Beruflich Dokumente
Kultur Dokumente
Topic 1.1 - Catalysis and Catalytic Reactors
Hochgeladen von
Norzaifee NizamudinCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Topic 1.1 - Catalysis and Catalytic Reactors
Hochgeladen von
Norzaifee NizamudinCopyright:
Verfügbare Formate
CHE625
Lecturer: Pn. Sharmeela bt Matali
Office
Advanced
Chemical
Reaction
Engineering
: PA-11-8C (level 11,
Tower 2)
Contact:
03-5543 6328 (O)
013-299 0609
sharmeela@salam.uitm.edu.my
Credit to: Cik Nor Faeqah
Pn Norhasyimi
CPE624
Course Learning Outcome:
By the end of this topic, you should be able to:
Explain the steps in catalytic reactions; write the mechanism and
chemical kinetics.
Able to describe rate limiting step.
Able to describe Langmuir-Hinshelwood Kinetic Mechanism.
Ref : The Eng. Of Chemical Reactions ( Schmidt) Chapter 7 (pg 269)
FACULTY OF CHEMICAL ENGINEERING
CPE624
INTRODUCTION
IDEAL REACTORS REVISION
FACULTY OF CHEMICAL ENGINEERING
CPE624
Design Equation for Ideal
Homogeneous Reactors
Ideal
Batch Reactor
Mass
Balance
Constant density system
FACULTY OF CHEMICAL ENGINEERING
CPE624
Design Equation for Ideal
Homogeneous Reactors
Ideal
Plug Flow Tubular Reactor (PFTR/PFR)
Ideal
Constant Stirred Tank Reactor (CSTR)
FACULTY OF CHEMICAL ENGINEERING
CPE624
TO TEST your BASICS of CRE
The elementary of liquid phase reaction A ->B + 2C is
carried out isothermally in a flow reactor with no
pressure drop. The rate constant at 50oC is 10-4min-1 and
the activation energy is 85 kJ/mol. Pure A enters at 10
atm and 127oC, and molar flow rate is 2.5 mol/min.
Calculate the space time and reactor volume for 90%
conversion in CSTR and PFR.
FACULTY OF CHEMICAL ENGINEERING
CPE624
CHAPTER 1
CATALYTIC REACTORS AND
MASS TRANSFER
FACULTY OF CHEMICAL ENGINEERING
CPE624
1.1 Catalytic Reactions
What is Catalyst and Catalysis?
A catalyst is a substance that affects the rate of
a reaction but does not enter into the
stoichiometry of the reaction.
Catalysis is the change in rate of a
chemical reaction due to the participation of a
substance called a catalyst.
Catalysis is the occurrence, study, and use of catalysts and catalytic
processes.
Catalysts that speed the reaction are called positive catalysts WHILE
slow the reaction are called inhibitors (or negative catalysts).
Substances that increase the activity of catalysts are called promoters,
and substances that deactivate catalysts are called catalytic poisons.
FACULTY OF CHEMICAL ENGINEERING
10
CPE624
Lets watch a video on catalysis:
http://www.youtube.com/watch?
v=A_PhvIktMOw&feature=endscreen
FACULTY OF CHEMICAL ENGINEERING
11
CPE624
Example of Catalysis
Gaseous H2 and O2 are inert at room temperature.
They react rapidly when exposed to Pt catalyst.
FACULTY OF CHEMICAL ENGINEERING
12
CPE624
Catalysts and Catalysis
Different shapes and sizes of catalyst.
FACULTY OF CHEMICAL ENGINEERING
13
CPE624
FACULTY OF CHEMICAL ENGINEERING
14
CPE624
Catalysts and Catalysis
Catalytic packed-bed reactor, schematic.
FACULTY OF CHEMICAL ENGINEERING
15
CPE624
Theory of Catalysis
Activated complex formation theory:
Catalysts generally react with one or more reactants
to form intermediates that subsequently give the
final reaction product, in the process regenerating
the catalyst. The following is a typical reaction
scheme, where C represents the catalyst, X and Y
are reactants, and Z is the product of the reaction of
X and Y:
X + C XC (1)
Y + XC XYC (2)
XYC CZ (3)
CZ C + Z (4)
X+YZ
FACULTY OF CHEMICAL ENGINEERING
16
CPE624
Theory of Catalysis
Adsorption theory (Old): The
reactants
in
gaseous
state
or
in
solu0ons,
are
adsorbed
on
the
surface
of
the
solid
catalyst.
The
increase
in
concentra0on
of
the
reactants
on
the
surface
increases
the
rate
of
reac0on.
Adsorp0on
being
an
exothermic
process,
the
heat
of
adsorp0on
is
u0lised
in
enhancing
the
rate
of
the
reac0on.
Adsorption theory (Modern):
The
modern
adsorp0on
theory
is
the
combina0on
of
intermediate
compound
forma0on
theory
and
the
old
adsorp0on
theory.
The
mechanism
involves
ve
steps:
(1)
Diusion
of
reactants
to
the
surface
of
the
catalyst.
(2)
Adsorp0on
of
reactant
molecules
on
the
surface
of
the
catalyst.
(3)
Occurrence
of
chemical
reac0on
on
the
catalysts
surface
through
forma0on
of
an
intermediate
(Figure
depicted
below).
(4)
Desorp0on
of
reac0on
products
from
the
catalyst
surface,
and
thereby,
OF CHEMICAL
ENGINEERING
making
the
surface
available
again
for
more
FACULTY
reac0on
to
occur.
(5)
Diusion
of
reac0on
products
away
from
the
catalysts
surface.
17
CPE624
Theory of Catalysis
Adsorption theory
H
Pt
H
Pt
Pt
Pt
Pt
O
Pt
Pt
Pt
Pt
Pt
Pt
Pt
Pt
FACULTY OF CHEMICAL ENGINEERING
18
CPE624
A Mechanism for the oxidation of carbon
monoxide
FACULTY OF CHEMICAL ENGINEERING
19
CPE624
What
is
adsorp1on?
Adsorption is a physical or chemical phenomenon by which
the molecules present in a liquid or a gas attach to the
surface of a solid.
Surface
means
both
external
and
internal
surface.
The substance on which surface adsorp1on occurs is termed as the
adsorbent, and the substance which adsorbed from the bulk phase is
known as the adsorbate.
Depending on the force of aBrac1on, adsorp1on is mainly two types: (1)
Physical adsorp1on (Physisorp1on) and (2) Chemical adsorp1on
FACULTY OF CHEMICAL ENGINEERING
(Chemisorp1on).
20
CPE624
Physical
Adsorp1on
Physical
adsorp1on
(Physisorp1on)
Physical
adsorp1on
is
a
phenomenon
which
takes
place
purely
due
to
the
van
der
Waals
forces
of
aBrac1on.
-
It
is
a
reversible
phenomenon.
-
Because
of
very
week
force
of
aBrac1on,
the
physical
adsorp1on
can
not
bring
to
any
change
of
chemical
structure
of
the
adsorbent
and
adsorbate.
-
It
can
be
compared
with
the
condensa1on
of
vapour
of
liquids.
FACULTY OF CHEMICAL ENGINEERING
21
CPE624
Chemical
Adsorp1on
Chemical
adsorp1on
(Chemisorp1on)
Chemical
adsorp1on
is
adsorp1on
which
results
from
chemical
bond
forma1on
(strong
interac1on)
between
the
adsorbent
and
the
adsorbate
in
a
monolayer
on
the
surface.
Example:
Organic
compound
get
adsorbed
on
the
solid
surface
with
chemical
bond
forma1on.
FACULTY OF CHEMICAL ENGINEERING
22
CPE624
Why does solid substance adsorb?
The adsorption phenomenon comes
from the existence of non-compensated
forces of a physical nature on the
surface of the solid.
Adsorbate
Adsorbent
Adsorbate
All the bonding requirements of the
constituent atoms of the material are
filled by other atoms in the material.
However, atoms on the surface of the
adsorbent are not wholly surrounded by
other adsorbent atoms and therefore can
attract adsorbates.
FACULTY OF CHEMICAL ENGINEERING
23
CPE624
Heterogeneous Reactor and
Voidage,
Reactor
volume, V = Volume of fluid plus
volume of catalyst
V = V fluid + V catalyst
Void
fraction or the fraction of the reactor
volume occupied by fluid
V fluid
Volume of fluid
=
=
Volume of reactor
V
FACULTY OF CHEMICAL ENGINEERING
24
CPE624
Different types of Rates in
Heterogeneous Catalytic Reactions
Homogeneous
reactions:
r=
Heterogeneous
moles
volume time
reactions:
moles
r =
area time
''
moles
r =
mass time
'
FACULTY OF CHEMICAL ENGINEERING
25
CPE624
Pseudo-homogeneous rate
r
x (volume ) = r x (area of catalyst)
Surface
area of catalyst
surface area of catalyst
sg =
mass of catalyst
r = s g c (1 )r"
FACULTY OF CHEMICAL ENGINEERING
26
CPE624
Space time,
volume of fluid in a reactor
=
volumetric flow rate passes through the reactor
Homogeneous
Reactors:
Heterogeneous
V fluid = V
volume of reactor
inlet volumetric flow rate
Reactors
=
V
0
FACULTY OF CHEMICAL ENGINEERING
27
CPE624
What are the criteria of the catalyst?
The following criteria dene a good quality catalyst
for a reac1on:
1. Only small quan1ty is needed for a
reac1on.
2. They are specic. One catalyst is needed for specic
reac1on only.
3. Physical proper1es may change during a reac1on
but it
does not take part in the reac1on.
4. No catalyst can change an equilibrium state of a
FACULTY OF CHEMICAL ENGINEERING
reac1on.
28
CPE624
What are the proper1es of the catalysts?
Porous catalyst catalyst that has a large area
Molecular sieves small pore that will admit small
molecule
Monolithic
catalyst
can
be
either
porous
or
non-porous
Supported
catalyst
-
consist
of
par1cles
of
an
ac1ve
material
dispersed
over
a
less
ac1ve
substance.
Unsupported
catalyst
Promoters
small
amount
of
ac1ve
ingredients
Deac1va1on
of
catalyst
-
Aging
-
Poisoning
-
Fouling
or
coking
FACULTY OF CHEMICAL ENGINEERING
29
CPE624
Catalyst Properties
Porous
catalyst catalyst that has a large area
Molecular sieves small pore that will admit small
molecule
Monolithic catalyst can be either porous or nonporous
Supported catalyst - consist of particles of an
active material dispersed over a less active
substance.
Unsupported catalyst
Promoters small amount of active ingredients
FACULTY OF CHEMICAL ENGINEERING
30
CPE624
Catalyst Properties
Deactivation
of catalyst
Aging
Poisoning
Fouling
Catalytic
or coking
reaction involved:
Physical
adsorption
Chemical adsorption
FACULTY OF CHEMICAL ENGINEERING
31
CPE624
Porous catalyst
Why
we need porous catalyst???
A few major catalyst:
Amorphous
silica
High area aluminas
Zeolites/aluminosilicates
Carbon
Supported noble metal catalysts (Pt, Pd, Ag,
Rh, Ni etc)
FACULTY OF CHEMICAL ENGINEERING
32
CPE624
Explanation of Catalytic Cracking through Zeolites- video
FACULTY OF CHEMICAL ENGINEERING
33
CPE624
Catalytic Reactors
Packed
Bed Reactor
Slurry reactor
Fluidized bed reactor
Riser reactor
FACULTY OF CHEMICAL ENGINEERING
34
CPE624
Catalytic Reactors
Packed
bed Reactor
Assume
no mixing
Mass balance:
dC j
dz
= jr
FACULTY OF CHEMICAL ENGINEERING
35
CPE624
Catalytic Reactors
Slurry
and Fludized Bed Reactor
Assume
well mixed
Mass Balance:
The image cannot be displayed. Your computer may not have enough memory to open the image, or the image may have been corrupted. Restart your computer, and then open the file again. If the red
x still appears, you may have to delete the image and then insert it again.
The image cannot be displayed. Your computer may not have enough memory to open the
image, or the image may have been corrupted. Restart your computer, and then open the
file again. If the red x still appears, you may have to delete the image and then insert it
again.
FACULTY OF CHEMICAL ENGINEERING
36
CPE624
Example and Exercise in
Class
QUESTION
1 Tutorial 1
Example 7-1 (Schmidt) page 277
Problem 7-2 pg 318
FACULTY OF CHEMICAL ENGINEERING
Das könnte Ihnen auch gefallen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- 0580 m17 QP 42 PDFDokument20 Seiten0580 m17 QP 42 PDFNorzaifee NizamudinNoch keine Bewertungen
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- 2018u009 PDFDokument14 Seiten2018u009 PDFNorzaifee NizamudinNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- No Id Mac: Borang Penilaian Ujian BleepDokument1 SeiteNo Id Mac: Borang Penilaian Ujian BleepNorzaifee NizamudinNoch keine Bewertungen
- Bukit Tinggi Booking Number: 990110 Jumanji: Welcome To The JungleDokument1 SeiteBukit Tinggi Booking Number: 990110 Jumanji: Welcome To The JungleNorzaifee NizamudinNoch keine Bewertungen
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Tips Service ChargesDokument7 SeitenTips Service ChargesNorzaifee NizamudinNoch keine Bewertungen
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Surds Past Edexcel Exam Questions: C Studywell Publications Ltd. 2015Dokument4 SeitenSurds Past Edexcel Exam Questions: C Studywell Publications Ltd. 2015Norzaifee NizamudinNoch keine Bewertungen
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Take5 OilnGasDokument8 SeitenTake5 OilnGasNorzaifee NizamudinNoch keine Bewertungen
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Flue Gas Treatment For Co2 Capture - ccc169Dokument61 SeitenFlue Gas Treatment For Co2 Capture - ccc169Norzaifee Nizamudin100% (1)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- Physical, Chemical and Biological Characteristics of WastewaterDokument45 SeitenPhysical, Chemical and Biological Characteristics of WastewaterNorzaifee Nizamudin100% (2)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Carta Organisasi AblePerfectDokument1 SeiteCarta Organisasi AblePerfectNorzaifee NizamudinNoch keine Bewertungen
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- The Financial Benefits of Cleaner Production (CP)Dokument11 SeitenThe Financial Benefits of Cleaner Production (CP)Norzaifee NizamudinNoch keine Bewertungen
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Guideline NadopodDokument28 SeitenGuideline NadopodNorzaifee NizamudinNoch keine Bewertungen
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- Yoko Zeki 2008Dokument10 SeitenYoko Zeki 2008Norzaifee NizamudinNoch keine Bewertungen
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
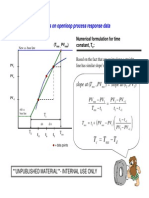
- Numerical Time ConstantDokument1 SeiteNumerical Time ConstantNorzaifee NizamudinNoch keine Bewertungen
- Delayed Coking: Chapter 5Dokument0 SeitenDelayed Coking: Chapter 5LTE002Noch keine Bewertungen
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Cracking (Chemistry) - WikipediaDokument3 SeitenCracking (Chemistry) - WikipediaDharmesh patelNoch keine Bewertungen
- Selective Hydrogenation of Vinyl Acetylene To 1,3-Butadiene in Concentrated Vinyl Acetylene Mixed C4 by PdAl2O3 CatalystDokument1 SeiteSelective Hydrogenation of Vinyl Acetylene To 1,3-Butadiene in Concentrated Vinyl Acetylene Mixed C4 by PdAl2O3 CatalystPaisan InsornNoch keine Bewertungen
- Adobe Scan 01-Mar-2022Dokument23 SeitenAdobe Scan 01-Mar-2022RamNoch keine Bewertungen
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- CHE246 - Technical Lab Report On Rotary PDFDokument5 SeitenCHE246 - Technical Lab Report On Rotary PDFanujaNoch keine Bewertungen
- Xmas Tree & SSTT DrawingDokument1 SeiteXmas Tree & SSTT DrawingAderobaki GbengaNoch keine Bewertungen
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- Cdiars3 Module Act. DinangwatanDokument2 SeitenCdiars3 Module Act. DinangwatanEngland Israel SevidalNoch keine Bewertungen
- Partition Coefficient EditedDokument17 SeitenPartition Coefficient EditedcoltalbNoch keine Bewertungen
- ALKYLATIONDokument6 SeitenALKYLATIONtariq fareedNoch keine Bewertungen
- Products of CombustionDokument27 SeitenProducts of CombustionLorlie Bernales Blanca-TanjayNoch keine Bewertungen
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- Hot Filtration Melting Point Determination: NOT ClosedDokument3 SeitenHot Filtration Melting Point Determination: NOT ClosedGrawpNoch keine Bewertungen
- AL-2 Flowdiagram SeparationDokument1 SeiteAL-2 Flowdiagram SeparationCitramanda ServicesNoch keine Bewertungen
- Company Profile 2023Dokument34 SeitenCompany Profile 2023Elrich Joseph JalandoniNoch keine Bewertungen
- 09highly Coupled Dist SystDokument19 Seiten09highly Coupled Dist SystJohn YuNoch keine Bewertungen
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- DGS 6600 010 Rev 2-48Dokument1 SeiteDGS 6600 010 Rev 2-48Mohamed NasifNoch keine Bewertungen
- Cathodic ProtectionDokument15 SeitenCathodic ProtectionmiraNoch keine Bewertungen
- MBR PresentaionDokument14 SeitenMBR PresentaionnurmaNoch keine Bewertungen
- BIO307 Lecture 7 (Enzyme Kinetics III)Dokument11 SeitenBIO307 Lecture 7 (Enzyme Kinetics III)Phenyo Mmereki100% (2)
- Recovery of Acetic Acid by Means of Liquid-Liquid ExtractionDokument5 SeitenRecovery of Acetic Acid by Means of Liquid-Liquid ExtractionAYALEYDENNoch keine Bewertungen
- HydrodesulfurisationDokument3 SeitenHydrodesulfurisationSO R ANNoch keine Bewertungen
- JIPO Catalogue CrucibleDokument1 SeiteJIPO Catalogue CrucibleSNP suvananNoch keine Bewertungen
- Classics in Total Synthesis - 5a19e8ef1723dd231dc62f63Dokument12 SeitenClassics in Total Synthesis - 5a19e8ef1723dd231dc62f63Preeti YadavNoch keine Bewertungen
- Quarter 1-Module 1-Lesson 3Dokument36 SeitenQuarter 1-Module 1-Lesson 3Charis Mades Maglasang- Alejo100% (3)
- Material Science Chapter 7.2 PDFDokument15 SeitenMaterial Science Chapter 7.2 PDFamraqstnaNoch keine Bewertungen
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Reaction Engineering Lectures - Part2Dokument38 SeitenReaction Engineering Lectures - Part2Ahmed AliNoch keine Bewertungen
- Enzyme Kinetics Questions and Answers: 2 Year Undergraduates-Biology 2018-2019Dokument8 SeitenEnzyme Kinetics Questions and Answers: 2 Year Undergraduates-Biology 2018-2019Emmanuel JoyNoch keine Bewertungen
- Nickel Chromium PlatingDokument10 SeitenNickel Chromium Platingselvamuthukumar100% (1)
- Sulfur Compound Extraction and Sweetening: P A R T 11Dokument14 SeitenSulfur Compound Extraction and Sweetening: P A R T 11Bharavi K SNoch keine Bewertungen
- Vulcanization - WikipediaDokument6 SeitenVulcanization - WikipediaAanand MishraNoch keine Bewertungen
- 1-2. Unit OperationsDokument45 Seiten1-2. Unit OperationsIqbalMananiNoch keine Bewertungen