Beruflich Dokumente
Kultur Dokumente
Tolerância Imunológica
Hochgeladen von
Akla CruzCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Tolerância Imunológica
Hochgeladen von
Akla CruzCopyright:
Verfügbare Formate
Review
|
doi: 10.1111/j.1365-2796.2007.01855.x
Immune tolerance: mechanisms and application in clinical
transplantation
M. Sykes
From the Transplantation Biology Research Center, Bone Marrow Transplantation Section, Massachusetts General
Hospital Harvard Medical School, Boston, MA, USA
Abstract. Sykes M (Massachusetts General Hospital Harvard Medical School, Boston, MA, USA).
Immune tolerance: mechanisms and application in
clinical transplantation (Review). J Intern Med 2007;
262: 288310.
The achievement of immune tolerance, a state of
specic unresponsiveness to the donor graft, has the
potential to overcome the current major limitations to
progress in organ transplantation, namely late graft
loss, organ shortage and the toxicities of chronic
nonspecic immumnosuppressive therapy. Advances
The need for immune tolerance in transplantation
Immune tolerance is a state in which the immune system is specically unresponsive to antigens of interest.
For example, most people enjoy a state of immune
tolerance to their own antigens, resulting in freedom
from autoimmune disease. In the case of organ and cell
transplantation, tolerance denotes a state of specic
immune unresponsiveness to the donor graft, with normal responses to other antigens. The ability to respond
normally to other antigens contrasts sharply with the
effect of nonspecic immunosuppressive agents that
are used clinically to prevent rejection, which are associated with increased risks of infection and malignancy. This paper will review the current status of
tolerance in the eld of organ and tissue transplantation. Achievement of transplantation tolerance is the
holy grail in clinical transplantation for three major
reasons. First, whilst improvements in nonspecic
immunosuppressive therapy have markedly improved
outcomes in organ transplantation, these drugs are
associated with many specic organ toxicities as well
as the life-long increased risks of infection and
288
2007 Blackwell Publishing Ltd
in our understanding of immunological processes,
mechanisms of rejection and tolerance have led
to encouraging developments in animal models,
which are just beginning to be translated into
clinical pilot studies. These advances are reviewed
here and the appropriate timing for clinical trials is
discussed.
Keywords: bone marrow transplantation, immunity,
immunology, immunosuppressive treatment, transplantation immunology.
malignancy mentioned above. Secondly, chronic
rejection is a major factor contributing to constantly
downsloping long-term survival curves for organ
allografts. The half-lives of this second, late phase of
graft loss have not changed signicantly with improvements in immunosuppressive therapy over the last
25 years. Chronic rejection can be avoided by
tolerance induction. Thirdly, there is a critical shortage
of allogeneic organs for transplantation, which could
be overcome by the use of other species as organ and
tissue sources, i.e. xenografts. However, immune
barriers to xenografts are even stronger than those to
allografts, and the induction of tolerance at both the
humoral and the cellular level is likely to be needed
for the successful application of xenotransplantation in
humans.
Numerous approaches to tolerance induction have
been developed in rodent models. Many of these largely reect the strong inherent tolerogenicity of
primarily vascularized heart, liver and kidney grafts in
these animals rather than the potency of the toleranceinducing regimens per se. A short course of many
M. Sykes
Review: Transplantation tolerance
types of immunosuppression can allow these tolerogenic effects to prevail over the rejection response,
leading to long-term graft acceptance. Because such
grafts are unfortunately less tolerogenic in large animals and humans, these tolerance strategies have not
been effectively applied in humans. Thus, before clinical evaluation is appropriate, tolerance strategies
should rst be tested in stringent animal models,
including strongly immunogenic grafts such as major
histocompatibility complex (MHC)-mismatched skin
in rodents and vascularized organ graft models in
large animals.
The simple denition of tolerance in the rst paragraph above includes several different immunological
states. In one, the allograft is accepted without chronic immunosuppression, but the recipient can reject a
second graft from the same donor. In a different state,
the immune system accepts any other organ or tissue
from the same donor without immunosuppression,
and in vitro studies reveal specic unresponsiveness
to the donor. This state of tolerance can be described
as systemic. There are intermediate forms of tolerance
in which some types of second graft, but not others
from the same donor, are accepted. In some forms of
tolerance, in vitro studies show normal or reduced
anti-donor responses without the complete unresponsiveness that characterizes systemic tolerance. In all
these states, the recipient can reject organs from a
third party donor.
The above discussion is focused on T-cell tolerance
because T cells clearly play a central role in allograft
rejection. In nave allograft recipients, B-cell tolerance
is not a separate concern, because in the absence of
help from donor-reactive T cells, de novo anti-donor
alloantibody responses are not generated. However,
there are several situations in which B-cell tolerance
would be advantageous. These include transplantation
to recipients with preexisting natural antibodies,
which are antibodies that are present without known
prior sensitization, against the donor. Examples are
anti-isohaemagglutinins against blood group antigens
and natural antibodies in sera of primates that
recognize porcine carbohydrate antigens, as is
discussed below. Additionally, a recipient may contain
anti-donor antibodies because of prior sensitization
to antigens of that donor, for example due to pregnancy, blood transfusions or prior transplants.
Mechanisms of T and B-cell tolerance
As discussed above, the achievement of T-cell tolerance would overcome the major barriers to successful
allografting discussed above. There are three major
mechanisms of T-cell tolerance, including clonal deletion, anergy and suppression (commonly referred to
as regulation). These mechanisms may act alone or
together to achieve tolerance. Clonal deletion implies
death of T cells with receptors recognizing donor
antigens. Deletion is the major mechanism of selftolerance induction during T-cell development in the
thymus. Mature T cells in the peripheral lymphoid
tissues can also be deleted under certain conditions.
Suppression, in which a cell population actively
downregulates the reactivity of T cells, has recently
been implicated in many rodent transplantation tolerance models and in the maintenance of self-tolerance.
Anergy denotes the inability of T cells to proliferate
and produce interleukin-2 (IL-2) in response to antigens they recognize. In addition, a graft may simply
be ignored by recipient T cells. These mechanisms
are discussed in more detail and in the context of
transplantation below.
T cell clonal deletion in transplantation
Most intrathymic T-cell tolerance results from deletion
of developing thymocytes whose receptors recognize
self antigens presented by haematopoietic cells and
thymic epithelial cells (reviewed in Ref. [1]). The processes involved in T-cell education in the thymus
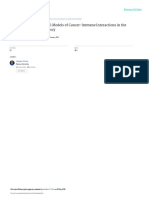
are depicted and explained in Fig. 1. High avidity
interactions between immature thymocytes due, at
least in part, to a relatively high afnity interaction
between a rearranged T-cell receptor (TCR) and a
peptide MHC complex on antigen-presenting cells
(APC) in the thymus, result in deletion of the thymocyte by apoptotic cell death [2, 3]. TCRs with lower
afnity for such complexes are more likely to survive
this process, and other mechanisms are required to
ensure their tolerance when they enter the periphery,
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 289
M. Sykes
(a)
Review: Transplantation tolerance
(b)
Fig. 1 Schematic depiction of T cell education in the thymus (a) and the role of mixed chimerism in achieving central deletional tolerance (b). In the normal situation (a) thymocyte and APC progenitors migrate to the thymus from the marrow (1) to
become double negative thymocytes (2) or APCs. Thymocyte progenitors may productively rearrange a and b T cell receptor
chains, which are rst expressed by CD4+CD8+ (double positive) thymocytes (3). These thymocytes are then subject to positive and negative selection processes. Positive selection (3), which rescues the double positive thymocyte from programmed cell
death, results from a low avidity interaction between thymocytes and thymic epithelial cells in the thymic cortex. This interaction requires a low-afnity interaction between the rearranged thymocyte TCR and a self MHC peptide complex presented by
the thymic epithelial cell. Depending on whether this MHC molecule is of class I or class II, the thymocyte will lose expression of CD4 or CD8, respectively, resulting in the generation of a CD8 or CD4 single positive thymocyte. The double-positive
or single-positive thymocyte may also die, however, if it interacts with higher afnity with a self MHC peptide complex on an
APC (4) or, possibly, a thymic epithelial cell, in the thymic medulla or corticomedullary junction. The surviving thymocytes
undergo further maturation (5), then leave the thymus and enter the peripheral lymphoid tissues. In mixed chimeras (b), thymocyte and APC progenitors of both donor and host origin migrate to the thymus from the marrow (1) to become double negative
thymocytes or APCs (2). Thymocyte progenitors of both types may productively rearrange a and b T cell receptor chains and
become CD4 CD8 double positive thymocytes (3). These thymocytes are then subject to positive and negative selection processes. Positive selection (3), which rescues the double positive thymocyte from programmed cell death, is mediated exclusively by thymic epithelial cells and hence MHC of host origin in the thymic cortex. Depending on whether this MHC
molecule is of class I or class II, the donor or recipient thymocyte will lose expression of CD4 or CD8, respectively, resulting
in the generation of donor and recipient CD8 and CD4 single positive thymocytes. The double positive or single positive
thymocyte may also die, however, if it interacts with higher afnity with an MHC peptide complex on a donor or recipientderived APC (4) or, possibly, a thymic epithelial cell, in the thymic medulla or corticomedullary junction. Consequently, only
mature T cells that lack strong reactivity to donor or host antigens survive this negative selection process, resulting in emergence from the thymus only of T cells (of both donor and recipient origin) that are tolerant of both the donor and the host (5).
particularly under conditions of inammation and
antigen upregulation. Indeed, deletion is not the only
mechanism of intrathymic tolerance induction: T cells
with receptors recognizing self antigens presented by
nonhaematopoietic thymic stromal cells [4] or even
haematopoietic cells [5] may be rendered anergic.
Additionally, presentation of antigens by the thymic
epithelium promotes the development of specic
regulatory cells that tolerize other T cells in the
periphery [6].
Intrathymic deletion is induced most potently by antigen presented on haematopoietic cell types, including
dendritic cells [7]. This is a major reason why
290
allogeneic haematopoietic transplantation (HCT)
provides a powerful approach to tolerance induction
in lymphoablated rodents. To avoid rejection of the
marrow, specic T-cell ablation in the thymus and the
periphery can be achieved with relatively nontoxic,
nonmyeloablative conditioning that includes T-celldepleting monoclonal antibodies (mAb) and local irradiation to the thymus [8, 9]. As might be expected,
tolerance induced by intrathymic deletion is systemic,
as shown both in vivo and in vitro (reviewed in Ref.
[1]). In this setting, the only signicant mechanism
involved in maintaining transplantation tolerance is
intrathymic clonal deletion [1012]. Anti-donor antibody can be given to established mixed chimeras to
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
Review: Transplantation tolerance
eliminate donor chimerism; this results in loss of
tolerance to donor skin grafts, and in the de novo
appearance in the blood of T cells with receptors that
recognize donor antigens. However, if the recipient
thymus is removed before chimerism is eliminated
with anti-donor antibody, specic tolerance to the
donor is preserved, and donor-reactive TCR do not
appear in the circulation [11]. These results show that
chimerism is needed only in the thymus and not in
the periphery to ensure persistent tolerance. Antigen
in the periphery is not required and, once the thymus
is removed, donor antigen is not required to maintain
tolerance at all. These results are consistent with a
purely central deletional tolerance mechanism, as
tolerance resulting from peripheral anergy or suppression requires persistent antigen [1315]. Thymic APC
continually turn over, emphasizing the need for
haematopoietic stem cell engraftment at sufcient
levels in order to ensure an uninterrupted supply of
donor APC in the recipient thymus for life when
tolerance depends solely on intrathymic deletion.
Because they lack active suppressive tolerance mechanisms, such animals are vulnerable to loss of tolerance if nontolerant T cells are allowed to emerge
from the thymus after intentional depletion of donor
antigen, or after exogenous administration of nontolerant host-type T cells [11, 16].
Exposure of mature T cells to antigen in the periphery can also result in T-cell clonal deletion [17].
Self antigen cross-presentation by lymph node dendritic cells under noninammatory conditions leads to
deletion of tissue antigen-specic CD8+ cytotoxic T
lymphocytes (CTL) [18]. CD8 cells may be deleted
because of exhaustion in the presence of a large,
persistent antigen load [19]. As an alternative to
global T-cell depletion, co-stimulatory blockade with
anti-CD154 (see below) can be used in combination
with bone marrow transplantation (BMT) to achieve
mixed chimerism and long-term central, deletional
tolerance [20, 21]. In such animals, the preexisting
alloreactive T-cell repertoire is not depleted with
mAb, and other mechanisms come into play.
Peripheral deletion, specically, of donor-reactive
CD8 [22, 23] and CD4 [16, 20, 24, 25] cells occurs
under these conditions. A similar phenomenon has
been demonstrated for peripheral CD8 cells in mice
receiving donor-specic transfusion (DST) combined
with anti-CD154 [26]. Peripheral T cell apoptosis has
been demonstrated, though without specic markers
for alloreactive T cells, in mice tolerized with
anti-CD154 mAb, rapamycin and cardiac allografts
[27].
Additional mechanisms of peripheral deletion, such as
the activity of veto cells, which are cells that kill
CTL that recognize them [28], can delete alloreactive
CTL precursors in the periphery. Recently,
CD4)CD8) cytotoxic regulatory cells have been
reported to delete alloreactive CD8+ T cells with the
same specicity as the regulatory cells [29].
B cell clonal deletion in transplantation
As discussed above, there are several transplant situations in which tolerance induction of B cells would
be of potential value. Immunoglobulin (Ig) receptor
transgenic mice have been widely used for the analysis of mechanisms of B-cell tolerance. Such studies
indicate that immature B cells are susceptible to
deletion when they encounter membrane-bound antigens expressed by haematopoietic or nonhaematopoietic cells [30]. Developing B cells whose
rearranged Ig receptors recognize a self antigen
undergo developmental arrest followed by Ig light
chain receptor editing. If this second chance rearrangement leads to the formation of a nonautoreactive
Ig receptor, the B cell survives; if not, the B cell
dies [31].
B cells have been divided into several subsets, including follicular, marginal zone and B-1 B cells. B-1
cells in mice produce natural antibodies recognizing important xenogeneic carbohydrate antigens
without known prior immunization [32]. Data suggest that a similar subset may produce such natural
antibodies in nonhuman primates and man [33].
Such antibodies are responsible for xenograft hyperacute rejection, and can be deleted in mice via apoptosis when their surface Ig receptors are cross-linked
by cell-bound antigens [34]. Deletion and or receptor
editing is responsible for the long-term tolerance of
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 291
M. Sykes
natural antibody-producing B-1 cells in mice when
mixed chimerism is induced using a bone marrow
donor that expresses an antigen for which natural
antibody-producing cells preexist in the recipient
[3537].
T cell anergy
In addition to a signal through their T-cell receptor, T
cells require stimulation of additional receptors,
termed co-stimulatory molecules, in order to be fully
activated. CD28 is a major co-stimulatory receptor,
whose ligands consist of B7-1 (CD80) and B7-2
(CD86) molecules expressed by APC. T cell anergy
develops when T cells encounter peptide MHC complexes without receiving adequate accessory or costimulatory signals [38]. T cells can also be rendered
anergic if they encounter peptide ligands for which
they have low afnity [3]. Certain APC, such as
macrophages [39] and tolerogenic dendritic cells that
may be immature or matured in a specic manner
[40] have the capacity to induce T cell anergy, in part
due to secretion of suppressive cytokines and lack of
adequate co-stimulation. Anergy is associated with
altered signalling and tyrosine phosphorylation patterns [38, 41]. T cell anergy can often [42], but not
always [16, 43], be overcome by providing exogenous
IL-2. Anergy has been associated with TCR downmodulation [44]. It should be borne in mind that
anergy is reversible under pro-inammatory conditions [45, 46], including the presence of infection, so
it is unlikely to be reliable as the sole long-term
tolerance mechanism.
Deletion has followed induction of an anergic state in
the continued presence of antigen in some, but not
all, models [47, 48]. In mice receiving BMT under
the cover of co-stimulatory blockade, peripheral
donor-reactive CD4 T cells are rendered anergic prior
to their deletion over a period of weeks [16]. Anergic
T cells may also down-regulate the activity of other T
cells, so that they function as regulatory T cells
(Treg), perhaps by conditioning APC such that they
tolerize T cells recognizing presenting the same or different antigens presented by these APC [49]. Moreover, Treg (see below) can promote the induction of
292
Review: Transplantation tolerance
T cell anergy [50] and may themselves have biochemical properties suggestive of an anergic state
[51].
B cell anergy
As many self-reactive B cells escape deletion during
development in the bone marrow, anergy is an
important tolerance mechanism. Many of these B
cells are anergic and die within the peripheral
lymphoid tissues when they encounter abundant but
low avidity antigens [30] (reviewed in Ref. [31]).
Similar to T cell anergy, B cell anergy requires
persistent antigen and is characterized by antigen
receptor downregulation [30], altered signalling patterns and increased apoptosis upon antigen encounter
[31]. T-cell tolerance and the consequent absence of
T cell help maintain B cell anergy. Anergic B cells
can nevertheless be activated in the presence of high
avidity antigen and T cell help [31]. Anergy is the
mechanism leading to early tolerance of natural antibody-producing B-1 cells in mice rendered mixed chimeric with bone marrow cells expressing an antigen
recognized by recipient natural antibody-producing
cells [3537].
Lymphocytes ignoring graft antigens (ignorance)
In some situations, antigens may simply be ignored
by T cells [44] or B cells [30] with receptors
recognizing them. This may occur when antigens
are presented by nonprofessional APC which are
unable to activate T cells, or when T cells fail to
migrate to the antigen-bearing tissue, as documented
in murine solid tumour models [52]. Several factors
appear to determine such T-cell behaviour, including
the level of antigen expression, how recently the
responding T cell has emerged from the thymus
[44], and the presence or absence of proinammatory cytokines [53] and co-stimulatory molecules
in peripheral tissues [54]. As might be easily
imagined, ignorance is a precarious state which
can be upset by additional immunological stimuli
provoked by inammation induced by infections
[55] or by presentation of antigen on professional
APCs [56].
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
Review: Transplantation tolerance
Active suppression of T-cell responses
It has become increasingly clear in recent years that
several mechanisms exist to down-modulate immune
responses once they are initiated, and that it is the
balance of activating and modulating functions that
determine the outcome of any response. Many mechanisms, including killing of APC by CTL, inhibitory
effects of cytokines, activation-induced cell death, etc.
contribute to this down-modulation of immune
responses. In addition, studies in the 1970s introduced
the concept that T cells themselves could actively suppress immune responses. Whilst certain T cell and
non-T cell populations were implicated in this suppression, it is only in the last decade or so that
molecular markers of suppressive T cells have been
identied and that suppressive cell populations
have been isolated, cultured in vitro and adoptively
transferred.
Regulatory CD4+ T cells
Suppressive CD25+CD4+ T cells have been strongly
implicated in the induction and maintenance of selftolerance [57, 58]. More recent studies have shown
that these cells are generated mainly in the thymus,
require specic positive selection (Fig. 1) [6] and
express FoxP3, a transcription factor that controls the
genetic programme associated with their suppressive
activity [59, 60]. These Treg may require an intermediate-afnity MHC peptide ligand (too low for negative selection) expressed on cortical epithelial cells of
the thymus for their survival and maturation [6]. Constitutively CD25+ Treg of this type have been termed
natural Treg [61]. In vitro suppression by these Treg
seems to require cell-to-cell contact [61]. Transforming growth factor-b (TGF-b) is a cytokine that has
been strongly implicated in the maintenance of Treg
and as a mediator of their suppressive activity [62
65]. Both CD4 and CD8 T cells are subject to
suppression by Treg, and memory as well as nave
responses have been shown to be suppressed. Several
reports indicate that the Treg require specic antigen
for their activation, but that the nal effector mechanism of suppression is nonantigen specic [6668].
Rechallenge with specic antigen induces c-interferon
(IFN-c) expression by Treg, which appears to be critical for their function [69]. Generation, expansion,
survival and possibly the function of Treg is highly
dependent on IL-2, which is not produced by the Treg
themselves [70].
Additional CD4+ T-cell populations with suppressive
function include FoxP3+ CD25+ cells that arise from
FoxP3-CD25- cells in the periphery following antigen-specic stimulation (adaptive Treg) [61], especially in the presence of TGF-b [71]. Additionally,
Tr1 regulatory cells are induced by chronic antigenic
stimulation in the presence of IL-10 and can suppress
autoimmune diseases in mice. These cells produce
high levels of IL-10 and low amounts of IL-2
(reviewed in Ref. [72]), and immature dendritic cells
can support their development in vitro [73]. Both natural Treg and Tr1 cells are hyporesponsive to TCRmediated stimulation but can be grown slowly in vitro
in the presence of certain cytokines, including IL-2.
The in vitro suppressive function of Tr1 is dependent
on IL-10 and TGF-b [72].
Transforming growth factor-b is clearly an important
cytokine for several suppressive populations. Besides
maintaining peripheral Treg populations and functions
[63, 65], TGF-b promotes adaptive Treg differentiation [74] and suppresses T-cell activation and Th1
differentiation through several Treg-independent
mechanisms [65, 75]. It can also modulate dendritic
cell function, rendering them tolerogenic for T cells
[40]. However, TGF-b has highly pleiotropic functions and cannot be viewed purely as an immunosuppressive cytokine. For example, TGF-b has been
implicated in chronic brotic conditions (e.g. chronic
graft-versus-host disease, GVHD; [76]) and in the differentiation of nave T cells to the pro-inammatory
IL-17-producing Th17 phenotype [77, 78].
Suppressive T cells have been implicated in numerous
experimental models leading to allograft tolerance
(reviewed in Refs. [79, 80]). Functional evidence for
specic suppressor cells was obtained in early models
of transplantation tolerance (reviewed in Ref. [81]) and
Hall et al. rst identied CD25+CD4+ T cells as a
specic suppressive population in rats receiving
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 293
M. Sykes
cardiac allografts with a short course of cyclosporin
[82]. Since then, Treg have been implicated in numerous models involving acceptance of vascularized
allografts in rodents receiving an initial immunosuppressive treatment. Regimens have included donorspecic cell infusions (termed DST), with [66] or
without [83, 84] co-stimulatory blockade [85, 86]
or partial T-cell depletion [87] or other combinations
of these. Treg promote the acceptance of MHCmatched, minor histocompatibility antigen-mismatched
skin grafts in mice receiving nondepleting anti-CD4
and CD8 antibodies with or without anti-CD154
[8890] or anti-CD154 and CD8 cell depletion
[91, 92]. Treg have been implicated in islet allograft
acceptance after treatment with CTLA4Ig [93].
Adaptive Treg [90, 94] have been implicated in some
of these studies.
The thymus plays an important role in several peripheral tolerance models, perhaps due to its role in generating Treg. For example, the thymus is needed for
tolerance induction in a porcine model involving a
short course of a high-dose calcineurin inhibitor in
combination with a renal allograft [95]. A similar phenomenon has been observed in rats receiving soluble
alloantigens in combination with a vascularized allograft [96] and active regulatory cell populations have
been described [96]. However, other mechanisms
involving recirculation of activated T cells to the thymus have also been implicated [97], and the circulation of peripheral dendritic cells to the thymus may
also play a role by promoting intrathymic deletion of
newly developing thymocytes [98] and possibly by
inducing positive selection of Treg.
There is considerable evidence for a role for natural
Treg in maintaining self-tolerance in humans. Congenital defects in FoxP3 in humans are associated
with an autoimmune syndrome, immune dysregulation, polyendocrinopathy, enteropathy, X-linked
(IPEX), that resembles its counterpart in mice (the
scurfy mutant) [99]. Defects in IL-2 signalling
through the STAT-5 transcription factor lead to similar
defects in Treg in mice and humans [100, 101].
These and the above experimental results have led to
considerable interest in the role of Treg in clinical
294
Review: Transplantation tolerance
transplantation, and correlative data have begun to
emerge. Chronic renal allograft rejection has been
associated with reduced circulating Treg concentrations [102]. Whilst discontinuation of immunosuppressive medications usually leads to rejection, a
small fraction of such patients accept their grafts nevertheless, i.e. they demonstrate spontaneous tolerance. These patients do not show increased circulating
Treg compared with controls [102]. Increased urinary
FoxP3 mRNA has been reported to predict improved
outcome of renal allograft rejection episodes [103].
The use of calcineurin inhibitors, but not rapamycin,
has been associated with reduced percentages of Treg
in blood of kidney allograft recipients [104].
Studies in mice have demonstrated the ability of Treg
to inhibit GVHD [105] and Treg have been implicated
in a mouse model in which GVHD has been inhibited
by pre-BMT exposure of donor T cells to recipient alloantigens in the presence of anti-CD40L [106]. In
humans, several studies have documented increased
[107, 108] or decreased [109] Treg concentrations in
association with chronic GVHD and decreased numbers in association with acute GVHD [110] in HCT
recipients, resulting in some confusion at the present
time. Treatment of severe autoimmune disease with
lymphoablative therapy followed by autologous HCT
has been associated with restoration of normal Treg
populations [111].
Other suppressive cell populations
In addition to the T cell populations discussed above,
other T-cell and non-T-cell suppressive cell populations can down-modulate immune responses. Fully
differentiated CD4+ helper T-(Th) cells may polarize
their cytokine secretion patterns to that of the Th1
subset, which secretes IL-2 and IFN-c, the Th17 subset that produces IL-17 [77] or the T-helper type 2
(Th2) subset that secretes IL-4 and IL-10 [112]. Th1
cells promote the generation of cytolytic CD8+ T
cells, whilst Th2 helps antibody responses but not
CTL responses [112]. A similar polarization of the
pattern of cytokine secretion occurs in CD8+ cytolytic
T cells [113]. In the early 1990s, there was considerable interest in the concept that polarization to Th2
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
Review: Transplantation tolerance
type of response from a pro-inammatory Th1 (IL-2and IFN-c-producing) response could promote allograft acceptance, and data associated Th2 responses
with such acceptance. However, only a few studies
directly demonstrated a role for Th2 cells in tolerance
induction and it is now clear that Th2 cells and their
cytokines can promote allograft rejection (reviewed in
Ref. [114]).
Natural killer (NK) T cells (T cells that express NK
cell-associated markers and may utilize an invariant
TCR-a chain) are another subset of T cells with regulatory activity, which may be mediated in part by
Th2-type cytokines [115]. NKT cells have recently
been shown to depend on TGF-b for their development [65, 75]. NKT cells are enriched in bone marrow and can suppress GVHD [116, 117], at least in
part via an IL-4-dependent mechanism [117]. Total
lymphoid irradiation (TLI) markedly enriches NKT
cells in the lymphoid tissues [118]. The markedly
reduced incidence of acute GVHD recently described
in patients receiving haematopoietic cell transplantation with a TLI-based regimen may be related to Th2
cytokine polarization induced by this population
[119].
A CD4)CD8) T cell population lacking NK cell
markers that suppresses skin graft rejection by CD8 T
cells with the same TCR has been described in a
mouse model [29], but the importance of this cell
population in other settings remains to be determined.
Human CD8+CD28) T cells have been reported to
suppress alloresponses and xenoresponses in vitro
[120], and recent studies have implicated CD8 cells
as regulatory cells in models of autoimmunity [121],
heart graft acceptance [122], skin grafting [123] and
GVHD [124126]. Natural [124] and adaptive
[126], FoxP3-expressing [122, 124], TGF-b-producing [127], and IL-10-producing [126] regulatory
CD8 cells have been described, and extensive data
are emerging on the role of these cells in various
models. One mechanism of immune down-modulation mediated by CD8 T cells is simply the killing
by alloreactive CTL of critical donor APC populations [128].
Some CD8+ CTL-mediated suppressive phenomena
might be attributable to veto activity of these cells.
Veto cells inactivate CTL recognizing antigens
expressed on the veto cell surface [28], resulting in
suppression of CTL responses to antigens shared by
the veto cells. CTL, various bone marrow cell subsets
and NK cells have been reported to have such activity. Veto cells may promote GVH tolerance, promote
allogeneic marrow engraftment and promote tolerance
induction with DST (reviewed in Refs [28, 81]). Veto
activity has been suggested to involve TGF-b [129].
Thus, whilst many types of Treg have been recently
described, much remains to be learned about the relative importance of each of these, their potential in
large animal models and the circumstances under
which they can be optimally generated. Several
groups are exploring the approach of expanding Treg
in vitro and then administering them in vivo to suppress alloimmunity or autoimmunity. Whilst methods
of nonspecically expanding mouse and human Treg
ex vivo have recently been developed [130, 131], animal studies suggest that antigen specicity is important for the achievement of effective suppression
following adoptive transfer [130]. As alloreactivity
includes many different donor antigens and donor
cells will not be available pretransplant for cadaveric
donor transplantation, this approach may be difcult
to apply.
Co-stimulatory blockade in transplantation
The discovery that TCR stimulation without co-stimulation can induce anergy [132] has led to intensive
evaluation of co-stimulatory blockade in the transplantation eld. Blockade of the CD28 co-stimulatory
pathway can be achieved with specic mAb or with a
soluble receptor for the B7-1 B7-2 ligands. CTLA4,
an alternate, inhibitory T receptor with a higher afnity than CD28 for these ligands, has been studied in
experimental models as a soluble CTLA4-Ig fusion
protein. Another pathway that has been targeted
recently involves the interaction between CD154 on
activated T cells with the CD40 receptor on APC.
This interaction plays an important role in
allowing APC to achieve full activating capacity by
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 295
M. Sykes
upregulating B7 molecules, MHC, antigen processing
pathways, cytokines and other molecules. Blockade of
the CD40CD154 pathway alone or in combination
with CTLA4-Ig can achieve marked prolongation of
fully MHC-mismatched skin graft survivals in some
mouse strain combinations. However, permanent tolerance of these grafts is not reliably achieved [133
136]. These treatments can more reliably induce permanent acceptance of tolerogenic rodent allografts
such as hearts [137]. Anergy of donor-reactive cells
and an important role for Treg have been implicated
in such models [138]. Rapamycin, a pharmacological
inhibitor of mammalian target of rapamycin, appears
to selectively allow expansion, activation and survival
of Treg whilst blocking proliferation of effector T
cells [139141]. This drug has been reported to
achieve robust allograft tolerance when used in combination with anti-CD154 [142] or with anti-IL-15
and a long-acting form of IL-2 [143].
The combination of DST and anti-CD154 leads to
long-term acceptance of several types of allografts
[144] and to prolongation of fully MHC-mismatched
skin graft survival, which can be permanent in
thymectomized mice [145]. Again, both anergy and a
role for Treg have been implicated [144, 145], as well
as peripheral CD8 cell deletion [26, 146]. However,
these mechanisms are apparently insufcient to prevent rejection of the skin graft by newly emerging alloreactive T cells in euthymic mice [147]. IFN-c,
which has traditionally been considered to be a Th1
proinammatory cytokine, has been shown to play an
important role in the tolerance achieved in this and
other models [145, 146, 148], possibly because of its
role in supporting Treg function [69]. DST with rapamycin and anti-CD154 has been reported to markedly
prolong islet allograft survival in nonhuman primates
[149]. The combination of anti-CD154, BMT and
DST allows the achievement of mixed chimerism and
robust tolerance; the durable chimerism ensures central deletion of donor-reactive T cells, preventing their
emergence from the thymus after the transplant [23,
150].
Despite the achievement of prolonged allograft survival (though not tolerance) in nonhuman primates
296
Review: Transplantation tolerance
[151154], attempts to apply co-stimulatory blockade
for the induction of tolerance clinically have not succeeded. The combination of rapamycin, DST and antiCD154 was reported to achieve tolerance in three of
ve nonhuman primate recipients [155]. However,
anti-CD154 use has been complicated by thromboembolic phenomena [156], resulting in termination of the
trials evaluating it. Anti-CD40 agents may have less
pro-thrombotic activity [157] and may be evaluated in
future trials. Whilst CTLA4Ig alone did not lead to
optimal renal allograft survival in nonhuman primates
[151], it is currently being evaluated as a calcineurin
inhibitor-sparing immunosuppressant in clinical trials
[158]. In another clinical trial, acute GVHD was
reduced in leukaemic patients receiving human leucocyte antigen (HLA)-mismatched bone marrow transplants that were exposed to recipient alloantigens ex
vivo in the presence of CD28 blockade with CTLA4Ig
[159]. Whilst several additional co-stimulatory pathways exist and combinations of blockers are showing
promise in rodent models, co-stimulatory blockade
alone has not yet proved to be sufciently powerful
to achieve tolerance in nonhuman primates or
humans.
Mixed chimerism as an approach to transplantation
tolerance
Bone marrow engraftment reliably induces tolerance
to the most immunogenic allografts, such as fully
MHC-mismatched skin and small bowel grafts, in animal models (reviewed in Ref. [160]). The ability to
achieve transplantation tolerance with HCT has been
well documented in patients who rst received HCT
with conventional myeloablative conditioning to treat
a haematological malignancy, and later accepted an
organ transplant from the same donor without chronic
immunosuppressive therapy (reviewed in Ref. [161]).
Haematopoietic cell administration in utero or neonatally, in immunologically immature hosts, has long
been known to be associated with transplantation
tolerance in animals (reviewed in Ref. [162]) and
durable chimerism and renal allograft tolerance have
recently been achieved in a porcine model involving
in utero transplantation of T-cell-depleted adult bone
marrow [163]. Both intrathymic and extrathymic
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
Review: Transplantation tolerance
mechanisms have been implicated in neonatally
induced tolerance [164, 165]. As prenatal diagnosis of
congenital diseases has become possible, injection of
allogeneic pluripotent haemopoietic stem cells to preimmune human fetuses has been used successfully to
correct immunodeciency diseases diagnosed in utero
[166168].
However, haematopoietic cell transplantation has not
yet been routinely applied for the intentional induction
of allograft tolerance in humans. The early protocols
that achieved tolerance in adult rodents involved lethal
total-body irradiation (TBI) as conditioning for marrow
engraftment. Removal of mature donor T cells before
transplantation was shown to reliably prevent GVHD
[169]. However, MHC-mismatched allogeneic HCT in
larger animals, including humans, has proved to be less
successful and more dangerous than in rodents because
of several factors, including the toxicity associated with
myeloablative conditioning and the inordinately high
risks of GVHD and engraftment failure (reviewed in
[170]). The incidence of marrow rejection is increased
when donor marrow is T-cell-depleted to prevent
GVHD. Even when MHC mismatching is avoided,
GVHD still aficts approximately 50% of patients who
undergo HLA-identical sibling HCT, even with posttransplant immunosuppressive pharmacotherapy and
reduced-intensity conditioning [171]. Although it is a
major cause of morbidity and mortality, this GVHD
risk is acceptable in individuals with malignant disease
because it is associated with benecial graft-versustumour responses [172]. However, the severe, opposing
risks of GVHD and graft failure have precluded the
routine performance of extensively HLA-mismatched
transplantation, so that many patients with no other
curative options are not transplanted because they lack
an appropriately matched donor. The risks of GVHD
and marrow aplasia caused by graft rejection would be
completely unacceptable in a patients receiving HCT
solely for the purpose of organ allograft tolerance
induction. Therefore, the development of more specic
and effective methods of overcoming the barriers to
marrow engraftment with minimal GVHD risk will be
essential before this approach can be routinely applied
to tolerance induction in patients needing organ
transplantation.
For the purpose of allograft tolerance induction,
achievement of a state of mixed, rather than full,
donor haematopoietic chimerism would be desirable.
Mixed chimerism means that donor and host elements
both contribute to haematopoietic repopulation at
readily detectable levels. Mixed chimerism can be
achieved with less toxic (nonmyeloablative) conditioning regimens than those that lead to full donor
chimerism. In addition to their reduced toxic side
effects, nonmyeloablative regimens allow recovery of
host haematopoiesis, so that life-threatening marrow
failure does not occur if donor marrow is rejected.
Furthermore, improved immunocompetence has been
observed in murine mixed compared to full allogeneic
chimeras when full MHC barriers are crossed. Peripheral reconstitution of mixed, but not full chimeras,
includes host-type APC, allowing optimal antigen
presentation to T cells that have developed in the host
thymus, and which therefore preferentially recognize
peptide antigens presented by host-type MHC molecules [173, 174]. Anti-viral CTL responses in mixed
chimeras showed exquisite specicity for recipientderived MHC restricting elements [175]. Additionally,
whilst nonhaematopoietic thymic stromal cells have
some capacity to induce deletional tolerance, host
haematopoietic cells present in mixed but not full chimeras most reliably assure the intrathymic deletion of
host-reactive cells [10, 12].
As discussed in the section on intrathymic clonal
deletion as a mechanism of transplantation tolerance,
a nonmyeloablative conditioning approach consisting
of low dose (3 Gy) TBI, T-cell-depleting mAbs and
thymic irradiation [8] reliably achieves a state of
mixed chimerism in which intrathymic deletion is the
major mechanism maintaining donor-specic tolerance
[10, 11]. T-cell alloreactivity preexisting in both the
thymus and periphery must be eliminated in order to
permit allogeneic stem cell engraftment and early
seeding of the thymus with allogeneic APC. Intrathymic alloreactivity can be eliminated using thymic
irradiation [8], high doses of T-cell-depleting antibodies [176], or co-stimulatory blockers such as antiCD154 or CTLA4Ig [20, 177]. Whilst it is unclear
whether the requirement to overcome intrathymic alloreactivity would apply to older humans with involuted
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 297
M. Sykes
thymi, the adult thymus remains functional at a low
level even with advanced age [178] and large animal
studies have shown the necessity for thymic irradiation in a mixed chimerism model [179]. Elimination
of peripheral anti-donor T-cell alloreactivity can be
achieved with global T-cell depletion with mAbs [8,
9] or with co-stimulatory blockers combined with
BMT [20]. Low-dose TBI or busulfan creates an
environment that facilitates engraftment and expansion of donor haematopoietic stem cells [176, 180].
Whilst the need for even mildly myelosuppressive
treatments can be avoided by administering very high
marrow doses [9, 21, 181], administration of such
high stem cell numbers is not currently clinically
feasible.
Durable, multilineage mixed chimerism leads to systemic tolerance, as specic unresponsiveness to the
donor is observed in in vitro assays of alloreactivity.
Moreover, donor skin grafts, which provide the most
stringent test of tolerance, are specically accepted at
any time post-BMT [8, 21, 177, 182].
Many regimens achieving mixed chimerism and tolerance have been described in rodents [183188], including the use of TLI plus BMT [189], which has been
evaluated in humans without success [190, 191]. Various combinations of anti-T-cell antibodies, irradiation
and immunosuppressive drugs have been used successfully in large animal models [192195]. However,
the mechanisms of tolerance in these models are more
complex than simple central deletion, as complete
depletion of recipient T cells has not been achieved
prior to BMT.
The ability to replace recipient T-cell depletion with
co-stimulatory blockade in murine models is important because of the difculty in using antibodies to
achieve T-cell depletion in large animals and humans.
Secondly, if truly exhaustive T-cell depletion could be
achieved in humans, T-cell recovery from the thymus
might be dangerously slow, especially in older individuals (reviewed in Ref. [178]). Replacement of
some [182] or all [20, 21, 181] T-cell-depleting antibodies with co-stimulatory blockade is therefore an
important advance.
298
Review: Transplantation tolerance
Graft-versus-host disease does not occur in the animal
models of mixed chimerism discussed above, despite
the use of unmodied donor bone marrow cells. This
is due in part to the continued presence of the T-celldepleting or co-stimulatory blocking antibodies in the
circulation of the recipients at the time of BMT [196].
These antibody levels readily prevent alloreactivity by
the relatively small number of mature T cells in the
donor marrow.
The role of intrathymic clonal deletion in the induction and maintenance of tolerance in mixed allogeneic
chimeras was discussed in an earlier section. It was
also pointed out that, in animals in which the preexisting alloreactive T-cell repertoire is not depleted
with mAb but is tolerized by the combination of
BMT and co-stimulatory blockade, specic peripheral
deletion of donor-reactive CD8 [22, 23] and CD4 [16,
20, 24, 25] cells is observed. The mechanisms of
deletion of the two subsets appear to be different in
this model. Peripheral donor-reactive CD8 cells are
deleted rapidly, whereas deletion of donor-reactive
CD4 cells occurs more slowly, over 45 weeks. Deletion is preceded by anergy towards donor antigens
[16, 197]. It is interesting that the peripheral tolerance
of CD8 cells in this model is dependent on the presence of CD4 cells, but only in the rst 10 days, after
which the donor-reactive CD8 cell deletion is complete. The CD4 cells that rapidly tolerize the CD8
population do not appear to be typical CD25+ natural Treg [22]. Regulatory cells do not appear to play
a signicant role in maintaining the long-term tolerance induced by anti-CD154 and low-dose TBI with
BMT, as tolerance and chimerism are obliterated by
the infusion of relatively small numbers of nontolerant
recipient-type spleen cells in this model and linked
suppression is not observed [16, 22], presumably
because the deletion of donor-reactive T cells is so
complete. We have speculated that the persistence of
donor-reactive T cells is required to expand and maintain a regulatory-response specic for that donor
[198]. If the donor-reactive cells are completely deleted, no suppressive reaction is maintained. In other
models of BMT with co-stimulatory blockade, on the
other hand, typical natural Treg appear to be involved
in the induction [199] and maintenance [200] of
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
Review: Transplantation tolerance
tolerance, possibly reecting less rapid or complete
peripheral deletion of donor-reactive T cells.
Bone marrow
conditioning
transplantation
without
host
In the regimens inducing mixed chimerism discussed
above, specic host conditioning is used to overcome
the immunological and physiological barriers to donor
marrow engraftment. Microchimerism, meaning chimerism at low levels that requires sensitive techniques
such as polymerase chain reaction to be detected, may
be detectable for many years in patients receiving
solid organ allografts without haematopoietic cell
transplantation [201]. Microchimerism should be distinguished from the mixed chimerism discussed
above, in which multilineage chimerism is readily
measurable by ow cytometry. The signicance of the
detection of spontaneous microchimerism is unclear
[202]. Lasting microchimerism is clearly not required
for tolerance in all models [203205], and tolerance is
by no means assured in the presence of microchimerism [206]. Microchimerism neither denotes a state of
tolerance nor is required to maintain an allograft
under all circumstances [202, 206208].
Based on the reports of microchimerism in organ allograft recipients, several groups have intentionally
boosted microchimerism in patients receiving conventional immunosuppression by infusing donor bone
marrow cells along with solid organ transplantation
[209, 210]. Whilst no clear-cut reduction in rejection
or immunosuppressive medication doses have been
observed in these trials [211213], ongoing studies
combining marrow infusion with T-cell-depleting antibodies should provide further information on the
potential of this approach.
Approaches to xenogeneic tolerance
Mixed chimerism
Mixed xenogeneic chimerism can also be achieved
with nonmyeloablative conditioning, leading to T-cell
tolerance [214]. However, innate immune barriers
posed by NK and cd T cells must be overcome to
achieve rat marrow engraftment in mice [215],
whereas these cells do not pose major barriers to engraftment of allogeneic haematopoietic cells given in
sufcient numbers with adequate T-cell immunosuppression [216]. Once mixed chimerism is achieved
in the rat to mouse species combination, both T cell
and B-cell tolerance is observed [214, 217220].
Pigs are considered to be the most suitable xenogeneic
donor species for transplantation to humans, but
progress in this area has been impeded due to the
presence in human sera of natural antibodies (Nab) that
cause hyperacute rejection of porcine vascularized
xenografts. The major specicity recognized by these
Nab is a ubiquitous carbohydrate epitope, Gala13Galb1-4GlcNAc-R (aGal). GalT knockout mice have
a targeted mutation of the a1-3Gal transferase (GalT)
enzyme and, like humans, produce anti-aGal Nab.
Both preexisting and newly developing B cells producing anti-aGal antibodies are tolerized by the induction
of mixed chimerism in GalT knockout mice receiving
aGal-expressing allogeneic or xenogeneic marrow [35,
36, 221]. The induction of mixed xenogeneic chimerism thereby prevents hyperacute rejection, a delayed
antibody-mediated form of rejection termed acute
vascular rejection, as well as cell-mediated rejection of
primarily vascularized cardiac xenografts [219]. AntiGal-producing cells are tolerized [35, 219] by an early
anergy mechanism and later by clonal deletion and or
receptor editing [37].
Mixed allogeneic chimeras [222] and mixed xenogeneic chimeras [223] also show tolerization of NK
cells towards the donor. Despite this T, B and NK cell
tolerance, the levels of xenogeneic donor chimerism
decline gradually over time in mixed xenogeneic chimeras, due to a competitive advantage of recipient
mouse marrow over xenogeneic rat marrow [224].
Species specicity or selectivity of haematopoietic
cytokines, adhesion molecules, etc. [225] probably
account for this advantage. Achievement of xenogeneic haematopoiesis is an even more formidable
challenge in highly disparate (discordant) species
combinations. Genetic engineering approaches [226
228] can overcome these barriers, and have allowed
the demonstration, using a humanized mouse model,
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 299
M. Sykes
that human T cells can be centrally tolerized to porcine xeonantigens via induction of mixed xenogeneic
chimerism [229].
Macrophages also impose an innate immune barrier to
the engraftment of xenogeneic marrow from highly
disparate species [230, 231] that may be overcome by
a genetic engineering approach to ensure adequate
inhibition of recipient macrophage activation by ligands on xenogeneic donor haematopoietic cells [232].
Review: Transplantation tolerance
the aGal knockout pig is a major advance in overcoming the obstacles posed by anti-aGal Nab [243,
245247], antibodies of other, less dominant specicities are a signicant obstacle to success using aGal
knockout pigs as organ source animals to nonhuman
primates [248]. The ability of mixed chimerism to
tolerize Nab-producing cells of all specicities [217]
may therefore be an important advantage of this
approach.
Current clinical trials of tolerance induction
Xenogeneic thymic transplantation
Xenogeneic T-cell tolerance can also be achieved
across highly disparate species barriers by replacing
the recipient thymus with a xenogeneic donor thymus
after host T-cell depletion and thymectomy [233
235]. Tolerance to both the donor and the host
develops at least in part by intrathymic deletional
mechanisms [235, 236]. Adequate immune function is
achieved [234], even though positive selection in such
grafts is mediated only by porcine thymic MHC, with
no inuence of mouse MHC [237, 238]. Likewise,
excellent immune function is achieved in humans
receiving HLA-mismatched allogeneic thymic transplantation for the treatment of congenital thymic aplasia (DiGeorge syndrome) [239, 240], suggesting that
restriction incompatibility resulting from MHC
disparity between the positive selecting epithelial cells
in the thymus and the APC in the periphery need not
be a major obstacle to the achievement of adequate
immune function. Signicantly, human T cells have
been shown to be tolerant of porcine donor antigens
when they develop in xenogeneic porcine thymus
grafts [241]. This approach has been applied and
demonstrated promise in pig-to-primate xenograft
models [242, 243]. In addition to intrathymic deletion,
regulatory cells probably play a role in the donorspecic tolerance achieved with xenogeneic thymic
transplantation [244]. Ultimately, success in clinical
xenotransplantation may require a combination
approach involving thymic transplantation to tolerize
residual host T cells and newly developing T cells,
and haematopoietic cell transplantation to tolerize the
innate immune system, including Nab-producing B
cells and NK cells. Whilst the recent development of
300
Most transplant clinicians have treated patients who
have chosen to withdraw their immunosuppressive
therapy. Whilst the majority of such patients reject
their allograft, occasional patients do not [248, 249],
demonstrating that tolerance can be achieved in
humans. Extensive efforts are underway in many centres to identify markers that would distinguish such
tolerant patients prospectively before withdrawal of
immunosuppression, but so far none have proved to
be reliable [102, 250]. Whilst studies are ongoing to
evaluate the ability to slowly withdraw immmunosuppression in cohorts of liver transplant recipients, it
is not yet clear whether this approach will successfully achieve tolerance. The only approach that has
been successfully used for the intentional induction
of tolerance in humans is the nonmyeloablative
induction of mixed haematopoietic chimerism. These
studies, which have so far involved only small
numbers of patients, were justied by a combination
of clinical data in patients with haematopoietic
malignancies and extensive animal data discussed
above, including the achievement of tolerance in
nonhuman primates receiving combined MHC-mismatched kidney and BMT with nonmyeloablative
conditioning [192].
At the Massachusetts General Hospital, we have
recently developed clinical regimens aimed at achieving mixed chimerism with nonmyeloablative conditioning in patients with haematological malignancies
[251]. This approach is based on observations in mice
that lymphohaematopoietic GVH reactions induced by
donor lymphocyte infusions given to established
mixed chimeras can mediate GVL without causing
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
Review: Transplantation tolerance
GVHD [252255]. GVH-reactive donor T cells that
are activated and expand in the lymphoid tissues do
not trafc to the GVHD target tissues (which include
skin, liver and gut) because of the absence of inammation in these tissues in established mixed chimeras
[256]. Attempts to replicate this approach in patients
with haematological malignancies demonstrated the
safety and anti-tumour potential of this approach
[251], providing an opportunity to evaluate the ability
of nonmyeloablative HCT to achieve transplantation
tolerance in patients with renal failure resulting from
multiple myeloma. Six patients have received a simultaneous nonmyeloablative bone marrow transplant and
renal allograft from HLA-identical sibling donors. All
six patients have accepted their kidney grafts, four
without any immunosuppression for periods of more
than 28 years. One of these four patients required
transient immunosuppression for a rejection episode
after initial immunosuppression withdrawal and
another patient later received a myeloablative transplant from the same donor to treat myeloma progression, and requires immunosuppression to treat GVHD.
The two patients who were not withdrawn from immunosuppression required these drugs for the treatment of GVHD, not for graft rejection. Similar to the
nonhuman primate model described above, in which
BMT plays an important role in inducing tolerance to
a simultaneously transplanted kidney from the same
donor [179], chimerism in these tolerant patients was
only transient [257]. Whilst not well understood, it is
possible that the kidney graft itself may participate in
tolerance induction and or maintenance after chimerism has played its initial role. In vitro studies performed in these patients suggest that tolerance may be
specic for donor antigens expressed by the kidney,
whilst responses to antigens expressed on haematopoietic cells but not the kidney may even be sensitized
[257]. Data also suggest a possible role for regulatory
cells other than Treg in maintaining tolerance [257].
The promising results obtained in these patients have
provided an important demonstration that tolerance
can be intentionally induced in humans. A second trial
has now been initiated at the Massachusetts General
Hospital for the transplantation of HLA-mismatched
haploidentical bone marrow and kidney grafts in
patients without malignancy. These patients receive a
regimen that was previously shown in patients with
malignancies to result in transient mixed chimerism
without GVHD [258]. With this critical safety parameter established and the above data showing that tolerance could be achieved with transient chimerism in
recipients of combined kidney and bone marrow
transplants in the HLA-identical setting, it was
deemed appropriate to evaluate this approach using
these extensively HLA-mismatched donors. Whilst
still in progress, the early results of this study are
promising.
Conclusions
As short-term results of most allogeneic organ transplants are excellent, it is this authors view that
several criteria should be met before new strategies
are justiably evaluated to replace chronic immunosuppressive therapy: (i) Studies in rodents should
demonstrate robust tolerance in multiple strain combinations using extensively histoincompatible, highly
immunogenic grafts such as skin. Most of the studies
implicating Treg in tolerance induction in rodent models utilize highly tolerogenic, poorly immunogenic
grafts such as primarily vascularized hearts. Whilst
treatment with a short course of many different
immunosuppressive agents allows the inherent Treginducing capacity to result in tolerance to such grafts,
cardiac allografts are far less tolerogenic in large
animals; (ii) Efcacy must be demonstrated in large
animal models; (iii) Acceptable toxicity must be
demonstrated in large animal models.
As discussed above, Treg and co-stimulatory blockade
have attracted considerable interest for the induction
of tolerance. However, large animal studies have not
yet been reported in which Treg have been administered or clearly shown to achieve transplantation tolerance. In fact, protocols that have led to marked graft
prolongation and have been associated with the development of Treg in rodent models have not led to
transplantation tolerance in large animals [151, 154].
The same is true of co-stimulatory blockade. Thus,
further understanding and the development of practical and effective approaches for tolerance induction
will be needed before these strategies can be attemp-
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 301
M. Sykes
ted clinically. Several recent attempts at tolerance
induction in humans, for which all three criteria above
had not been met, have failed [191, 259, 260].
In the case of HCT for the induction of tolerance, the
three criteria described above have all been met, permitting the evaluation of this approach in pilot clinical
trials. When complete removal of immunosuppression
can be successfully achieved, the increased short-term
toxicity associated with the conditioning for HCT
may be acceptable. Nevertheless, it will be important
to proceed cautiously to allow adequate assessment of
the risk : benet ratio of this tolerance strategy over
time.
Conflict of interest statement
No conict of interest was declared.
Acknowledgements
I thank Dr Nina Tolkoff-Rubin and Dr Thomas R.
Spitzer for helpful review of the manuscript and Ms
Kelly Walsh for expert assistance with its preparation.
This work was supported by NIH grants RO1
HL49915, RO1 CA79989, PO1 P01 AI39755, PO1
HL018646, PO1 CA11159, the Immune Tolerance
Network, the Juvenile Diabetes Research Foundation
and the Multiple Myeloma Foundation.
References
1 Wekerle T, Sykes M. Mixed chimerism as an approach for the
induction of transplantation tolerance. Transplantation 1999;
68: 45967.
2 Allen PM. Peptides in positive and negative selection: a delicate balance. Cell 1994; 76: 5936.
3 Alam SM, Travers PJ, Wung JL et al. T-cell-receptor afnity
and thymocyte positive selection. Nature 1996; 381: 61620.
4 Ramsdell F, Fowlkes BJ. Clonal deletion versus clonal anergy:
the role of the thymus in inducing self tolerance. Science
1990; 248: 13428.
5 Inaba M, Inaba K, Hosono M et al. Distinct mechanisms of
neonatal tolerance induced by dendritic cells and thymic B
cells. J Exp Med 1991; 173: 54959.
6 Baldwin TA, Hogquist KA, Jameson SC. The fourth way?
Harnessing aggressive tendencies in the thymus. J Immunol
2004; 173: 651520.
302
Review: Transplantation tolerance
7 Brocker T, Riedinger M, Karjalainen K. Targeted expression
of major histocompatibiity complex (MHC) class II molecules
demonstrates that dendritic cells can induce negative but not
positive selection of thymocytes in vivo. J Exp Med 1997;
185: 54150.
8 Sharabi Y, Sachs DH. Mixed chimerism and permanent specic transplantation tolerance induced by a non-lethal preparative regimen. J Exp Med 1989; 169: 493502.
9 Sykes M, Szot GL, Swenson K, Pearson DA. Induction of
high levels of allogeneic hematopoietic reconstitution and
donor-specic tolerance without myelosuppressive conditioning. Nat Med 1997; 3: 7837.
10 Tomita Y, Khan A, Sykes M. Role of intrathymic clonal deletion and peripheral anergy in transplantation tolerance induced
by bone marrow transplantation in mice conditioned with a
non-myeloablative regimen. J Immunol 1994; 153: 108798.
11 Khan A, Tomita Y, Sykes M. Thymic dependence of loss of
tolerance in mixed allogeneic bone marrow chimeras after
depletion of donor antigen. Peripheral mechanisms do not contribute to maintenance of tolerance. Transplantation 1996; 62:
3807.
12 Manilay JO, Pearson DA, Sergio JJ, Swenson KG, Sykes M.
Intrathymic deletion of alloreactive T cells in mixed bone marrow chimeras prepared with a nonmyeloablative conditioning
regimen. Transplantation 1998; 66: 96102.
13 Morecki S, Leshem B, Eid A, Slavin S. Alloantigen persistence in induction and maintenance of transplantation tolerance.
J Exp Med 1987; 165: 146880.
14 Rocha B, Tanchot C, Von Boehmer H. Clonal anergy blocks
in vivo growth of mature T cells and can be reversed in the
absence of antigen. J Exp Med 1993; 177: 151721.
15 Pape KA, Merica R, Mondino A, Khoruts A, Jenkins MK.
Direct evidence that functionally impaired CD4+ T cells persist
in vivo following induction of peripheral tolerance. J Immunol
1998; 160: 471929.
16 Kurtz J, Shaffer J, Anosova N, Benichou G, Sykes M. Mechanisms of early peripheral CD4 T cell tolerance induction by
anti-CD154 monoclonal antibody and allogeneic bone marrow
transplantation: evidence for anergy and deletion, but not
regulatory cells. Blood 2004; 103: 433643.
17 Ferber I, Schonrich G, Schenkel J, Mellor AL, Hammerling
GJ, Arnold B. Levels of peripheral T cell tolerance induced by
different doses of tolerogen. Science 1994; 263: 6746.
18 Heath WR, Kurts C, Miller JFAP, Carbone FR. Cross-tolerance: a pathway for inducing tolerance to peripheral tissue
antigens. J Exp Med 1998; 187: 154953.
19 Moskophidis D, Lechner F, Pircher H, Zinkernagel RM. Virus
persistence in acutely infected immunocompetent mice by
exhaustion of antiviral cytotoxic effector T cells. Nature 1993;
362: 75861.
20 Wekerle T, Sayegh MH, Hill J et al. Extrathymic T cell
deletion and allogeneic stem cell engraftment induced with
costimulatory blockade is followed by central T cell tolerance.
J Exp Med 1998; 187: 203744.
21 Wekerle T, Kurtz J, Ito H et al. Allogeneic bone marrow transplantation with costimulatory blockade induces macrochimerism
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
Review: Transplantation tolerance
and tolerance without cytoreductive host treatment. Nat Med
2000; 6: 4649.
Fehr T, Takeuchi Y, Kurtz J, Sykes M. Rapid peripheral deletion of donor-reactive CD8 T cells permits mixed allogeneic
chimerism and tolerance induction in recipients of anti-CD40
L and allogeneic BMT: short-term regulation by CD4 cells.
Eur J Immunol 2005; 35: 267990.
Seung E, Mordes JP, Rossini AA, Greiner DL. Hematopoietic
chimerism and central tolerance created by peripheral-tolerance
induction without myeloablative conditioning. J Clin Invest
2003; 112: 795808.
Wekerle T, Sayegh MH, Chandraker A, Swenson KG, Zhao Y,
Sykes M. Role of peripheral clonal deletion in tolerance induction with bone marrow transplantation and costimulatory
blockade. Transplant Proc 1999; 31: 680.
Wekerle T, Kurtz J, Sayegh MH et al. Peripheral deletion after
bone marrow transplantation with costimulatory blockade has
features of both activation-induced cell death and passive cell
death. J Immunol 2001; 166: 23116.
Iwakoshi NN, Markees TG, Turgeon N et al. Skin allograft
maintenance in a new synchimeric model system of tolerance.
J Immunol 2001; 167: 662330.
Wells AD, Li XC, Li Y et al. Requirement for T cell apoptosis
in the induction of peripheral transplantation tolerance.
Nat Med 1999; 5: 130312.
Miller RG. The veto phenomenon and T-cell regulation. Immunol Today 1986; 7: 1124.
Zhang ZX, Yang L, Young KJ, DuTemple B, Zhang L. Identication of a previously unknown antigen-specic regulatory
T cell and its mechanism of suppression. Nat Med 2000; 6:
7829.
Goodnow CC. Transgenic mice and analysis of B-cell tolerance. Annu Rev Immunol 1992; 10: 489518.
Ferry H, Leung JC, Lewis G et al. B-cell tolerance. Transplantation 2006; 81: 30815.
Ohdan H, Swenson KG, Kruger-Gray HW et al. Mac-1negative B-1b phenotype of natural antibody-producing cells,
including those responding to Gala1,3Gal epitopes in a1,3galactosyltransferase decient mice. J Immunol 2000; 165:
551829.
Xu Y, Yang YG, Ohdan H et al. Characterization of anti-Gal
antibody-producing cells of baboons and humans. Transplantation 2006; 81: 9408.
Murakami M, Tsubata T, Okamoto M et al. Antigen-induced
apoptotic death of Ly-1 B cells responsible for autoimmune
disease in transgenic mice. Nature 1992; 357: 7780.
Ohdan H, Yang Y-G, Shimizu A, Swenson KG, Sykes M.
Mixed bone marrow chimerism induced without lethal
conditioning prevents T cell and anti-Gala1,3Gal antibodymediated heart graft rejection. J Clin Invest 1999; 104:
28190.
Ohdan H, Swenson KG, Kitamura H, Yang Y-G, Sykes M.
Tolerization of Gala1,3Gal-reactive B cells in presensitized
a1,3-galactosyltransferase-decient mice by nonmyeloablative
induction of mixed chimerism. Xenotransplantation 2002; 8:
22738.
37 Kawahara T, Shimizu I, Ohdan H, Zhao G, Sykes M. Differing
mechanisms of early and late B-cell tolerance induced by
mixed chimerism. Am J Transplant 2005; 5: 28219.
38 Appleman LJ, Boussiotis VA. T cell anergy and costimulation.
Immunol Rev 2003; 192: 16180.
39 Hoves S, Krause SW, Schutz C et al. Monocyte-derived
human macrophages mediate anergy in allogeneic T cells and
induce regulatory T cells. J Immunol 2006; 177: 26918.
40 Rutella S, Danese S, Leone G. Tolerogenic dendritic cells:
cytokine modulation comes of age. Blood 2006; 108: 143540.
41 Chiodetti L, Choi S, Barber DL, Schwartz RH. Adaptive tolerance and clonal anergy are distinct biochemical states. J Immunol 2006; 176: 227991.
42 Schwartz RH. Costimulation of T lymphocytes: the role of
CD28, CTLA-4, and B7 BB1 in interleukin-2 production and
immunotherapy. Cell 1992; 71: 10658.
43 Wells AD, Walsh MC, Bluestone JA, Turka LA. Signaling
through CD28 and CTLA-4 controls two distinct forms of T
cell anergy. J Clin Invest 2001; 108: 895903.
44 Arnold B, Schonrich G, Hammerling GJ. Multiple levels of
peripheral tolerance. Immunol Today 1993; 14: 124.
45 Knoechel B, Lohr J, Zhu S et al. Functional and molecular
comparison of anergic and regulatory T lymphocytes. J Immunol 2006; 176: 647383.
46 Rocken M, Urban JF, Shevach EM. Infection breaks T cell
tolerance. Nature 1992; 359: 7982.
47 Rocha B, Grandien A, Freitas AA. Anergy and exhaustion are
independent mechanisms of peripheral T cell tolerance. J Exp
Med 1995; 181: 9931003.
48 Chai J-G, Bartok I, Scott D, Dyson J, Lechler R. T:T antigen
presentation by activated murine CD8+ T cells induces anergy
and apoptosis. J Immunol 1998; 160: 365565.
49 Vendetti S, Chai JG, Dyson J, Simpson E, Lombardi G,
Lechler R. Anergic T cells inhibit the antigen-presenting function of dendritic cells. J Immunol 2000; 165: 117581.
50 Vanasek TL, Nandiwada SL, Jenkins MK, Mueller DL.
CD25+Foxp3+ regulatory T cells facilitate CD4+ T cell clonal
anergy induction during the recovery from lymphopenia.
J Immunol 2006; 176: 58809.
51 Li L, Godfrey WR, Porter SB et al. CD4+CD25+ regulatory
T-cell lines from human cord blood have functional and
molecular properties of T-cell anergy. Blood 2005; 106: 3068
73.
52 Ochsenbein AF, Sierro S, Odermatt B et al. Roles of tumour
localization, second signals and cross priming in cytotoxic
T-cell induction. Nature 2001; 411: 105864.
53 Heath WR, Allison J, Hoffman MW et al. Autoimmune diabetes as a consequence of locally produced interleukin-2. Nature
1992; 359: 5479.
54 von Herrath MG, Guerder S, Lewicki H, Flavell RA, Oldstone
MBA. Coexpression of B7-1 and viral (self) transgenes in
pancreatic b cells can break peripheral ignorance and lead to
spontaneous autoimmune diabetes. Immunity 1995; 3: 72738.
55 Ohashi PS, Oehen S, Buerki K et al. Ablation of tolerance
and induction of diabetes by virus infection in viral antigen
transgenic mice. Cell 1991; 65: 30517.
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 303
M. Sykes
56 Lafferty KJ, Babcock SK, Gill RG. Prevention of rejection by
treatment of the graft: an overview. Prog Clin Biol Res 1986;
224: 87117.
57 Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. Immunologic self-tolerance maintained by activated T cells expressing
IL-2 receptor a-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases.
J Immunol 1995; 155: 115164.
58 Groux H, Powrie F. Regulatory T cells and inammatory
bowel disease. Immunol Today 1999; 20: 4426.
59 Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell
development by the transcription factor foxp3. Science 2003;
299: 105761.
60 Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the
development and function of CD4(+)CD25(+) regulatory T
cells. Nat Immunol 2003; 4: 3306.
61 OGarra A, Vieira P. Regulatory T cells and mechanisms of
immune system control. Nat Med 2004; 10: 8015.
62 Huber S, Schramm C, Lehr HA et al. Cutting edge: TGF-beta
signaling is required for the in vivo expansion and immunosuppressive capacity of regulatory CD4+CD25+ T cells.
J Immunol 2004; 173: 652631.
63 Marie JC, Letterio JJ, Gavin M, Rudensky AY. TGF-{beta}1
maintains suppressor function and Foxp3 expression in
CD4+CD25+ regulatory T cells. J Exp Med 2005; 201: 10617.
64 Huang X, Zhu J, Yang Y. Protection against autoimmunity in
nonlymphopenic hosts by CD4+ CD25+ regulatory T cells is
antigen-specic and requires IL-10 and TGF-beta. J Immunol
2005; 175: 428391.
65 Li MO, Sanjabi S, Flavell RA. Transforming growth factorbeta controls development, homeostasis, and tolerance of T
Cells by regulatory T cell-dependent and -independent mechanisms. Immunity 2006; 25: 45571.
66 Hara M, Kingsley CI, Niimi M et al. IL-10 is required for regulatory T cells to mediate tolerance to alloantigens in vivo.
J Immunol 2001; 166: 378996.
67 Thornton AM, Shevach EM. Suppressor effector function of
CD4+CD25+ immunoregulatory T cells is antigen nonspecic.
J Immunol 2000; 164: 18390.
68 Karim M, Feng G, Wood KJ, Bushell AR. CD25+CD4+ regulatory T cells generated by exposure to a model protein antigen
prevent allograft rejection: antigen-specic reactivation in vivo
is critical for bystander regulation. Blood 2005; 105: 48717.
69 Sawitzki B, Kingsley CI, Oliveira V, Karim M, Herber M,
Wood KJ. IFN-{gamma} production by alloantigen-reactive
regulatory T cells is important for their regulatory function in
vivo. J Exp Med 2005; 201: 192535.
70 Malek TR, Bayer AL. Tolerance, not immunity, crucially
depends on IL-2. Nat Rev Immunol 2004; 4: 66574.
71 Fu S, Zhang N, Yopp AC et al. TGF-beta induces Foxp3 +
T-regulatory cells from CD4 + CD25- precursors. Am J Transplant 2004; 4: 161427.
72 Roncarolo MG, Gregori S, Battaglia M, Bacchetta R, Fleischhauer K, Levings MK. Interleukin-10-secreting type 1 regulatory T cells in rodents and humans. Immunol Rev 2006;
212: 2850.
304
Review: Transplantation tolerance
73 Jonuleit H, Schmitt E, Schuler G, Knop J, Enk AH. Induction
of interleukin 10-producing, nonproliferating CD4(+) T cells
with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J Exp Med 2000; 192:
121322.
74 Zheng SG, Wang JH, Stohl W, Kim KS, Gray JD, Horwitz
DA. TGF-beta requires CTLA-4 early after T cell activation to
induce FoxP3 and generate adaptive CD4+CD25+ regulatory
cells. J Immunol 2006; 176: 33219.
75 Marie JC, Liggitt D, Rudensky AY. Cellular mechanisms of
fatal early-onset autoimmunity in mice with the T cell-specic
targeting of transforming growth factor-beta receptor. Immunity
2006; 25: 44154.
76 Banovic T, MacDonald KP, Morris ES et al. TGF-beta in allogeneic stem cell transplantation: friend or foe? Blood 2005;
106: 220614.
77 Bettelli E, Carrier Y, Gao W et al. Reciprocal developmental
pathways for the generation of pathogenic effector TH17 and
regulatory T cells. Nature 2006; 441: 2358.
78 Mangan PR, Harrington LE, OQuinn DB et al. Transforming
growth factor-beta induces development of the T(H)17 lineage.
Nature 2006; 441: 2314.
79 Wood KJ, Sakaguchi S. Regulatory lymphocytes: regulatory T
cells in transplantation tolerance. Nat Rev Immunol 2003; 3:
199210.
80 Battaglia M, Roncarolo MG. Induction of transplantation tolerance via regulatory T cells. Inamm Allergy Drug Targets
2006; 5: 15765.
81 Sykes M, Auchincloss Jr H, Sachs DH. Transplantation
immunology. In: Paul WE, ed. Fundamental Immunology.
Philadelphia, PA: Lippincott-Raven, 2003; 1481555.
82 Hall BM, Pearce NW, Gurley K, Dorsch SE. Specic unresponsiveness in rats with prolonged cardiac allograft survival
after treatment with cyclosporine. J Exp Med 1990; 171: 141
57.
83 Bushell A, Karim M, Kingsley CI, Wood KJ. Pretransplant
blood transfusion without additional immunotherapy generates CD25+CD4+ regulatory T cells: a potential explanation
for the blood-transfusion effect. Transplantation 2003; 76:
44955.
84 Kitade H, Kawai M, Rutgeerts O et al. Early presence of
regulatory cells in transplanted rats rendered tolerant by donorspecic blood transfusion. J Immunol 2005; 175: 496370.
85 Zhai Y, Kupiec-Weglinski JW. What is the role of regulatory T
cells in transplantation tolerance? Curr Opin Immunol 1999;
11: 497503.
86 Zhai Y, Meng L, Gao F, Wang Y, Busuttil RW, KupiecWeglinski JW. CD4+ T regulatory cell induction and function
in transplant recipients after CD154 blockade is TLR4
independent. J Immunol 2006; 176: 598894.
87 Karim M, Bushell AR, Wood KJ. Regulatory T cells in transplantation. Curr Opin Immunol 2002; 14: 58491.
88 Graca L, Thompson S, Lin CY, Adams E, Cobbold SP,
Waldmann H. Both CD4(+)CD25(+) and CD4(+)CD25())
regulatory cells mediate dominant transplantation tolerance.
J Immunol 2002; 168: 555865.
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
Review: Transplantation tolerance
89 Graca L, Cobbold SP, Waldmann H. Identication of regulatory T cells in tolerated allografts. J Exp Med 2002; 195:
16416.
90 Cobbold SP, Castejon R, Adams E et al. Induction of foxP3(+)
regulatory T cells in the periphery of T cell receptor transgenic
mice tolerized to transplants. J Immunol 2004; 172: 600310.
91 Honey K, Cobbold SP, Waldmann H. CD40 ligand blockade
induces CD4+ T cell tolerance and linked suppression.
J Immunol 1999; 163: 480510.
92 Graca L, Honey K, Adams E, Cobbold SP, Waldmann H. Cutting edge: anti-CD154 therapeutic antibodies induce infectious
transplantation tolerance. J Immunol 2000; 165: 47836.
93 Tran HM, Nickerson PW, Restifo AC et al. Distinct mechanisms for the induction and maintenance of allograft tolerance
with CTLA4-Fc treatment. J Immunol 1997; 159: 22329.
94 Karim M, Kingsley CI, Bushell AR, Sawitzki BS, Wood KJ.
Alloantigen-induced CD25+CD4+ regulatory T cells can
develop in vivo from CD25-CD4+ precursors in a thymusindependent process. J Immunol 2004; 172: 9238.
95 Yamada K, Iereno F, Gianello P, Shimizu A, Colvin RB, Sachs
DH. Role of the thymus in transplantation tolerance in miniature swine. III. Surgical manipulation of the thymus interferes
with stable induction of tolerance to class I-mismatched renal
allografts. Transplantation 1999; 67: 11129.
96 Odorico JS, OConnor T, Campos L, Barker CF, Posselt AM,
Naji A. Examination of the mechanisms responsible for tolerance induction after intrathymic inoculation of allogeneic bone
marrow. Ann Surg 1993; 218: 52531.
97 Ali A, Garrovillo M, Oluwole OO et al. Mechanisms of
acquired thymic tolerance: induction of transplant tolerance by
adoptive transfer of in vivo alloMHC peptide activated syngeneic T cells. Transplantation 2001; 71: 14428.
98 Bonasio R, Scimone ML, Schaerli P, Grabie N, Lichtman AH,
von Andrian UH. Clonal deletion of thymocytes by circulating
dendritic cells homing to the thymus. Nat Immunol 2006; 7:
1092100.
99 Bacchetta R, Passerini L, Gambineri E et al. Defective regulatory and effector T cell functions in patients with FOXP3
mutations. J Clin Invest 2006; 116: 171322.
100 Zorn E, Nelson EA, Mohseni M et al. IL-2 regulates FOXP3
expression in human CD4+CD25+ regulatory T cells through
a STAT-dependent mechanism and induces the expansion of
these cells in vivo. Blood 2006; 108: 15719.
101 Cohen AC, Nadeau KC, Tu W et al. Cutting edge: decreased
accumulation and regulatory function of CD4+ CD25(high) T
cells in human STAT5b deciency. J Immunol 2006; 177:
27704.
102 Louis S, Braudeau C, Giral M et al. Contrasting
CD25hiCD4+T cells FOXP3 patterns in chronic rejection and
operational drug-free tolerance. Transplantation 2006; 81:
398407.
103 Muthukumar T, Dadhania D, Ding R et al. Messenger RNA
for FOXP3 in the urine of renal-allograft recipients. N Engl J
Med 2005; 353: 234251.
104 Segundo DS, Ruiz JC, Izquierdo M et al. Calcineurin inhibitors, but not rapamycin, reduce percentages of
105
106
107
108
109
110
111
112
113
114
115
116
117
118
119
CD4+CD25+FOXP3+ regulatory T cells in renal transplant
recipients. Transplantation 2006; 82: 5507.
Edinger M, Hoffmann P, Ermann J et al. CD4(+)CD25(+)
regulatory T cells preserve graft-versus-tumor activity while
inhibiting graft-versus-host disease after bone marrow transplantation. Nat Med 2003; 9: 114450.
Taylor PA, Noelle RJ, Blazar BR. CD4(+)CD25(+) immune
regulatory cells are required for induction of tolerance to alloantigen via costimulatory blockade. J Exp Med 2001; 193:
13118.
Clark FJ, Gregg R, Piper K et al. Chronic graft-versus-host disease is associated with increased numbers of peripheral blood
CD4+CD25high regulatory T cells. Blood 2004; 103: 24106.
Sanchez J, Casano J, Alvarez MA et al. Kinetics of regulatory
CD25high and activated CD134+ (OX40) T lymphocytes during acute and chronic graft-versus-host disease after allogeneic
bone marrow transplantation. Br J Haematol 2004; 126:
697703.
Zorn E, Kim HT, Lee SJ et al. Reduced frequency of FOXP3+
CD4+CD25+ regulatory T cells in patients with chronic graftversus-host disease. Blood 2005; 106: 290311.
Miura Y, Thoburn CJ, Bright EC et al. Association of Foxp3
regulatory gene expression with graft-versus-host disease.
Blood 2004; 104: 218793.
de Kleer I, Vastert B, Klein M et al. Autologous stem cell
transplantation for autoimmunity induces immunologic selftolerance by reprogramming autoreactive T cells and restoring
the CD4+CD25+ immune regulatory network. Blood 2006;
107: 1696702.
Mossman TR, Coffman RL. Th1 and Th2 cells: different
patterns of lymphokine secretion lead to different functional
properties. Annu Rev Immunol 1989; 7: 14573.
Croft M, Carter L, Swain SL, Dutton RW. Generation of polarized antigen-specic CD8 effector populations: reciprocal
action of interleukin (IL)-4 and IL-12 in promoting type 2 versus type 1 cytokine proles. J Exp Med 1994; 180: 171528.
Le Moine A, Goldman M, Abramowicz D. Multiple pathways
to allograft rejection. Transplantation 2002; 73: 137381.
Higuchi M, Zeng D, Shizuru J et al. Immune tolerance to
combined organ and bone marrow transplants after fractionated
lymphoid irradiation involves regulatory NK T cells and clonal
deletion. J Immunol 2002; 169: 556470.
Sykes M, Hoyles KA, Romick ML, Sachs DH. In vitro and in
vivo analysis of bone marrow derived CD3+, CD4), CD8),
NK1.1+ cell lines. Cell Immunol 1990; 129: 47893.
Zeng D, Lewis D, Dejbakhsh-Jones S et al. Bone marrow
NK1.1- and NK1.1+ T cells reciprocally regulate acute graft
versus host disease. J Exp Med 1999; 189: 107381.
Lan F, Zeng D, Higuchi M, Huie P, Higgins JP, Strober S. Predominance of NK1.1(+)TCRalphabeta(+) or DX5(+)TCRalphabeta(+) T cells in mice conditioned with fractionated lymphoid
irradiation protects against graft-versus-host disease: natural
suppressor cells. J Immunol 2001; 167: 208796.
Lowsky R, Takahashi T, Liu YP et al. Protective conditioning
for acute graft-versus-host disease. N Engl J Med 2005; 353:
132131.
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 305
M. Sykes
120 Ciubotariu R, Colovai AI, Pennesi G et al. Specic suppression of human CD4+ Th cell responses to pig MHC antigens
by CD8+CD28- regulatory T cells. J Immunol 1998; 161:
5193202.
121 Faunce DE, Terajewicz A, Stein-Streilein J. Cutting edge: in
vitro-generated tolerogenic APC induce CD8(+) T regulatory
cells that can suppress ongoing experimental autoimmune
encephalomyelitis. J Immunol 2004; 172: 19915.
122 Liu J, Liu Z, Witkowski P et al. Rat CD8+ FOXP3+ T suppressor cells mediate tolerance to allogeneic heart transplants,
inducing PIR-B in APC and rendering the graft invulnerable
to rejection. Transpl Immunol 2004; 13: 23947.
123 Hoglund P. Induced peripheral regulatory T cells: the family
grows larger. Eur J Immunol 2006; 36: 2646.
124 Xystrakis E, Dejean AS, Bernard I et al. Identication of a
novel natural regulatory CD8 T-cell subset and analysis of its
mechanism of regulation. Blood 2004; 104: 3294301.
125 Xia G, Kovochich M, Truitt RL, Johnson BD. Tracking ex
vivo-expanded CD4+CD25+ and CD8+CD25+ regulatory T
cells after infusion to prevent donor lymphocyte infusioninduced lethal acute graft-versus-host disease. Biol Blood
Marrow Transplant 2004; 10: 74860.
126 Noble A, Giorgini A, Leggat JA. Cytokine-induced IL10-secreting CD8 T cells represent a phenotypically distinct
suppressor T-cell lineage. Blood 2006; 107: 447583.
127 Myers L, Takahashi C, Mittler RS, Rossi RJ, Vella AT. Effector CD8 T cells possess suppressor function after 4-1BB and
Toll-like receptor triggering. Proc Natl Acad Sci U S A 2003;
100: 5348 53.
128 Laffont S, Coudert JD, Garidou L et al. CD8+ T-cell-mediated
killing of donor dendritic cells prevents alloreactive T helper
type-2 responses in vivo. Blood 2006; 108: 225764.
129 Verbanac KM, Carver FM, Haisch CE, Thomas JM. A role
for transforming growth factor-beta in the veto
mechanism in transplant tolerance. Transplantation 1994; 57:
893900.
130 Tang Q, Henriksen KJ, Bi M et al. In vitro-expanded antigenspecic regulatory T cells suppress autoimmune diabetes.
J Exp Med 2004; 199: 145565.
131 Godfrey WR, Spoden DJ, Ge YG et al. Cord blood
CD4(+)CD25(+)-derived T regulatory cell lines express FoxP3
protein and manifest potent suppressor function. Blood 2005;
105: 7508.
132 Gribben JG, Guinan EC, Boussiotis VA et al. Complete blockade of B7 family-mediated costimulation is necessary to
induce human alloantigen-specic anergy: a method to ameliorate graft-versus-host disease and extend the donor pool.
Blood 1996; 87: 488793.
133 Larsen CP, Elwood ET, Alexander DZ et al. Long-term acceptance of skin and cardiac allografts after blocking CD40 and
CD28 pathways. Nature 1996; 381: 4348.
134 Larsen CP, Pearson TC. The CD40 pathway in allograft rejection, acceptance, and tolerance. Curr Opin Immunol 1997; 9:
6417.
135 Williams MA, Trambley J, Ha J et al. Genetic characterization
of strain differences in the ability to mediate CD40 CD28-
306
Review: Transplantation tolerance
136
137
138
139
140
141
142
143
144
145
146
147
148
149
independent rejection of skin allografts. J Immunol 2000; 165:
684957.
Lu LF, Lind EF, Gondek DC et al. Mast cells are essential
intermediaries in regulatory T-cell tolerance. Nature 2006; 442:
9971002.
Larsen CP, Alexander DZ, Hollenbaugh D et al. CD40-GP39
interactions play a critical role during allograft rejection: suppression of allograft rejection by blockade of the CD40-gp39
pathway. Transplantation 1996; 61: 49.
Quezada SA, Bennett K, Blazar BR, Rudensky AY, Sakaguchi
S, Noelle RJ. Analysis of the underlying cellular mechanisms
of anti-CD154-induced graft tolerance: the interplay of clonal
anergy and immune regulation. J Immunol 2005; 175: 7719.
Tian L, Lu L, Yuan Z, Lamb JR, Tam PK. Acceleration of
apoptosis in CD4+CD8+ thymocytes by rapamycin accompanied by increased CD4+CD25+ T cells in the periphery. Transplantation 2004; 77: 1839.
Battaglia M, Stabilini A, Roncarolo MG. Rapamycin selectively expands CD4+CD25+FoxP3+ regulatory T cells. Blood
2005; 105: 47438.
Baan CC, Van der Mast BJ, Klepper M et al. Differential
effect of calcineurin inhibitors, anti-CD25 antibodies and rapamycin on the induction of FOXP3 in human T cells. Transplantation 2005; 80: 1107.
Li Y, Zheng XX, Li XC, Zand MS, Strom TB. Combined
costimulation blockade plus rapamycin but not cyclosporine
produces permanent engraftment. Transplantation 1998; 66:
13878.
Zheng XX, Sanchez-Fueyo A, Sho M, Domenig C, Sayegh
MH, Strom TB. Favorably tipping the balance between cytopathic and regulatory T cells to create transplantation tolerance.
Immunity 2003; 19: 50314.
Quezada SA, Fuller B, Jarvinen LZ et al. Mechanisms of
donor-specic transfusion tolerance: preemptive induction of
clonal T-cell exhaustion via indirect presentation. Blood 2003;
102: 19206.
Markees TG, Phillips NE, Gordon EJ et al. Long-term survival
of skin allografts induced by donor splenocytes and antiCD154 antibody in thymectomized mice requires CD4+ T
cells, interferon-c, and CTLA4. J Clin Invest 1998; 101:
244655.
Iwakoshi NN, Mordes JP, Markees TG, Phillips NE, Rossini
AA, Greiner DL. Treatment of allograft recipients with donorspecic transfusion and anti-CD154 antibody leads to deletion
of alloreactive CD8+ T cells and prolonged graft survival in a
CTLA4-dependent manner. J Immunol 2000; 164: 51221.
Markees TG, Phillips NE, Noelle RJ et al. Prolonged survival
of mouse skin allografts in recipients treated with donor
splenocytes and antibody to CD40 ligand. Transplantation
1997; 64: 32935.
Zheng X-X, Markees TG, Hancock WW et al. CTLA4 signals
are required to optimally induce allograft tolerance with combined donor-specic transfusion and anti-CD154 monoclonal
antibody treatment. J Immunol 1999; 162: 498390.
Koulmanda M, Smith RN, Qipo A, Weir G, Auchincloss H,
Strom TB. Prolonged survival of allogeneic islets in cynomol-
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
150
151
152
153
154
155
156
157
158
159
160
161
162
163
164
165
Review: Transplantation tolerance
gus monkeys after short-term anti-CD154-based therapy: nonimmunologic graft failure? Am J Transplant 2006; 6: 68796.
Takeuchi Y, Ito H, Kurtz J, Wekerle T, Ho L, Sykes M. Earlier
low-dose TBI or DST overcomes CD8+ T-cell-mediated alloresistance to allogeneic marrow in recipients of anti-CD40L.
Am J Transplant 2004; 4: 3140.
Kirk AD, Harlan DM, Armstrong NN et al. CTLA4-Ig and
anti-CD40 ligand prevent renal allograft rejection in primates.
Proc Natl Acad Sci U S A 1997; 94: 878998.
Kirk AD, Burkly LC, Batty DS et al. Treatment with humanized monoclonal antibody against CD154 prevents acute renal
allograft rejection in nonhuman primates. Nat Med 1999; 5:
68693.
Kenyon NS, Chatzipetrou M, Masetti M et al. Long-term survival and function of intrahepatic islet allografts in rhesus
monkeys treated with humanized anti-CD154. Proc Natl Acad
Sci U S A 1999; 96: 81327.
Kirk AD, Tadaki DK, Celniker A et al. Induction therapy with
monoclonal antibodies specic for cd80 and cd86 delays the
onset of acute renal allograft rejection in non-human primates.
Transplantation 2001; 72: 37784.
Preston EH, Xu H, Dhanireddy KK et al. IDEC-131 (antiCD154), sirolimus and donor-specic transfusion facilitate
operational tolerance in non-human primates. Am J Transplant
2005; 5: 103241.
Koyama I, Kawai T, Andrews D et al. Thrombophilia associated with anti-CD154 monoclonal antibody treatment and its
prophylaxis in nonhuman primates. Transplantation 2004; 77:
4602.
Adams AB, Shirasugi N, Jones TR et al. Development of a
chimeric anti-CD40 monoclonal antibody that synergizes with
LEA29Y to prolong islet allograft survival. J Immunol 2005;
174: 54250.
Vincenti F, Larsen C, Durrbach A et al. Costimulation blockade with belatacept in renal transplantation. N Engl J Med
2005; 353: 77081.
Guinan EC, Boussiotis VA, Neuberg D et al. Transplantation
of anergic histoincompatible bone marrow allografts. N Engl J
Med 1999; 340: 170414.
Sykes M. Mixed chimerism and transplant tolerance. Immunity
2001; 14: 41724.
Dey B, Sykes M, Spitzer TR. Outcomes of combined bone
marrow and solid organ transplants: a review. Medicine (Baltimore) 1998; 77: 35569.
Charlton B, Auchincloss H Jr, Fathman CG. Mechanisms
of transplantation tolerance. Annu Rev Immunol 1994; 12:
70734.
Mathes DW, Solari MG, Randolph MA et al. Long-term
acceptance of renal allografts following prenatal inoculation
with adult bone marrow. Transplantation 2005; 80: 13008.
Streilein JW. Neonatal tolerance of H-2 alloantigens. Transplantation 1991; 52: 110.
Borenstein SH, Tao KS, West LJ, Chamberlain JW. Extrathymic deletion of CD8+ alloreactive T cells in a transgenic T cell
receptor model of neonatal tolerance. Transplantation 2001;
72: 180716.
166 Bacchetta R, Vandekerckhove BAE, Touraine J-L et al. Chimerism and tolerance to host and donor in severe combined
immunodeciencies transplanted with fetal liver stem cells.
J Clin Invest 1993; 91: 106778.
167 Touraine JL. Treatment of human fetuses and induction of
immunological tolerance in humans by in utero transplantation
of stem cells into fetal recipients. Acta Haematol 1996; 96:
1159.
168 Flake AW, Roncarolo M-G, Puck JM et al. Treatment of
X-linked severe combined immunodeciency by in utero transplantation of paternal bone marrow. N Engl J Med 1996; 335:
180610.
169 Poynton CH. T cell depletion in bone marrow transplantation.
Bone Marrow Transplant 1988; 3: 26579.
170 Martin P. Overview of marrow transplantation immunology.
In: Forman SJ, Blume KG, Thomas ED, eds. Hematopoietic
Cell Transplantation. Cambridge, UK: Blackwell Scientic
Publications, 1999; 1927.
171 Alyea EP, Kim HT, Ho V et al. Comparative outcome of nonmyeloablative and myeloablative allogeneic hematopoietic cell
transplantation for patients older than 50 years of age. Blood
2005; 105: 104.
172 Kolb HJ, Schmid C, Barrett AJ, Schendel DJ. Graft-versusleukemia reactions in allogeneic chimeras. Blood 2004; 103:
76776.
173 Singer A, Hathcock KS, Hodes RJ. Self recognition in allogeneic radiation chimeras. A radiation resistant host element
dictates the self specicity and immune response gene phenotype of T-helper cells. J Exp Med 1981; 153: 1286301.
174 Ildstad ST, Wren SM, Bluestone JA, Barbieri SA, Sachs DH.
Characterization of mixed allogeneic chimeras. Immunocompetence, in vitro reactivity, and genetic specicity of tolerance.
J Exp Med 1985; 162: 23144.
175 Ruedi E, Sykes M, Ildstad ST et al. Antiviral T cell competence and restriction specicity of mixed allogeneic (P1+P2
>P1) irradiation chimeras. Cell Immuol 1989; 121: 18595.
176 Tomita Y, Sachs DH, Sykes M. Myelosuppressive conditioning
is required to achieve engraftment of pluripotent stem cells
contained in moderate doses of syngeneic bone marrow. Blood
1994; 83: 93948.
177 Wekerle T, Sayegh MH, Ito H et al. Anti-CD154 or CTLA4Ig
obviates the need for thymic irradiation in a non-myeloablative
conditioning regimen for the induction of mixed hematopoietic
chimerism and tolerance. Transplantation 1999; 68: 134855.
178 Haynes BF, Markert ML, Sempowski GD, Patel DD, Hale LP.
The role of the thymus in immune reconstitution in aging,
bone marrow transplantation, and HIV-1 infection. Annu Rev
Immunol 2000; 18: 52960.
179 Kimikawa M, Sachs DH, Colvin RB, Bartholemew A, Kawai
T, Cosimi AB. Modications of the conditioning regimen for
achieving mixed chimerism and donor-specic tolerance in
cynomolgus monkeys. Transplantation 1997; 64: 70916.
180 Ito H, Takeuchi Y, Shaffer J, Sykes M. Local irradiation
enhances congenic donor pluripotent hematopoietic stem cell
engraftment similarly in irradiated and non-irradiated sites.
Blood 2003; 103: 194954.
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 307
M. Sykes
181 Durham MM, Bingaman AW, Adams AB et al. Cutting edge:
administration of anti-CD40 ligand and donor bone marrow
leads to hemopoietic chimerism and donor-specic tolerance
without cytoreductive conditioning. J Immunol 2000; 165:
14.
182 Ito H, Kurtz J, Shaffer J, Sykes M. CD4 T cell-mediated alloresistance to fully MHC-mismatched allogeneic bone marrow
engraftment is dependent on CD40-CD40 L interactions, and
lasting T cell tolerance is induced by bone marrow transplantation with initial blockade of this pathway. J Immunol 2001;
166: 2981.
183 Hale DA, Gottschalk R, Umemura A, Maki T, Monaco AP.
Establishment of stable multilineage hematopoietic chimerism
and donor-specic tolerance without irradiation. Transplantation 2000; 69: 124251.
184 Neipp M, Gammie JS, Exner BG et al. A partial conditioning
approach to achieve mixed chimerism in the rat: Depletion of
host natural killer cells signicantly reduces the amount of
total body irradiation required for engraftment. Transplantation
1999; 68: 36978.
185 Kean LS, Durham MM, Adams AB et al. A cure for murine
sickle cell disease through stable mixed chimerism and tolerance induction after nonmyeloablative conditioning and major
histocompatibility complex-mismatched bone marrow transplantation. Blood 2002; 99: 18409.
186 Liang Y, Huang T, Zhang C et al. Donor CD8+ T cells facilitate induction of chimerism and tolerance without GVHD in
autoimmune NOD mice conditioned with anti-CD3 mAb.
Blood 2005; 105: 21808.
187 Camirand G, Rousseau J, Ducharme ME, Rothstein DM,
Tremblay JP. Novel duchenne muscular dystrophy treatment
through myoblast transplantation tolerance with anti-CD45RB,
anti-CD154 and mixed chimerism. Am J Transplant 2004; 4:
125565.
188 Anam K, Akpinar E, Craighead N, Black AT, Hale DA. Targeted T-cell depletion or CD154 blockade generates mixed
hemopoietic chimerism and donor-specic tolerance in mice
treated with sirolimus and donor bone marrow. Transplantation
2004; 78: 12908.
189 Slavin S, Strober S, Fuks Z, Kaplan HS. Induction of specic
tissue transplantation tolerance using fractionated total lymphoid
irradiation in adult mice: long-term survival of allogeneic bone
marrow and skin grafts. J Exp Med 1977; 146: 34.
190 Najarian JS, Ferguson RM, Sutherland DER et al. Fractionated total lymphoid irradiation as preparative immunosuppression in high risk renal transplantation. Ann Surg 1982;
196: 44251.
191 Millan MT, Shizuru JA, Hoffmann P et al. Mixed chimerism
and immunosuppressive drug withdrawal after HLA- mismatched kidney and hematopoietic progenitor transplantation.
Transplantation 2002; 73: 138691.
192 Kawai T, Cosimi AB, Colvin RB et al. Mixed allogeneic
chimerism and renal allograft tolerance in cynomologous
monkeys. Transplantation 1995; 59: 25662.
193 Huang CA, Fuchimoto Y, Sheir-Dolberg R, Murphy MC, Neville DM Jr, Sachs DH. Stable mixed chimerism and tolerance
308
Review: Transplantation tolerance
194
195
196
197
198
199
200
201
202
203
204
205
206
207
208
using a non-myeloablative preparative regimen in a large
animal model. J Clin Invest 2000; 105: 17381.
Fuchimoto Y, Huang CA, Yamada K et al. Mixed chimerism
and tolerance without whole body irradiation in a large animal
model. J Clin Invest 2000; 105: 177989.
Hettiaratchy S, Melendy E, Randolph MA et al. Tolerance
to composite tissue allografts across a major histocompatibility barrier in miniature swine. Transplantation 2004; 77:
51421.
Tomita Y, Khan A, Sykes M. Mechanism by which additional
monoclonal antibody injections overcome the requirement for
thymic irradiation to achieve mixed chimerism in mice receiving bone marrow transplantation after conditioning with anti-T
cell mAbs and 3 Gy whole body irradiation. Transplantation
1996; 61: 47785.
Kurtz J, Ito H, Wekerle T, Shaffer J, Sykes M. Mechanisms
involved in the establishment of tolerance through costimulatory blockade and BMT: lack of requirement for CD40L-mediated signaling for tolerance or deletion of donor-reactive
CD4+ cells. Am J Transplant 2001; 1: 33949.
Kurtz J, Wekerle T, Sykes M. Tolerance in mixed chimerism
a role for regulatory cells? Trends Immunol 2004; 25: 51823.
Bigenzahn S, Blaha P, Koporc Z et al. The role of nondeletional tolerance mechanisms in a murine model of mixed
chimerism with costimulation blockade. Am J Transplant
2005; 5: 123747.
Domenig C, Sanchez-Fueyo A, Kurtz J et al. Roles of deletion
and regulation in creating mixed chimerism and allograft tolerance using a nonlymphoablative irradiation-free protocol.
J Immunol 2005; 175: 5160.
Starzl TE, Demetris AJ, Trucco M et al. Chimerism and
donor-specic nonreactivity 27 to 29 years after kidney allotransplantation. Transplantation 1993; 55: 12727.
Wood K, Sachs DH. Chimerism and transplantation tolerance:
cause and effect. Immunol Today 1996; 17: 5848.
Bushell A, Pearson TC, Morris PJ, Wood KJ. Donor-recipient
microchimerism is not required for tolerance induction following recipient pretreatment with donor-specic transfusion and
anti-CD4 antibody. Transplantation 1995; 59: 136771.
Shirwan H, Wang HK, Barwari L, Makowka L, Cramer DV.
Pretransplant injection of allograft recipients with donor blood
or lymphocytes permits allograft tolerance without the presence of persistent donor microchimerism. Transplantation
1996; 61: 13826.
Fisher RA, Cohen DS, Ben-Ezra JM, Sallade RE, Tawes JW,
Tarry WC. Induction of long-term graft tolerance and
donor recipient chimerism. J Surg Res 1996; 60: 1815.
Schlitt HJ, Hundreiser J, Ringe B, Pichlmayr R. Systemic microchimerism of donor-type associated with irreversible acute
liver graft rejection eight years after transplantation. N Engl J
Med 1994; 330: 6467.
Schlitt HJ. Is microchimerism needed for allograft tolerance?
Transplant Proc 1997; 29: 824.
Hisanaga M, Hundrieser J, Boker K et al. Development, stability, and clinical correlations of allogeneic microchimerism after
solid organ transplantation. Transplantation 1996; 61: 405.
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
M. Sykes
Review: Transplantation tolerance
209 Shapiro R, Rao AS, Fontes P et al. Combined simultaneous
kidney bone marrow transplantation. Transplantation 1995;
60: 14215.
210 Rolles K, Burroughs AK, Davidson BR, Karatapanis S, Prentice HG, Hamon MD. Donor-specic bone marrow infusion
after orthotopic liver transplantation. Lancet 1994; 343: 2635.
211 Rugeles MT, Aitouche A, Zeevi A et al. Evidence for the
presence of multilineage chimerism and progenitors of donor
dendritic cells in the peripheral blood of bone marrow-augmented organ transplant recipients. Transplantation 1997; 64:
73541.
212 Ciancio G, Burke GW, Garcia-Morales R et al. Effect of living-related donor bone marrow infusion on chimerism and in
vitro immunoregulatory activity in kidney transplant recipients.
Transplantation 2002; 74: 48896.
213 Tryphonopoulos P, Tzakis AG, Weppler D et al. The role of
donor bone marrow infusions in withdrawal of immunosuppression in adult liver allotransplantation. Am J Transplant
2005; 5: 60813.
214 Sharabi Y, Aksentijevich I, Sundt TM III, Sachs DH, Sykes
M. Specic tolerance induction across a xenogeneic barrier:
production of mixed rat mouse lymphohematopoietic chimeras
using a nonlethal preparative regimen. J Exp Med 1990; 172:
195202.
215 Nikolic B, Cooke DT, Zhao G, Sykes M. Both c d T cells
and NK cells inhibit the engraftment of xenogeneic rat bone
marrow cells in mice. J Immunol 2001; 166: 1398404.
216 Lee LA, Sergio JJ, Sykes M. Natural killer cells weakly resist
engraftment of allogeneic long-term multilineage-repopulating
hematopoietic stem cells. Transplantation 1996; 61: 12532.
217 Aksentijevich I, Sachs DH, Sykes M. Humoral tolerance in
xenogeneic BMT recipients conditioned with a non-myeloablative regimen. Transplantation 1992; 53: 110814.
218 Lee LA, Sergio JJ, Sachs DH, Sykes M. Mechanism of tolerance
in mixed xenogeneic chimeras prepared with a non-myeloablative conditioning regimen. Transplant Proc 1994; 26: 11978.
219 Ohdan H, Yang Y-G, Swenson KG, Kitamura H, Sykes M. T
cell and B-cell tolerance to gala1,3gal-expressing heart xenografts is achieved in a1,3-galactosyltransferase-decient mice
by non-myeloablative induction of mixed chimerism. Transplantation 2001; 71: 153242.
220 Nikolic B, Lei H, Pearson DA, Sergio JJ, Swenson KA, Sykes
M. Role of intrathymic rat class II cells in maintaining deletional tolerance in xenogeneic rat>mouse bone marrow
chimeras. Transplantation 1998; 65: 121624.
221 Yang Y-G, deGoma E, Ohdan H et al. Tolerization of antigala1-3gal natural antibody-forming B cells by induction of
mixed chimerism. J Exp Med 1998; 187: 133542.
222 Zhao Y, Ohdan H, Manilay JO, Sykes M. NK cell tolerance in
mixed allogeneic chimeras. J Immunol 2003; 170: 5398405.
223 Kawahara T, Rodriguez-Barbosa JI, Zhao Y, Zhao G, Sykes
M. Global unresponsiveness as a mechanism of natural killer
cell tolerance in mixed xenogeneic chimetas. Am J Transplant
2007; (in press).
224 Gritsch HA, Sykes M. Host marrow has a competitive advantage which limits donor hematopietic repopulation in mixed
xenogeneic chimeras. Xenotransplantation 1996; 3: 31220.
225 Simon AR, Warrens AN, Sykes M. Efcacy of adhesive interactions in pig-to-human xenotransplantation. Immunol Today
1999; 20: 291338.
226 Lapidot T, Pumia F, Doedens M, Murdoch B, Williams DE,
Dick JE. Cytokine stimulation of multilineage hematopoiesis
from immature human cells engrafted in SCID mice. Science
1992; 255: 113743.
227 Yang Y-G, Sergio JJ, Swenson K, Glaser RM, Monroy R,
Sykes M. Donor-specic growth factors promote swine
hematopoiesis in SCID mice. Xenotransplantation 1996;
3: 92101.
228 Chen AM, Zhou Y, Swenson K, Sachs DH, Sykes M, Yang
Y-G. Porcine stem cell engraftment and seeding of murine thymus with class II+ cells in mice expressing porcine cytokines:
toward tolerance induction across discordant xenogeneic barriers. Transplantation 2000; 69: 248490.
229 Lan P, Wang L, Diouf B et al. Induction of human T cell tolerance to porcine xenoantigens through mixed hematopoietic
chimerism. Blood 2004; 103: 39649.
230 Terpstra W, Leenen PJ, van den Bos C et al. Facilitated
engraftment of human hematopoietic cells in severe combined
immunodecient mice following a single injection of
C12MDP liposomes. Leukemia 1997; 11: 104954.
231 Abe M, Cheng J, Qi J et al. Elimination of porcine hematopoietic cells by macrophages in mice. J Immunol 2002; 168:
6218.
232 Wang H, Verhalen J, Madariaga ML et al. Attenuation of phagocytosis of xenogeneic cells by manipulating CD47. Blood
2006; 109: 83642.
233 Lee LA, Gritsch HA, Sergio JJ et al. Specic tolerance across
a discordant xenogeneic transplantation barrier. Proc Natl Acad
Sci U S A 1994; 91: 108647.
234 Zhao Y, Fishman JA, Sergio JJ et al. Immune restoration by
fetal pig thymus grafts in T cell-depleted, thymectomized mice.
J Immunol 1997; 158: 16419.
235 Zhao Y, Swenson K, Sergio JJ, Arn JS, Sachs DH, Sykes M.
Skin graft tolerance across a discordant xenogeneic barrier.
Nat Med 1996; 2: 12116.
236 Zhao Y, Sergio JJ, Swenson KA, Arn JS, Sachs DH, Sykes M.
Positive and negative selection of functional mouse CD4 cells
by porcine MHC in pig thymus grafts. J Immunol 1997; 159:
21007.
237 Zhao Y, Swenson K, Sergio JJ, Sykes M. Pig MHC mediates
positive selection of mouse CD4+ T cells with a mouse MHCrestricted TCR in pig thymus grafts. J Immunol 1998; 161:
13206.
238 Zhao Y, Rodriguez-Barbosa JI, Zhao G, Shaffer J, Arn JS, Sykes M. Maturation and function of mouse T cells with a transgeneic TCR positively selected by highly disparate xenogeneic
porcine MHC. Cell Mol Biol 2000; 47: 21728.
239 Markert ML, Kostyu DD, Ward FE et al. Successful formation
of a chimeric human thymus allograft following transplantation
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310 309
M. Sykes
240
241
242
243
244
245
246
247
248
249
250
251
310
of cultured postnatal human thymus. J Immunol 1997; 158:
9981005.
Markert ML, Boeck A, Hale LP et al. Transplantation of
thymus tissue in complete DiGeorge syndrome. N Engl J
Med 1999; 341: 11809.
Nikolic B, Gardner JP, Scadden DT, Arn JS, Sachs DH, Sykes
M. Normal development in porcine thymus grafts and specic
tolerance of human T cells to porcine donor MHC. J Immunol
1999; 162: 34027.
Wu A, Yamada K, Neveille DM et al. Xenogeneic thymic
transplantation in a pig-to-baboon model. Transplantation
2002; 75: 28291.
Yamada K, Yazawa K, Shimizu A et al. Marked prolongation
of porcine renal xenograft survival in baboons through the use
of alpha1,3-galactosyltransferase gene-knockout donors and
the cotransplantation of vascularized thymic tissue. Nat Med
2005; 11: 324.
Rodriguez-Barbosa JI, Zhao Y, Barth R et al. Enhanced CD4
reconstitution by grafting neonatal porcine tissue in alternative
locations is associated with donor-specic tolerance and suppression of pre-existing xenoreactive T cells. Transplantation
2001; 72: 122331.
Phelps CJ, Koike C, Vaught TD et al. Production of alpha
1,3-galactosyltransferase-decient pigs. Science 2003; 299:
4114.
Kolber-Simonds D, Lai L, Watt SR et al. Production of
{alpha}-1,3-galactosyltransferase null pigs by means of nuclear
transfer with broblasts bearing loss of heterozygosity mutations. Proc Natl Acad Sci U S A 2004; 101: 733540.
Kuwaki K, Tseng YL, Dor FJ et al. Heart transplantation in
baboons using alpha1,3-galactosyltransferase gene-knockout
pigs as donors: initial experience. Nat Med 2005; 11: 2931.
Chen G, Qian H, Starzl T et al. Acute rejection is associated
with antibodies to non-Gal antigens in baboons using
Gal-knockout pig kidneys. Nat Med 2005; 11: 12958.
Strober S, Dhillon M, Schubert M et al. Acquired immune tolerance to cadaveric renal allografts. A study of three patients
treated with total lymphoid irradiation. N Engl J Med 1989;
321: 2833.
Roussey-Kesler G, Giral M, Moreau A et al. Clinical operational tolerance after kidney transplantation. Am J Transplant
2006; 6: 73646.
Spitzer TR, MCafee S, Sackstein R et al. The intentional
induction of mixed chimerism and achievement of anti-tumor
responses following non-myeloablative conditioning therapy
and HLA-matched and mismatched donor bone marrow trans-
Review: Transplantation tolerance
252
253
254
255
256
257
258
259
260
plantation for refractory hematologic malignancies. Biol Blood
Marrow Transplant 2000; 6: 30920.
Sykes M, Sheard MA, Sachs DH. Graft-versus-host-related
immunosuppression is induced in mixed chimeras by alloresponses against either host or donor lymphohematopoietic
cells. J Exp Med 1988; 168: 23916.
Mapara MY, Kim Y-M, Wang S-P, Bronson R, Sachs DH,
Sykes M. Donor lymphocyte infusions mediate superior graftversus-leukemia effects in mixed compared to fully allogeneic
chimeras: a critical role for host antigen-presenting cells. Blood
2002; 100: 19039.
Mapara MY, Kim Y-M, Marx J, Sykes M. DLI-mediated GVL
effects in mixed chimeras established with a non-myeloablative
conditioning regimen: extinction of GVL effects coincides
with loss of alloreactive cells following conversion to full
donor chimerism. Transplantation 2003; 76: 297305.
Chakraverty R, Eom HS, Sachs J et al. Host MHC class II+
antigen-presenting cells and CD4 cells are required for CD8mediated graft-versus-leukemia responses following delayed
donor leukocyte infusions. Blood 2006; 108: 210613.
Chakraverty R, Cote D, Buchli J et al. An inammatory
checkpoint regulates recruitment of graft-versus-host-reactive T
cells to peripheral tissues. J Exp Med 2006; 203: 202131.
Fudaba Y, Spitzer TR, Shaffer J et al. Myeloma responses and
tolerance following combined kidney and nonmyeloablative
marrow transplantation: in vivo and in vitro analyses. Am J
Transplant 2006; 6: 212133.
Spitzer TR, MCafee S, Dey BR et al. Non-myeloablative haploidentical stem cell transplantation using anti-CD2 monoclonal antibody (MEDI-507)-based conditioning for refractory
hematologic malignancies. Transplantation 2003; 75: 174851.
Kirk AD, Hale DA, Mannon RB et al. Results from a human
renal allograft tolerance trial evaluating the humanized CD52specic monoclonal antibody alemtuzumab (CAMPATH-1H).
Transplantation 2003; 76: 1209.
Kirk AD, Mannon RB, Kleiner DE et al. Results from a
human renal allograft tolerance trial evaluating T-cell depletion
with alemtuzumab combined with deoxyspergualin. Transplantation 2005; 80: 10519.
Correspondence: Megan Sykes, MD, Transplantation Biology
Research Center, Massachusetts General Hospital Harvard Medical
School, MGH-East Building 149-5102, 13th Street, Boston, MA
02129, USA.
(fax: +617 724 9892; e-mail: megan.sykes@tbrc.mgh.harvard.edu).
2007 Blackwell Publishing Ltd Journal of Internal Medicine 262; 288310
Das könnte Ihnen auch gefallen
- Immunology of Transplant Rejection: More..Dokument7 SeitenImmunology of Transplant Rejection: More..kusumrajaiNoch keine Bewertungen
- 263-270 RemuzziDokument8 Seiten263-270 Remuzzimishrak20Noch keine Bewertungen
- Transplantation ImmunologyDokument23 SeitenTransplantation ImmunologyCatherine RajanNoch keine Bewertungen
- Allorecognition and Alloresponse 2007Dokument12 SeitenAllorecognition and Alloresponse 2007CherryLollipop25Noch keine Bewertungen
- Immunology of Transplant RejectionDokument8 SeitenImmunology of Transplant Rejectionxplaind100% (1)
- Immuo AnswersDokument13 SeitenImmuo Answersaditya sahuNoch keine Bewertungen
- Piis0272638615005120 1Dokument11 SeitenPiis0272638615005120 1KRIZARA MARIA VALVERDE RIVERANoch keine Bewertungen
- TransplantationDokument34 SeitenTransplantationIlham KuNoch keine Bewertungen
- Transplantation Immunology: Jun Dou (窦骏)Dokument39 SeitenTransplantation Immunology: Jun Dou (窦骏)KlisjanaNoch keine Bewertungen
- 10 1056@NEJMra1911109 PDFDokument11 Seiten10 1056@NEJMra1911109 PDFMартин ДончевNoch keine Bewertungen
- LECTURE 17 Major Histocompatibility MoleculesDokument7 SeitenLECTURE 17 Major Histocompatibility MoleculesDomenica Mishel Sandoval SegoviaNoch keine Bewertungen
- Immunology 7Dokument25 SeitenImmunology 7ukashazam19Noch keine Bewertungen
- Cellular Immune Responses in Red Blood Cell AlloimmunizationDokument5 SeitenCellular Immune Responses in Red Blood Cell AlloimmunizationВладимир ДружининNoch keine Bewertungen
- 2.2. Cancer Immunotherapy Co-Stimulatory Agonists and Co-Inhibitory AntagonistsDokument11 Seiten2.2. Cancer Immunotherapy Co-Stimulatory Agonists and Co-Inhibitory AntagonistsDiego EskinaziNoch keine Bewertungen
- toleranceDokument5 SeitentoleranceAfaq AhmadNoch keine Bewertungen
- Immune Tolerance in Multiple SclerosisDokument19 SeitenImmune Tolerance in Multiple SclerosisRafael3462Noch keine Bewertungen
- Vonderheide Et Al-2014-Immunological ReviewsDokument7 SeitenVonderheide Et Al-2014-Immunological ReviewsMarjorie AlemanNoch keine Bewertungen
- Immune ToleranceDokument7 SeitenImmune TolerancehamaadaNoch keine Bewertungen
- 1 s2.0 S108387911300387X MainDokument9 Seiten1 s2.0 S108387911300387X MainIsmail FadlurrohmanNoch keine Bewertungen
- +++kpwilkie Chapter RevisedDokument35 Seiten+++kpwilkie Chapter RevisedEnes ÇakmakNoch keine Bewertungen
- TransplantationDokument8 SeitenTransplantationMd Sanaullah AlamNoch keine Bewertungen
- 18 JL 7Dokument15 Seiten18 JL 7Irma SihotangNoch keine Bewertungen
- Immuno TherapyDokument20 SeitenImmuno TherapyAttis PhrygiaNoch keine Bewertungen
- Immune Responses To Human Papillomavirus: Margaret StanleyDokument7 SeitenImmune Responses To Human Papillomavirus: Margaret StanleyDina A. ŠabićNoch keine Bewertungen
- Rheumatoid Arthritis Linked to Premature Aging of Immune SystemDokument9 SeitenRheumatoid Arthritis Linked to Premature Aging of Immune SystemKeerthi SagarNoch keine Bewertungen
- TransplatationDokument3 SeitenTransplatationMalavika MaluNoch keine Bewertungen
- Transplant Immunology OverviewDokument47 SeitenTransplant Immunology OverviewViena Mae MaglupayNoch keine Bewertungen
- Graft Rejection Immunological Suppression PDFDokument7 SeitenGraft Rejection Immunological Suppression PDFmanoj_rkl_07Noch keine Bewertungen
- Transfusion-Related Immunomodulation (TRIM) : An Update: Eleftherios C. Vamvakas, Morris A. BlajchmanDokument22 SeitenTransfusion-Related Immunomodulation (TRIM) : An Update: Eleftherios C. Vamvakas, Morris A. BlajchmanLaura Francisca Urrutia IbacetaNoch keine Bewertungen
- Graft RejectionDokument8 SeitenGraft Rejectionasmaa100% (1)
- 8ra19 (2009) Omar A. Ali: Sci Transl Med Et AlDokument12 Seiten8ra19 (2009) Omar A. Ali: Sci Transl Med Et AlSage RadachowskyNoch keine Bewertungen
- Chapter 3 Blood Transfusions and The Immune SystemDokument14 SeitenChapter 3 Blood Transfusions and The Immune SystemLulu Afifah OctaviaNoch keine Bewertungen
- 1 s2.0 S221138352100099X MainDokument14 Seiten1 s2.0 S221138352100099X Mainsara.madkour99Noch keine Bewertungen
- Att 5KJBXUZs14QHOXaS8FJcB5Fkf0M-SsmsUCS5rCUA7t8Dokument9 SeitenAtt 5KJBXUZs14QHOXaS8FJcB5Fkf0M-SsmsUCS5rCUA7t8mackienmao1999Noch keine Bewertungen
- Targeting T Cell Activation in Immuno-Oncology: S.D. Saibil and P.S. OhashiDokument8 SeitenTargeting T Cell Activation in Immuno-Oncology: S.D. Saibil and P.S. OhashiNBME testNoch keine Bewertungen
- Cancer and The Immune System Basic Concepts and Targets For InterventionDokument38 SeitenCancer and The Immune System Basic Concepts and Targets For InterventionMariana NannettiNoch keine Bewertungen
- Host Defence Mechanisms Against TumorsDokument7 SeitenHost Defence Mechanisms Against TumorsHamam AmastrdamNoch keine Bewertungen
- Cancer Immunology and ImmunotherapyDokument15 SeitenCancer Immunology and ImmunotherapyRamona PalalogosNoch keine Bewertungen
- Fast Facts: CAR T-Cell Therapy: Insight into current and future applicationsVon EverandFast Facts: CAR T-Cell Therapy: Insight into current and future applicationsNoch keine Bewertungen
- Reading 8Dokument23 SeitenReading 8Loredana DobreaNoch keine Bewertungen
- Tolerance, Suppression and The Fetal Allograft: ReviewDokument9 SeitenTolerance, Suppression and The Fetal Allograft: Reviewshine8395Noch keine Bewertungen
- Therapeutic Application of T Regulatory Cells in Composite Tissue AllotransplantationDokument15 SeitenTherapeutic Application of T Regulatory Cells in Composite Tissue AllotransplantationAngelica SerbulNoch keine Bewertungen
- 2a PDFDokument11 Seiten2a PDFAndrea Il MiodoNoch keine Bewertungen
- CLINICS 2019 - Adoptive T-Cell Therapy For Solid MalignanciesDokument15 SeitenCLINICS 2019 - Adoptive T-Cell Therapy For Solid MalignanciesWilverCarbonelLuyoNoch keine Bewertungen
- Terapia Inmunomoduladora 1Dokument9 SeitenTerapia Inmunomoduladora 1Nathy Pasapera AlbanNoch keine Bewertungen
- Kuby Immunology TransplantationDokument21 SeitenKuby Immunology TransplantationMalki kawtarNoch keine Bewertungen
- Artículo BaseDokument10 SeitenArtículo BaseSöö FiiNoch keine Bewertungen
- Transplantation: Saba Shahzadi 1721138054Dokument6 SeitenTransplantation: Saba Shahzadi 1721138054sandy candyNoch keine Bewertungen
- Dendritic Cell Gene TherapyDokument16 SeitenDendritic Cell Gene TherapyazharbattooNoch keine Bewertungen
- Treg CellsDokument15 SeitenTreg Cellsmr.hakku.jetaNoch keine Bewertungen
- Ca Immunotherapy Beyond PDFDokument25 SeitenCa Immunotherapy Beyond PDFJamil ANoch keine Bewertungen
- GRAFT VS HOST AND TRANSPLANT INFECTIONDokument44 SeitenGRAFT VS HOST AND TRANSPLANT INFECTIONHasna Mirda AmazanNoch keine Bewertungen
- Periodontal TreatmentDokument21 SeitenPeriodontal TreatmentAdyas AdrianaNoch keine Bewertungen
- Hogquist 2005Dokument11 SeitenHogquist 2005murat tosunNoch keine Bewertungen
- R 2022 - 代谢Metabolic Profiles &TME of Regulatory TDokument20 SeitenR 2022 - 代谢Metabolic Profiles &TME of Regulatory T1262615286Noch keine Bewertungen
- envhper00522-0014Dokument5 Seitenenvhper00522-0014Afaq AhmadNoch keine Bewertungen
- Transplantation and Rejection 2022Dokument25 SeitenTransplantation and Rejection 2022malkalhmaidh03Noch keine Bewertungen
- Reviews: T Cell-Based Therapies: Challenges and PerspectivesDokument15 SeitenReviews: T Cell-Based Therapies: Challenges and PerspectivesDaniel Luis Meléndez ChoqueNoch keine Bewertungen
- Modulation Mine DocomentDokument6 SeitenModulation Mine DocomentKala KingNoch keine Bewertungen
- PIIS1074761313004378Dokument14 SeitenPIIS1074761313004378ZadorxsNoch keine Bewertungen
- SFGH Infusion Guide RV 05-5-16 FinalDokument17 SeitenSFGH Infusion Guide RV 05-5-16 Finalapi-249622140Noch keine Bewertungen
- Portfolio Presentation DeckDokument44 SeitenPortfolio Presentation Deckapi-338954319Noch keine Bewertungen
- TOFT ThyroidDokument72 SeitenTOFT Thyroidkgrhoads0% (1)
- BPH (Benign Prostatic Hyperplasia)Dokument11 SeitenBPH (Benign Prostatic Hyperplasia)AviZhafiraNoch keine Bewertungen
- Event ProposalDokument2 SeitenEvent ProposalSudhakar Parthasarathy50% (2)
- Ihe680 032911Dokument62 SeitenIhe680 032911cecsdistancelab100% (2)
- Interpretasi Lesi SpesifikDokument27 SeitenInterpretasi Lesi SpesifikSTEFFI MIFTANoch keine Bewertungen
- The Royal Pentagon Review Specialist Inc. Pediatric NursingDokument55 SeitenThe Royal Pentagon Review Specialist Inc. Pediatric NursingDavid PunzalanNoch keine Bewertungen
- World Clinics (Acne) PDFDokument267 SeitenWorld Clinics (Acne) PDFAisyahMuftihaturrahmahNoch keine Bewertungen
- Stop Smoking: A Bad HabitDokument3 SeitenStop Smoking: A Bad HabitPrhameswati Cahyaning KinasihNoch keine Bewertungen
- MiasmsDokument2 SeitenMiasmsAshu Ashi100% (1)
- CTBDokument2 SeitenCTBmike307Noch keine Bewertungen
- Advantages of High-Dose Rate (HDR) Brachytherapy in Treatment of Prostate CancerDokument6 SeitenAdvantages of High-Dose Rate (HDR) Brachytherapy in Treatment of Prostate CancerPiotr JankowskiNoch keine Bewertungen
- Drug-Lab Test Interaction - Full PDFDokument9 SeitenDrug-Lab Test Interaction - Full PDFmanikNoch keine Bewertungen
- A, B, C, Us 2018Dokument6 SeitenA, B, C, Us 2018tulus ikhlasNoch keine Bewertungen
- Hereditary Cancer PanelDokument7 SeitenHereditary Cancer PanelAjay MahalkaNoch keine Bewertungen
- Unit Plan Owaisi College of NursingDokument5 SeitenUnit Plan Owaisi College of NursingSiva0726Noch keine Bewertungen
- Essiac Formula OriginaleDokument16 SeitenEssiac Formula OriginaleRoberto De Jesus100% (1)
- Cholesterol TCM PDFDokument6 SeitenCholesterol TCM PDFMartijn JohanNoch keine Bewertungen
- Macnally 2008Dokument10 SeitenMacnally 2008Saad ELMI MOUSSANoch keine Bewertungen
- 12 Signs of The ZodiacDokument9 Seiten12 Signs of The ZodiacProfessorAsim Kumar MishraNoch keine Bewertungen
- Cholelithiasis: Group MembersDokument12 SeitenCholelithiasis: Group MembersShiela GutierrezNoch keine Bewertungen
- Hand PosturesDokument6 SeitenHand Posturesmadream7Noch keine Bewertungen
- Plantar Wart Treatment and CausesDokument7 SeitenPlantar Wart Treatment and CausesulfaraudhaNoch keine Bewertungen
- Asps 2013Dokument205 SeitenAsps 2013faissal jameel100% (1)
- Determination of Heavy Metals Concentration in Drinking Water of Potiskum Metropolitan, North Eastern, NigeriaDokument9 SeitenDetermination of Heavy Metals Concentration in Drinking Water of Potiskum Metropolitan, North Eastern, NigeriaAli Abubakar AuduNoch keine Bewertungen
- Adrenergic AntagonistsDokument29 SeitenAdrenergic AntagonistsBenedict Brashi100% (1)
- DK Chakra Chart 2017Dokument2 SeitenDK Chakra Chart 2017Tuxita SuryaNoch keine Bewertungen
- Radiography in Veterinary MedicineDokument68 SeitenRadiography in Veterinary MedicineGarry Lasaga100% (1)
- Life Sciences p1 Gr10 QP Nov2019 - Eng DDokument16 SeitenLife Sciences p1 Gr10 QP Nov2019 - Eng DSkhethelo NdlangisaNoch keine Bewertungen