Beruflich Dokumente
Kultur Dokumente
Ie00064a001 PDF
Hochgeladen von
Juan Camilo Henao0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
28 Ansichten9 SeitenVanadium-titanium oxides are well-known catalytic systems for the selective oxidation of different substrates, such as NH3 with NOx. Vanadia species strongly interacting with the Ti 02 and soluble bulk vanadium oxide are formed on these catalysts. Different kinds of active sites are suggested as being responsible for toluene activation and ammonia combustion.
Originalbeschreibung:
Originaltitel
ie00064a001.pdf
Copyright
© © All Rights Reserved
Verfügbare Formate
PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenVanadium-titanium oxides are well-known catalytic systems for the selective oxidation of different substrates, such as NH3 with NOx. Vanadia species strongly interacting with the Ti 02 and soluble bulk vanadium oxide are formed on these catalysts. Different kinds of active sites are suggested as being responsible for toluene activation and ammonia combustion.
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
28 Ansichten9 SeitenIe00064a001 PDF
Hochgeladen von
Juan Camilo HenaoVanadium-titanium oxides are well-known catalytic systems for the selective oxidation of different substrates, such as NH3 with NOx. Vanadia species strongly interacting with the Ti 02 and soluble bulk vanadium oxide are formed on these catalysts. Different kinds of active sites are suggested as being responsible for toluene activation and ammonia combustion.
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 9
Ind. Eng. Chem. Res.
1987,26, 639-647 639
Ammoxidation of Toluene to Benzonitrile on
Vanadium-Titanium Oxides Catalysts Prepared by
Precipitation. The Role of Catalyst Composition
Patrizia Cavalli, Fabrizio Cavani, Italo Manenti, and Ferruccio Trifirb*
Zst i t ut o di Tecnologie Chi mi che Speci al i , 40136 Bologna, I t al y
The catalytic behavior of V205/Ti 02 catalysts prepared by the contemporaneous precipitation of
vanadium and titanium oxides was tested in the reaction of toluene ammoxidation to benzonitrile.
Generally, two kinds of vanadia species are formed on these catalysts: a vanadia species strongly
interacting with the Ti 02 (insoluble in a basic medium) and soluble bulk vanadium oxide, which
does not interact with the support. The relative amounts of the two species depend on the crystalline
structure of the Ti 02. I n particular, the latter species is prevalent in V205/Ti 02 (rutile) catalysts,
while the former predominates in V205/Ti 02 (anatase) catalysts. However, during reaction a re-
distribution of the two species occurs, and the samples become more similar. Different kinds of
active sites are suggested as being responsible for toluene activation and ammonia combustion. For
a vanadia content corresponding approximately to the amount necessary for the monolayer coverage
of Ti 02, higher yields in benzonitrile were obtained. The formation of an upper layer of vanadia
over the Ti 02 surface causes a strong increase in ammonia combustion and therefore a decrease in
the yield of benzonitrile.
Vanadium-titanium oxides (V-Ti-0) are well-known
catalytic systems for the selective oxidation of different
substrates, both inorganic, such as NH3 with NOx (Inge-
mar Odenbrand et al., 1985; Wongard and Nobe, 1984),
and organic, such as toluene (van der Berg et al., 1984; van
Hengstum et al., 1983a), xylenes (Cole et al., 1978; Saleh
et al., 1986; Wachs et al., 1985; Wainwright and Foster,
1979), butene (Ai, 1976), and butadiene (Bond et al., 1979).
Their catalytic properties in the ammoxidation of or-
ganic substrates also have been investigated, especially as
regards the conversion of picoline to nicotinonitrile (An-
demon, 1982; Andersson and Andersson, 1985), while very
little attention has been devoted to the characterization
of these catalysts for the ammoxidation of alkylaromatics
such as toluene or xylenes (Suvorov et al., 1972).
For this kind of reaction, more suitable catalysts seem
to be pure V205 or V205 supported on Al,03 (Ito and Sano,
1967; Niwa et al., 1977; Niwa and Murakami, 1982) and
they have received more attention. In our opinion, V-Ti-0
catalysts should be studied in more detail in regard to the
reaction of ammoxidation for the following reasons: (a)
I t is known that V-Ti-0 systems are able to activate se-
lectively alkylaromatic substrates, giving rise to partial
oxidation products. I t has been suggested that the slow
step in ammoxidation is the same as for oxidation (Brill
and Finely, 1964; Hadley, 1961; Ogata and Sakanishi, 1968;
Simon and Germain, 1975), and therefore it can be in-
teresting to verify the suitability of this system for the
ammoxidation reaction. (b) Much work has been devoted
to obtaining the understanding of the interaction between
vanadia and Ti 02 (Bond and Bruckman, 1981; Bond et al.,
1986; Gqsior et al., 1984; Haber, 1984; Roozeboom et al.,
1980; Slinkard and de Groot, 1979; Wachs et al., 1985),
which plays a role greater than that of just acting as a
support for the active phase. This interaction gives rise
to a more active and selective catalyst than pure vanadia
in the mild oxidation of o-xylene to phthalic anhydride.
(c) A detailed study of the ammoxidation reaction of al-
kylaromatics, involving the effect of NH3 combustion and
the effect of varying the vanadia concentration in the
catalyst, has not yet been made.
Some authors have stated that in o-xylene oxidation it
is necessary to have the anatase structure of TiOz in order
0888-58851 871 2626-0639$01.50/0
to obtain a good catalyst (Gpsior et al., 1984; Haber, 1984;
Wachs et al., 1985). Many attempts have been made to
explain the mechanism of the modifying action of anatase
on vanadia; particularly, some authors ascribe it to the
selective exposure of the (010) face of vanadia on the
surface (Inomata et al., 19831, due to the epitactic match
between the same planes of the two oxides (Vejux and
Courtine, 1978). This causes an increase in the number
of V=O bonds exposed on the surface-active sites for
hydrocarbon activation (Gpsior and Machey, 1983). This
effect can also enhance weakening of the vanadyl bond
(Roozeboom et al., 1980; Tarama et al., 1964) and favors
its reducibility (Wachs et al., 1985). On the other hand,
some authors (Cole et al., 1978; van Hengstum et al., 1983a)
find that the crystalline form of Ti 02 is not important in
determining the activity and selectivity of the catalyst, and
the only important factor is the surface area of the catalyst
itself. In same cases, the rutile form of titania gives better
performances than the supported vanadia (Bond et al.,
1979; Slinkard and de Groot, 1979).
Moreover, we do think that the way to prepare the
catalyst can be very important in determining the catalytic
performance. V-Ti-0 catalysts usually characterized in
the literature are prepared by the impregnation technique
(Bond et al., 1979; Busca et al., 1986; Roozeboom et al.,
1980; van Hengstum et al., 1983b) or by the mixing of
vanadium and titanium oxides, followed by heating of the
mixture at high temperatures (Andersson, 1982) or by the
grafting technique (Bond and Bruckman, 1981; Busca et
al., 1985). The aim of this work was to carry out an in-
vestigation of the catalytic properties of V-Ti-0, prepared
by the coprecipitation technique, in the ammoxidation of
toluene to benzonitrile and, specifically, to study the fol-
lowing problems: (1) What is the optimum amount of
vanadia on titania? Many authors have found that a
monolayer of vanadia on titania is the necessary quantity
to obtain good performances for the oxidation of o-xylene
(Bond and Konig, 1982; van Hengstum et al., 1983a; Wachs
et al., 1985); an excess of vanadia had practically no effect
on the performance. Somewhat different results were
obtained by other authors (Andersson, 1982; Andersson
and Andersson, 1985) in the ammoxidation of picoline.
Indeed, in the ammoxidation reaction, a third reactant,
0 1987 American Chemical Society
640 Ind. Eng. Chem. Res., Vol. 26, No. 4, 1987
ammonia, is involved, and therefore it is interesting to
study the role of surface and bulk vanadia in these con-
ditions. (2) What is the effect of the TiO, crystalline
structure on the activity and selectivity? (3) What is the
role of the different vanadia species formed on the cata-
Experimental Section
Preparation of the Catalysts. The catalysts were
prepared by contemporaneous precipitation of vanadium
and titanium oxides from a solution containing both V(1V)
and Ti(1V) ions. Specifically, the V(1V) solution was
prepared by reducing V205 with an oxalic acid solution,
until a deep blue solution was obtained. TiC1, was partially
hydrolyzed in water (pH <1 by addition of HC1). The two
solutions were then mixed in the right amounts necessary
to obtain the desired amount of V205 in the final catalyst.
The successive coprecipitation was carried out by a hy-
drolysis at T >60 "C; depending on the temperature of
hydrolysis, the anatase or rutile structure can be obtained.
Characterization of the Catalysts. The catalysts were
characterized by means of X-ray diffraction analyses,
chemical analyses, and BET surface area measurements.
I t is well known (Inomata et al., 1983; Yoshida et al.,
1972) that treatment of the catalysts with a basic solution
(30% NH40H/H20) leads to the dissolution of vanadium
oxide which is not chemically bound to the titania. This
method has been successfully used recently (Busca et al.,
1986) to distinguish between the different species that are
formed by the wet impregnation and grafting techniques
of vanadia on preformed TiO,, and the data obtained were
confirmed by spectroscopic analyses. Detailed studies are
in course to more thoroughly characterize the nature of
the vanadia species formed in V-Ti-0 catalysts prepared
by the coprecipitation technique. Following this procedure,
the calcined catalyst was treated with an ammonia solution
(pH =10) to dissolve the vanadium oxide not strongly
interacting with the support; the remaining solid was then
dissolved in hot concentrated sulfuric acid. The two so-
lutions were analyzed separately, with the manganometric
method, to determine the amounts of vanadium in the
samples.
A Carlo Erba Sorpty 1700 was used for the BET surface
area measurements (one-point method).
The X-ray diffraction analyses were carried out with a
Philips diffractometer, using Ni-filtered Cu Kcuradiation.
Catalytic Tests. Catalytic tests were carried out by
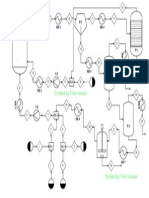
using a fixed-bed flow reactor at atmospheric pressure.
The reactor was a stainless steel tube (4-mm diameter,
600-mm long) placed in a copper bar and externally heated
by electric resistances.
The effluents, kept at 270 "C to prevent condensation,
were analyzed by using two gas chromatographs on-line
to the reactor. The first gas chromatograph analyzed the
unconverted toluene and the benzonitrile formed; traces
of other byproducts (benzene and benzaldehyde) were also
detected. The column was a silicon oil DC 550, supported
on Chromosorb W 80-100; the oven temperature was
programmed from 80 to 220 "C (heating rate 16 deg/min).
The uncondensable gases (O,, N2, CO, and C02) were an-
alyzed by the second gas chromatograph, using a Carbo-
sieve S column; the oven temperature was programmed
from 20 to 240 "C (heating rate 8 deg/min).
The catalyst was used either in the powder form (for
V205/Ti02 (rutile) samples) or in a form supported on
ceramic pellets (for V205/Ti02 (anatase) samples). In the
former case, the particle size ranged from 0.125 to 0.250
mm, and 1 cm3 of catalyst (0.64 g) was used. In the second
case, the active phase was a thin layer (60-pm thick) de-
lyst?
posited over the cylindrical pellets (670 by weight of active
phase); 40 pellets (diameter, 4.0 mm; height, 4.1 mm) were
used for each series of catalytic tests, corresponding to 0.38
g of active phase. The pellets were overlaid to constitute
the catalytic bed, and a thermocouple was inserted along
the channel formed by the overlaid pellets, possessing a
central axial hole.
In order to verify whether the use of the catalyst in the
form of a thin layer supported on the pellets could lead
to some differences in the catalytic behavior as compared
with the conventional use in the form of powder, prelim-
inary tests were made on a 2570 V205/Ti02 (anatase)
sample, using it in both forms. The dependence of the
selectivities on the reactant conversion are the same in the
two cases. The only difference lies in the range of tem-
perature of activity, which is slightly different; this dif-
ference however is related to the different amounts of
active phases in the two cases and to the different relative
residence times.
The pressure drop along the catalytic bed was in all cases
lower than 0.15 kg/cm2. The absence of significant phe-
nomena of bypass of the reactants was verified using H20
and N2 as gas tracers. The absence of homogeneous com-
bustion of the reactants or of the products at high tem-
peratures was verified.
Pore diffusion was verified not to be rate limiting by
varying the size of the particles (catalyst in the form of the
powder). The absence of significant interparticle transport
effects was verified by varying the amount of catalyst, at
a constant W/ F ratio.
Before catalytic tests, the catalysts were calcined in air
at 400 "C and then left in the reactant mixture at the same
temperature for about 10 h. After this period, the catalyst
exhibits a stable catalytic behavior.
Results
Characterization of V205/ Ti 02 Catalysts. X-ray
diffraction analyses of samples calcined at 400 "C show
for the V2OS/Ti0, (rutile) samples, in addition to the lines
relative to the rutile phase ( d =3.24, 2.49, 2.18, 1.69, 1.62,
1.47, 1.36 A), the most intense diffraction lines of V205 (d
=4.38, 3.40,2.88 A) but only in the 25% V2OS/TiO2 (rutile)
catalyst.
A more detailed analysis of the maxima attributable to
the rutile phase shows a shift of the reflections toward
lower d values, with a parallel contraction of the cell
volume and of the lattice parameter a. This indicates the
formation of a substitutional solid solution of V4+in the
rutile lattice (Andersson and Andersson, 1985; Bond et al.,
1979); since the ionic radius of V4+is slightly smaller than
that of Ti4+, a contraction of the unit cell occurs, to an
extent which depends on the amount of vanadium incor-
porated.
In the case of the V205/Ti02 (anatase) catalysts, only
the lines attributable to Ti02 can be distinguished, even
at higher V20, contents (d =3.51, 2.38,1.89, 1.70, 1.66, 1.48
A). No modification of the cell volume or parameters was
observed.
Reported in Table I are the chemical analyses of three
V205/Ti02 (rutile) samples, containing 5%, 15%, and 25%
by weight V205, and in Table I1 are the analyses for three
Vz05/Ti 02 (anatase) samples, containing 5%, 1070, and
25% by weight V,Ob All samples were calcined at 400 "C
for 3 h before analysis. In particular the absolute amounts
of soluble and insoluble vanadia are reported; also reported
in the same tables are the values of surface area of the
catalysts and the amount of V205 necessary to have a
complete monolayer coverage of the support. These values
are, of course, functions of the surface area of the supports
Ind. Eng. Chem. Res., Vol. 26, No. 4, 1987 641
8
Y) 40-
n
%
a -
m-
Table I. Chemical Analyses of VzO5/TiO2 (Rutile)
Catalysts
/ i
/:,./,
--./ -e
nominal
V25 surface
content, area, theoretical
% w t m2/g soluble insoluble monolayere
50 49 traces 5.0 7.2
5b 49 traces 5.0 7.2
15' 68 8.4 6.6 10.0
15b 68 3.9 11.1 10.0
25" 92 13.3 11.7 13.5
25b 92 4.0 21.0 13.5
% wt Vz05 (vanadia)
a After calcination. After catalytic tests. Bond and Bruck-
man, 1981.
Table 11. Chemical Analyses of V205/Ti02 (Anatase)
Catalysts
nominal
v25 surface
content, area, theore tical
% w t m2/g soluble insoluble monolayerc
50 57 0.6 4.4 8.4
5b 57 0.9 4.1 8.4
10" 52 0.2 9.8 7.6
1 Ob 52 2.0 8.0 7.6
25" 50 3.0 22.0 7.3
25b 50 10.0 15.0 7.3
% wt V205 (vanadia)
"After calcination. *After catalytic tests. 'Bond and Bruck-
man, 1981.
and were calculated based on the considerations made by
Bond and Bruckman (1981) and by Roozeboom et al.
(1980). These authors took into consideration the surface
area occupied by one V02,5 species and calculated the
number of these species necessary to cover a unit surface
area of the support.
These kinds of considerations have often been used for
characterizing V2O5/TiO2 catalysts (Busca et al., 1986;
Inomata et al., 1983; Roozeboom et d., 1980). The analyses
of the catalysts after reaction are also reported in the same
tables. The reacted samples do not show appreciable
modifications in the values of surface area or in the X-ray
diffraction patterns as compared to the fresh ones.
Effect of Ammonia on Toluene Conversion. The
dependence of benzonitrile and C0 2 productivities (the
main products of toluene ammoxidation), on the NH3
partial pressure, on a catalyst containing 25% by weight
Vz05 on anatase is reported in Figure 1. With amounts
of ammonia up to the stoichiometric amount required for
the reaction (which is not in the ratio 1:l because of the
combustion of ammonia to nitrogen), the yield in benzo-
nitrile is directly dependent on the ammonia partial
pressure. On the contrary, the formation of COz decreases
considerably with increasing amounts of ammonia in the
feed. The overall conversion of toluene also decreases in
the whole range of ammonia partial pressures tested, until
it reaches an approximately constant value at high NH3
concentrations. At very low NH3 partial pressure, small
amounts of benzaldehyde are also detected.
Catalytic Activity of TiOz. Pure Ti02, prepared in
the same way as the Vz05/Ti02 samples, was loaded in the
reactor, both as anatase and as rutile. The yields in carbon
oxides and nitrogen obtained for TiOz (rutile) as functions
of the reaction temperature are reported in Figure 2. The
sample showed formation of both carbon oxides and ni-
trogen. Moreover, a lack in the carbon balance indicated
the formation of heavy products, undetectable by gas
chromatography; no trace of benzonitrile was observed.
On the contrary, Ti 02 (anatase) did not show any ac-
tivity in toluene or ammonia overoxidation, from 270 to
\ \
0.05 0.1 (
0
0
ammonia part,pressure, atm.
5
Figure 1. Benzonitrile (A) and C02 productivities (0) as functions
of the ammonia partial pressure at the reactor inlet; catalyst, 25%
V205/Ti0, (anatase). Experimental conditions: reaction tempera-
ture, 310 O C ; residence time, 1.79 s; Rtoluene). 0.0117 atm; P( 02) ,
0.135 atm.
-i i
--
" m - 340 360
temperature, C
0
Figure 2. CO (A), COP (e), and N2 (m) yields as functions of the
reaction temperature: catalyst, TiO, (rutile) (0.56 9). Experimental
conditions: P(toluene), 0.0117 atm; P(NHJ , 0.075 atm; P( 02) , 0.0135
atm; residence time, 0.73 s.
0 2 4 6 8 1 0
ammonia part pressure , atm XIO*
Figure 3. CO (0) and COz (0) yields from benzonitrile combustion
as functions of the ammonia partial pressure at the inlet of the
reactor: catalyst, Ti02 (0.56 g) as rutile (full symbols) and as anatase
(open symbols). Experimental conditions: reaction temperature, 370
O C ; P(benzonitrile), 0.0125 atm; P( 02) , 0.135 atm; residence time,
0.73 s.
400 "C; only heavy products were formed. Other tests were
made by feeding benzonitrile into the reactor, in the
presence of oxygen and increasing amounts of ammonia
(Figure 3). The benzonitrile burned on the Ti 02 surface,
especially when low amounts of ammonia were present.
Vz05/Ti02 (Anatase) Catalysts: Effect of Varying
the Amount of Vanadium. A comparison between the
catalytic behaviors of two catalysts containing different
642 Ind. Eng. Chem. Res., Vol. 26, No. 4, 1987
s . 0
0
s
:i :4
temperature C
Figure 4. Toluene conversion (0) and benzonitrile (A), CO (01, and
COz (0) yields as functions of the reaction temperature, for the 10%
Vz06/Ti02 (anatase) (full symbols) and the 25% VzO5/TiO2 (ana-
tase) (open symbols) catalysta. Experimental conditions: P(toluene),
0.0117 atm; P(O,), 0.135 atm; P(NH3), 0.075 atm; residence time, 1.79
S.
3
5 60-
0
>.
40 -
temperature C
Fi gure 5. Nz (0) and NzO (0) yields as functions of the reaction
temperature; catalysts and experimental conditions as in Figure 4.
amounts of nominal Vz05 (10% and 25% by weight) on
anatase is reported in Figures 4 and 5.
The yields in benzonitrile and COz as functions of the
reaction temperature are reported in Figure 4, while the
yields in Nz and NzO are reported in Figure 5. The be-
haviors of the two catalysts are very similar at low tem-
peratures as regards the formation of benzonitrile. On the
contrary, they exhibit different behaviors regarding the
combustion of ammonia. I t can be seen that the formation
of nitrogen is lower on the catalyst containing 10% Vz05
and that also the formation of NzO is shifted toward higher
temperatures.
Another difference lies in the range of temperatures
where the product formed is stable. For the 10% Vz05/
TiOz (anatase) sample the product is stable over a wide
range of temperatures (up to 380 "C), while for the 25%
VZO5/TiO2 catalyst a sharper maximum in the yield is
observed.
The behavior of the 5% VzO5/TiO2 catalyst is shown in
Figure 6. The very low number of active sites in the
sample does not allow high levels of conversion to be
reached. A comparison at conversions lower than 60%
shows that the combustion of ammonia to Nz on this
sample is lower, in correspondence to the same levels of
toluene conversion. On the other hand, the selectivity is
kept lower by the formation of heavy products, whose
presence is not detectable by gas chromatography.
In order to distinguish the role of the two species iden-
tified by chemical analyses, an extraction of the vanadia
which did not interact with the support was made on the
spent catalyst (25% VzO5/TiO2, in the form of powder);
and its catalytic activity was again verified. The catalytic
behaviors of the unwashed (full lines) and washed catalysts
(dotted lines) are reported in Figure 7. The latter sample
temperature , C
Figure 6. Toluene conversion (O), benzonitrile (A), CO ( O) , COz (W)
N, (o), and NzO (A) yields, and lack of carbon balance (0) as
functions of the reaction temperature; catalyst, 5% Vz06/Ti02
(anatase). Experimental conditions as in Figure 4.
100 0-0-
temperature , C
Fi gure 7. Toluene conversions and products yields as functions of
the reaction temperature: catalyst, 25% VzO5/TiO2 (anatase) as
powder (1.0 cm3, 0.639 g) (full lines) and washed 25% V2O5/TiO2
(anatase) (dotted lines). Experimental conditions: P(toluene),
0.0117 atm; P(O,), 0.135 atm; P(NH,), 0.075 atm; residence time, 0.73
s; symbols as in Figure 6.
temperature , C
Fi gure 8. Toluene conversion and products yields as functions of
the reaction temperature: catalysts, 15% VzOS/TiOz (rutile) (full
symbols) and 25% VzO5/TiO2 (rutile) (open symbols). Experimental
conditions as in Figure 7; symbols as in Figure 4.
exhibits the same behavior as the former one, thus showing
that the vanadia not strongly interacting with the support
does not have a specific role in the mechanism of the
ammoxidation reaction, either in the activation and oxi-
Ind. Eng. Chem. Res., Vol. 26, No. 4, 1987 643
loot
P;
O 3 0 0 320 340 360 380
temperature, C
Figure 9. Nz (0) and N20 (0) yields as functions of the reaction
temperature; catalysts and experimental conditions as in Figure 8.
Figure 10. Toluene conversion and products yields as functions of
the reaction temperature: catalyst, 5% VzO5/TiO2 (rutile). Exper-
imental conditions as in Figure 8; symbols as in Figure 6.
dation of toluene or in the oxidation of ammonia.
Vz0,/TiO2 (Rutile) Catalysts: Effect of Varying
the Amount of Vanadium. The yields in benzonitrile
and carbon oxides from toluene and of Nz and NzO from
ammonia are reported in Figures 8 and 9 as functions of
the reaction temperature for two Vz05/Ti02 (rutile) cat-
alysts containing 15% and 25% VzO,, respectively. Figure
10 shows the behavior of a 5% VZ0,/TiO2 catalyst.
The 25% VzOs/TiOz sample is the most active in toluene
conversion and shows high selectivities in benzonitrile
formation (about 80% at total conversion).
The 15% V205/Ti02 sample is less active but shows
comparable selectivities in the benzonitrile. For the 5%
VzO5/TiO2 sample, the overall toluene conversion is similar
to that of the 15% VZO5/TiOz catalyst. I t can also be
noted that the 5% VZ0,/TiOz catalyst shows a high for-
mation, at low temperatures, of undetected heavy products,
the formation of which decreases with increasing tem-
perature, due to their combustion to carbon oxides.
Another consideration that may be made concerns the
shape of the curve representing the benzonitrile yield. In
the 5% VzO5/TiO2 sample, the yield starts decreasing at
340 "C, with an increase in the formation of COz (due also
to the combustion of heavy products) and CO. The 15%
VZO5/TiOz sample exhibits a much wider maximum for
the desired product. Only at very high temperatures does
the yield start to decrease, in correspondence with the lack
of ammonia in the reaction medium, due to its combustion
to Nz and N20. The 25% V20,/TiOZ sample which shows
the highest amount of ammonia combustion, shows a
sharper maximum in benzonitrile.
The 25% V205/Ti02 sample also was washed with a
basic solution, to dissolve the vanadia not interacting with
t A- I
Figure 11. Benzonitrile (A), COz (O), and N2 (0) yields as functions
of the reaction temperature: catalyst, 10% V2O6/TiO2 (anatase),
undoped (full symbols) and doped with Cs (V/Cs =18/1) (open
symbols). Experimental conditions as in Figure 4.
the support; the catalyst showed the same degree of am-
monia combustion, toluene conversion, and benzonitrile
yield.
Finally, some tests were made (for comparison) on a
10% Vz05/Ti02 (anatase) catalyst calcined at 560 "C in
the reaction atmosphere, in order to transform the anatase
into the rutile structure. The catalyst showed a consid-
erable decrease in activity (the conversion was about 15%
at 350 "C), while the selectivity of the catalyst remained
approximately constant. Analyses of the catalyst showed
that a considerable decrease in surface area (from 50 m2/g
to less than 1 m2/g) occurred in correspondence with the
crystalline transformation.
Effect of Doping with Cesium. In order to verify
whether or not doping our catalysts could lead to some
enhancement in the catalytic behavior, for example,
causing a variation in ammonia combustion, the 10%
V2O5/TiOZ (anatase) catalyst was doped by adding CsCl
during its preparation.
The catalytic behavior of a 10% V20,/TiO2 (anatase)
catalyst containing cesium in the amount necessary to
obtain a V/Cs atomic weight ratio equal to 18:l is reported
in Figure 11along with that of the undoped catalyst. I t
is shown that the activity of the alkali-doped catalyst is
lower and the attainment of the maximum benzonitrile
yield is shifted to higher temperatures. On the contrary,
no modification is observed in the ammonia combustion;
the benzonitrile yield starts to decrease at the same tem-
perature for the two catalysts, that is, in correspondence
to the same degree of ammonia combustion.
Discussion
Comparison between V205/ Ti 02 (Anatase) and
V205/Ti02 (Rutile) Catalysts. I t has been established
in the scientific literature that a bidimensional vanadia
structure is formed in VzO5/TiO2 catalysts on the surface
of the TiOz (the so-called "monolayer") (Bond et al., 1986;
Inomata et al., 1983; Saleh et al., 1986; Roozeboom et al.,
1980). However, there is little agreement in the literature
about the nature of this structure. The only certain con-
sideration is that a strong interaction occurs with the
centers on the Ti02 surface, leading to some kind of va-
nadia species characterized by unique properties, such as
higher reactivity with respect to vanadia as the bulk oxide
(Niwa and Murakami, 1982; Roozeboom et al., 1980; Wachs
et al., 1985). Over this monolayer coverage, both upper
layers and aggregates of crystalline V205 are formed (In-
omata et al., 1983; Bond et al., 1986); the latter species not
directly interacting with the TiOz surface can be selectively
644 Ind. Eng. Chem. Res., Vol. 26, No. 4, 1987
dissolved by means of a washing with a basic solution
(Yoshida et al., 1972).
A very important consideration arising from our data
is that the soluble vanadia has no catalytic activity, as
shown by the tests on the washed catalysts. Therefore,
all the correlations between the activity and the amount
of vanadium in the catalyst must be made taking into
account only the insoluble part. This is in agreement with
the suggestions of some authors about the role of excess
vanadia in the oxidation of o-xylene (Bond et al., 1986;
Vanhove and Blanchard, 1971; van Hengstum et al.,
1983a).
The two series of catalysts, characterized by the different
natures of the supports, have similar surface areas, in the
range 50-90 m2/g. However, there are remarkable dif-
ferences in the relative distribution of the two vanadia
species. In particular, the amount of interacting vanadia
is higher for the VzO5/TiO2 (anatase) samples, except for
the 5% Vz05/Ti02 one. This is in agreement with the
indications given in the scientific literature about the su-
perior ability of the anatase structure in the formation of
the monolayer as compared with the rutile structure
(Haber, 1984; Saleh et al., 1986). This difference has been
related to the presence, in the former case, of a crystallo-
graphic fit between some planes of Vz05 and the growth
planes of anatase (Vejux and Courtine, 1978).
It is interesting, however, to compare these data with
those of the samples after reaction. In both series of
samples, a modification of the chemical situation occurred,
with a redistribution of the two vanadia species. In par-
ticular, for the V2O5/TiO2 (anatase) catalysts, the amount
of soluble vanadia generally increases for the three samples
during the permanence in the reaction medium, while the
amount of insoluble vanadia decreases. The opposite oc-
curs for the VzO5/TiO2 (rutile) samples. An explanation
of the phenomenon may be obtained by comparing the
amounts of the vanadium species with those corresponding
to the theoretical monolayer. The values reported indicate
that before reaction in the VzO5/TiO2 (rutile) samples, the
amount of interacting vanadia is lower than the theoretical
amount necessary for the formation of a monolayer.
Moreover, it must be considered that part of the insoluble
vanadia is probably present in solid solution in the rutile
lattice, as indicated by the X-ray diffraction analyses.
Following the indications of Bond et al. (1979) and of
Andersson and Andersson (1985), the amount of vanadium
in solid solution should not exceed 3-4% by weight, ex-
pressed as Vz05.
The opposite is true for the VzO5/TiO2 (anatase) Sam-
ples. The amount of insoluble vanadia is higher, before
reaction (except for the 5% VzO5/TiO2 sample), than the
theoretical amount for the monolayer; work by Bond et
al. (1986) and by Inomata et al. (1983) indicates the pos-
sibility of forming more than one monolayer of vanadium
(thus, of multilayers or towers of vanadium oxide) in
V2O5/TiO2 (anatase) catalysts.
I t is interesting to note that after reactions the two series
of catalysts appear very similar; thus, during reaction, the
weakly interacting vanadia spreads over the free rutile
surface. In the case of the VzO5/TiO2 (anatase) catalysts,
on the contrary, the insoluble vanadium partly segregates,
forming soluble bulk Vz05. Therefore, after reaction all
the samples reach a chemical situation that is a function
(for the same amount of nominal V205) of the surface area
and not of the crystalline structure of the TiOP More in
detail, after reaction, in the case of the lowest amount of
VZO5 ( 5%) , in both V205/Ti02 (anatase) and VzO,/TiOz
(rutile) samples, the vanadia is overall lower than the
theoretical amount for monolayer coverage; therefore, it
can be thought that part of the TiOz surface remains ex-
posed to the gas phase. In the case of the 10% VZO5/TiOz
(anatase) and 15% VzO5/TiO2 (rutile), the amount of va-
nadia which strongly interacts with the TiOz (and therefore
can not be dissolved) is in both cases very near to the
theoretical monolayer. Finally, for the highest Vz05-con-
taining catalysts, the amount of vanadia considerably ex-
ceeds the theoretical amount for monolayer formation; in
particular, the amount of strongly interacting vanadium
is in both cases about double that necessary to form a
monolayer.
The similarity of V205/Ti02 (anatase) and V2O5/TiO2
(rutile) catalysts is accompanied by a very similar catalytic
behavior of the samples. A comparison between the 25%
V2O5/TiOZ (anatase) and the 25% V2O5/TiOZ (rutile)
catalysts can be made by comparing tests reported in
Figures 7 and 8. In fact, in both cases tests were made with
the same weight of active phase and by using the catalysts
in the form of particles with the same dimensions.
I t is seen that they both are rather selective in benzo-
nitrile and exhibit a decrease in yield when the combustion
of ammonia becomes relevant. A difference lies instead
in the activity of the two samples. The 25% VzO5/TiO2
(rutile) catalyst is more active than the 25% VzO5/TiO2
(anatase), but this difference can be attributed to the
higher surface area of the latter sample.
Effect of NH3 Partial Pressure. Figure 1 shows that
ammonia has an inhibiting effect over the toluene con-
version. This effect can be explained by competition be-
tween ammonia and the hydrocarbon for the same active
sites (Cavalli et al., 1986); a similar effect also occurs in
the case of paraffin ammoxidation, with a shift of the
activity toward higher temperatures with respect to the
oxidation when ammonia is added to the feed (Cavani et
al., 1986).
Another effect of adding ammonia to the feed is the very
strong decrease in the products of combustion. The am-
monia stabilizes the intermediate of toluene activation
(which in these conditions would be otherwise burnt to
carbon oxides) with the formation of benzonitrile (Cavalli
et al., 1986); this product is very stable in the reaction
environment, allowing high selectivities to be obtained.
Effect of Varying the Amount of Vanadium. a.
Pure TiOp Figures 2 and 3 show that pure TiOz possesses
some activity in the toluene or ammonia conversion. In
particular, both pure anatase and rutile activate toluene,
giving rise to the formation of heavy products, undetect-
able by gas chromatography. I t has been reported that on
acid supports the formation of heavy products from aro-
matic condensation can easily occur (Niwa et al., 1981).
Moreover, the rutile shows activity in the combustion of
toluene to carbon oxides and in the oxidation of ammonia
to nitrogen.
Similar activity is not shown by pure anatase. This
difference between the two crystalline modifications of
TiOz has also been found for other kinds of reactions
(Cunningham et al., 1981; Onishi and Hamamura, 1970;
Svintsova et al., 1977); it has been attributed to the greater
reducibility and Lewis acidity of coordinatively unsatu-
rated Ti4+ions at the rutile surface (Woning and van
Santen, 1983). Finally, both supports show some activity
in the combustion of benzonitrile, above all in the absence
of ammonia. This seems to indicate that the acid centers
on the surface of titania can activate the basic benzonitrile,
causing its combustion. The presence of ammonia can
decrease this phenomenon, probably through a competition
with the product for the acid centers. Similarly, it has been
Ind. Eng. Chem. Res., Vol. 26, No. 4, 1987 645
found that the TiOz surface is responsible for phthalic
anhydride combustion in the oxidation of o-xylene (Ggsior
et al., 1984; Wachs et al., 1985).
b. Submonolayer Region. As indicated before, the
5% VZ0,/TiO2 (anatase) and 5% V2O5/TiO2 (rutile) sam-
ples probably possess some fraction of exposed Ti 02 sur-
face, because of the low amount of V205, with respect to
their areas. I n agreement, both samples form heavy
products. The low selectivity in benzonitrile (due to tol-
uene combustion) and the relatively high ammonia com-
bustion of the 5% Vz05/Ti02 (rutile) sample also are at-
tributable to the exposed rutile surface. The same sample
shows a decrease at high temperatures in the yield in
benzonitrile. This can be due either to the exposed rutile,
which has been shown to be active in the product over-
oxidation, or to the combustion of the intermediate of
toluene activation. The latter, in the absence of ammonia
(mainly burnt to nitrogen), cannot be converted to the
stable product; Figure 1 shows that at low ammonia partial
pressure the main products obtained are carbon oxides.
In the case of the 5% Vz05/Ti02 (anatase), the low
number of active sites (a lower amount of catalyst was
used) does not allow considerations to be made at high
conversion. It is, however, seen that at low conversion the
combustion of ammonia is particularly low; this is in
agreement with the tests made on pure anatase, which was
found to be inactive in this kind of reaction.
c. Monolayer Region. The 10% V2O5/TiO2 (anatase)
and 15% V2O5/TiO2 (rutile) catalysts have an amount of
insoluble vanadia which corresponds to the theoretical
monolayer coverage. Therefore, in the two cases the
fraction of exposed titania surface must be very low, and
no effect due to the support should be present. In
agreement, no formation of heavy products is observed in
the two samples, since the carbon balance is very good.
Moreover, in the case of the 15% V205/Ti02 (rutile)
catalyst, the oxidation of ammonia to nitrogen decreases
considerably with respect to the 5% VzO5/TiO2 (rutile),
along with an increase in the selectivity in benzonitrile (due
to the absence of the parallel reaction of toluene com-
bustion over the free rutile surface). In both samples, the
low degree of ammonia overoxidation allows a wider range
of temperatures where the benzonitrile can be obtained
with high yields. Thus, the ammonia in the gas phase is
always sufficient (except at very high temperatures) to
stabilize the intermediate with the formation of benzo-
nitrile.
d. Supramonol ayer Region. The 25% V205/Ti02
(anatase) and 25% VZO5/TiOz (rutile) catalysts have high
amounts of insoluble vanadia, about twice as much as that
necessary for obtaining total monolayer coverage. It is
therefore probable that some kind of structure is formed
over the first coverage of the support, which also interacts
chemically with the latter.
The catalytic data show that both 25% Vz05/Ti02
(anatase) and 25% VzO5/TiO2 (rutile) are more active in
the ammonia overoxidation to N2 and NzO than the 10%
VzO5/TiO2 (anatase) and the 15% V205/Ti02 (rutile)
samples, respectively. Because of the increased ammonia
combustion, the range of high yield in benzonitrile becomes
narrower for both of the catalysts.
Moreover, while the 25% Vz05/Ti02 (rutile) is more
active than the 15% V2O5/TiO2 (rutile) in toluene con-
version, the 25% and 10% VzO5/TiO2 (anatase) show the
same activity. It is possible to explain this different be-
havior considering that the 25% V205/Ti02 (rutile) sample
has a surface area higher than the 15% V2O5/TiOZ (rutile)
one; the amount of vanadium constituting the first cov-
Scheme I. General Network of the Reaction
p NzO1N2
NH,
\
Ph CHO \
heavy
products - co. co,
erage of the Ti 02 is also higher (Bond and Bruckman,
1981) and leads to enhanced activity. On the contrary, the
25% V205/Ti02 (anatase) sample has the same surface
area as the 10% VZO5/TiOz (anatase) one, and therefore,
the amount of vanadium constituting the complete support
coverage is the same in the two samples.
These data indicate, therefore, that the activity in tol-
uene conversion is a function of the amount of vanadium
oxide constituting the first TiOz coverage and, therefore,
of the surface area of the sample.
A common peculiarity of the 25% V2O5/TiO2 (anatase)
and 25% V205/Ti02 (rutile) samples is that they both
exhibit a strong increase in N2 and NzO formation with
respect to the samples containing lower amounts of va-
nadium. For instance, in correspondence to an increase
from 20% to 60% toluene conversion at 290 "C (for the
15% V205/Ti02 (rutile) and the 25% Vz05/Ti02 (rutile)
samples, respectively), there is an increase in Nz yield from
about 5% to 45%. The 10% Vz05/Ti02 (anatase) and the
25% V2O5/TiO2 (anatase) catalysts possess the same ac-
tivity in toluene conversion, while the latter sample is more
active in the ammonia overoxidation. This means that
higher vanadium oxide contents than the amounts nec-
essary for the formation of the monolayer lead to the
formation of specific sites for ammonia overoxidation, in
both V2O5/TiO2 (anatase) and VzO5/TiO2 (rutile) catalysts.
On the contrary, the amounts of vanadium oxide corre-
sponding to the formation of only one layer coverage
(monolayer). lead to the formation of sites responsible for
toluene and ammonia selective activation. The hypothesis
of the existence of two different kinds of active sites is also
supported by a kinetic analysis, which showed that while
toluene conversion is influenced by the ammonia partial
pressure (Figure l), the ammonia overoxidation is not
affected by the toluene partial pressure (Cavalli et al.,
1987).
Effect of Cesium. Alkali metals are known to influence
the catalytic activity of V205 (Mross, 1983). Their pro-
moting action was found to increase from lithium to cesium
(Krupay and Ross, 1977). Also in the case of alkylaromatic
ammoxidation, alkali-doped vanadium-based catalysts
have been suggested to be more selective than the undoped
ones (Ogata and Sakanishi, 1966). In particular, alkali
metals are known to lower the surface acidity (Wainwright
and Foster, 1979) and, therefore, in our case, should modify
the interaction of ammonia with the catalyst.
The experimental evidence is that the overoxidation of
ammonia is not influenced by the addition of small
amounts of cesium, while the centers that selectively ac-
tivate toluene and ammonia are partially inhibited. This
is a further confirmation of the existence of two different
kinds of active sites in these catalysts, one for toluene and
ammonia selective activation (and formation of benzo-
nitrile) and one for ammonia overoxidation to N2 and N20.
General Network of Reaction. The results can be
finally correlated with the general network reported in
Scheme I. The toluene is first activated, forming probably
646 Ind. Eng. Chem. Res., Vol. 26. No. 4, 1987
some kind of radical species (Ogata and Sakanishi, 1968;
Simon and Germain, 1975; Weiss et al., 1973); this reaction
is the slow step, and the intermediate is quickly converted
to a first product, such as benzaldehyde or benzylamine
(Cavalli et al., 1986; Andersson, 1986).
The data at low ammonia concentration indicate that
low yields of benzaldehyde are formed in these conditions.
However, tests at very low residence times showed the
formation of benzylamine, suggesting it could be the first
product formed (Cavalli et al., 1986). Whichever it is, this
product is then quickly converted to benzonitrile. The
latter is very stable and doesnt undergo successive reac-
tions of combustion; this explains the generally high se-
lectivities that are obtained. Only in the case when an
exposed Ti 02 surface is present on the catalyst and when
there is a lack of ammonia in the gas phase can the ben-
zonitrile be overoxidized to carbon oxides (Figure 3).
Together with the reaction of benzonitrile formation,
there is a parallel one of combustion over the reactant itself
or over the intermediate. This reaction generally occurs
to a low extent and becomes important only in the fol-
lowing cases: (1) the presence of an exposed rutile surface
in V205/Ti02 (rutile) catalysts; (2) the absence of ammonia
in the gas phase. The latter phenomenon occurs when the
catalyst is particularly active in the overoxidation of am-
monia, thus in the presence of a rutile-free surface or with
catalysts containing high amounts of vanadium oxide
(higher than the one necessary for the formation of a
monolayer). In these conditions, the intermediate of
toluene activation (or alternatively the benzaldehyde
formed) that is rather unstable in these conditions cannot
react with the ammonia to form the benzonitrile and is
mainly burnt to carbon oxides.
Another source of decreasing selectivity is the parallel
formation of heavy products, on the free TiOa surface; at
high temperature, the products are burnt to carbon oxides.
In conclusion, the best catalyst for this kind of reaction
is the one with the amount of vanadium oxide necessary
for obtaining only a monolayer coverage of Ti02, regardless
of the crystalline structure of the latter.
Conclusions
The following considerations can be made based on the
results reported.
(1) The contemporaneous precipitation of vanadium and
titanium oxides leads to catalysts characterized by either
the anatase or rutile structures for TiO,; V205/Ti 02
(anatase) and V205/Ti02 (rutile) samples have, after
calcination, similar values of surface areas, in the range
50-90 m2/g.
(2) Two species of vanadia are present in the catalysts
after calcination: a vanadium species that is soluble in a
basic medium (weakly interacting with the support) and
an insoluble vanadia, strongly interacting with Ti02. The
former species is prevalent in V205/Ti02 (rutile) catalysts,
while the contrary is true in VzO5/TiO2 (anatase) catalysts.
In the case of V205/Ti02 (rutile) samples, a solid solution
of vanadium in the rutile lattice also is formed.
(3) The permanence in the reaction medium changes the
relative distribution of the two species in the samples; thus,
after reaction, the V20,/Ti02 (anatase) and V205/Ti02
(rutile) samples are more similar and have similar relative
amounts of the two species.
(4) With increasing nominal amounts of vanadia in the
preparation, the quantity of the insoluble species increases,
up to the formation of more than one theoretical layer
coverage of the Ti 02 surface.
(5) Pure TiO,, both as anatase and rutile, exhibits ac-
tivity in heavy products formation and benzonitrile ov-
eroxidation; rutile is also active in toluene combustion and
ammonia overoxidation to Nz and N20.
(6) Excess vanadium oxide, present as soluble bulk V205,
is not active in toluene ammoxidation and ammonia ov-
eroxidation.
(7) For lower amounts of vanadium oxide, which do not
lead to a complete monolayer coverage of the Ti02, the
catalysts are less active in benzonitrile formation; in the
case of the 5% V205/Ti02 (rutile) catalyst, the exposed
rutile surface leads to a high activity in N2 formation.
(8) For amounts of vanadium oxide corresponding to a
monolayer coverage of TiOz, the catalysts are active and
selective in benzonitrile formation, while the activity in
ammonia overoxidation is relatively low; the desired
product can be obtained with high yields over a wide range
of temperatures.
(9) For both Vz05/Ti02 (anatase) and V205/Ti02 (rutile)
catalysts with amounts of vanadia higher than that nec-
essary for the formation of the monolayer, a strong increase
in N2 formation from NH, occurs. This leads to a decrease
in the yield in benzonitrile at high temperatures, because
of the instability of the intermediate of toluene activation
in the absence of ammonia.
(10) The catalytic behaviors of the Vz05/Ti02 (anatase)
and V205/Ti02 (rutile) catalysts are very similar, sug-
gesting that the crystalline structure of Ti02 is not im-
portant in determining the catalytic performance.
(11) The best performances in the reaction of toluene
ammoxidation are therefore obtained, for both Vz05/Ti02
(anatase) and V2O5/TiOZ (rutile) catalysts, when approx-
imately a monolayer coverage of the Ti02 surface is ob-
tained.
Acknowledgment
The present work was sponsored by ENICHEM Sintesi,
Italy.
Registry No. PhCH,, 108-88-3; PhCN, 100-47-0; V,06,
1314-62-1; Ti02, 13463-67-7.
Literature Cited
Ai, M. Bull. Soc. Chem. Jpn. 1976, 49, 1328.
Andersson, A. J. Catal. 1982, 76, 144.
Andersson, A. J . Catal. 1986, 100, 414.
Andersson, A.; Andersson, S. L. T. In Solid St at e Chemistry i n
Catalysis; Grasselli, R. K., Brazdil, J . F., Eds.; ACS Symposium
Series 279; American Chemical Society: Washington, DC, 1985;
p 121.
Bond, G. C.; Bruckman, K. Faraday Discuss. Chem. Soc. 1981, 72,
Bond, G. C.; Konig, P. J. Catal. 1982, 77, 309.
Bond, G. C.; Sarkany, A. J .; Parfitt, G. D. J. Catal. 1979, 57, 476.
Bond, G. C.; Zurita, J . P.; Flamerz, S.; Gellings, P. J .; Bosch, H.; van
Brill, UT. F.; Finely, J . H. I nd. Eng. Chem. Prod. Res. Deu. 1964,3(2),
Busca, G.; Centi, G.; Marchetti, L.; TrifirB, F. Langmui r 1986, in
Busca, G.; Marchetti, L.; Centi, G.; Trifirb, F. J. Chem. SOC., Faraday
Cavalli, P.; Cavani, F.; Manenti, I.; Trifirb, F. Catal. Today 1987, in
Cavalli, P.; Cavani, F.; El-Sawi, Manenti, I.; Trifirb, F. Ind. Eng.
Cavani, F.; Centi, G.; Riva, A.; TrifirB, F. Catal. Today 1987, in press.
Cole, D. J .; Cullis, Ch. F.; Hucknall, D. J . J. Chem. Soc., Faraday
237.
Ommen, J . G.; Kip, B. J . Appl . Catal. 1986, 22, 361.
89.
press.
Trans. 1 1985, 81, 1003.
press.
Chem. Res. 1987, in press.
Trans. 1 1978, 72, 2185.
Ind. Eng. Chem. Res. 1987,26,647-652 647
Cunningham, J .; Hodnett, B. K.; Ilyas, M.; Tobin, J .; Leahy, E. L.;
Fierro, J . L. G. Faraday Discuss. Chem. SOC. 1981, 72, 283.
Gpior, M.; Gpior, I.; Grzybowska, B. Appl . Catal. 1984, 10, 87.
Gpior, M.; Machey, T. J. Catal. 1983,83, 472.
Haber, J. Pure Appl . Chem. 1984,56(12), 1663.
Hadley, D. J . Chem. Ind. (London) 1961, 238.
Ingemar Odenbrand, C. U.; Lundin, S. T.; Andersson, L. A. H. Appl.
Inomata, M.; Mori, K.; Miyamoto, A,; Ui, T.; Murakami, Y. J. Phys.
Ito, M.; Sano, K. Bull. Chem. SOC. Jpn. 1967,40, 1307.
Krupay, B. W.; Ross, R. A. J. Catal. 1977,49, 92.
Mross, W. D. Catal. Rev.-S'ci. Eng. 1983,25(4), 591.
Niwa, M.; Ando, H.; Murakami, Y. J. Catal. 1977,49, 92.
Niwa, M.; Murakami, Y. J. Catal. 1982, 76,9.
Niwa, M.; Sago, M.; Ando, H.; Murakami, Y. J. Catal. 1981,69,69.
Ogata, Y.; Sakanishi, K. Chem. Ind. (London) 1966,49, 2055.
Ogata, Y.; Sakanishi, K. Tetrahedron 1968,24, 3788.
Onishi, Y.; Hamamura, T. Bull. Chem. SOC. Jpn. 1970, 43, 996.
Roozeboom, F.; Mittelmeyer-Hazeleger, M. C.; Mouljin, J . A,; Me-
dema, J .; de Beer, V. H. J .; Gellings, P. J . J. Phys. Chem. 1980,
84, 2783.
Saleh, R. Y.; Wachs, I. E.; Chan, S. S.; Chersich, C. C. J. Catal. 1986,
98, 102.
Simon, G.; Germain, J . E. Bull. SOC. Chim. Fr. 1975,11, 2617.
Slinkard, W. E.; de Groot, P. B. J. Catal. 1979,57, 476.
Suvorov, B. V.; Gutsalyuk, G. N.; Bukeikhanov, N. R. Akad. Nauk.
Catal. 1985, 18, 335.
Chem. 1983,87,754.
Kaz. SSR Ser. Khim. 1972,22(2), 87.
Svintsova, L. G.; Boldyrev, N. A.; Korneichuck, G. P. Kinet. Katal.
1977, 18, 590.
Tarama, K.; Teranishi, S.; Yoshida, S.; Tamura, N. Proceedings, 3rd
International Congress on Catalysis; Elsevier: Amsterdam, 1964;
Vol. I, p 282.
van der Berg, P. J.; van der Wiele, K.; den Ridder, J . J . Proceedings,
8t h Znternational Congress on Catalysis; Dechema: West Berlin,
J uly 1984; Vol. V, p 393.
van Hengstum, A. J .; van Ommen, J . G.; Bosch, H.; Gellings, P. J .
Appl . Catal. 1983a, 8, 369.
van Hengstum, A. J .; van Ommen, J . G.; Bosch, H.; Gellings, P. J .
Appl . Catal. 1983b, 5, 207.
Vanhove, D.; Blanchard, M. Bull. SOC. Chim. Fr. 1971, 3391.
Vejux, A.; Courtine, P. J. Solid State Chem. 1978, 23, 93.
Wachs, I. E.; Saleh, R. Y.; Chan, S. S.; Chersich, C. C. Aml . Catal.
- _
1985, 15, 339.
Wainwright, M. S.; Foster, N. R. Catal. Rev.-Sci. Eng. 1979,19,211.
Weiss, F.; Marion, J.; Metzger, J .; Cognion, J. M. Kinet. Katal. 1973,
14, 45.
564.
Wongard, W. C.; Nobe, K. Ind. Eng. Chem. Prod. Res. Dev. 1984,23,
Woning, J.; van Santen, R. A. Chem. Phys. Let t . 1983, 101(6), 541.
Yoshida, S.; Iguchi, T.; Ishida, S.; Tarama, K. Bull. Chem. SOC. Jpn.
1972, 45, 376.
Received f or review December 26, 1985
Revised manuscript received September 15, 1986
Accepted November 3, 1986
Selective Aromatization of Light Paraffins on
Platinum-Ion-Exchanged Gallium-Silicate Bifunctional
Cat a1 ys t s
Tomoyuki Inui,* Yoshinori Makino, Fumio Okazumi, Susumu Nagano, and
Akira Miyamoto
Depart ment of Hydrocarbon Chemi st ry, Faculty of Engineering, Kyot o Uni versi t y, Sakyo- ku, Kyot o 606,
Japan
Direct conversion of propane to aromatics was investigated on Ga-ion-exchanged H-ZSM-5 (Ga/
H-ZSM-5), Pt-ion-exchanged H-ZSM-5 (Pt/H-ZSM-5), gallium-silicate having the pentasil pore-
opening structure (Ga-silicate), and Pt-ion-exchanged Ga-silicate (Pt/Ga-silicate). Among the
catalysts, Pt/Ga-silicate exhibited the best performance; the conversion of propane and selectivity
to aromatics attained 92.3% and 60.770, respectively, at 600 OC. Although Ga/H-ZSM-5 with low
Ga content was active and selective for the propane aromatization, the increase in Ga content did
not improve the performance. As for Ga-silicate, various amounts of Ga were incorporated in the
framework of silicate crystal, and it was therefore more active and selective than the Ga/H-ZSM-5.
Although Pt/H-ZSM-5 was much more active than Ga-silicate, the former catalyst was not selective
for the aromatization. The Pt/Ga-silicate was also effective for the aromatization of such light
paraffins as ethane, butane, and pentane. It was also found that Pt in Pt/Ga-silicate plays an
important role to reduce the catalyst decay and to regenerate the decayed catalyst with air. Bi-
functional roles of Pt and Ga were proposed to understand catalytic behaviors of the Pt/Gasilicate.
Effective utilization of C24, light paraffins is one of the
most important subjects of investigation in both chemistry
and industry of hydrocarbons. Much attention has re-
cently been given to a catalytic process for the direct
conversion of the light paraffins to aromatic hydrocarbons
such as benzene, toluene, and xylenes (e.g., Chu, 1978;
Chester and Chu, 1982; Dave et al., 1982; J ahnson and
Hilder, 1984; Inui and Okazumi, 1984a; Mole et al., 1985;
Engelen et al., 1985; Inomata et al., 1985; Inui et al.,
1986a,b). These aromatics can be utilized as a booster for
the high-octane-number gasoline and are important as
fundamental raw materials in petrochemical industry. It
has been found that Ga- or Zn-ion-exchanged H-ZSM-5
(Ga/H-ZSM-5 or Zn/H-ZSM-5) is useful for this reaction
(e.g., Chu, 1978; Chester and Chu, 1982; Dave et al., 1982;
Mole et al., 1985). We have also shown that Pt-ion-ex-
changed H-ZSM-5 (Pt/H-ZSM-5) is effective for the re-
action (Inui and Okazumi, 1984a; Inui et al., 198613).
However, further improvements of catalysts seem neces-
sary to establish the process for the direct conversion of
the light paraffins to aromatics.
ZSM-5 is an aluminosilicate crystal having the pentasil
pore-opening structure. The aluminum ingredient in
ZSM-5 is responsible for the formation of strong acid sites
which play active sites for various reactions. The pore
structure of ZSM-5 leads to various types of shape selec-
tivity reactant shape selectivity, product shape selectivity,
and transition-state shape selectivity (e.g., Weisz, 1981;
08S8-5885/87/2626-0647~0~.50~0 0 1987 American Chemical Society
Das könnte Ihnen auch gefallen
- Childhood Dream Job - AstronautDokument1 SeiteChildhood Dream Job - AstronautJuan Camilo HenaoNoch keine Bewertungen
- Nitrogen BlanketingDokument7 SeitenNitrogen Blanketing3bandhuNoch keine Bewertungen
- Purge With NitrogenDokument5 SeitenPurge With Nitrogendeion29100% (1)
- Creative Science & Research - Screen Printing Press (2004)Dokument21 SeitenCreative Science & Research - Screen Printing Press (2004)amrandconan100% (2)
- Algebra Angel Cap9Dokument66 SeitenAlgebra Angel Cap9Ariel GomezNoch keine Bewertungen
- Parker Tank Blanketing White Paper PDFDokument4 SeitenParker Tank Blanketing White Paper PDFshashi kant kumarNoch keine Bewertungen
- Uniteolstates Patent Office: "133.2535.lftii?iiii'tfiiiw 7 'Dokument2 SeitenUniteolstates Patent Office: "133.2535.lftii?iiii'tfiiiw 7 'Juan Camilo HenaoNoch keine Bewertungen
- Dow Filmtec BW30XFR 400 34iDokument2 SeitenDow Filmtec BW30XFR 400 34iJuan Camilo HenaoNoch keine Bewertungen
- Batista 2013Dokument13 SeitenBatista 2013Juan Camilo HenaoNoch keine Bewertungen
- Dow Filmtec BW30XFR 400 34iDokument2 SeitenDow Filmtec BW30XFR 400 34iJuan Camilo HenaoNoch keine Bewertungen
- Learn English Podcasts Elementary 01 01 Support Pack TranscriptDokument22 SeitenLearn English Podcasts Elementary 01 01 Support Pack Transcriptpramithbuddika100% (1)
- 02bfe50dacb1fe39e0000000 PDFDokument50 Seiten02bfe50dacb1fe39e0000000 PDFJuan Camilo HenaoNoch keine Bewertungen
- Created by Trial Version: HE-3 HE-4Dokument1 SeiteCreated by Trial Version: HE-3 HE-4Juan Camilo HenaoNoch keine Bewertungen
- Papper Co2Dokument15 SeitenPapper Co2Juan Camilo HenaoNoch keine Bewertungen
- Flash ConceptsDokument2 SeitenFlash ConceptsJuan Camilo HenaoNoch keine Bewertungen
- Extreme Independence - All Combinations of 4 LimbsDokument3 SeitenExtreme Independence - All Combinations of 4 LimbsJuan Camilo HenaoNoch keine Bewertungen
- 1 PBDokument11 Seiten1 PBJuan Camilo HenaoNoch keine Bewertungen
- Design and Operation of Fractionating ColumnsDokument6 SeitenDesign and Operation of Fractionating ColumnsJuan Camilo HenaoNoch keine Bewertungen
- CLASS 300 Welding Neck & Slip-On Flanges: ASTM A-105 & A181-CLASS 60Dokument3 SeitenCLASS 300 Welding Neck & Slip-On Flanges: ASTM A-105 & A181-CLASS 60Juan Camilo HenaoNoch keine Bewertungen
- Aspen HYSYS DynamicsDokument3 SeitenAspen HYSYS DynamicsJuan Camilo HenaoNoch keine Bewertungen
- IT H Unit IGDokument19 SeitenIT H Unit IGJuan Camilo HenaoNoch keine Bewertungen
- Zirconia-Supported Vanadium Oxide Catalysts For Ammoxidation and Oxidation of Toluene: A Characterization and Activity StudyDokument22 SeitenZirconia-Supported Vanadium Oxide Catalysts For Ammoxidation and Oxidation of Toluene: A Characterization and Activity StudyJuan Camilo HenaoNoch keine Bewertungen
- Poços de CaldasDokument8 SeitenPoços de CaldasAnninha FlavioNoch keine Bewertungen
- United States' Patent Office: Patented July 20, 1948Dokument5 SeitenUnited States' Patent Office: Patented July 20, 1948Juan Camilo HenaoNoch keine Bewertungen
- Dowtherm A PropertiesDokument2 SeitenDowtherm A Propertiesflashiitm100% (1)
- Benzoguanamine: Product Specification SheetDokument1 SeiteBenzoguanamine: Product Specification SheetJuan Camilo HenaoNoch keine Bewertungen
- Benzoguanamine: Product Specification SheetDokument1 SeiteBenzoguanamine: Product Specification SheetJuan Camilo HenaoNoch keine Bewertungen
- AbsorptionStripping PDFDokument25 SeitenAbsorptionStripping PDFJuan Camilo HenaoNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5784)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (890)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (399)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (587)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (265)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (72)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2219)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (119)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Structure-First YearDokument12 SeitenStructure-First Yearioemk251Noch keine Bewertungen
- Astm D 2036-98 Standard Test Methods For Cyanides in Water PDFDokument20 SeitenAstm D 2036-98 Standard Test Methods For Cyanides in Water PDFGianinaRoncalChávezNoch keine Bewertungen
- Summative Test (Physics)Dokument3 SeitenSummative Test (Physics)Allen Paul GamazonNoch keine Bewertungen
- Rheopecty: Confusion Between Rheopectic and Dilatant FluidsDokument2 SeitenRheopecty: Confusion Between Rheopectic and Dilatant Fluidsengineer bilalNoch keine Bewertungen
- Channel Tut v1Dokument11 SeitenChannel Tut v1Umair IsmailNoch keine Bewertungen
- Induction MotorDokument27 SeitenInduction MotorNandhini SaranathanNoch keine Bewertungen
- Ch-27.2 Crystalline Materials - Detects in Crystalline MaterialsDokument102 SeitenCh-27.2 Crystalline Materials - Detects in Crystalline MaterialsasjfgauojfgfNoch keine Bewertungen
- 100 Years Werner HeisenbergDokument308 Seiten100 Years Werner Heisenbergmvmbapple100% (3)
- Orifice SizingDokument2 SeitenOrifice SizingAvinav Kumar100% (1)
- Triggering and Dynamic Evolution of The LUSI Mud Volcano, IndonesiaDokument14 SeitenTriggering and Dynamic Evolution of The LUSI Mud Volcano, IndonesiaAndika Bayu AjiNoch keine Bewertungen
- Engleski Jezik-Seminarski RadDokument18 SeitenEngleski Jezik-Seminarski Radfahreta_hasicNoch keine Bewertungen
- Roller Coaster Physics Coursework GcseDokument7 SeitenRoller Coaster Physics Coursework Gcseafjwdbaekycbaa100% (2)
- Active Suspension Using SkyhookDokument14 SeitenActive Suspension Using SkyhookSandor CleganeNoch keine Bewertungen
- Lukac Perkowski Book Introduction and Quantum MechanicsDokument104 SeitenLukac Perkowski Book Introduction and Quantum MechanicsSitalk TeresNoch keine Bewertungen
- Eco Brick Paper 140314 - Full Paperr2Dokument30 SeitenEco Brick Paper 140314 - Full Paperr2zero kakumaruNoch keine Bewertungen
- WhatReallyisUHPC TowardsaGlobalDefinition UHPC2018FuzhouChina July292018Dokument14 SeitenWhatReallyisUHPC TowardsaGlobalDefinition UHPC2018FuzhouChina July292018Данило ГадайчукNoch keine Bewertungen
- Parameter Adaptation of Reduced Order Models For Three-Dimensional Flutter AnalysisDokument9 SeitenParameter Adaptation of Reduced Order Models For Three-Dimensional Flutter AnalysisAtsis PapadopoulosNoch keine Bewertungen
- SCH 305Dokument34 SeitenSCH 305Qwin NajyaNoch keine Bewertungen
- Abutment Design CalculationsDokument13 SeitenAbutment Design CalculationsFitsum Abebe100% (1)
- Power Transmission: Components Used To Transmit Power: Gears, Belt, Clutch and BrakesDokument17 SeitenPower Transmission: Components Used To Transmit Power: Gears, Belt, Clutch and Brakesrip111176Noch keine Bewertungen
- Rife Energy Medicine Cures DiseasesDokument5 SeitenRife Energy Medicine Cures DiseasesOna OnićNoch keine Bewertungen
- Case Study: Multi Life Cycle Center: R. Knoth, M. Brandstötter, B. Kopacek, P. KopacekDokument5 SeitenCase Study: Multi Life Cycle Center: R. Knoth, M. Brandstötter, B. Kopacek, P. KopacekgroventoNoch keine Bewertungen
- Chemistry Dictionary PDFDokument356 SeitenChemistry Dictionary PDFMuhammad YounusNoch keine Bewertungen
- NX Nastran 3 Release GuideDokument28 SeitenNX Nastran 3 Release GuideMSC Nastran BeginnerNoch keine Bewertungen
- DipolesDokument42 SeitenDipolesRajat GiriNoch keine Bewertungen
- Lect 4Dokument28 SeitenLect 4george samyNoch keine Bewertungen
- Computer Simulation For Large Scale Bioprocess Design: S.A. Rouf, P.L. Douglas, M. Moo-Young, J.M. ScharerDokument6 SeitenComputer Simulation For Large Scale Bioprocess Design: S.A. Rouf, P.L. Douglas, M. Moo-Young, J.M. Scharerdaimon_pNoch keine Bewertungen
- Chap3 (D) MillDokument22 SeitenChap3 (D) MilljojoNoch keine Bewertungen
- BS en 10297 2Dokument36 SeitenBS en 10297 2Sharma ShailenNoch keine Bewertungen
- Finite Element Analysis of A Cracked Cantilever Beam: Research PaperDokument5 SeitenFinite Element Analysis of A Cracked Cantilever Beam: Research PaperSanketNoch keine Bewertungen