Beruflich Dokumente
Kultur Dokumente
Article Wjpps 1386011514
Hochgeladen von
Donig FermanianOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Article Wjpps 1386011514
Hochgeladen von
Donig FermanianCopyright:
Verfügbare Formate
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES
P.Ramasubramaniyan et al.
World Journal of Pharmacy and Pharmaceutical Sciences
Volume 2, Issue 6, 5412-5418.
Research Article
ISSN 2278 4357
EQUIPMENT QUALIFICATION AND VALIDATION OF TABLET
COMPRESSION MACHINE
P.Ramasubramaniyan*, N.Sharanya, Palanichamy.S, T.Srinag, P.Pavani, Solairaj.P
Department of Pharmaceutics, Sankaralingam Bhuvaneswari College of Pharmacy, Sivakasi,
Tamil Nadu, India.
Article Received on
16 August 2013,
ABSTRACT
Revised on 15 Sept 2013,
Accepted on 05 November
2013
undertaken to demonstrate the intended use and performance of the
Qualification an ideal step for equipment validation is the action
utilities and equipment. The individual steps of qualification such as
design, installation, operational and performance qualification were
*Correspondence for
done in order to qualify the equipment. Blue print of equipment
Author:
validation was also included. In this article the compression parameters
* Dr. P. Ramasubramaniyan,
such as compression force, turret rpm and feeder rpm were studied.
Professor, Department of
The process capability of the equipment was studied by observing the
Pharmaceutics, Sankaralingam
weight of 20 tablets, disintegration time, friability, individual weight
Bhuvaneswari College of
variation, thickness and hardness. These parameters were studied at
Pharmacy, Sivakasi, Tamil
Nadu, INDIA.
ramskit@yahoo.com
three different speeds and the best results were obtained at the
optimum speed (40 rpm).
KEYWORDS:
Qualification,
validation,
design,
installation,
operational, performance, compression
INTRODUCTION
Qualification: It refers to activities undertaken to demonstrate that utilities and equipment
are suitable for their intended use and perform properly. The individual qualification steps are
defined as follow:
Design Qualification (DQ): It is the documented verification that the proposed design of the
facilities, systems and equipment is suitable for the intended purpose.
www.wjpps.com
5412
P.Ramasubramaniyan et al.
World Journal of Pharmacy and Pharmaceutical Sciences
Installation Qualification (IQ): It is documented evidence that the premises, supporting
utilities, the equipment have been built and installed in compliance with design
specifications.
Operational Qualification (OQ): In this phase the process parameters are challenged to
assure that product meets all defined requirements under all anticipated conditions of
manufacturing, i.e., worst case testing.
Performance Qualification (PQ): The performance qualification is carried out to establish
the performance and efficiency of the tablet machine during tablet compression operation.
The key objective of this phase is to demonstrate that the process will produce acceptable
product under normal operating conditions.
A Typical Validation Blueprint of Equipment Validation:
1. Installation qualification
Facilities
Utilities
Equipment
2. Operation qualification
Testing Protocols for Utilities and Equipment
3. Validation
Testing protocols for products and cleaning systems
4. Documentation
5. Validation of the QA testing laboratory
6. SOPs
7. Training of personnel
8. Organization charts, Schedule of events
The main aim and objective of this research is to qualify the equipment based on its
process capability by observing the compression parameters.
METHODOLOGY
Execution of Design Qualification
Documentation and verification of procedures required to fulfill the protocol.
Execution of protocol and data collection, interpretation and review of data for accuracy,
completeness and cGMP compliance.
Approval of original protocol formats, approval for the final summaries and system
qualification statement.
www.wjpps.com
5413
P.Ramasubramaniyan et al.
World Journal of Pharmacy and Pharmaceutical Sciences
Execution of IQ, OQ and PQ:
It includes the development and approval of an IQ, OQ, PQ protocols followed by the
performance of IQ, OQ, PQ. The further step includes the workout and approval of IQ, OQ,
PQ reports.
Procedure Followed for Performance Qualification:
The blend/dummy material was unloaded into the hoppers on the both sides and the
compression machine was operated at low speed (20RPM) as per operating instructions. Then
the machine was set to run for 20 minutes continuously after adjusting the following
parameters.
1. Individual tablet weight variation
2. Weight of 20 tablets
3. Hardness
4. Thickness
5. Disintegration time, Friability
Then the experiment was repeated at both medium (40RPM) and high speed (60RPM)
with same set of parameters and the parameters were checked at different speeds (low,
medium and high rpm). The results were reported
.
Sampling Plan
Collect 100 tablets for every 05 minutes.
RESULTS & DISCUSSION
TABLE No. 1:
Speed: Medium (40rpm)
Feeder rpm: LHS: 52, RHS: 56
Turret rpm:40RPM
Compression Force (KN): LHS: Main:16.9, Pre:5.7 RHS: Main:17.1, Pre:6.2
S.
PARAMETER
ACCEPTANCE
TIME INTERVAL (LHS)
No.
CRITERIA
0 min 05
10
15
20 min
min
min
min
1
Weight of 20 tablets
21.28 2%
21.24
21.26 21.26
21.28 21.25
(20.85 21.71g)
2
Disintegration Time
NMT 20 min
10'52" 12'31" 09'59" 11'02" 12'15"
3
Friability
NMT 0.8% w/w
0.34
0.41
0.37
0.33
0.39
The compression parameters such as weight of 20 tablets, disintegration time, friability,
individual weight variation, thickness and hardness are taken in to consideration at slow
www.wjpps.com
5414
P.Ramasubramaniyan et al.
World Journal of Pharmacy and Pharmaceutical Sciences
(20RPM), medium (40RPM) and high (60RPM) speeds of machine. The results shown here
are the one observed at medium speed (40RPM) which is found to be optimum.
TABLE No. 2
Speed: Medium (40rpm)
Feeder rpm: LHS: 52, RHS: 56
Turret rpm:40RPM
Compression Force (KN): LHS: Main:16.9, Pre:5.7 RHS: Main:17.1, Pre:6.2
S.
PARAMETER
ACCEPTANCE
TIME INTERVAL (RHS)
No.
CRITERIA
0 min 05
10
15
20 min
min
min
min
1
Weight of 20 tablets
21.28 2%
21.28
21.29 21.28
21.24 21.26
(20.85 21.71g)
2
Disintegration Time
NMT 20 min
11'31" 13'03" 10'31" 11'15" 12'52"
3
Friability
NMT 0.8% w/w
0.36
0.44
0.40
0.34
0.41
TABLE No. 3
Time / No. Individual Weight Variation of the tablet : 1.064 5.0% (1.011 1.117 g)
of Tablets Speed: 40rpm
1
2
3
4
5
6
7
8
9
10
0
LHS 1.061 1.060 1.057 1.067 1.070 1.059 1.059 1.062 1.059 1.059
min RHS 1.053 1.064 1.076 1.060 1.058 1.061 1.057 1.064 1.059 1.065
05
LHS 1.054 1.045 1.061 1.067 1.063 1.061 1.060 1.059 1.066 1.055
min RHS 1.063 1.067 1.070 1.059 1.072 1.071 1.075 1.065 1.051 1.055
10
LHS 1.057 1.066 1.053 1.059 1.053 1.071 1.069 1.050 1.070 1.059
min RHS 1.052 1.065 1.067 1.057 1.056 1.064 1.066 1.055 1.067 1.056
15
LHS 1.065 1.066 1.059 1.058 1.063 1.060 1.064 1.047 1.063 1.061
min RHS 1.054 1.064 1.063 1.058 1.055 1.063 1.059 1.057 1.075 1.059
20
LHS 1.042 1.049 1.056 1.056 1.060 1.051 1.064 1.056 1.059 1.056
min RHS 1.054 1.056 1.043 1.043 1.065 1.054 1.056 1.053 1.046 1.058
TABLE No. 4
Time / No.
of Tablets
0
LHS
min RHS
05
LHS
min RHS
10
LHS
min RHS
15
LHS
min RHS
Thickness: 7.1 0.3mm (6.8 7.4mm)
1
2
3
4
5
6.98
6.96
6.98
6.97
6.94
6.96
6.94
6.94
6.93
6.94
6.98
6.94
6.96
6.98
6.97
6.94
6.98
6.94
6.98
6.97
6.91
6.94
6.98
6.94
6.89
6.97
6.98
6.94
6.98
6.94
6.94
6.92
6.94
6.94
6.97
6.89
6.97
6.98
6.94
6.87
6
6.94
6.98
6.84
6.96
6.88
6.92
7.01
6.94
7
6.93
6.98
6.86
6.94
6.89
6.93
7.0
6.93
8
6.94
7.01
6.89
6.98
6.98
6.94
6.98
6.94
9
6.90
6.96
6.86
6.97
6.93
6.94
6.99
6.93
10
6.92
6.96
6.92
6.87
6.92
7.00
6.94
7.00
20
min
6.99
6.89
6.93
7.0
6.94
6.96
6.98
7.01
6.98
7.0
6.94
6.98
LHS
RHS
www.wjpps.com
6.89
6.96
6.94
6.98
6.95
6.94
6.98
6.89
5415
P.Ramasubramaniyan et al.
World Journal of Pharmacy and Pharmaceutical Sciences
TABLE No. 5
Time / No. Hardness: 10.0 - 16.0 kp
of Tablets
1
2
3
4
12.8
14.0
14.6
0
LHS 13.6
min RHS 12.6
13.1
13.6
12.8
5
15.2
13.4
6
13.7
13.6
7
13.8
12.8
8
14.2
14.6
9
14.6
14.8
10
14.8
13.8
10
min
LHS
RHS
LHS
RHS
14.0
12.8
13.6
13.8
14.6
12.6
13.8
13.8
14.2
12.4
14.1
14.2
13.6
13.8
14.7
14.7
13.8
13.8
13.5
14.9
13.2
14.2
13.4
13.6
12.6
14.4
14.9
13.4
12.2
13.8
15.0
13.4
14.0
14.2
12.8
12.6
14.2
14.6
13.3
12.7
15
min
20
min
LHS
RHS
LHS
RHS
12.7
14.8
14.9
14.8
12.7
14.1
12.9
12.8
13.6
14.6
12.8
12.6
13.5
13.6
14.6
14.6
14.2
13.6
14.7
14.1
14.5
13.2
14.1
14.2
14.6
14.4
14.4
14.5
14.9
14.5
14.4
14.6
14.8
14.3
13.9
13.9
14.8
14.2
13.6
13.8
05
min
SUMMARY & CONCLUSION
The parameters studied under the compression process are such as compression force, turret
rpm and feeder rpm. These are set at three different speeds and analyzed on both sides to
assess the exact parameters.
Turret was set at the speed of 20rpm with Feeder at LHS as 32rpm, at RHS as 36rpm and
Compression Force (KN) Main at LHS as 17.0, at RHS as 16.1 and Pre at LHS as 5.7, at RHS
as 5.3.
Turret was set at the speed of 40rpm with Feeder at LHS as 52rpm, at RHS as 56rpm and
Compression Force (KN) Main at LHS as 16.9, at RHS as 17.1 and Pre at LHS as 5.7, at RHS
as 6.2.
Turret was set at the speed of 60rpm with Feeder at LHS as 70rpm, at RHS as 75rpm and
Compression Force (KN) Main at LHS as 15.8, at RHS as 16.3 and Pre at LHS as 5.6, at RHS
as 6.5.
The samples were collected from both the sides (LHS, RHS) to qualify the two press stations
and the observations made from the results were found to be in specified limits as per the IH
specifications.
From this we conclude that the equipment is successfully qualified and can be used for
production of further batches.
www.wjpps.com
5416
P.Ramasubramaniyan et al.
World Journal of Pharmacy and Pharmaceutical Sciences
ACKNOWLEDGMENT
We express our sincere thanks to our Principal Dr.P.Solairaj and management of
Sankaralingam Bhuvaneswari College of Pharmacy, Sivakasi for providing us the required
facilities. We also like to thank all the members involved in this work for their immense
support and cooperation.
REFERENCES
1. Annex 4, Supplementary guidelines on good manufacturing practices: validation, World
Health Organization, WHO Technical Report Series, No. 937, 2006, P-9.
2. Ashish Mudbidri, Tablet Compression Principles, Pharma Times - Vol. 42 - No. 11 November, P-44, 2010.
3. Frederick j. Carleton, James P. Agalloco, Validation of pharmaceutical processes sterile
products second edition, Marcel Dekker, Inc, 2006.
4. Good Manufacturing Practices for pharmaceutical products. In: WHO Expert Committee
on Specifications for Pharmaceutical Preparations. Thirty-second report. Geneva, World
Health Organization, 1992, Annex 1 (WHO Technical Report Series, No. 823).
5. Guidance for Industry Process Validation: General Principles and Practices, U.S.
Department of Health and Human Services, Food and Drug Administration, Center for
Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research
(CBER), Center for Veterinary Medicine (CVM), January 2011, Current Good
Manufacturing Practices (CGMP), Revision 1, P-13.
6. J.Agalloco, Master Planning of Validation, PDA Training and Research Institute, April,
1988.
7. Kenneth W. Sigvardson, Joseph A. Manalo, Robert W. Roller, Fatieh Saless, and David
Wasserman, Laboratory Equipment qualification, pharmaceutical technology, October,
2001.
8. L. Nandhakumar, G. Dharmamoorthy, S. Rameshkumar and S. Chandrasekaran, An
Overview of Pharmaceutical Validation: Quality Assurance View Point, International
Journal of Research in Pharmacy and Chemistry, ISSN: 2231 - 2781, 1(4), P - 1003
1014, 2011.
9. Leon Lachman, Herbert A. Lieberman, The Theory and Practice of Industrial Pharmacy,
Special Indian Edition, 2009.
10. Maas & Peither AG, GMP Publishing, LOGFILE No. 8 / August, P-2, 2011.
www.wjpps.com
5417
P.Ramasubramaniyan et al.
World Journal of Pharmacy and Pharmaceutical Sciences
11. National Conference of Standards Laboratories, Establishment and Adjustment of
Calibration Intervals, NCSL RP-1, 1996.
12. P.A. Cloud, 1997, Validating a Laboratory Incubator, BioPharm 10 (11), 3042.
13. PDA, Validation of Computer-Related Systems, Technical Report No. 18, PDA J. Sci.
Technol. 49 (1), S1S17, 1995.
14. Robert A. Nash, Alfred H. Wachter, Pharmaceutical Process Validation. 3rd Ed., volume
129, Marcel Dekker, Inc, 2003.
15. Satyabrata Jena, K.Sravya, Arjun Goje, D. Narendra Prasad and Syed Sameer,
Equipment Qualification for RMG, FBD, Blender & Compression Machines- An
Overview, International Journal of Novel Drug Delivery Technology; Vol-1 | Issue-3 |
Jul-Sep 2011.
www.wjpps.com
5418
Das könnte Ihnen auch gefallen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5795)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1091)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Temperature Mapping Study Protocol Procedure PDFDokument15 SeitenTemperature Mapping Study Protocol Procedure PDFJhonnnnnnNoch keine Bewertungen
- D2493Dokument5 SeitenD2493Donig FermanianNoch keine Bewertungen
- BLP 530 Gas Porosimeter127 20 - InstructionsDokument11 SeitenBLP 530 Gas Porosimeter127 20 - InstructionsDonig FermanianNoch keine Bewertungen
- Annotated BibliographyDokument5 SeitenAnnotated Bibliographyapi-273602090Noch keine Bewertungen
- CC:PC DpcoDokument8 SeitenCC:PC DpcoRuchitaa MisshraNoch keine Bewertungen
- ATI Nursing ModulesDokument9 SeitenATI Nursing ModulestgushikaNoch keine Bewertungen
- NISTIR 6969 20140926 20160121rev PDFDokument210 SeitenNISTIR 6969 20140926 20160121rev PDFDonig FermanianNoch keine Bewertungen
- BS en Iso 00787-5-1995 (2000)Dokument10 SeitenBS en Iso 00787-5-1995 (2000)Donig FermanianNoch keine Bewertungen
- This Publ Icati On Is Avai Labl e Free of Cha Rge Fro M: S://D Oi.o rg/1 0.6 028 /NI St.I R.6 969 - 201 9Dokument11 SeitenThis Publ Icati On Is Avai Labl e Free of Cha Rge Fro M: S://D Oi.o rg/1 0.6 028 /NI St.I R.6 969 - 201 9Donig FermanianNoch keine Bewertungen
- Process Monitoring Systems PDFDokument4 SeitenProcess Monitoring Systems PDFDonig FermanianNoch keine Bewertungen
- ImpartialityDokument2 SeitenImpartialitysujud100% (2)
- ISO 14644 - Revised Cleanroom Standard: February 2016Dokument36 SeitenISO 14644 - Revised Cleanroom Standard: February 2016Donig FermanianNoch keine Bewertungen
- SF500 InstructionDokument142 SeitenSF500 InstructionDonig FermanianNoch keine Bewertungen
- Impartiality PDFDokument2 SeitenImpartiality PDFDonig FermanianNoch keine Bewertungen
- TG14 Version 3 July 2019Dokument7 SeitenTG14 Version 3 July 2019Donig FermanianNoch keine Bewertungen
- TGGuideline 114 Viscosity of LiquidsDokument7 SeitenTGGuideline 114 Viscosity of LiquidsDonig FermanianNoch keine Bewertungen
- Fuel Handling Jet Fuel Quality and Test Procedure Manual Petrovalue Aviation 2008.2 0-1 PDFDokument45 SeitenFuel Handling Jet Fuel Quality and Test Procedure Manual Petrovalue Aviation 2008.2 0-1 PDFDonig FermanianNoch keine Bewertungen
- Rotronic HL 1D TL 1D ManualDokument4 SeitenRotronic HL 1D TL 1D ManualDonig FermanianNoch keine Bewertungen
- I-CAL-GUI-013 Calibration Guide No. 13 WebDokument13 SeitenI-CAL-GUI-013 Calibration Guide No. 13 WebDonig FermanianNoch keine Bewertungen
- Types of Pressure:: When and Why Are They Used?Dokument4 SeitenTypes of Pressure:: When and Why Are They Used?Donig FermanianNoch keine Bewertungen
- Calibration Kit Dw-3 F-Mod 35 ViscometerDokument1 SeiteCalibration Kit Dw-3 F-Mod 35 ViscometerDonig FermanianNoch keine Bewertungen
- Adm 870 PDFDokument54 SeitenAdm 870 PDFDonig FermanianNoch keine Bewertungen
- Density of Water (G/ML) vs. Temperature (°C) : (From Handbook of Chemistry and Physics, 53rd Edition, P. F4)Dokument1 SeiteDensity of Water (G/ML) vs. Temperature (°C) : (From Handbook of Chemistry and Physics, 53rd Edition, P. F4)Donig FermanianNoch keine Bewertungen
- 3 Theory Calibration JKBHJGHJGHHFGHFGDokument88 Seiten3 Theory Calibration JKBHJGHJGHHFGHFGDonig FermanianNoch keine Bewertungen
- 0 G UGo T4 HZWL7 H9 WVN L13 A M4 S F9 T QYMh LRQUi YCXVDokument5 Seiten0 G UGo T4 HZWL7 H9 WVN L13 A M4 S F9 T QYMh LRQUi YCXVGajanan Vinayak NaikNoch keine Bewertungen
- Drugs in Pregnancy: A Handbook For Pharmacists and PhysiciansDokument3 SeitenDrugs in Pregnancy: A Handbook For Pharmacists and PhysiciansSeno Adi WicaksonoNoch keine Bewertungen
- PT. Bina Mitra Jaya BersamaDokument25 SeitenPT. Bina Mitra Jaya BersamaHADI PRAYITNONoch keine Bewertungen
- Part I - Sample Questions: COMPETENCY 1: Patient CareDokument20 SeitenPart I - Sample Questions: COMPETENCY 1: Patient CareAbdallah ElmenshawyNoch keine Bewertungen
- 7877978979Dokument16 Seiten7877978979Alembante MollaNoch keine Bewertungen
- Crash Cart Check ListDokument2 SeitenCrash Cart Check ListNDH PHARMACYNoch keine Bewertungen
- Checklist EmergencyDokument5 SeitenChecklist EmergencyGabriella graziani50% (2)
- LPLPO JKN DonyDokument18 SeitenLPLPO JKN DonyAbdurroziNoch keine Bewertungen
- Price List MOLEX AYUS Per 18.11.2020 (Cabang)Dokument24 SeitenPrice List MOLEX AYUS Per 18.11.2020 (Cabang)Apotek Hilda AlnairaNoch keine Bewertungen
- Multiallergen Immunotherapy For Allergic Rhinitis and AsthmaDokument7 SeitenMultiallergen Immunotherapy For Allergic Rhinitis and AsthmaPeter SalimNoch keine Bewertungen
- Talak KejangDokument52 SeitenTalak KejangRiga Medina IINoch keine Bewertungen
- Ranitidine HCLDokument9 SeitenRanitidine HCLMochammad FerdiansyahNoch keine Bewertungen
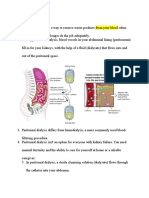
- Peritoneal DialysisDokument3 SeitenPeritoneal DialysisGoh Kek BoonNoch keine Bewertungen
- Zarbo (2016) - Integrative Psychotherapy WorksDokument3 SeitenZarbo (2016) - Integrative Psychotherapy WorksAnonymous Fqa80pKNoch keine Bewertungen
- REVISI TARIF OBAT & ALKES 9 JUNI 2020 GhifarDokument322 SeitenREVISI TARIF OBAT & ALKES 9 JUNI 2020 GhifarwisnuNoch keine Bewertungen
- Kanker Payudara PDFDokument20 SeitenKanker Payudara PDFbabehNoch keine Bewertungen
- Hospitals in Eastern Province, Sri LankaDokument4 SeitenHospitals in Eastern Province, Sri LankaJeewajothy JeewaNoch keine Bewertungen
- Lisdexamfetamine Chemistry Pharmacodynamics Pharmacokinetics and Clinical Efficacy Safety and Tolerability in The Treatment of Binge Eating DisorderDokument11 SeitenLisdexamfetamine Chemistry Pharmacodynamics Pharmacokinetics and Clinical Efficacy Safety and Tolerability in The Treatment of Binge Eating DisorderAR AoarNoch keine Bewertungen
- Arte Sun AteDokument9 SeitenArte Sun Atebaitur_rohmah8372Noch keine Bewertungen
- Pharmacology 1Dokument35 SeitenPharmacology 1prabhuNoch keine Bewertungen
- PrescriptionDokument3 SeitenPrescriptionMurali KrishnaNoch keine Bewertungen
- 2023 07 03Dokument11 Seiten2023 07 03maksumahNoch keine Bewertungen
- Medication Order ReviewDokument34 SeitenMedication Order ReviewVidho RiveraNoch keine Bewertungen
- Pedia Recommended DosageDokument5 SeitenPedia Recommended DosageNerak LuNoch keine Bewertungen
- NST and Their FunctionsDokument5 SeitenNST and Their FunctionsyazNoch keine Bewertungen
- Poultry Disease Diagnosis - Picture Book-2Dokument2 SeitenPoultry Disease Diagnosis - Picture Book-2बनकर परिवाराचा लाडका गोट्याNoch keine Bewertungen