Beruflich Dokumente
Kultur Dokumente
Proposal Vitamin C Content in Fruit
Hochgeladen von
Nurul AshikinOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Proposal Vitamin C Content in Fruit
Hochgeladen von
Nurul AshikinCopyright:
Verfügbare Formate
PROPOSAL
ANALYSIS OF VITAMIN C (ASCORBIC ACID) IN 10
FRUITS USING UV- SPECTROPHOTOMETER
ROYAL COLLEGE OF MEDICINE PERAK (UNIKL-RCMP)
SSM MODULE YEARR 2 2012
GROUP 33
RESEARCHERS
NURUL ATIQAH MAHMUD
57260211083
NUR HIDAYU FHATIHAH GHADZALI @GHAZALI
57260211062
NUR SYUHAINA ABU BAKAR
57260211126
MAZRIA AMANI MAZLAN
57260211130
SITI RASHIDAH ABU BAKAR
57260211051
SUPERVISOR
Checked by:
Approved by:
1.
INTRODUCTION
Vitamin C or ascorbic acid is a six-carbon lactone, which is synthesized from glucose
by many animals. Vitamin C is synthesized in the liver in some mammals and in the
kidney in birds and reptiles. However, this is not synthesized in a human body, which
makes it an essential nutrient which human obtained through dietary. Due to
deficiency of vitamin C, human suffer from the potentially lethal disease scurvy.
Vitamin C is largely distributed in fruits and plants that make it accessible to be
obtained. It is abundantly found in watermelon, papaya, oranges, apples, strawberries,
pineapple, mango and guava. It is also found in green leafy vegetables, cabbages,
cauliflower, broccoli and tomatoes.
Vitamin C is one of the important water-soluble vitamins. It is essential for
collagen, carnitine as well as neurotransmitter biosynthesis. Hence, vitamin C is
undeniably an important nutrient for metabolic processes in human. The current US
recommended daily allowance (RDA) for ascorbic acid ranges 100-120-mg/per day
for adults. Many health benefits have been attributed such as being a powerful
antioxidant, anti-atherogenic, anti-carcinogenic, immune-modulator and prevent cold
(K.A Naidu et. al, 2003).
Lately there has been mast production of synthetic or nutraceutical ascorbic
acid in the market to keep up with the demand of vitamin C supplement by the
consumers. A study showed that adults taking synthetic vitamin C supplementation
had serious effect. The result shows that synthetic vitamin C supplementation
decreases the exercise or training efficiency. This is because it depreciates the cellular
adaptations of exercise in the mitochondrial level.
Therefore, it is always best to consume natural vitamin C. Due to that, a
research to measure the content of vitamin C in five local fruits in Malaysia is carried
out to compare the ascorbic acid content between these fruits.
2.
LITERATURE REVIEW
A research study had been done regarding iodimetric determination of ascorbic acid
(vitamin C) in citrus fruits in year 2007. The study is conducted to determine the
ascorbic acid content of the juices from four different types of citrus fruits which are
orange, tangerine, grapefruit and lime. The study showed that orange has the highest
value of ascorbic acid (600 g /mL) followed by grape (446 g /mL) and then
tangerine (415 g /mL). Lime had the least value (306 g /mL). It is concluded that
orange would supply more ascorbic acid per milliliter for body need. From this
research, it is also concluded that the juices of oranges and tangerine are hereby
recommended, preferably, to be taken daily during meal at quantities sufficient to
meet daily vitamin C need (Izuagie et al. , 2007).
Besides, another research is carried out to quantify the total phenolics,
flavonoids and carotenoides, vitamin C contents, reported as antioxidants, in the
extracts of four locally available varieties of durian. In the research, the fruits are
extracted using dichloromethane: pentane (1:1 v/v) and subjected to study the various
quality parameters. The result showed the total of vitamin C contents.
This research also proved that caffeic acid and quercetin are the dominant
antioxidant substances found in durian. From this research, we know that the
bioactivity of ripe durian is high and the total polyphenols are the main contributors to
the overall antioxidant capacity. It is thus recommended to consume durian fruit for
uptake of antioxidant phytochemicals and vitamin C (Ashraf et al. , 2011).
3.
STUDY RATIONAL
Diets rich in fresh fruits and vegetables can protect us against chronic and
degenerative diseases (Joshipura et al. , 1999). The significant of this study is to study
the content of vitamin C in fresh fruits highly available in the market. Due to the side
effects of the synthetic supplementation of vitamin C, this study is carried out to find
the content of naturally available vitamin C in fresh fruits. This is significant to
educate the public that vitamin C supplementation can be taken naturally from fruits.
It is without a doubt that synthetic, nutraceutical vitamin C capsule available in the
market is modified chemically to prolong their time of consume. With this study, we
can help educate the public about the significant of eating fresh fruits that is readily
available in the market and therefore create awareness to take natural
supplementation, which is cheaper and healthier.
4.
OBJECTIVES
4.1.
GENERAL OBJECTIVES
4.1.1.
To determine the content of vitamin C in nine local fruits and an import fruit
in comparison with supplement vitamin C.
4.2.
SPECIFIC OBJECTIVES
4.2.1. To study the concentration of vitamin C using UV spectrophotometer method
with sodium oxalate acts as stabilizer.
4.2.2. To compare the ascorbic acid content between Orange and nine local fruits in
Malaysia which are:
4.2.2.1.
4.2.2.2.
4.2.2.3.
4.2.2.4.
4.2.2.5.
4.2.2.6.
4.2.2.7.
4.2.2.8.
4.2.2.9.
Guava
Mango
Durian
Pamelo
Banana
Pineapple
Star fruit
Papaya
Water apple/ bell fruit
4.2.3. To compare all the 10 fruits with the supplement vitamin C available in the
market.
4.2.4. To determine which fruits have the highest concentration of vitamin C.
4.2.5. To determine the pH value in each fruits used in the experiment.
4.2.6. To educate the public about the importance of vitamin C in daily diet.
4.2.7. To provide alternative for the public who want cheaper and healthier supply of
vitamin C.
5.0.
METHODOLOGY
There are many analytical methods have been reported to determine vitamin C
concentration such as titrimetry (Gunjan, 2012), electrochemical (Kim, 1989) and
potentiometric method (Leslie et al. , 1941). Some methods are available for
determination of ascorbic acid but only few are found to be used to determine both
forms of vitamin C (L-ascorbic acid and dehydroascorbic acid). It is very difficult to
choose a suitable method to determine the vitamin C contents in fruit juice. All the
methods have their own limitation for different purpose. (Mohammad et al. , 2007).
In this study, the method chosen is direct spectrophotometric method. It is
simple and highly sensitive method for determination of L-ascorbic acid in pure form.
This method used sodium oxalate (0.0056 mol/dm 3) as a stabilizer and molar
absorptivity (which does not require an extraction procedure) was 1.42x104 dm3mol1
cm-1 at 266nm. Beers law is obeyed in concentration range of 0.857-12.0 g ascorbic
acid/cm3 (Amra et al. , 2011).
5.1.
INSTRUMENTS
UV-Visible spectrophotometer using 1cm path length is used to measure the content
of vitamin C in each sample solutions. Digital pH meter is used to determine the pH
value in each sample solutions.
5.2.
REAGENTS
All reagents used are of analytical-reagent grade. Buffer solution (pH = 5.4), Sodium
oxalate solution (0.0056 mol/dm3) and L-ascorbic acid solution (1.13x10-3 mol/dm3).
5.3.
REAGENT PREPARATION
Buffer solution (pH = 5.4)
A mixture of potassium dihydrogenphosphate (0.03 mol/dm 3) and disodium
hydrogenphosphate (8.99x10-4 mol/dm3) is prepared by dissolving 4.08 g of KH 2PO4
(Fluka) and 0.16 g of Na2HPO4.2H2O (Merck) in 100 cm3 of distilled water.
Sodium oxalate solution (0.0056 mol/dm3)
This solution is prepared by dissolving 0.75 g of sodium oxalate (Sigma) in 1000 cm 3
of the buffer solution.
L-Ascorbic acid solution (1.13x10-3 mol/dm3)
A 0.05 g amount of L-ascorbic acid (Riedel-de Han) was dissolved in 250 cm 3 of the
sodium oxalate solution.
5.4.
SAMPLE
10 raw material of fresh fruits used in this study are purchased from a local
supermarket on the day of analysis. Guava (Psidium guajava), mango (Mangifera
indica), durian (Durio zibethinus), pamelo (Citrus grandis), banana (Musa spp),
pineapple (Ananas comosus), star fruit (Averrhoa carambola), papaya (Carica
papaya), water apple (Syzygium samarangense). The fruits chosen are in ripest state
(according to assumption).
5.5.
SAMPLE PREPARATION
All the 10 sample of fruits are washed thoroughly with water and the juices are
extracted manually using juice squeezer. All samples are filtered to remove pulp and
seed. After that they are stored in already labelled plastic containers. A small amount
of fruit juice is sufficient for the determination of ascorbic acid in these samples.
5.6.
GENERAL PROCEDURE
Transfer a portion of the sample solution containing 60 300 g of L-ascorbic acid to
a 25 cm3 standard flask. Dilute to the mark with the (0.0056 mol/dm 3) sodium oxalate
solution and measure the absorbance at 266 nm against the sodium oxalate solution as
a blank.
5.7.
DETERMINATION OF L-ASCORBIC ACID IN FRUIT SAMPLES
Transfer an accurately weighed amount of powder obtained from several tablets into a
100 cm3 volumetric flask, dissolve and make up to the mark with the (0.0056
mol/dm3) sodium oxalate solution. Filter and dilute a suitable aliquot of the filtrate to
50 cm3 with the stabilizer solution. Take an aliquot of the final solution and determine
the ascorbic acid content as described under general procedure.
5.8.
DETERMINATION pH IN FRUIT SAMPLES
Set the digital pH meter to set mode and insert the electrode into the solution samples.
Wait the reading to stabilize before record the value appeared on the display. Always
rinse the electrode before use it to test other sample solutions.
6.0.
RESULTS
Plan for data analysis and interpretation
Table 1: Biochemical characteristics of different types of fruits in Malaysia
Types of fruits
Vitamin C Contents(mg/100g)
pH
Orange
Bell fruit
Guava
Banana
Mango
Papaya
Starfruit
Pineapple
Durian
Pomelo
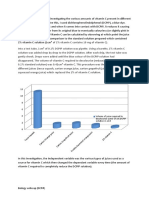
Figure 1: Experimental and literature values of vitamin C content for fresh
tropical fruits
7.0.
REFERENCES
K.A Naidu (2003) Vitamin C in human health and disease is still a mystery? An
overview, Nutrition Journal, 2(7), 1.
Joshipura, Alberto. A, JoAnn E.M, Meir J. S, Eric B.R, Frank E. S, Charles H.H,
Donna S, Walter C.W (1999) Fruits and vegetables Intake in Relation to Risk
of Ischemic Stroke, The Journal of American Medical Association (JAMA),
282(13), 1237-1239.
Ashraf, M. A., Maah, M. J., Yusoff, I., Mahmood, K., & Wajid, A. (2011). Study of
Antioxidant Potential of Tropical Fruit. International Journal of Bioscience,
Biochemistry and Bioinformatics, 1(1), 53-57.
Izuagie, A. A., & Izuagie, F. O. (2007). Iodimetric Determination of Ascorbic Acid
(Vitamin C) in Citrus Fruits. Research Journal of Agriculture and Biological
Sciences, 3(5), 367-369).
Gunjan Kashyap & Mangla Dave Gautam (2012). Analysis of Vitamin C in
Commercial and Naturals substances by Iodometric Titration found in Nimar
and Malwa region. Journal of Scientific Research in Pharmacy, 2277-9469.
Kim HJ. (1989). Determination of total vitamin C by ion exclusion chromatography
with electrochemical detection. US National Library of Medicine National
Institutes of Health Journal, Jul-Aug; 72(4): 681-6.
Leslie J. Harris, L. W. Mapson & Y. L. Wang (1941). A Simple Potentiometric
Method for Determining Ascorbic Acid, Suitable for Use with Coloured
Extracts. Nutritional Laboratory, University of Cambridge and Medical
Research Council.
Mohammad Mizanur Rahman Khan & Mohammad Mazedul Hosain (2007). Analysis
of Vitamin C (ascorbic acid) Contents in Various Fruits and Vegetables by
UV-spectrophotometry. Bangladesh J. Sci. Ind. Res. 42(4), 417-424, 2007.
Amra Selimovi, Mirsad Salki & Amel Selimovi (2011). Direct Spectrophotometric
Determination of L-ascorbic acid in Pharmaceutical Preparations Using
Sodium oxalate as a Stabilizer. International Journal of Basic & Applied
Sciences IJBAS-IJENS Vol: 11 No: 02.
Das könnte Ihnen auch gefallen
- Jaggery ProcessDokument4 SeitenJaggery ProcessAsh1ScribdNoch keine Bewertungen
- Atomic Adsorption SpectrosDokument14 SeitenAtomic Adsorption SpectrosRzi Danil Ishutin0% (1)
- Electrogravimetry: Electrogravimetric Analysis V Semester Inorganic ChemistryDokument2 SeitenElectrogravimetry: Electrogravimetric Analysis V Semester Inorganic Chemistryvandv printsNoch keine Bewertungen
- Gravimetric Sulfate DeterminationDokument3 SeitenGravimetric Sulfate DeterminationkatlegoNoch keine Bewertungen
- Research ProposalDokument6 SeitenResearch ProposalrubyshreeNoch keine Bewertungen
- Green Chemistry and Its Role For Sustainability: PresenterDokument16 SeitenGreen Chemistry and Its Role For Sustainability: PresenterMandeep BatraNoch keine Bewertungen
- Bio ReportDokument8 SeitenBio ReportTharshini_Indr_6713Noch keine Bewertungen
- ConclusionDokument1 SeiteConclusionNitin KashyapNoch keine Bewertungen
- Biology November ReportDokument3 SeitenBiology November ReportIndrani GoswamiNoch keine Bewertungen
- Gobena's Thesis Commented 1Dokument69 SeitenGobena's Thesis Commented 1Gobena AdebaNoch keine Bewertungen
- S Determination of Caffeine in BeveragesDokument5 SeitenS Determination of Caffeine in BeveragesVioleta Grigoras100% (1)
- Purification of Surface Water From Orange Peel WastesDokument4 SeitenPurification of Surface Water From Orange Peel WastesJohn Bryan Aldovino100% (1)
- Simultaneous Determination of Salicylic Acid and Acetylsalicylic AciDokument5 SeitenSimultaneous Determination of Salicylic Acid and Acetylsalicylic Aciiabureid7460Noch keine Bewertungen
- Yeast LabDokument4 SeitenYeast LabAtikah OlivaNoch keine Bewertungen
- LONG CAFFEINE UVLong Caffeine UvDokument8 SeitenLONG CAFFEINE UVLong Caffeine UvzaNoch keine Bewertungen
- Computer Investigatory Project CBSE Class 12Dokument10 SeitenComputer Investigatory Project CBSE Class 12sidharth1196100% (3)
- Biology ProjectDokument9 SeitenBiology ProjectAbdul Sami0% (1)
- 6 - Toxicants Formed During ProcessingDokument45 Seiten6 - Toxicants Formed During Processingismailmannur100% (1)
- Ascorbic Acid ExperimentDokument10 SeitenAscorbic Acid ExperimentJoa YupNoch keine Bewertungen
- Chemistry Investigatory Project OnDokument15 SeitenChemistry Investigatory Project OnjibinNoch keine Bewertungen
- Dokumen - Tips - Toxicants Formed During Food Processing 2 PDFDokument45 SeitenDokumen - Tips - Toxicants Formed During Food Processing 2 PDFHồ Ngọc Thanh HiềnNoch keine Bewertungen
- I EVALUATION OF THE TOXICITY OF THE METHANOLIC EXTRACT OF MORINGA OLEIFERA LEAVES ON MICEDokument53 SeitenI EVALUATION OF THE TOXICITY OF THE METHANOLIC EXTRACT OF MORINGA OLEIFERA LEAVES ON MICEAdetolaNoch keine Bewertungen
- Chromatography - The Most Versatile Method of Chemical AnalysisDokument438 SeitenChromatography - The Most Versatile Method of Chemical AnalysisdnabrasilNoch keine Bewertungen
- Vitamin C Write-UpDokument5 SeitenVitamin C Write-UpannafiiNoch keine Bewertungen
- 4A F16 Exp 04 Absorbance and Fluorescence Spectros PDFDokument27 Seiten4A F16 Exp 04 Absorbance and Fluorescence Spectros PDFWanqing HeNoch keine Bewertungen
- Food MCB II NotesDokument73 SeitenFood MCB II NotesRichard Simon KisituNoch keine Bewertungen
- ThesisDokument175 SeitenThesisselvaraj2482100% (2)
- Chemical Equilibrium in A Liquid PhaseDokument7 SeitenChemical Equilibrium in A Liquid PhasePevie Anne Tenebroso100% (1)
- Enzyme, A Substance That Acts As ADokument7 SeitenEnzyme, A Substance That Acts As AAnanyaNoch keine Bewertungen
- Estimation of Sugar by HAGEDORN - JensonDokument3 SeitenEstimation of Sugar by HAGEDORN - JensonTrung Mai VanNoch keine Bewertungen
- Determination of The PH Value of Water Using A PH MeterDokument7 SeitenDetermination of The PH Value of Water Using A PH MeterRome Dionisio Komicho100% (1)
- Advantages of Liquid Liquid Extraction SystemDokument5 SeitenAdvantages of Liquid Liquid Extraction Systemkumarsachin3801100% (1)
- Complete AAS N ICPDokument14 SeitenComplete AAS N ICPMaxvicklye Rayner100% (1)
- DCPIP Write UpDokument2 SeitenDCPIP Write UpHashim Chishty0% (1)
- Cutm Examination HandbookDokument73 SeitenCutm Examination HandbookTarun Kumar PradhanNoch keine Bewertungen
- Lab Manual For Down Stream Process LabDokument31 SeitenLab Manual For Down Stream Process LabShaasvath AishvaryaNoch keine Bewertungen
- Chemistry Project On Presence of Insecticides Pesticides in Fruits VegetablesDokument9 SeitenChemistry Project On Presence of Insecticides Pesticides in Fruits VegetablesIshu SinghNoch keine Bewertungen
- Chemistry Investigatory Project ReportDokument9 SeitenChemistry Investigatory Project ReportRaj ChaudhariNoch keine Bewertungen
- Atr Ft-Ir Imaging of Acetic AcidDokument10 SeitenAtr Ft-Ir Imaging of Acetic AcidMon RonquilloNoch keine Bewertungen
- C FABCON FINALEtotooHD720pDokument39 SeitenC FABCON FINALEtotooHD720pChloegelo MendozaNoch keine Bewertungen
- Experiment On Vitamins - CONGSONDokument3 SeitenExperiment On Vitamins - CONGSONShayne Angelique CongsonNoch keine Bewertungen
- Biology ProjectDokument4 SeitenBiology ProjectAnthony FalconNoch keine Bewertungen
- Vitmin C ReportDokument14 SeitenVitmin C ReportOdongo TonnyNoch keine Bewertungen
- Make Biochemistry Manual: Serum Calcium EstimateDokument3 SeitenMake Biochemistry Manual: Serum Calcium Estimatehaseeb ShafaatNoch keine Bewertungen
- Chemistry Investigatory ProjectDokument12 SeitenChemistry Investigatory ProjectHuzaifa LokhandwalaNoch keine Bewertungen
- Chemistry Investigatory ProjectDokument16 SeitenChemistry Investigatory Project10A 1 ANSH PANARANoch keine Bewertungen
- FTIR Spectrometer AnalysisDokument16 SeitenFTIR Spectrometer AnalysisßraiñlĕsšȜĭnšteĭñNoch keine Bewertungen
- Harmonized FTPE Curriculum-FinalDokument222 SeitenHarmonized FTPE Curriculum-Finalprashant_cool_4_u100% (1)
- Main Project TopicsDokument1 SeiteMain Project Topics1234cbriNoch keine Bewertungen
- Measuring The Vitamin C Content of Foods and Fruit Juices Ss 36Dokument1 SeiteMeasuring The Vitamin C Content of Foods and Fruit Juices Ss 36Aji CuteMadNoch keine Bewertungen
- Tissue CultureDokument32 SeitenTissue CultureYash WardhanNoch keine Bewertungen
- Difference Between AC and DC MotorDokument2 SeitenDifference Between AC and DC MotorYrty KdfgNoch keine Bewertungen
- (18530) Sheet 1 Electrochemistry B PDFDokument99 Seiten(18530) Sheet 1 Electrochemistry B PDFAnuragPandey100% (1)
- Prac 4 ReportDokument15 SeitenPrac 4 ReportAvanthi Moodley100% (1)
- Chemistry Investigatory ProjectDokument11 SeitenChemistry Investigatory ProjectAmalendu Sundar MandalNoch keine Bewertungen
- CHE555 Assignment 1 Mac 2015Dokument2 SeitenCHE555 Assignment 1 Mac 2015Jaja TeukieNoch keine Bewertungen
- Ref 3Dokument12 SeitenRef 3ANDRES FELIPE MOSQUERA MARTINEZNoch keine Bewertungen
- 265 2416 3 PB PDFDokument5 Seiten265 2416 3 PB PDFjessica sidhartaNoch keine Bewertungen
- Yarkwanand OketundeDokument8 SeitenYarkwanand Oketundejihansalmaazhaarfirdaus00Noch keine Bewertungen
- Acido Ascorbico en Jugos de Fruta y Su Rata de Perdida Por AlamacenamientoDokument4 SeitenAcido Ascorbico en Jugos de Fruta y Su Rata de Perdida Por AlamacenamientoYuliana PerezNoch keine Bewertungen
- HFMD N MeningitisDokument19 SeitenHFMD N MeningitisNurul AshikinNoch keine Bewertungen
- Fracture Classification and Eponym Fractures: Supervisor: DR SyarifahDokument56 SeitenFracture Classification and Eponym Fractures: Supervisor: DR SyarifahNurul AshikinNoch keine Bewertungen
- Obsteric EmergenciesDokument48 SeitenObsteric EmergenciesNurul AshikinNoch keine Bewertungen
- Alzheimer FolioDokument15 SeitenAlzheimer FolioNurul AshikinNoch keine Bewertungen
- Jyotish - 2003 Sri Jagannath Center - Narayan Iyer - Divisional Charts - Divining Through DivisionsDokument36 SeitenJyotish - 2003 Sri Jagannath Center - Narayan Iyer - Divisional Charts - Divining Through DivisionsDeepa MishraNoch keine Bewertungen
- E TN SWD Csa A23 3 94 009 PDFDokument5 SeitenE TN SWD Csa A23 3 94 009 PDFRazvan RobertNoch keine Bewertungen
- Nursing Care Plan: Pt.'s Data Nursing Diagnosis GoalsDokument1 SeiteNursing Care Plan: Pt.'s Data Nursing Diagnosis GoalsKiran Ali100% (3)
- GearsDokument14 SeitenGearsZulhilmi Chik TakNoch keine Bewertungen
- Database Management Systems Lab ManualDokument40 SeitenDatabase Management Systems Lab ManualBanumathi JayarajNoch keine Bewertungen
- BedDokument17 SeitenBedprasadum2321Noch keine Bewertungen
- Counselling Goes To The Movies: Antwone Fisher (2002)Dokument12 SeitenCounselling Goes To The Movies: Antwone Fisher (2002)Azizul MohamadNoch keine Bewertungen
- Essay EnglishDokument4 SeitenEssay Englishkiera.kassellNoch keine Bewertungen
- Shielded Metal Arc Welding Summative TestDokument4 SeitenShielded Metal Arc Welding Summative TestFelix MilanNoch keine Bewertungen
- ID2b8b72671-2013 Apush Exam Answer KeyDokument2 SeitenID2b8b72671-2013 Apush Exam Answer KeyAnonymous ajlhvocNoch keine Bewertungen
- tf00001054 WacDokument22 Seitentf00001054 WacHritik RawatNoch keine Bewertungen
- Concentrating Partial Entanglement by Local OperationsDokument21 SeitenConcentrating Partial Entanglement by Local OperationsbhpliaoNoch keine Bewertungen
- Musculoskeletan Problems in Soccer PlayersDokument5 SeitenMusculoskeletan Problems in Soccer PlayersAlexandru ChivaranNoch keine Bewertungen
- City/ The Countryside: VocabularyDokument2 SeitenCity/ The Countryside: VocabularyHương Phạm QuỳnhNoch keine Bewertungen
- The Changeling by Thomas MiddletonDokument47 SeitenThe Changeling by Thomas MiddletonPaulinaOdeth RothNoch keine Bewertungen
- MINIMENTAL, Puntos de Corte ColombianosDokument5 SeitenMINIMENTAL, Puntos de Corte ColombianosCatalina GutiérrezNoch keine Bewertungen
- Chhabra, D., Healy, R., & Sills, E. (2003) - Staged Authenticity and Heritage Tourism. Annals of Tourism Research, 30 (3), 702-719 PDFDokument18 SeitenChhabra, D., Healy, R., & Sills, E. (2003) - Staged Authenticity and Heritage Tourism. Annals of Tourism Research, 30 (3), 702-719 PDF余鸿潇Noch keine Bewertungen
- Win Tensor-UserGuide Optimization FunctionsDokument11 SeitenWin Tensor-UserGuide Optimization FunctionsadetriyunitaNoch keine Bewertungen
- 28Dokument12 Seiten28physicsdocsNoch keine Bewertungen
- Department of Chemistry Ramakrishna Mission V. C. College, RaharaDokument16 SeitenDepartment of Chemistry Ramakrishna Mission V. C. College, RaharaSubhro ChatterjeeNoch keine Bewertungen
- Signalling in Telecom Network &SSTPDokument39 SeitenSignalling in Telecom Network &SSTPDilan TuderNoch keine Bewertungen
- Del Monte Usa Vs CaDokument3 SeitenDel Monte Usa Vs CaChe Poblete CardenasNoch keine Bewertungen
- Types of Numbers: SeriesDokument13 SeitenTypes of Numbers: SeriesAnonymous NhQAPh5toNoch keine Bewertungen
- Barangay Sindalan v. CA G.R. No. 150640, March 22, 2007Dokument17 SeitenBarangay Sindalan v. CA G.R. No. 150640, March 22, 2007FD BalitaNoch keine Bewertungen
- Klabin Reports 2nd Quarter Earnings of R$ 15 Million: HighlightsDokument10 SeitenKlabin Reports 2nd Quarter Earnings of R$ 15 Million: HighlightsKlabin_RINoch keine Bewertungen
- Abnormal PsychologyDokument13 SeitenAbnormal PsychologyBai B. UsmanNoch keine Bewertungen
- Lesson 3 The Prodigal SonDokument2 SeitenLesson 3 The Prodigal Sonapi-241115908Noch keine Bewertungen
- Goal Ball Lesson PlanDokument4 SeitenGoal Ball Lesson Planapi-378557749100% (1)
- MAT2355 Final 2002Dokument8 SeitenMAT2355 Final 2002bojie_97965Noch keine Bewertungen
- 2023-Tutorial 02Dokument6 Seiten2023-Tutorial 02chyhyhyNoch keine Bewertungen