2 4 - (Aq) 4 2 - (Aq) 2Dokument5 SeitenChem 40.1 - Midterms Reviewer Discussion: Buffers: - (Aq) 2 4 - (Aq) 4 2 - (Aq) 2Steffi GatdulaNoch keine Bewertungen
- PH and Buffer System - NotesDokument29 SeitenPH and Buffer System - Noteskatherine morenoNoch keine Bewertungen
- Buffer SolutionDokument7 SeitenBuffer SolutionFerisa Wisuda NingtyasNoch keine Bewertungen
- Buffers SolutionsDokument4 SeitenBuffers SolutionsAmrit RamroopNoch keine Bewertungen
- Adc Invoice 400Dokument2 SeitenAdc Invoice 400Altaf Ur RehmanNoch keine Bewertungen
- File IndexDokument113 SeitenFile IndexAltaf Ur RehmanNoch keine Bewertungen
- Enquiry ListDokument12 SeitenEnquiry ListAltaf Ur RehmanNoch keine Bewertungen
- Weboc 1Dokument30 SeitenWeboc 1Altaf Ur RehmanNoch keine Bewertungen
- Documentation Description: For Import & Export GoodsDokument8 SeitenDocumentation Description: For Import & Export GoodsAltaf Ur RehmanNoch keine Bewertungen
- Bill of Entry Bill of Export Baggage Declaration Transshipment PermitDokument2 SeitenBill of Entry Bill of Export Baggage Declaration Transshipment PermitAltaf Ur RehmanNoch keine Bewertungen
- 1) Sales Contract 1) Sales Contract Importer or Buyer Importer or BuyerDokument5 Seiten1) Sales Contract 1) Sales Contract Importer or Buyer Importer or BuyerAltaf Ur RehmanNoch keine Bewertungen
- CondolenceDokument23 SeitenCondolenceAltaf Ur RehmanNoch keine Bewertungen
- CSS Everyday Science Notes PDFDokument50 SeitenCSS Everyday Science Notes PDFWaqas Gul100% (1)
- Sami Pharmaceuticals (PVT) LTD Costing / Comparison: Butylated Hydroxy AnisolDokument5 SeitenSami Pharmaceuticals (PVT) LTD Costing / Comparison: Butylated Hydroxy AnisolAltaf Ur RehmanNoch keine Bewertungen
- Clarification LetterDokument1 SeiteClarification LetterAltaf Ur RehmanNoch keine Bewertungen
- Acids and Alkalis Workbook Pages 89 To 97Dokument9 SeitenAcids and Alkalis Workbook Pages 89 To 97Dev AgarwalNoch keine Bewertungen
- Chapter 8Dokument44 SeitenChapter 8Pirate CaribbeanNoch keine Bewertungen
- PKa Values For Organic and Inorganic Brönsted Acids at 25Dokument4 SeitenPKa Values For Organic and Inorganic Brönsted Acids at 25Gabriela Nohemy Nuñez RodriguezNoch keine Bewertungen
- Chapter 2 SolucionarioDokument24 SeitenChapter 2 SolucionariomonicaNoch keine Bewertungen
- PH Measurement and Buffer PreparationDokument3 SeitenPH Measurement and Buffer PreparationaayaaaNoch keine Bewertungen
- Buffer KSP ProbDokument4 SeitenBuffer KSP ProbFitria Rahma DewiNoch keine Bewertungen
- Lab ReportDokument10 SeitenLab Reportapi-327825157Noch keine Bewertungen
- AcidBase PEQ1Dokument2 SeitenAcidBase PEQ1Aaron LeQuangNoch keine Bewertungen
- BuffersDokument22 SeitenBufferslana alarnabNoch keine Bewertungen
- Week6 11 Titration PHDokument99 SeitenWeek6 11 Titration PH62100959Noch keine Bewertungen
- Acids and Alkalis PDFDokument37 SeitenAcids and Alkalis PDFKizzy Anne Boatswain CarbonNoch keine Bewertungen
- 16 Acid Base TitrationDokument27 Seiten16 Acid Base TitrationManni Piyush Sharma100% (1)
- Plant Dose Chart PDFDokument1 SeitePlant Dose Chart PDFMarco Sanchez MurilloNoch keine Bewertungen
- Experiment 13Dokument4 SeitenExperiment 13Karl Vincent Radin TanNoch keine Bewertungen
- LAB 4 (Total Acidity) (DMT30093)Dokument3 SeitenLAB 4 (Total Acidity) (DMT30093)Teresa YanNoch keine Bewertungen
- Ionization of Weak Monoprotic AcidDokument4 SeitenIonization of Weak Monoprotic Acidmcris101Noch keine Bewertungen
- Acids Base Salt QuestionsDokument3 SeitenAcids Base Salt QuestionsraipujaNoch keine Bewertungen
- Aromatic Organic TestsDokument3 SeitenAromatic Organic TestsErica BugejaNoch keine Bewertungen
- PharmacyDokument3 SeitenPharmacykarimakki100% (1)
- Pharmaceutical Aids and NecessitiesDokument25 SeitenPharmaceutical Aids and NecessitiesKyle Angelic DavasolNoch keine Bewertungen
- CH 7 ObjDokument4 SeitenCH 7 ObjchongpeisiNoch keine Bewertungen
- Indicators & PH CurvesDokument35 SeitenIndicators & PH CurvesSairam PrasathNoch keine Bewertungen
- CBSE Class 10 Science Lab Manual - Properties of Acetic AcidDokument9 SeitenCBSE Class 10 Science Lab Manual - Properties of Acetic AcidPratyush Gohain BaruahNoch keine Bewertungen
- Peka Chemistry Form 4 Student S and Teacher S Manual 02 Acid Base TitrationDokument6 SeitenPeka Chemistry Form 4 Student S and Teacher S Manual 02 Acid Base TitrationWaNnur WanEe Mohd YusofNoch keine Bewertungen
- Ion Exchange Resin TypesDokument2 SeitenIon Exchange Resin Typesjdiosbernal10Noch keine Bewertungen
- PH MeterDokument37 SeitenPH MeterMelroy Castalino100% (1)
- Worksheet Acid Base Titration CurveDokument2 SeitenWorksheet Acid Base Titration CurveMark MagoNoch keine Bewertungen
- EducationalDokument4 SeitenEducationalwondimuNoch keine Bewertungen
- ABG Compensations Answers - 2Dokument1 SeiteABG Compensations Answers - 2khryssNoch keine Bewertungen
- PH Meter DraftDokument121 SeitenPH Meter DraftHiba BashirNoch keine Bewertungen
- Sodium Bicarbonate: Nature's Unique First Aid RemedyVon EverandSodium Bicarbonate: Nature's Unique First Aid RemedyBewertung: 5 von 5 Sternen5/5 (21)
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincVon EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincBewertung: 3.5 von 5 Sternen3.5/5 (137)
- Process Plant Equipment: Operation, Control, and ReliabilityVon EverandProcess Plant Equipment: Operation, Control, and ReliabilityBewertung: 5 von 5 Sternen5/5 (1)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactVon EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactBewertung: 5 von 5 Sternen5/5 (5)
- Guidelines for Defining Process Safety Competency RequirementsVon EverandGuidelines for Defining Process Safety Competency RequirementsBewertung: 3 von 5 Sternen3/5 (1)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeVon EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeBewertung: 4 von 5 Sternen4/5 (1)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeVon EverandChemistry for Breakfast: The Amazing Science of Everyday LifeBewertung: 4.5 von 5 Sternen4.5/5 (14)
- It's Elemental: The Hidden Chemistry in EverythingVon EverandIt's Elemental: The Hidden Chemistry in EverythingBewertung: 4 von 5 Sternen4/5 (10)
- Taste: Surprising Stories and Science About Why Food Tastes GoodVon EverandTaste: Surprising Stories and Science About Why Food Tastes GoodBewertung: 3 von 5 Sternen3/5 (20)
- AP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeVon EverandAP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeNoch keine Bewertungen
- Guidelines for Chemical Process Quantitative Risk AnalysisVon EverandGuidelines for Chemical Process Quantitative Risk AnalysisBewertung: 5 von 5 Sternen5/5 (1)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeVon EverandChemistry for Breakfast: The Amazing Science of Everyday LifeBewertung: 4.5 von 5 Sternen4.5/5 (90)
- AP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeVon EverandAP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeBewertung: 5 von 5 Sternen5/5 (1)
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideVon EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNoch keine Bewertungen
- Well Control for Completions and InterventionsVon EverandWell Control for Completions and InterventionsBewertung: 4 von 5 Sternen4/5 (10)
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeVon EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeBewertung: 5 von 5 Sternen5/5 (4)
- The Periodic Table: A Very Short IntroductionVon EverandThe Periodic Table: A Very Short IntroductionBewertung: 4.5 von 5 Sternen4.5/5 (3)
- An Applied Guide to Water and Effluent Treatment Plant DesignVon EverandAn Applied Guide to Water and Effluent Treatment Plant DesignBewertung: 5 von 5 Sternen5/5 (4)
- Process Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersVon EverandProcess Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersNoch keine Bewertungen
- Formulating, Packaging, and Marketing of Natural Cosmetic ProductsVon EverandFormulating, Packaging, and Marketing of Natural Cosmetic ProductsNoch keine Bewertungen