Beruflich Dokumente
Kultur Dokumente
HSV-1 Therapy of Primary Tumors Reduces The Number of Metastases in An Immune-Competent Model of Metastatic Breast Cancer
Hochgeladen von
higginscribdOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
HSV-1 Therapy of Primary Tumors Reduces The Number of Metastases in An Immune-Competent Model of Metastatic Breast Cancer
Hochgeladen von
higginscribdCopyright:
Verfügbare Formate
ARTICLE
doi:10.1016/S1525-0016(03)00236-3
HSV-1 Therapy of Primary Tumors Reduces the Number
of Metastases in an Immune-Competent Model
of Metastatic Breast Cancer
Darby L. Thomas and Nigel W. Fraser*
Department of Microbiology, University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania 19104-6076
*
To whom correspondence and reprint requests should be addressed at the Department of Microbiology, University of Pennsylvania School of Medicine,
319a Johnson Pavilion, 3610 Hamilton Walk, Philadelphia, PA 19104-6067. Fax: (215) 898-3849. E-mail: nfraser@mail.med.upenn.edu.
The HSV-1 1716 mutant virus and similar oncolytic herpesviruses deficient in the 34.5 neurovirulence gene are able to reduce the growth of tumors in mice. Here we demonstrate that HSV-1
1716 therapy moderately reduced the growth of tumors of the highly malignant, spontaneously
metastasizing 4T1 mouse mammary carcinoma model. This moderate effect on 4T1 tumor growth
was likely due to poor replication kinetics of HSV-1 1716 in 4T1 cells. Interestingly, HSV-1 therapy
of the primary tumor increased the survival time of mice. Coincident with this increase was a
reduction in metastases as determined by quantification of the number of metastatic cells in the
lungs. HSV-1 therapy of the primary tumor was also able to reduce the establishment of a second
challenge of 4T1 tumors. Moreover, infiltrates of both CD4 and CD8 T cells were detected in
HSV-1 1716-treated tumors. An important role for the T cell infiltrates was confirmed when HSV-1
therapy did not reduce the growth of 4T1 tumors in SCID mice. Collectively, these results
demonstrate that an HSV-dependent anti-tumor immune response is required for the reduction
in primary 4T1 tumor growth and for the reduction in the establishment of metastases in this
tumor model.
Key Words: HSV-1, gene therapy, 4T1 mammary tumor, metastasis, immune response
INTRODUCTION
Herpes simplex viruses with mutations in the 34.5 neurovirulence gene-encoding infected cell protein (ICP34.5)
do not replicate in terminally differentiated cells, such as
cells of the central nervous system, but can replicate in
dividing cells, such as cancer cells [13]. ICP34.5 functions to inactivate the dsRNA-induced PKR pathway, to
prevent host protein synthesis shutoff, by complexing
with protein phosphatase 1, which results in the dephosphorylation of eIF-2 [4]. The selective replication of
34.5-mutant viruses has been exploited to design replicating HSV-1 vectors for cancer therapy. HSV-1 therapy
can increase the survival of mice harboring established
brain tumors and reduce the growth (sometimes completely) of established subcutaneous tumors in mice after
intraneoplastic injection. Herpes vectors like G207,
R3616, and 1716 have been shown to be effective in therapy
of human tumors in immune-deficient mouse models, such
as colon, ovarian, glioma, breast, prostate, and melanoma,
as well as in therapy of immune-competent mouse models of glioma, colorectal, and melanoma [59 and re-
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
1525-0016/03 $30.00
viewed in 10]. Consequently, results of completed phase I
clinical trials for both G207 and 1716 demonstrated no
toxicity at doses up to 109 and 105, respectively, in patients with malignant glioma and melanoma [1113].
It is now well established that an important component of HSV-1 cancer therapy is an immune response
elicited by the virus. For example, Toda et al. [14] demonstrated that G207 therapy of a colon carcinoma model
acts as an in situ vaccine by eliciting a tumor-specific
immune response. Similarly, HSV-1 1716 therapy of a
brain tumor model of metastatic melanoma requires multiple components of the immune system [15]. In addition,
preexisting immunity to HSV-1 can enhance HSV-1 1716
therapy of brain tumors [16]. Considering these studies,
we selected an immune-competent cancer modelthe
4T1 mouse mammary carcinoma model [1719]and
asked whether oncolytic HSV-1 therapy would be effective in this model of an aggressive metastatic cancer.
4T1 tumors established in the mouse mammary gland
closely resemble human breast cancer in their immunogenicity and their growth and metastatic properties [20].
Derived from Balb/c mice, 4T1 cells are considered very
543
ARTICLE
doi:10.1016/S1525-0016(03)00236-3
FIG. 1. Replication of HSV-1 17 and HSV-1 1716 in 4T1 and Vero cells. 4T1 and Vero cells were infected at a m.o.i. of 0.1 for 1 h with (A) HSV-1 17 or (B)
HSV-1 1716 as described under Materials and Methods. The zero time point was taken after washing of the cell monolayers immediately following the 1-h
infection. Time points were taken in duplicate and were titered on Vero cells in triplicate. The mean pfu at each time point is presented in log scale standard
error.
weakly immunogenic with a relative antigenic strength of
less than 0.01 (with 9.9 being the most immunogenic)
[21,22]. Tumors derived from 4T1 cells spontaneously
metastasize primarily to the lungs (95%), but also spread
to the liver (75%) and brain (40%) as well as the blood
and lymph nodes [20]. Because 4T1 cells are 6-thioguanine resistant, micrometastases to different organs are
easily amplified and the number of metastatic cells is
easily quantified [17,20].
Here we demonstrated that HSV-1 1716 therapy of
established 4T1 mammary tumors moderately reduced
the growth rate of these primary tumors compared to
those treated with culture medium (mock). However, the
therapy of the primary tumor significantly reduced the
number of clonogenic lung metastases and the mean survival time of the mice compared to mock therapy. In
addition, HSV-1 1716 therapy of primary 4T1 tumors
inhibited the establishment of a second challenge of 4T1
tumor cells, implicating HSV stimulation of an anti-tumor immune response. This implication was supported
by the presence of infiltrating CD4 and CD8 T cells.
Furthermore, HSV-1 1716 therapy did not affect the
growth of primary tumors in immune-deficient SCID
mice, verifying that an immune response is required for
effective HSV-1 therapy of the primary 4T1 tumors as well
as for the reduction in the establishment of metastases.
RESULTS
HSV-1 1716 Replicates Poorly in 4T1 Mammary
Tumor Cells
Although Balb/c mice are susceptible to HSV-1 infection,
not all murine cell lines derived from susceptible mice can
544
be efficiently infected with HSV-1 [23,24]. Therefore, we
examined the ability of wild-type HSV-1 17 and the
34.5-mutant HSV-1 1716 to replicate in 4T1 cells compared to HSV-susceptible Vero cells. We infected Vero and
4T1 cells with HSV-1 17 and 1716 at a multiplicity of
infection (m.o.i.) of 0.1. Infected monolayers were harvested over time and the number of plaque-forming units
(pfu) was determined by plaque assay. HSV-1 17 replicated to high levels within 3 days in both Vero and 4T1
cells (Fig. 1A). In contrast, HSV-1 1716 yields were three
logs lower in 4T1 cells than in Vero cells in the same time
period (Fig. 1B), suggesting that, unlike Vero cells, 4T1
cells cannot complement the 34.5 mutation to support
replication of this virus. Although 4T1 cells are susceptible to killing by HSV-1 1716 at a high m.o.i. (data not
shown), HSV-1 1716 replication is not as efficient in 4T1
cells as in Vero cells.
HSV-1 1716 Only Moderately Reduces the Growth of
Primary Mammary Tumors
Though HSV-1 1716 does not replicate well in 4T1 cells,
we still wanted to examine the potential of HSV-1 1716 to
reduce the growth of established 4T1 tumors. A single
injection of HSV-1 1716 had little effect on tumor growth
(data not shown); therefore we chose to analyze the outcome of two and three separate injections of HSV-1 1716.
Similarly, Chahlavi et al. [25] demonstrated that multiple
injections of lower quantities of virus are more effective
than a single injection of a high dose of virus. Tumors
established subcutaneously in the abdominal mammary
gland received intratumor injections of 5.4 105 pfu of
HSV-1 1716 virus on days 9 and 13 (double therapy), or
on days 9, 13, and 16 (triple therapy), post-injection of
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
doi:10.1016/S1525-0016(03)00236-3
ARTICLE
FIG. 2. HSV-1 1716 therapy of 4T1 mammary tumors. Balb/c mice were injected subcutaneously in the abdominal mammary gland with 1 104 viable 4T1 cells.
Mean tumor diameters were determined as described under Materials and Methods. Palpable tumors were injected with 20 l containing either 5.4 105 pfu
of HSV-1 1716 or DMEM (mock-infected) on (A) days 9 and 13 (double therapy; mock, n 24; 1716, n 23) or (B) days 9, 13, and 16 (triple therapy; mock,
n 22; 1716, n 19). Arrows indicate the days when therapy was administered. *Significant differences between mock- and 1716-treated tumors. Error bars
represent standard error.
tumor cells. We determined the mean tumor diameter
(calculated as the square root of the product of two perpendicular diameters) for each mouse every 3 to 4 days
until day 27 post-injection of tumor cells. Between days
13 and 16, we found only a small increase in the mean
diameter of tumors receiving double HSV-1 1716 therapy
(Fig. 2A), and no increase in the mean diameter of tumors
receiving triple HSV-1 1716 therapy (Fig. 2B), in comparison to tumors treated with culture medium (mock
treated). Beginning at day 16, the difference in the mean
diameters between HSV-1 1716- and mock-treated tumors
was significant each day until the endpoint for both double (Fig. 2A, P 0.025) and triple therapy (Fig. 2B, P
0.002). Despite this significance, the mean diameter of
HSV-1 1716-treated tumors continued to increase after
day 16 at either a similar rate (Fig. 2A, double therapy)
or a reduced rate (Fig. 2B, triple therapy) compared to
mock-treated tumors, suggesting that HSV-1 therapy increased in effectiveness with increased inoculations. However, therapy was not totally effective.
HSV-1 1716 Replicates Poorly in 4T1 Mammary
Tumors
HSV-1 1716 therapy has been demonstrated to treat brain
tumors in mice effectively, and replication of HSV-1 1716
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
in these tumors is characterized by an increase in virus
detection (by plaque assay) in the days following injection [6,9]. Because HSV-1 1716 therapy was not very effective in reducing the growth of primary 4T1 tumors, we
next wanted to determine whether HSV-1 1716 was replicating within the tumor. We repeated triple therapy of
primary 4T1 tumors and harvested the tumors at the time
points indicated in Fig. 3. On the days of therapy, we
harvested the tumors immediately following injection of
virus to determine the amount of input virus detectable
by plaque assay. We found that, on the days following
each injection of HSV-1 1716, virus was detected by
plaque assay (Fig. 3). However, the quantity of virus decreased instead of increased over time, though infectious
virus was still detectable for up to 3 days. This result is
similar to results seen when HSV-1 1716 is injected directly into the brain [6,26]. In these studies, Kesari et al.
[26] found that, when injected into the ventricles where
HSV-1 1716 replicates in the ependymal cells, infectious
virus can be detected by titration for up to 2 days. When
injected into the brain parenchyma, where HSV-1 1716
cannot replicate, infectious virus is not detected at any
time postinjection. Considering these studies in the brain,
our results suggest that inefficient viral replication is occurring in the 4T1 tumors.
545
ARTICLE
doi:10.1016/S1525-0016(03)00236-3
that received double mock therapy (Fig. 4A) or triple
mock therapy (Fig. 4B) survived a mean of 37.6 5.2 days
or 48.9 10.7 days, respectively. The difference we observed between the two therapies, which is not statistically significant, is likely due to experimental variability,
though we cannot formally rule out the effects of manipulating the tumor. One double-mock-therapy mouse survived past day 100 post-injection of tumor cells (Fig. 4A).
We presume that this mouse did not get metastases,
which is in accordance with previous observations that
metastasis (e.g., to the lungs) is less than 100% [20,28].
Mice with primary tumors that received either double
(Fig. 4A) or triple (Fig. 4B) HSV-1 1716 therapy survived
significantly longer, with a mean of 48.4 12.3 (P
0.014) or 58.1 7.7 days (P 0.005), respectively.
FIG. 3. HSV-1 1716 replication in 4T1 tumors. Balb/c mice with established
4T1 tumors were given triple therapy as described for Fig. 2. Two tumors per
time point were harvested on the days indicated post-injection of virus and
prepared as described under Materials and Methods. Tumor lysates were
assayed for HSV-1 1716 levels by plaque assay on Vero cells in triplicate. The
mean pfu at each time point is presented in log scale standard error. Arrows
indicate the days when therapy was administered.
HSV-1 1716 Therapy of Primary Mammary Tumors
Increases the Survival of Mice
A recent study demonstrated that therapy of primary 4T1
tumors with IL-12, delivered by gene gun, did not reduce
the growth of primary tumors, yet increased the survival
time of mice [27]presumably due to a decrease in metastasis. Therefore, we reasoned that it was still possible
that HSV-1 therapy, though only moderately effective on
primary 4T1 tumors, could prolong the survival time of
treated mice. To address this possibility, we divided the
mice described in Fig. 2 with primary tumors treated with
either two or three rounds of HSV-1 1716 therapy into
two groups. We monitored the first group for survival
following surgical removal of the primary tumor at day 27
post-injection of tumor cells. Mice with primary tumors
HSV-1 1716 Therapy of Primary Mammary Tumors
Reduces the Number of Metastatic Cells in the Lungs
Established 4T1 tumors form disseminated metastases
throughout the body of the mouse [17,20]. The increase
in survival time of mice with HSV-1 1716-treated primary
4T1 tumors suggests that therapy reduced the extent of
metastases. We therefore utilized the resistance of 4T1
cells to 6-thioguanine to quantify the number of metastatic cells in the lungs. We chose to look exclusively at
the lungs since previous studies have demonstrated that
metastasis to the lungs best represents the extent of 4T1
cell metastasis throughout the mouse [20]. We sacrificed
the second group of mice from the primary tumor experiments described in Fig. 2 on day 27 and removed, dissociated, and cultured their lungs in 6-thioguanine to assay
the number of 4T1 colonies representing the number of
metastatic cells. We determined the natural log (ln) of the
number of metastatic colonies from each mouse to give
normality for statistical evaluation (data not shown).
Mice receiving double mock therapy had a mean of
69,665 66,434 metastatic colonies and mice receiving
triple mock therapy had a mean of 58,051 94,962
metastatic colonies (Table 1). Significantly, mice receiving
double or triple HSV-1 1716 therapy had a mean of 6781
12,353 (P 0.004) or 13,033 32,597 (P 0.033)
FIG. 4. Survival of mice with HSV-1 1716-treated 4T1 mammary tumors. On day 27 post-injection of tumor cells, tumors were surgically removed from mice
described in Fig. 2 as described under Materials and Methods and the mice were monitored for survival. KaplanMeier survival curves are presented. (A) Double
therapy: mock, n 12; 1716, n 12; P 0.014. (B) Triple therapy: mock, n 12; 1716, n 10; P 0.005.
546
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
ARTICLE
doi:10.1016/S1525-0016(03)00236-3
TABLE 1: Quantitation of clonogenic lung metastases from mice with HSV-1 1716-treated 4T1 mammary tumors
No. of mice with clonogenic lung metastasesa
b
Therapy
No. of injections
01000
100120,000
20,00150,000
50,000
Mean (ln)e
Mock
11
69,665 66,434 (10.10)
HSV-1 1716
12
6,781 12,353 (7.05)
Mock
58,051 94,962 (9.06)
HSV-1 1716
13,033 32,577 (5.75)
p-valuef
0.004
0.033
a
Mice described in Figure 2 were sacrificed on day 27 post-injection of 4T1 cells and the lungs were removed, processed, and cultured as described in Materials and Methods. The number
of 6-thioguanine-resistant 4T1 colonies stained with methelene blue were counted for each mouse. The number of mice with lung 4T1 colony numbers within each range is presented.
b
Mock-treated mice received culture media and HSV-1 1716-treated mice received 5.4 105 pfu per injection.
c
Established 4T1 tumors received injections of either HSV-1 1716 or culture media (mock) either two (days 9 and 13 post-injection of 4T1 cells) or three times (days 9, 13, and 16
post-injection of 4T1 cells) as described in Figure 2.
d
Total number of mice per group.
e
Mean number of 6-thioguanine-resistant 4T1 colonies standard deviation. (ln) is the natural log of the mean number of metastases.
f
The natural log (ln) of the number of metastases in the lungs of each mouse was used to determine the p-value for either two or three injections as described in Materials and Methods.
metastatic colonies, respectively (Table 1). These results
demonstrate that HSV-1 1716 therapy of the primary tumor can reduce the burden of metastases in the lungs. In
addition, we see a strong correlation between tumor size
and the number of metastatic colonies for each mouse
(data not shown), consistent with other studies using this
tumor model [20].
HSV-1 1716 Therapy of 4T1 Tumors Partially Protects
against a Second Challenge of 4T1 Cells
A possible explanation as to how HSV-1 1716 therapy of
the primary 4T1 tumor could reduce the number of metastases is that viral therapy stimulated an immune response to the tumor cells. We addressed this possibility by
asking whether treating established primary 4T1 tumors
with HSV-1 1716 inhibited the growth of a second challenge of 4T1 tumor cells. Specifically, established 4T1
tumors were given triple therapy and then surgically removed. We then injected additional 4T1 tumor cells on
the contralateral side of the abdomen and monitored
them for growth. Because 4T1 cells are very weakly immunogenic, secondary tumors were able to grow in the 10
mice given triple mock therapy and 6 of these 10 mice
died before the endpoint at 38 days after the first injection
of tumor cells, presumably due to metastases (Fig. 5).
Interestingly, 3 of 10 mice receiving triple HSV-1 1716
therapy did not develop secondary tumors. Although the
tumors that did form grew at a rate similar to that of
mock-treated tumors, only 1 of 10 mice receiving triple
HSV-1 1716 therapy did not survive until the endpoint of
the experiment (Fig. 5). These results suggest that HSV-1
therapy of primary 4T1 tumors can stimulate an antitumor immune response that reduces the establishment
of a second challenge of tumor cells.
CD4 and CD8 T Cells Are Present in HSV-1 1716Treated Tumors
It is well established that both CD4 and CD8 T cells are
essential for immune-mediated tumor rejection [reviewed
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
in 29]. To examine further a role for an anti-tumor immune response in HSV-1 1716 therapy of 4T1 tumors, we
performed immunohistochemical analyses on sections of
HSV-1 1716- and mock-treated tumors. Mice with established 4T1 tumors received triple HSV-1 1716 and mock
therapy as described above and we harvested and froze
tumors for sectioning during and after the course of therapy. Tumor sections were then analyzed for histology and
for the presence of CD4 and CD8 T cells. As early as day
12 post-injection of tumor cells, inflammatory cells, such
as neutrophils, could be detected throughout the mass of
HSV-1 1716-treated tumors (Fig. 6). CD4 and CD8 T
FIG. 5. Growth of a second challenge of 4T1 tumor cells after HSV-1 1716
therapy of primary 4T1 tumors. 4T1 tumors were established, received HSV-1
1716 or mock therapy on days 9, 13, and 16, and were measured periodically
as described for Fig. 2. On day 18 post-injection of 4T1 cells, the primary
tumors were surgically removed, and another 1 104 4T1 cells were injected
into the contralateral side. Seven days following the second injection, growth
of tumors was again measured periodically until the endpoint of 38 days. n
10 each group until days 32, 35, and 38 when two mock-treated mice each
day and day 38 when one HSV-1 1716-treated mouse was euthanized because
of illness. Arrows indicate the days when therapy was administered. *Significant differences between mock- and 1716-treated tumors. Error bars represent
standard error.
547
ARTICLE
doi:10.1016/S1525-0016(03)00236-3
cells could also be detected throughout the HSV-1 1716treated tumors. Peak staining for CD4 and CD8 T cells
appeared between day 17 (shown in Fig. 6) and day 20
post-injection of tumor cells and began to subside by day
23 (data not shown). Very few inflammatory cells could
be detected in mock-treated tumors (data not shown).
HSV-1 1716 Therapy Has No Effect on 4T1 Tumor
Growth and Metastases in SCID Mice
We wanted to further confirm the importance of the
immune system in the reduction of metastases by examining HSV-1 therapy of primary 4T1 tumors in immuneincompetent SCID mice, which are deficient in both T
cells and B cells. In addition, we wanted to address the
possibility that, without an adaptive immune response,
HSV-1 1716 would grow uninhibited throughout the tumor and, therefore, more efficiently destroy the tumor. As
above, we injected SCID mice with 1 104 viable 4T1
cells subcutaneously in the abdominal mammary gland.
Mice received intratumoral injections of either HSV-1
1716 or mock therapy on days 9, 13, and 16 post-injection
of cells, and we monitored the mean tumor diameter over
time. Interestingly, there was no difference in the growth
between HSV-1 1716-treated and mock-treated primary
4T1 tumors (Fig. 7). Consequently, there was also no
significant difference in the number of metastatic cells in
the lung (data not shown). These results indicate that an
intact immune response is needed for HSV-1 therapy of
the primary tumor, as well as for the reduction of metastases.
DISCUSSION
The use of replication-restricted HSV-1 vectors has gained
acceptance as plausible for cancer therapy. This acceptance is due to its efficacy in numerous rodent models of
cancer and its lack of toxicity in early clinical trials for
glioma and melanoma [1113]. Here we wanted to determine whether oncolytic HSV-1 therapy of an aggressive,
spontaneously metastasizing mammary carcinoma model
would be effective. It was found that HSV-1 therapy had
only a moderate effect on growth of the primary tumor,
yet primary tumor therapy led to an increase in survival
times and a reduction of metastatic cells in the lungs.
Tumor therapy using 34.5-mutant viruses in only
some models leads to complete regression of the tumor,
demonstrating that the efficacy of HSV-1 therapy will
likely be tumor-type dependent. In the 4T1 mouse mammary carcinoma model, HSV-1 1716 only temporarily
FIG. 6. Presence of immune infiltrates in HSV-1 1716-treated 4T1 tumors. 4T1
tumors were established and received HSV-1 1716 or mock therapy on days 9,
13, and 16 as described for Fig. 2. During and after the course of therapy, mice
were sacrificed and tumors harvested, frozen, and sectioned as described
under Materials and Methods. Sections of HSV-1 1716-treated 4T1 tumors
were either stained with hematoxylin and eosin (H&E) or reacted with antiCD4 or anti-CD8 antibodies and counterstained with methyl green to visualize
548
nuclei. Representative sections of tumors from days 12 and 17 are shown.
Some of the lymphocytes and plasma cells in the day 17 section are indicated
with white arrowheads. The day 17 photos are serial sections of the same
tumor area. Cells positive for CD4 and CD8 are stained purple. Magnification
equals 200.
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
doi:10.1016/S1525-0016(03)00236-3
FIG. 7. HSV-1 1716 therapy of 4T1 mammary tumors in SCID mice. 4T1
mammary tumors were established in SCID mice and received either HSV-1
1716 or mock therapy on days 9, 13, and 16 post-injection of tumor cells as
described for Fig. 2. n 10 each. Arrows indicate the days when therapy was
administered. No significant difference was found at any time point. Error bars
represent standard error.
reduced the growth of primary tumors. This lack of efficacy is likely due to the reduced replication kinetics of
HSV-1 1716 in these tumor cells and the poor replication
in 4T1 tumors as determined by plaque assay. Interestingly, wild-type 17 was able to replicate efficiently in
4T1 cells, indicating that 4T1 cells are not able to complement the replication defect of HSV-1 1716.
In human breast cancer models, inefficient HSV-1 replication in vitro also correlated with the poor oncolysis of
tumors in vivo. In these studies, Toda et al. [30] found that
the MDA-MB-435 and MCF-7 breast cancer cell lines are
more susceptible to HSV-1 mutant G207 infection at a
m.o.i. of 0.1 than T47D cells and that MDA-MB-231 cells
were not very susceptible to G207 infection at the same
m.o.i. This differing susceptibility correlated with the
ability of G207 to reduce the growth of MDA-MB-435
tumors but not of MDA-MB-231 tumors. Also similar to
4T1 cells, mouse CT-26 colorectal tumor cells have a reduced ability to support G207 replication at a low m.o.i.
(0.1), but can be efficiently killed at a higher m.o.i. (1.0)
[14,31]. Why HSV-1 has a reduced ability to replicate in
these different cell types has not been determined, yet the
block is not at the point of viral entry, like in B16 melanoma cells [8], since 4T1 and CT-26 cells are capable of
being infected and lysed by HSV-1. Studies by Farassati et
al. [32] have shown that efficient replication of 34.5-
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
ARTICLE
mutant viruses is dependent on an activated Ras signaling
pathway and they propose that components of the Ras
signaling pathway can inactivate PKR, thus complementing the lack of anti-PKR activity in the 34.5-mutant
viruses. We speculate that the oncogenic nature of 4T1
cells is not due to an activated Ras pathway. In this regard,
it is possible that other selectively oncolytic herpesviruses
that do not have mutations in 34.5, such as hrR3 [33,34]
and the recently described HF10 [35], might be more
efficacious in these tumor models and in other tumor
types that show resistance to replication and killing by
34.5-mutant viruses.
Interestingly, therapy of primary 4T1 mammary tumors in the absence of T and B cells is completely ineffective, indicating that viral oncolysis alone is not sufficient for the reduction in tumor diameters observed with
HSV-1 1716 therapy of 4T1 tumors in immune-competent
mice. Similar observations have been made in other models. We previously demonstrated that HSV-1 1716 therapy
did not prolong survival of SCID mice with brain tumors
in an experimental model of melanoma [16]. In addition,
therapy of CT-26 colorectal tumors, both flank and intracranial, with the HSV-1 mutant G207 was also ineffective
in athymic Balb/c mice [14,36]. However, in immunecompetent mice, HSV therapy of CT-26 tumors acts as an
in situ vaccine by inhibiting the growth of contralateral
tumors or of a second challenge of tumor cells [14,31,36].
In the 4T1 model, we found that HSV-1 1716 therapy of
the primary tumor increased the mean survival time of
mice and reduced the metastatic burden in the lungs.
Moreover, primary tumor therapy also can inhibit the
establishment of a second challenge of 4T1 tumors. It is
possible that the growth of secondary tumors could also
have been inhibited if more time had been allowed between removal of the primary tumor and the second
injection of 4T1 cells, which would have potentially allowed more time to develop a proper anti-tumor immune
response. We were also able to detect an infiltration of
both CD4 and CD8 T cells in HSV-1 1716-treated tumors. Collectively, these results, confirmed by the SCID
mice results, demonstrate that the immune system is essential for HSV-1 therapy of 4T1 mammary tumors and
that HSV-1 replication can act as a flag for the development of an anti-tumor immune response that can inhibit
the growth of primary tumors and the establishment of
metastases in this model system.
The use of rodent models for the study of breast cancer
has significantly contributed to the understanding of the
biology of cancer and to the development of cancer therapeutics. Although there are limitations to the use of
rodent models, there are many similarities in structure
and function between the mammary glands of humans
and rodents [37], further stressing the importance of analyses of these models. For the studies presented here, we
examined oncolytic HSV-1 therapy on tumors of the 4T1
mouse mammary carcinoma model [1719]. 4T1 tumors
549
ARTICLE
doi:10.1016/S1525-0016(03)00236-3
established in the mammary gland of syngeneic Balb/c
mice form disseminated metastases throughout the body,
most frequently in the lungs (9295%), as well as in the
liver (75%), brain (40%), blood, and lymph nodes
[20,28]. This metastatic pattern shares some commonality
with that of human breast cancer with metastases to the
lungs (24 77%), liver (22 62%), and brain (30%)
[20,38,39]. The 4T1 model has been used extensively in
immunotherapy studies [20,28,40,41] as well as antiangiogenic therapy studies [42,43]. Although HSV-1 1716
therapy on primary tumors was only moderately effective,
we propose that mouse 4T1 mammary tumors are a good
model for HSV-1 therapy studies. Accordingly, the studies
herein with this model should serve as a therapeutic baseline from which HSV-1 1716 therapy can be improved. It
is likely that other oncolytic HSV-1 vectors that express
immune response-stimulating molecules [44,45] or angiogenesis inhibitors would be more effective in this model.
MATERIALS
AND
METHODS
Cell culture, viruses, and virus titrations. The 4T1 tumor cells were a kind
gift from Dr. Fred Miller, Karmanos Cancer Center (Detroit, MI). 4T1 cells
and Vero cells were cultured in Dulbeccos modified Eagle medium
(DMEM) (GIBCO) supplemented with 5% fetal calf serum and were maintained in 0.05% penicillin/streptomycin, at 37C, in a humidified chamber, and in an atmosphere of 5% CO2. For the studies described here, we
used the HSV-1 1716 virus, which contains a 759-bp deletion within both
copies of ICP34.5, located in the terminal 1 kb of the long repeat region of
the wild-type HSV-1 strain 17 [2]. Wild-type HSV-1 17 and the HSV-1
1716 mutant were propagated in Vero cells, purified, concentrated by
high-speed centrifugation, and titered by black plaque assay as described
elsewhere using standard methods [46].
Replication of HSV-1 in cells. The efficiency of replication of HSV-1 17
and HSV-1 1716 in cells was as previously described [9]. Briefly, both 4T1
and Vero cells at 80 85% confluency were infected at a m.o.i. of 0.1 with
HSV-1 17 and HSV-1 1716. Cells were infected for 1 h at 37C. The
inoculum was removed and the cell monolayers were washed twice with
DMEM and overlaid with 2 ml of culture medium. The zero time point
was taken after washing of the cell monolayers immediately following
infection. At the appropriate time points, the cells were scraped into the
medium and frozen at 70C. Samples were thawed and refrozen twice
prior to titering on Vero cells as described above.
Establishment and therapy of 4T1 mouse mammary tumors. Female
Balb/c mice (Taconic, Germantown, NY) or SCID mice (Wistar Institute
Animal Facility, Philadelphia, PA) at 6 10 weeks of age were injected
subcutaneously in the abdominal mammary gland with 1 104 viable 4T1
cells. Tumors were palpable by day 7 8 post-injection of 4T1 cells and had
a mean tumor diameter of 3 to 5 mm. The mean tumor diameter was
calculated as the square root of the product of two perpendicular diameters
[20]. On days 9 and 13 (double therapy) or days 9, 13, and 16 (triple
therapy), each tumor was injected using a 27.5-gauge needle in two or
three places with a total volume of 20 l containing either 5.4 105 pfu
of HSV-1 1716 or DMEM (mock-infected). The mean tumor diameter was
measured every 2 4 days using a caliper. Tumor diameters of each mouse
were normalized by subtracting the mean tumor diameter measured on
day 8 post-injection of tumor cells from the mean tumor diameter measured on each subsequent day. On day 27 post-injection of 4T1 cells, mice
were divided into two groups. The first group was assayed for survival and
the second group assayed for lung metastases as described below. SCID
mice were sacrificed and assayed for lung metastases as described below on
day 26 post-injection of 4T1 cells.
For the second challenge of tumor cells, 4T1 tumors were established
550
and received triple therapy as described above. On day 18 post-injection of
4T1 cells, mice were anesthetized using 70 mg/kg ketamine/xylazine and
the primary tumors were surgically removed. The wounds were sutured
using wound clips. Another 1 104 viable 4T1 cells were then injected
into the contralateral side of the abdomen. The second challenge of cells
was injected on the same day as the primary tumor was removed to allow
time to monitor growth of the secondary tumors before the mock-treated
mice began to succumb to metastases starting approximately 38 days
post-injection of primary tumor cells. Seven days following the second
injection, the size of the tumors was again measured on the days indicated.
Mice that became moribund were euthanized according to IACUC guidelines of both the University of Pennsylvania and the Wistar Institute.
Replication of HSV-1 1716 in 4T1 tumors. Balb/c mice with established
4T1 tumors were given triple therapy as described above. Tumors were
harvested on different days post-injection of virus and snap frozen. On the
days of viral therapy, tumors were harvested immediately after injection of
virus. Tumors were thawed at 37C in 3 ml of DMEM and homogenized
using a tissue homogenizer. Tumor-cell suspensions were then refrozen
and thawed twice. Following centrifugation, the tumor supernatant was
assayed for HSV-1 1716 levels by plaque assay as described above.
Survival analyses. On day 27 post-injection of tumor cells, mice were
anesthetized using 70 mg/kg ketamine/xylazine and the tumors were
surgically removed such that the tumor would not regrow. The wounds
were sutured using wound clips. The mice were then monitored regularly
for any signs of illness and were euthanized when moribund according to
IACUC guidelines of both the University of Pennsylvania and the Wistar
Institute.
Quantitation of clonogenic lung metastases. Metastases of 4T1 cells to the
lungs were quantified as described [20]. Briefly, mice were sacrificed to
remove the lungs, which were then treated with elastase and collagenase
and filtered through a 70-m nylon cell strainer to isolate cells. Dilutions
of lung cells were plated in tissue culture medium in the presence of 60 M
6-thioguanine to allow for clonogenic growth of 4T1 cell colonies. Thioguanine-resistant cell colonies, which appear in 10 12 days, were fixed in
methanol and stained with methylene blue for counting.
Immunohistochemistry. 4T1 tumors were established and received either
HSV-1 1716 or mock injections on days 9, 13, and 16 post-injection of
tumor cells. Two mice for each group were sacrificed and their tumors
removed on days 10, 12, 14, 15, 17, 18, 20, 22, and 23 post-injection of
tumor cells. The harvested tumors were embedded in Tissue-Tek OCT
Compound (Fisher Scientific), snap frozen in 2-methylbutane, and sectioned at 6 m. Sections were stained with standard hematoxylin and
eosin or used in immunohistochemical staining. For immunohistochemical staining, briefly, sections were fixed in a descending series of ethanol
washes, quenched with 0.3% peroxide in PBS, and blocked in 5% goat
serum. Primary antibody, either rat anti-mouse CD4 (L3T4) or CD8 (Ly-2)
(1:100, 1:80, respectively; BD Biosciences, San Diego, CA) diluted in PBS,
was incubated on sections at room temperature for 1 h. Secondary antibody, biotinylated anti-rat made in goat (1:1500; Vector Laboratories,
Burlingame, CA), diluted in PBS, was incubated on sections at room
temperature for 45 min. Positive antibody reactivity was detected using an
indirect avidin biotin immunoperoxidase method (Vectastain ABC kit)
with VectorVIP as the chromogen, and nuclei were then counterstained
using methyl green, all used per the manufacturers instructions (Vector
Laboratories). Sections were washed in between each step with PBS and
were blocked before each step except the chromogen and counterstaining
steps using 5% goat serum in PBS. As a negative control for specificity, 5%
goat serum was used in place of primary antibody.
Statistical analyses. The unpaired t test was used to analyze differences in
mean tumor diameters, and KaplanMeier curves, using the Wilcoxon test
for significance, were used for analyses of survival experiments. Statistical
analyses of lung metastases were performed on the ln of the number of
metastases to give a valid P value since the normal distribution of the
number of metastases is very broad. All analyses were performed using
Statview, and a P value less than 0.05 was considered significant.
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
ARTICLE
doi:10.1016/S1525-0016(03)00236-3
ACKNOWLEDGMENTS
We thank Kevin OBrien at East Carolina University (Greenville, NC) for guidance with statistical analyses, Srikanth Yellayi for help with histological analyses, Daniel Ruge for technical assistance, and the members of the Fraser
laboratory for helpful discussions. This work was supported by a grant from the
National Institutes of Health (NS37516) to N.W.F. D.L.T. was supported by a
NRSA (CA93034) from the National Cancer Institute.
23.
24.
25.
RECEIVED FOR PUBLICATION JANUARY 10; ACCEPTED JULY 5, 2003.
REFERENCES
1. Chou, J., Kern, E. R., Whitley, R. J., and Roizman, B. (1990). Mapping of herpes simplex
virus neurovirulence to gamma 1 34.5, a gene nonessential for growth in culture.
Science 252: 12621266.
2. MacLean, A. R., Ul-Fareed, M., Robertson, L., Harland, J., and Brown, S. M. (1991).
Herpes simplex virus type 1 deletion variants 1714 and 1716 pinpoint neurovirulencerelated sequences in Glasgow strain 17 between immediate early gene 1 and the a
sequence. J. Gen. Virol. 72: 631 639.
3. Taha, M. Y., Brown, S. M., Clements, G. B., and Graham, D. I. (1990). The JH2604
deletion variant of herpes simplex virus type 2 (HG52) fails to produce necrotizing
encephalitis following intracranial inoculation of mice. J. Gen. Virol. 71: 15971601.
4. He, B., Gross, M., and Roizman, B. (1997). The 134.5 protein of herpes simplex virus
1 complexes with protein phosphatase 1 to dephosphorylate the subunit of the
eukaryotic translation initiation factor 2 and preclude the shutoff of protein synthesis by
double-stranded RNA-activated protein kinase. Proc. Natl. Acad. Sci. USA 94: 843 848.
5. Chambers, R., et al. (1995). Comparison of genetically engineered herpes simplex
viruses for the treatment of brain tumors in a scid mouse model of human malignant
glioma. Proc. Natl. Acad. Sci. USA 92: 14111415.
6. Kesari, S., et al. (1995). Therapy of experimental human brain tumors using a neuroattenuated herpes simplex virus mutant. Lab. Invest. 73: 636 648.
7. Martuza, R. L., Malick, A., Markert, J. M., Ruffner, K. L., and Coen, D. M. (1991).
Experimental therapy of human glioma by means of a genetically engineered virus
mutant. Science 252: 854 856.
8. Miller, C. G., Krummenacher, C., Eisenberg, R. J., Cohen, G. H., and Fraser, N. W.
(2001). Development of a syngeneic murine B16 cell line derived melanoma tumor
susceptible to destruction by neuro-attenuated HSV-1. Mol. Ther. 3: 160 168.
9. Randazzo, B. P., et al. (1995). Treatment of experimental intracranial murine melanoma
with a neuroattenuated herpes simplex virus 1 mutant. Virology 211: 94 101.
10. Martuza, R. L. (2000). Conditionally replicating herpes vectors for cancer therapy.
J. Clin. Invest. 105: 841 846.
11. Markert, J. M., et al. (2000). Conditionally replicating herpes simplex virus mutant,
G207 for the treatment of malignant glioma: results of a phase I trial. Gene Ther. 7:
867 874.
12. MacKie, R. M., Stewart, B., and Brown, S. M. (2001). Intralesional injection of herpes
simplex virus 1716 in metastatic melanoma. Lancet 357: 525526.
13. Rampling, R., et al. (2000). Toxicity evaluation of replication-competent herpes simplex
virus (ICP 34.5 null mutant 1716) in patients with recurrent malignant glioma. Gene
Ther. 7: 859 866.
14. Toda, M., Rabkin, S. D., Kojima, H., and Martuza, R. L. (1999). Herpes simplex virus as
an in situ cancer vaccine for the induction of specific anti-tumor immunity. Hum. Gene
Ther. 10: 38593.
15. Miller, C. G., and Fraser, N. W. (2003). Requirement of an integrated immune response
for successful neuroattenuated HSV-1 therapy in an intracranial metastatic melanoma
model. Mol. Ther. 7: 741747.
16. Miller, C. G., and Fraser, N. W. (2000). Role of the immune response during neuroattenuated herpes simplex virus-mediated tumor destruction in a murine intracranial
melanoma model. Cancer Res. 60: 5714 5722.
17. Aslakson, C. J., and Miller, F. R. (1992). Selective events in the metastatic process
defined by analysis of the sequential dissemination of subpopulations of a mouse
mammary tumor. Cancer Res. 52: 1399 405.
18. Dexter, D. L., Kowalski, H. M., Blazar, B. A., Fligiel, Z., Vogel, R., and Heppner, G. H.
(1978). Heterogeneity of tumor cells from a single mouse mammary tumor. Cancer Res.
38: 3174 3181.
19. Miller, F. R., Miller, B. E., and Heppner, G. H. (1983). Characterization of metastatic
heterogeneity among subpopulations of a single mouse mammary tumor: heterogeneity in phenotypic stability. Invasion Metastasis 3: 2231.
20. Pulaski, B. A., and Ostrand-Rosenberg, S. (1998). Reduction of established spontaneous
mammary carcinoma metastases following immunotherapy with major histocompatibility complex class II and B7.1 cell-based tumor vaccines. Cancer Res. 58: 1486 1493.
21. Reif, A. E. (1985). Some key problems for success of classical immunotherapy. Immunity
to Cancer (In A. E. Reif, and M. S. Mitchell, Eds.), pp. 316. Academic Press, New York.
22. Miller, F. R. (1996). Models of progression spanning preneoplasia and metastasis: the
MOLECULAR THERAPY Vol. 8, No. 4, October 2003
Copyright The American Society of Gene Therapy
26.
27.
28.
29.
30.
31.
32.
33.
34.
35.
36.
37.
38.
39.
40.
41.
42.
43.
44.
45.
46.
human MCF10neoT.Tgn series and a panel of mouse mammary tumors subpopulations. Mammary Tumor Cell Cycle, Differentiation, and Metastasis (In R. B. Dickson, and
M. E. Lippman, Eds.), pp. 243263. Kluwer Academic, Boston.
Lopez, C. (1975). Genetics of natural resistance to herpes simplex virus infections in
mice. Nature 258: 152153.
Kastrukoff, L. F., Lau, A. S., and Puterman, M. L. (1986). Genetics of natural resistance
to herpes simplex virus type 1 latent infection of the peripheral nervous system in mice.
J. Gen. Virol. 67: 613 621.
Chahlavi, A., Rabkin, S., Todo, T., Sundaresan, P., and Martuza, R. (1999). Effect of prior
exposure to herpes simplex virus 1 on viral vector-mediated tumor therapy in immunocompetent mice. Gene Ther. 6: 17511758.
Kesari, S., et al. (1998). A neuroattenuated ICP34.5-deficient herpes simplex virus type
1 replicates in ependymal cells of the murine central nervous system. J. Gen. Virol. 79:
525536.
Rakhmilevich, A. L., Janssen, K., Hao, Z., Sondel, P. M., and Yang, N. S. (2000).
Interleukin-12 gene therapy of a weakly immunogenic mouse mammary carcinoma
results in reduction of spontaneous lung metastases via a T-cell-independent mechanism. Cancer Gene Ther. 7: 826 838.
Pulaski, B. A., Terman, D. S., Khan, S., Muller, E., and Ostrand-Rosenberg, S. (2000).
Cooperativity of Staphylococcal aureus enterotoxin B superantigen, major histocompatibility complex class II, and CD80 for immunotherapy of advanced spontaneous
metastases in a clinically relevant postoperative mouse breast cancer model. Cancer Res.
60: 2710 2715.
Ostrand-Rosenberg, S., Gunther, V., Armstrong, T., Pulaski, B., Pipeling, M., and
Clements, V. (1999). Immunologic targets for the gene therapy of cancer. Gene Therapy
of Cancer (In E. Lattime, and S. Gerson, Eds.), p. 33. Academic Press, San Diego, CA.
Toda, M., Rabkin, S. D., and Martuza, R. L. (1998). Treatment of human breast cancer
in a brain metastatic model by G207, a replication-competent multimutated herpes
simplex virus 1. Hum. Gene Ther. 9: 21772185.
Toda, M., Martuza, R. L., Kojima, H., and Rabkin, S. D. (1998). In situ cancer vaccination: an IL-12 defective vector/replication-competent herpes simplex virus combination
induces local and systemic antitumor activity. J. Immunol. 160: 4457 4464.
Farassati, F., Yang, A.-D., and Lee, P. W. K. (2001). Oncogenes in Ras signalling
pathway dictate host cell permissiveness to herpes simplex virus 1. Nat. Cell Biol. 3:
745750.
Goldstein, D. J., and Weller, S. K. (1988). Herpes simplex virus type 1-induced ribonucleotide reductase activity is dispensable for virus growth and DNA synthesis: isolation
and characterization of an ICP6 lacZ insertion mutant. J. Virol. 62: 196 205.
Mineta, T., Rabkin, S. D., and Martuza, R. L. (1994). Treatment of malignant gliomas
using ganciclovir-hypersensitive, ribonucleotide reductase-deficient herpes simplex viral mutant. Cancer Res. 54: 39633966.
Takakuwa, H., et al. (2003). Oncolytic viral therapy using a spontaneously generated
herpes simplex virus type 1 variant for disseminated peritoneal tumor in immunocompetent mice. Arch. Virol. 148: 813 825.
Toda, M., Iizuka, Y., Kawase, T., Uyemura, K., and Kawakami, Y. (2002). Immuno-viral
therapy of brain tumors by combination of viral therapy with cancer vaccination using
a replication-conditional HSV. Cancer Gene Ther. 9: 356 364.
Cardiff, R. D., and Wellings, S. R. (1999). The comparative pathology of human and
mouse mammary glands. J. Mammary Gland Biol. Neoplasia 4: 105122.
Kamby, C., et al. (1987). Incidence and methodologic aspects of the occurrence of liver
metastases in recurrent breast cancer. Cancer 59: 1524 1529.
Tomin, R., and Donegan, W. L. (1987). Screening for recurrent breast cancerits
effectiveness and prognostic value. J. Clin. Oncol. 5: 62 67.
Brockstedt, D. G., et al. (2002). Development of anti-tumor immunity against a
non-immunogenic mammary carcinoma through in vivo somatic GM-CSF, IL-2, and
HSVtk combination gene therapy. Mol. Ther. 6: 627 636.
Majumdar, A. S., et al. (2000). Efficacy of herpes simplex virus thymidine kinase in
combination with cytokine gene therapy in an experimental metastatic breast cancer
model. Cancer Gene Ther. 7: 1086 1099.
Huang, X., et al. (2002). Combined therapy of local and metastatic 4T1 breast tumor
in mice using SU6668, an inhibitor of angiogenic receptor tyrosine kinases, and the
immunostimulator B7.2IgG fusion protein. Cancer Res. 62: 57275735.
Lin, P., et al. (1998). Antiangiogenic gene therapy targeting the endothelium-specific
receptor tyrosine kinase Tie2. Proc. Natl. Acad. Sci. USA 95: 8829 8834.
Andreansky, S., et al. (1998). Treatment of intracranial gliomas in immunocompetent
mice using herpes simplex viruses that express murine interleukins. Gene Ther. 5:
121130.
Parker, J. N., Gillespie, G. Y., Love, C. E., Randall, S., Whitley, R. J., and Markert, J. M.
(2000). Engineered herpes simplex virus expressing IL-12 in the treatment of experimental murine brain tumors. Proc. Natl. Acad. Sci. USA 97: 2208 2213.
Spivack, J. G., and Fraser, N. W. (1987). Detection of herpes simplex virus type 1
transcripts during a latent infection in mice. J. Virol. 61: 38413847 [Erratum appears
in J. Virol., 1988, 62: 663].
551
Das könnte Ihnen auch gefallen
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Abstracts For The 27th Annual Scientific Meeting of The Society For Immunotherapy of Cancer (SITC) PDFDokument71 SeitenAbstracts For The 27th Annual Scientific Meeting of The Society For Immunotherapy of Cancer (SITC) PDFhigginscribdNoch keine Bewertungen
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- ELP Language Biography Checklists For Young Learners enDokument8 SeitenELP Language Biography Checklists For Young Learners enhigginscribdNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5795)
- ALTE CEFR Speaking Grid Tests2014Dokument11 SeitenALTE CEFR Speaking Grid Tests2014higginscribdNoch keine Bewertungen
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- ELP Language Biography Plurilingual Profile enDokument5 SeitenELP Language Biography Plurilingual Profile enhigginscribdNoch keine Bewertungen
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- ELP What Is A Portfolio ENDokument1 SeiteELP What Is A Portfolio ENhigginscribdNoch keine Bewertungen
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- Praepositiones LatinaeDokument1 SeitePraepositiones LatinaeMagister Optimus MaximusNoch keine Bewertungen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Catalog Sep 2014Dokument14 SeitenCatalog Sep 2014higginscribdNoch keine Bewertungen
- ELP Language Biography Generic Checklists enDokument18 SeitenELP Language Biography Generic Checklists enhigginscribdNoch keine Bewertungen
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Luraghi Narrog 4 Luraghi-LibreDokument76 SeitenLuraghi Narrog 4 Luraghi-LibrehigginscribdNoch keine Bewertungen
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- 02.04.quiz 1 SolutionsDokument1 Seite02.04.quiz 1 SolutionshigginscribdNoch keine Bewertungen
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- What Are Complex Systems?Dokument18 SeitenWhat Are Complex Systems?higginscribdNoch keine Bewertungen
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- Differential Equations: Some Notes On Terminology: DX DT F (X)Dokument4 SeitenDifferential Equations: Some Notes On Terminology: DX DT F (X)higginscribdNoch keine Bewertungen
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Solutions To Quiz 2, Unit 2.4Dokument1 SeiteSolutions To Quiz 2, Unit 2.4higginscribdNoch keine Bewertungen
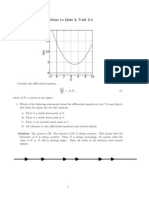
- Solutions To Quiz 1, Unit 2.2Dokument1 SeiteSolutions To Quiz 1, Unit 2.2higginscribdNoch keine Bewertungen
- 02.05AlgorithmicSolutions Summary 2Dokument3 Seiten02.05AlgorithmicSolutions Summary 2higginscribdNoch keine Bewertungen
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
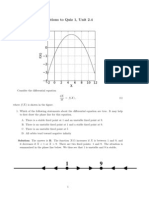
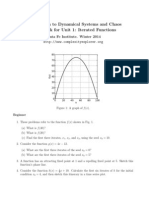
- Introduction To Dynamical Systems and Chaos Homework For Unit 1: Iterated FunctionsDokument2 SeitenIntroduction To Dynamical Systems and Chaos Homework For Unit 1: Iterated FunctionshigginscribdNoch keine Bewertungen
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Self-Organization and Cooperation in Social SystemsDokument29 SeitenSelf-Organization and Cooperation in Social SystemshigginscribdNoch keine Bewertungen
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Cellular Automata Are Idealized Models of Complex SystemsDokument58 SeitenCellular Automata Are Idealized Models of Complex SystemshigginscribdNoch keine Bewertungen
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1091)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)