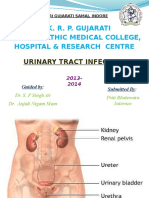
Beruflich Dokumente
Kultur Dokumente
The Investigation and Management of Interstitial Cystitis PDF
Hochgeladen von
Arif AshariOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
The Investigation and Management of Interstitial Cystitis PDF
Hochgeladen von
Arif AshariCopyright:
Verfügbare Formate
Review
The investigation and management of
interstitial cystitis
Matthew Parsons and Philip Toozs-Hobson
Department of Urogynaecology, Birmingham Womens Hospital, Birmingham, UK
Abstract
Interstitial cystitis (IC) is a chronic inflammatory disorder
of the bladder that is notoriously difficult to manage and
can result in considerable morbidity. It very likely overlaps
with painful bladder syndrome, but they are different
conditions. The aetiology remains obscure, and the definition
and diagnostic criteria are debated. The diagnosis of IC
is one of exclusion, frequently based on symptoms and
cystoscopy findings. Typical symptoms include frequency,
urgency, dysuria and lower abdominal, bladder, vaginal,
urethral or perineal pain, in the absence of bacterial cystitis.
Voiding often relieves the suprapubic discomfort, and
drinking alcohol- and caffeine-containing drinks frequently
exacerbates it. Many treatments have been tried, with little
sustained success. Proposed systemic treatments include antihistamines, heparin, amitriptyline and pentosan polysulfate
(a synthetic analogue of glycosaminoglycan which augments
the mucous protective layer of the bladder). In many patients
symptoms are improved following cysto-distension but the
benefits are short-lived. Instillations of dimethyl sulfoxide,
hyaluronic acid or chondroitin also show promise. Where
treatments have failed and symptom severity is such that the
patients quality of life is poor, a urological opinion should
be sought and reconstructive surgery considered. Available
options include partial cystectomy, augmentation cystoplasty,
and urinary diversion with or without cystectomy.
Keywords: Bladder, chronic pelvic pain syndrome, inflammation, interstitial cystitis, painful bladder syndrome
Introduction
Interstitial cystitis (IC) is a chronic inflammatory disorder
of the bladder that is notoriously difficult to manage
and can result in considerable morbidity. It can cause
symptoms of frequency and urgency, with pain as a
predominant feature in one or more regions of the pelvis
typically in the bladder, vagina or perineum leading to a
poor quality of life.
Correspondence: Matthew Parsons, Consultant,
Department of Urogynaecology, Birmingham Womens
Hospital, Metchley Park Road, Edgbaston, Birmingham
B15 2TG, UK. Email: matthew.parsons@bwhct.nhs.uk
132
Painful bladder syndrome (PBS) was defined only in
2002 and thus most of the literature in the area of bladder
pain refers to IC specifically, as will this article.1 There is
undoubtedly some overlap between the two, but at present
they remain distinct.
IC is a disease of extremes extremes of severe symptoms, of underdiagnosis and overdiagnosis, of aetiologic
theories that vary from the abstruse to the fashionable, of
treatment that varies from the alpha of vitamin prescriptions to the omega of radical bladder substitution surgery,
and sadly often, of confusion in medical thinking. 2 The
aim of this article is to summarize the presentation, investigation and treatment of women with IC.
Incidence
Women between the ages of 40 and 60 years are most
commonly affected. The condition occurs more frequently
in Caucasians and there is a 9:1 female predominance. 3
Reported prevalence rates for this condition vary widely
as there is no universally accepted definition.
In the Nurses Health Study I and II (n = 184,583) the
prevalence of IC was 5267/100,000, with no substantial
age variation.4 Although the results must be interpreted
in the light of the higher proportion of Caucasians in
this study than the background population, this is 50%
higher than was previously reported in the United States,
and three times greater than in European studies. In a
Finnish study of 2000 participants randomly selected
from the population register, the prevalence of symptoms
corresponding to probable IC was 450/100,000 (95% CI
100800), which is an order of magnitude higher again. 5
Aetiology
A brief summary is presented of some hypotheses relating
to the management and investigation of IC.6
Bladder wall dysfunction and GAG layer
deficiency
Dysfunctional epithelium in the lower urinary tract
is characterized by damage to the glycosaminoglycan
Journal of the British Menopause Society Vol. 11 No. 4 December 2005
M Parsons & P Toozs-Hobson
(GAG) layer, which leads to increased permeability to
potassium. Potassium can then permeate the bladder
musculature, triggering urgency and bladder contractions.
Mast cell activation is considered to be a result of GAG
layer deficiency, which induces neurogenic inflammation,
with over-expression of neurotransmitters. This proposal
remains controversial and as yet unconfirmed by electron
microscopy.
Infection
An infective cause for IC has been postulated repeatedly
since 1915 (see under Cystoscopy, below). However,
according to the criteria of the National Institute of
Diabetes and Digestive and Kidney Diseases (NIDDK), 3
infection is a cause for the exclusion of a diagnosis of IC,
and an infective agent has not been consistently identified.
Interstitial cystitis
chronic in the absence of urinary tract infection. It may
be experienced as sharp pain or as a pressure, or as a
burning. It may also be related to sexual intercourse. The
relationship of pain to voiding is important pain related
to filling, relieved by voiding, is a classic symptom of IC.
Pain during voiding is more suggestive of urinary tract
infection, vulval or vaginal disorders, or urethral diverticulum.9
Urinary frequency depends on many environmental
factors, and so a normal frequency does not preclude a
diagnosis of IC if urgency is present. In a study of women
with IC, none reported simultaneous onset of urgency, frequency, pain and nocturia. Sixty per cent presented with
urinary tract infection, 27% with urgencyfrequency and
13% with pain.11 All symptoms were present at a mean of
11 months after the onset of the first.
Autoimmune system disorder
Investigation and assessment
Disorders of the immune system are suspected as
being causative because of the association with certain
groups of human leukocyte antigen (HLA), allergies
and autoimmune processes, and sporadic successes with
immune suppressants.
Pelvic examination
Urotoxins
Urinary toxins and allergens have been postulated to be
involved in the deficiency of the GAG layer.
Hypoxia
Vascular, hormonal or toxic factors leading to hypoxia
of the bladder wall could affect the integrity of the GAG
layer. A reduced blood flow in the sub-urothelial layers
(but not deeper layers) has been seen in women with IC.
Clinical features
The symptom complex that should define IC is controversial. Typical symptoms include frequency, urgency, dysuria,
and lower abdominal, bladder, vaginal, urethral or perineal
pain, in the absence of bacterial cystitis. Voiding often
relieves the suprapubic discomfort, and drinking alcoholand caffeine-containing drinks frequently exacerbates the
pain. In a UK-based postal questionnaire survey, 64% of
respondents described daily pain when symptoms were at
their worst, and 37% described daily pain at the time of
survey. Most reported frequency, urgency and nocturia.7
Sixty-seven per cent reported a considerable impact
or more on their lifestyle and 46% reported moderate
depression or worse. Forty-nine per cent reported at least
considerable difficulties with sexual intercourse.
Incontinence tends not to be a major feature of IC,
although 14% of IC patients have evidence of detrusor
overactivity on subtracted cystometry.8 What is not clear
is whether the presenting complaint affects the likely
outcome of treatment.9 At a consensus workshop in Kyoto,
attendees were of the opinion that pelvic pain, urgency
and urinary frequency are necessary for IC to be a differential diagnosis.10
Pain typically occurs in the pelvic area in the bladder,
vagina, urethra, rectum or perineum and must be
Pelvic examination is often unhelpful. The bladder may
be tender on palpation, but examination is rather more
helpful in excluding other conditions that can cause
similar symptoms.
Urine studies
Urinalysis is usually normal but haematuria may be
present. Urine culture is essential to exclude simple
urinary tract infection, as well as atypical infections,
such as Ureaplasma urealyticum, Mycoplasma hominis or
Chlamydia trachomatis.
Urine cytology is recommended if haematuria is
present, or if there are risk factors for bladder cancer
(smoking, age, family history, occupational exposure to
certain industrial chemicals such as aromatic amines). In
a study of 128 women with irritative bladder symptoms,
and a total of 202 urine specimens, no positive cytology
nor transitional cell carcinomas were found in women
without haematuria.12 The cost-effectiveness of urinary
cytology in women without haematuria or risk factors is
therefore questionable.
Questionnaires and symptom scales
There are three published questionnaires for use in IC:
the University of Wisconsin IC Scale, the OLearySant
IC Symptom Index and IC Problem Index, and the Pelvic
Pain and Urgency/Frequency (PUF) Scale.1317
The University of Wisconsin IC Scale has not yet been
validated or published in a format for general use.14,15
However, it addresses quality of life and could therefore
be very useful.
The OLearySant indices were developed using a
focus group and validated using IC patients and asymptomatic controls.16,17 They are useful because they focus
on symptoms of urgency and bladder-related pain, but
they do little to assess the relationship of pain to more
general issues, or sexual activity, on the grounds that these
are not needed to distinguish IC.13
The PUF Scale is the most recently published and
widely studied in a urological and a gynaecological
pain setting. A large study utilizing the PUF Scale has
Journal of the British Menopause Society Vol. 11 No. 4 December 2005
133
M Parsons & P Toozs-Hobson
Interstitial cystitis
concluded that up to 23% of US females have IC, and this
had made many experts wary of its use.18
The general consensus of the expert panel in Kyoto,
and in the opinion of the authors of an expert review
of the literature, is that none of the scales can be used
for diagnosis, although they may be more valuable in
monitoring the progress of treatment outcomes.13,19
Urine markers
Certain markers are significantly increased in IC,
including anti-proliferative factor, epidermal growth factor,
insulin-like growth factor (IGF) binding protein-3 and
interleukin (IL)-6. Anti-proliferative factor is the most
likely candidate to become a diagnostic test, as it gives the
least overlap between the IC and control groups.20
Luminal nitric oxide (NO) can be used as a marker
to differentiate inflammation, which defines IC, from
urgency, frequency, nocturia and pain due to noninflammatory disorders, such as outflow obstruction
and neurogenic dysfunction.21 There is a nearly 20-fold
increase in mean bladder NO concentration in patients
with IC compared with those with detrusor overactivity,
outflow obstruction or sensory urgency, and compared
with asymptomatic controls. Further study has shown a
statistically significant correlation between changes in
symptom/problem scores and changes in luminal bladder
NO in individual patients, which suggests that NO could
be used not only to measure inflammation but also as an
objective evaluation of treatment response.22
Voiding diary
The voiding diary is an important tool in the investigation of lower urinary tract symptoms.23 It may range in
complexity from simple records of intake and output to
more complex diaries that include symptoms and incontinence episodes, and pad use, to facilitate history taking
about the degree of frequency, nocturia and volumes
voided at each episode. They are useful in the identification of polydipsia and polyuria as a cause of urinary
frequency.
In order for a voiding diary to have value, it must be
completed correctly. If the diary is too long then compliance is likely to be poor. Day-to-day variability may
compromise diaries that cover only one or two days,
and so a three-day diary has been suggested as optimal,
although others advocate a single-day diary. 24,25
Patients with IC have lower volumes at first sensation
to void and lower functional capacity than those without
the condition.26 Maximum voided volume is usually taken
to represent functional capacity. A significant positive
correlation has been found between cystometric bladder
capacity and maximum voided volume as recorded in a
home diary (rr = 0.4938, P < 0.01), which establishes the
validity of the latter.27 It is therefore usual to see women
with IC having frequent voids of small volume.
Urodynamics
The role of urodynamics is to rule out alternative
pathology, such as obstructed voiding or detrusor overactivity, rather than to provide a positive diagnosis of IC.
The relationship of uninhibited bladder contractions
with IC or painful bladder syndrome is not clear. In a
134
study correlating cystometric findings with presenting
symptoms, uninhibited detrusor contractions (UDCs)
were seen in 56 of 384 patients (14.6%).26
In practice, the diagnosis of IC is as much a clinical,
symptom-based one as one based on any particular test,
and many believe that urodynamics should be reserved
for selected cases. However, the inclusion of patients in
research studies requires some degree of standardization
and studies using the NIDDK criteria therefore require
urodynamics.
Potassium sensitivity testing (PST)
PST involves a comparison of pain and urgency sensation
after a 5 min instillation of 40 ml water and 5 min instillation of 40 ml 40 mEq/100 ml potassium chloride (KCl)
in the bladder. A positive test result is an increase in
pain with KCl and this occurs in a large majority of IC
sufferers.28 Intravesical PST is thought to be able to
detect abnormal epithelial permeability, and to discriminate patients with IC and normal subjects.29 No specific
urodynamic parameter or specific symptoms can predict
a positive KCl test in patients with frequencyurgency
syndrome or IC. 30
The role of PST is uncertain and is not part of routine
assessment in the UK. It may be useful in the assessment
of pain of uncertain origin but an empirical instillation
of 10 ml 1% lidocaine and 10 ml 8.4% bicarbonate has
been proposed as an alternative. Lidocaine alone is not
absorbed from the bladder and so needs to be alkalinized.
It is a very effective, but short-lasting, treatment for
bladder pain in a rescue scenario; if the pain persists
despite this, the patient is unlikely to get relief from
bladder treatments for IC. 31
Cystoscopy
The pursuit of a definition and diagnosis of IC began in
1887, when Skene described the disease as an inflammation of the bladder that destroyed the mucosa and
extended to the muscular parietes. In 1915 Hunner
described an ulcer. Glomerulations small, discrete,
purple haemorrhages of the bladder mucosa that are often
found in IC patients were first described in 1949.2
The purpose of cystoscopy is the exclusion of local,
intravesical abnormalities. The majority of patients with
IC have a urothelium that appears normal. In a study
of 196 women suffering from frequency, urgency and/or
bladder pain, patients with bladder pain at full bladder
and a positive KCl test had only a 45.2% chance of having
the characteristic appearances of the urothelium at
cystoscopy (see Figure 1). 30
The decision for cystoscopy, then, is frequently based
on the presence or absence of risk factors for conditions
such as bladder cancer. Since 1% of IC patients in a tertiary unit in the United States were eventually diagnosed
with transitional cell carcinoma of the bladder, some
authorities advocate cystoscopy as a mandatory investigation. 32,33
Rigid cystoscopy, under general or regional anaesthesia, is preferred to facilitate biopsy glycine should
be used as a filling medium if diathermy is to be used
for coagulation, although saline is adequate where
Versapoint (Bipolar Electrosurgery System) is available.
Infusion height should be approximately 80 cm above the
Journal of the British Menopause Society Vol. 11 No. 4 December 2005
M Parsons & P Toozs-Hobson
Interstitial cystitis
Box 1 An experts view of the debate31
Mandatory investigations
History and physical examination
Urinalysis, urine culture, urine cytology
Optional investigations
Symptom questionnaires
Cystoscopy
Further investigations for specific patients, where
indicated
PST
Hydrodistension
Bladder biopsy
Urodynamics
Figure 1 Typical appearances of the bladder at cystoscopy in a
patient with IC.
symphysis pubis, using a dripping chamber until the flow
stops, observing for blood vessels, deposits, white spots,
hyperaemia, scars, cracks or mucosal changes.
When maximum capacity is reached, distension is
maintained for three minutes; the bladder is then emptied
and refilled to around a third of the maximum for visualization of haemorrhage and for possible biopsy, which
should include detrusor muscle.
The commonest pathological findings are epithelial
denudation or ulceration, mononuclear inflammation,
oedema, congestion, haemorrhage and mast cell activation. The mast cell count is calculated. If it is over 28/mm 2
it is thought significant, although no histological features
are considered pathognomonic for IC, and the prognostic
features of many inflammatory markers are not known. 33
Mast cell activity, however, is thought to relate to disease
activity, and long term to result in fibrosis. 34
Cystoscopic appearances do not appear to identify
a distinct pathophysiological subset of patients with IC
symptoms. Patients who have typical cystoscopic findings
for IC have worse daytime frequency and nocturia, and a
lower bladder capacity under anaesthesia than those who
do not. However, the groups have similar urine markers
and bladder biopsy findings. 35
Diagnosis
There have been three major conferences in recent years
to try to establish a consensus on the diagnosis of IC:
International Consultation on Interstitial Cystitis Japan
(ICICJ) Kyoto, Japan, 2830 March 2003;36
European Copenhagen Workshop on Interstitial
Cystitis (IC) Copenhagen, Denmark, 2224 May
2003;37
National Institute of Diabetes and Digestive and
Kidney Diseases (NIDDK)/Interstitial Cystitis
Association (ICA) International Scientific
Symposium Virginia, USA, 30 October 1 November
2003. 38
Box 1 provides an experts view of the debate, which is
summarized below.
History and examination
At all three conferences it was agreed that a history of
urgency, frequency and pain associated with bladder
filling is typical of IC, and that systemic and pelvic
examination is necessary to exclude other major
pathology.
Questionnaires and symptom scales
The proceedings of the Kyoto conference record
discussion of the use of Wisconsin IC Scale, the OLeary
Sant IC Symptom Index and IC Problem Index and the
PUF Scale (see above). They were not advocated for
diagnosis but rather as markers of disease progression.1317
At the Copenhagen conference it was decided to use the
OLearySant indices, supplemented by a sexual score
to be defined. The use of voiding diaries and of a visual
analogue scale for grading of pain was agreed. At the
Virginia conference the PUF Scale was advocated as
a screening tool for epidemiological studies, with the
addition of some exclusion criteria.
Urine tests and urodynamics
At both the Kyoto and the Virginia conferences there was
support for the use of urine cytology in the diagnostic
workup, as well as exclusion of infection by culture and
microanalysis of the urine. The need for urodynamics was
discussed at all three conferences at Kyoto and Virginia
it was agreed that urodynamics are not essential. The
proceedings of the Kyoto conference go so far as to state:
not practised, no value, unnecessary, and painful for IC
patients. And, in addition: bladder hyperactivity should not
exclude IC. Yet the majority of attendees voted to include
urodynamic investigation in a future research protocol.
At Virginia, however, it was agreed that a voiding diary
is less expensive and provides more useful information.
At Copenhagen filling cystometry was strongly advocated
for both sexes, with pressureflow studies in men whose
maximum flow rate is less than 20 ml/s.
The Kyoto conference discussed the PST but it was felt
that, in view of the potential bias in some patients, the
Journal of the British Menopause Society Vol. 11 No. 4 December 2005
135
M Parsons & P Toozs-Hobson
Interstitial cystitis
difficulty in interpreting the result and the extreme pain
for severely affected patients, it should not be undertaken.
inhibiting neurological activity;
suppression of allergies.
Cystoscopy and biopsy
Single-drug therapy is sufficient for most patients, but
multi-modal therapy may be appropriate for those with
severe or long-standing disease.
No consensus, nor even a majority decision, was reached
in Kyoto with regard to the need for cystoscopy. There was
agreement, however, that bladder biopsy is not required.
Attendees in Virginia disagreed as to whether cystoscopy
is required and over whether it should not be needed in
epidemiological studies, owing to the difficulty of standardization. The group agreed, however, that cystoscopy
is useful in determining treatment strategies and is
necessary when a patient has a high risk of bladder cancer.
At Copenhagen there was a consensus that bladder biopsy,
including muscle for mast cell analysis, is mandatory.
Terminology
All three conferences debated their dissatisfaction with
the term interstitial cystitis. In Kyoto the suggestion
was that the term chronic pelvic pain syndrome
should be used, but this was countered by the argument
that some women respond to anticholinergics while
others do not, and that this therefore distinguishes an
overactive bladder from IC. The compromise IC as
part of a chronic pelvic pain syndrome (IC/CPPS) was
unanimously agreed. In Virginia there was disagreement
as to whether the term IC detracts from the diagnosis,
but it was felt that, as IC is prevalent in the medical
literature, dropping it would be too confusing. Rather,
it should be coupled with chronic pelvic pain (IC/
CPPB) and eventually IC dropped. In Copenhagen, the
definition provided by the International Continence
Society was adhered to more closely: of painful bladder
syndrome (PBS) as suprapubic pain related to bladder
filling, accompanied by other symptoms, such as
increased daytime and night-time frequency, in the
absence of proven urinary infection or other obvious
pathology.1
Management
Management options can seem as bewildering as the
aetiology to those clinicians trying to manage patients
with IC. Management begins with the first visit and
assessment, by acknowledging and validating the pain,
and setting patient expectations. 39 A wide range of
treatments are frequently offered, but in the UK drugs
of proven efficacy are typically offered to only a third or
less of patients. These include cimetidine (offered to 36%
of patients in one survey); antihistamines (5%); pentosan
polysulfate (12%); dimethyl sulfoxide (DMSO) (33%);
and anticonvulsants (2%).7 The five most commonly used
therapies in the United States have been reported to be
cystoscopy and hydrodistension, amitriptyline, phenazopyridine (a urinary tract analgesic not available in the
UK), special diet and intravesical heparin.40 A total of 183
different types of therapy were recorded!
Pharmacotherapy in IC is based on three principles:
controlling a dysfunctional urothelium by restoring the
mucous/GAG layer with GAG or GAG-like drugs;
136
Behavioural treatments
A major part of the management of patients with IC is
behavioural and non-pharmacological: physical therapy,
avoidance of flare-inducing foods, bladder training,
and stress management techniques can supplement
pharmacological treatment and improve clinical
response.41
Patients need to become involved in their management
and should be offered advice about accurate educational
resources. The Internet provides an easy way to access
a support group42,43 and gain valuable information, for
example regarding diet (dietary modification is usually
recommended, although there is little evidence from
randomized trials as to which foods are best avoided).
Avoiding foods with a high acidity or high potassium
content is desirable (see Table 1).
Hydrodistension
Cystoscopy with hydrodistension is the most commonly
performed diagnostic test and procedure and is thought
to work by disrupting bladder neuronal pathways and thus
pain transmission. The benefits are short-lived: in one
casecontrol series the benefits of hydrodistension lasted
a mean of only two months.44 It is therefore no longer
recommended.
Pharmacological treatments
Dimethyl sulfoxide (DMSO) and intravesical heparin
DMSO, a by-product of the wood industry, has been in
use as a commercial solvent since 1953. DMSO reduces
inflammation by several mechanisms. It is an antioxidant
and a scavenger of the free radicals that gather at the site
of injury; DMSO also stabilizes membranes and slows or
stops leakage from injured cells, and so may be useful in
restoring the GAG layer.
In a study evaluating the effectiveness of DMSO in the
treatment of cystoscopy-positive IC, patients were randomly allocated to receive either 50% DMSO or placebo
(saline) intravesically every two weeks for two sessions of
four treatments each. Subjectively, 53% of DMSO-treated
patients were markedly improved, compared with 18% in
the placebo group. Objectively, 93% of the DMSO group
were improved versus 35% in the placebo group.45 While
there were no significant side-effects, many patients complained of garlic halitus. In 25 patients followed for 12
months the relapse rate was 59%.
Heparin is a GAG that may afford protection to
the urothelium and reduce the relapse rate. It is better
tolerated than DMSO and does not produce garlic
halitus. It is not associated with coagulation anomalies
when administered intravesically, and may be useful in
up to 56% of patients.46 It also produces a significant
reduction in the relapse rate among patients who respond
to DMSO.47
Journal of the British Menopause Society Vol. 11 No. 4 December 2005
M Parsons & P Toozs-Hobson
Interstitial cystitis
Table 1 Dietary advice for IC sufferers 42,43
Food group
Permitted
Best avoided
Beverages
Bottled water, red bush tea, pear juice
Breads
White bread, rice bread/cakes, millet, buck wheat,
matzo, plain pita bread
Refined rice, plain pasta
Butter, margarine, vegetable oils
Soups made with allowed vegetables
Beef, chicken, lamb, veal, flounder, halibut, salmon,
snapper, scallops, sole, mackerel, tuna, crab, shrimp
Alcoholic beverages (e.g. beer and wine), carbonated
drinks such as sodas, coffee, tea, and fruit juices,
especially citrus or cranberry juice
Rye and sourdough bread
Cereals
Fats
Soups
Meat, fish,
poultry
Dairy
Vegetables
Beans
Fruits
Sweets
Dressings,
herbs and
spices
Nuts
Breakfast cereals
Aged, canned, cured, processed or smoked meats and
fish, anchovies, caviar, chicken livers, corned beef, and
meats which contain nitrates or nitrites
Cream cheese, cottage cheese, ricotta
Aged cheeses, sour cream, yogurt
Alfalfa, beets, broccoli, Brussels sprouts, chicory, carrots, Fava beans, lima beans, onions, tofu, soy beans and
celery, cucumber, cauliflower, okra (ladies fingers),
tomatoes
courgette, squash, turnips, kale, leeks
Aduki, navy, chick peas, split peas, kidney, peas
Melons other than cantaloupes, blueberries and pears
Apples, apricots, avocados, bananas, cantaloupes,
citrus fruits, cranberries, grapes, nectarines, peaches,
pineapples, plums, pomegranates, rhubarb, strawberries
and juices made from these fruits
White chocolate, frozen yogurt and milk
Other chocolate, sugars, artificial sweeteners
Rosemary, thyme, oregano, basil, marjoram, fennel,
Mayonnaise, ketchup, mustard, salsa, spicy foods
sage, dill, mixed herbs
(especially such ethnic foods as Chinese, Indian, Mexican
and Thai), soy sauce, salad dressing and vinegar,
including balsamic and flavoured vinegars
Pine, cashews, almonds
Most other nuts
Sodium pentosanpolysulfate
Sodium pentosanpolysulfate is chemically and structurally
similar to naturally occurring GAGs that are produced
by, and form a protective layer covering, the epithelium
of the urinary tract. It inhibits complement reactions in
inflammatory processes.48 It is the only oral agent used to
treat IC that has been rigorously investigated in doubleblind trials.
In a randomized, prospective, double-blind, placebocontrolled study conducted in 148 patients, 32% of the
group on sodium pentosanpolysulfate (100 mg three times
daily) showed significant improvement, compared with
16% on placebo ((P
P = 0.01). They also experienced a significant decrease in pain and urgency ((P
P = 0.04 and 0.01,
respectively).49
Further, in a randomized, double-blind, doubledummy, parallel-group, multicentre, 32-week study of
380 adults examining 300 mg, 600 mg and 900 mg doses
of sodium pentosanpolysulfate, improvement was not
dose dependent. The duration of therapy appeared more
important. 50
Hyaluronic acid and chondroitin sulfate
Hyaluronic acid is an important GAG that is present
throughout the body in connective tissues. In 25 patients
with the characteristic picture of IC and refractory
to other medical treatments, intravesical hyaluronic
acid produced a positive response rate of 56% at week
4, increasing to 71% by week 12. This response was
maintained until week 20; beyond week 24 there appeared
to be a moderate decrease in the effectiveness of the
medication. 51 There was no significant toxicity attributable to the presence of hyaluronic acid in the bladder. In
a small Danish follow-up study, monthly instillation of
sodium hyaluronate solution over three years gave benefit
in approximately two-thirds of patients, and apparent
recovery in 20%. 52
Chondroitin sulfate is a major component of the GAG
layer. An open-label study of chondroitin sulfate was
undertaken to determine the response of patients with IC
and positive PST, of whom 67% responded favourably. 53
Bacilli CalmetteGuerin (BCG)
Case reports suggested that BCG might be effective in the
treatment of IC, by affecting the GAG layer. Non-specific
inflammation of the urothelium is stimulated, which is
thought to promote its regeneration.
In 265 patients with treatment-refractory IC comparing
intravesical BCG to placebo instillations, the response
rate was not significant: 12% for placebo and 21% for
BCG ((P
P = 0.062). 54 A much smaller study (n = 30) found
a 60% BCG response rate, compared with a 27% placebo
response rate. 55 However, in the light of the larger study
this must be interpreted with caution.
Resiniferatoxin
Intravesical instillation of capsaicin, a neurotoxin
extracted from red chilli peppers, has significant effect
over placebo in the treatment of neurogenic detrusor
overactivity. 56 It exerts a biphasic effect on sensory nerves,
with initial excitation followed by a long-lasting block of
C-fibres, which are rendered resistant to natural stimuli. 57
Resiniferatoxin (RTX) is an analogue of capsaicin
extracted from euphorbia, a cactus-like plant. When given
intravesically, it is 1000 times more potent than capsaicin
in stimulating bladder activity; it is hypothesized that
such drugs will be effective in the treatment of IC by
decreasing the pain that leads to urinary frequency and
urgency. However, a randomized controlled trial found no
evidence of efficacy in IC. 58
Journal of the British Menopause Society Vol. 11 No. 4 December 2005
137
M Parsons & P Toozs-Hobson
Interstitial cystitis
Box 2 Key clinical messages
There is a 9:1 female-to-male preponderance.
Typical features are of bladder pain related to filling,
urgency and frequency.
Urine cytology is needed only if haematuria is present,
or where risk factors for bladder cancer are identified.
Urinary tract infection must be excluded.
The role of urodynamics is to rule out alternative
pathology.
The purpose of cystoscopy in the investigation of
bladder pain is the exclusion of local, intravesical abnormalities.
The majority of patients with IC have a normal
appearance to the urothelium.
Non-pharmacological strategies can improve clinical
response.
Sodium pentosanpolysulfate is the only oral agent used
to treat IC that has been rigorously investigated in large
double-blind trials.
Bladder instillations of DMSO may be very effective and
should be considered for use in patients with IC.
Other bladder instillations need further investigation
but show promise.
combination of bladder or pelvic pain, urinary urgency
and frequency.
The investigation is based very heavily on history and
examination clinical investigation is used to exclude
other pathology rather than to make a positive diagnosis.
The management is as varied as the list of possible
aetiologies, as one might expect. It is essential that the
patient be involved with the diagnosis and treatment, as
non-pharmacological dietary and behavioural treatment is
very important. Multiple modes of therapy are frequently
employed, although many of the newer agents have yet to
be subjected to large randomized studies. This reflects the
difficulty of identifying and standardizing a study population for such a heterogeneous condition.
Competing interests: MP has acted as an adviser for and
received support to attend scientific meetings from UCB,
Astellas and BioControl. PTH has received support from
Pfizer, AstraZeneca, UCB, Astellas, Eli Lilly, Molliston,
Gynecare and Galen.
References
Amitriptyline
Amitriptyline is frequently used in pain management. 59
Anecdotal experiences with the tricyclic class of antidepressants suggest that amitriptyline may be an effective
treatment modality in non-ulcerative IC.60 It was shown to
be effective in a small placebo-controlled study.61
Antihistamines
Increased numbers of activated mast cells have been
documented close to nerve endings containing substance
P in the bladders of patients with IC. Bladder mast cells
can be activated by carbachol, while hydroxyzine reduces
carbachol-induced serotonin release from rat bladder.62 In
an open-label case series of patients treated by their local
doctor, a 40% reduction in symptom scores was reported.
This rose to 55% in patients with a history of allergies,
suggesting that hydroxyzine is a useful drug for the
symptomatic treatment of IC, especially in patients with
documented allergies or evidence of bladder mast cell
activation.63 However, larger randomized studies would be
required to support a strong recommendation.
Surgery
Where other treatments fail and symptom severity is such
that the patients quality of life is very poor, a urological
opinion should be sought and reconstructive surgery
considered. Available options include partial cystectomy,
augmentation cystoplasty, and urinary diversion with or
without cystectomy.
Conclusions
IC is a chronic, disabling condition affecting predominantly women (Box 2). The aetiology is uncertain,
but almost certainly multi-modal. It presents with a
138
1 Abrams P, Cardozo L, Fall M, et al. The standardisation of
terminology of lower urinary tract function: report from the
Standardisation Sub-Committee of the International Continence
Society. Neurourol Urodyn 2002;21:16778
2 Hanno P. Definition and Diagnosis: Where We Are in 2003?
Interstitial Cystitis Epidemiology Task Force Meeting, National
Institute of Diabetes and Digestive and Kidney Diseases.
October 2003, Maryland USA. See http://www.niddk.nih.gov/
fund/other/archived-conferences/2003/ic/TaskForce_Meeting_
Report.pdf (last accessed 6 October 2005)
3 Parsons CL. Interstitial cystitis. In: Kursh ED, Mcguire EJ, eds.
Female Urology. Philadelphia: Lippincott, 1994: 42138
4 Curhan GC, Speizer FE, Hunter DJ, et al. Epidemiology of
interstitial cystitis: a population based study. J Urol 1999;161:
54952
5 Leppilahti M, Tammela TL, Huhtala H, Auvinen A. Prevalence
of symptoms related to interstitial cystitis in women: a
population based study in Finland. J Urol 2002;168:13943
6 Kurth KH. Interstital Cystitis. Berlin: Ergebnisse Verlag, 2004:
3953
7 Tincello DG, Walker AC. Interstitial cystitis in the UK:
results of a questionnaire survey of members of the Interstitial
Cystitis Support Group. Europ J Obstet Gynecol Reprod Biol
2005;118:915
8 Irwin PP, Takei M, Sugino Y. Summary of the urodynamics
workshops on IC Kyoto, Japan. Int J Urol 2003;10:S1923
9 Erickson DR. Painful bladder syndrome. In: Cardozo LD,
Staskin D, eds. Textbook of Female Urology and Urogynaecology.
2nd edn. Oxford: Isis Medical Media (in press)
10 Diokno AC, Homma Y, Sekiguchi Y, Suzuki Y. Interstitial
cystitis, gynaecologic pelvic pain, prostatitis, and their epidemiology. Int J Urol 2003;10:S36
11 Porru D, Politano VA, Gerardini M. Different clinical presentations of interstitial cystitis syndrome. Int Urogynecol J Pelvic
Floor Dysfunct 2004;15:198202
12 Duldulao KE, Diokno AC, Mitchell B. Value of urinary cytology
in women presenting with urge incontinence and/or irritative
voiding symptoms. J Urol 1997;157:11316
13 Ito T, Tomoe H, Ueda T, Yoshimura N, Sant G, Hanno P.
Clinical symptoms scale for interstitial cystitis for diagnosis
and for following the course of the disease. Int J Urol 2003;10
(Suppl):S246
14 Keller ML, McCarthy DO, Neider RS. Measurement of
symptoms of interstitial cystitis: a pilot study. Urol Clin North Am
1994;21:6772
15 Goin JE, Olalye D, Peters KM, Steinert B, Habicht K, Wynant
G. Psychometric analysis of the University of Wisconsin inter-
Journal of the British Menopause Society Vol. 11 No. 4 December 2005
M Parsons & P Toozs-Hobson
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
stitial cystitis scale: implications for use in randomized clinical
trials. J Urol 1998;159:108590
OLeary MP, Sant GR, Fowler FJ Jr, Whitmore KE, SpolarichKroll J. The Interstitial Cystitis Symptom Index and Problem
Index. Urol 1997;49 (Suppl 5A):5863
Lubeck DP, Whitmore K, Sant GR, Alvarez-Horine S, Lai C.
Psychometric validation of the OLearySant Interstitial Cystitis
Symptom Index in a clinical trial of pentosan polysulfate sodium.
Urol 2003;57 (Suppl 6A):626
Parsons CL, Dell J, Stanford EJ, et al. Increased prevalence of
interstitial cystitis: previously unrecognized urologic and gynaecologic cases identified using a new symptom questionnaire and
intravesical potassium sensitivity. Urol 2002;60:5738
Nordling J. Interstitial cystitis: how should we diagnose it and
treat it in 2004? Curr Opin Urol 2004;14:3237
Erickson DR, Xie SX, Bhavanandan VP, et al. A comparison of
multiple urine markers for interstitial cystitis. J Urol 2002;167:
24619
Ehren I, Hosseini A, Lundberg JO. Nitric oxide: a useful gas
in the detection of lower urinary tract inflammation. J Urol
1999;162:3279
Hosseini A, Ehren I, Wiklund NP. Nitric oxide as an objective
marker for evaluation of treatment response in patients with
classic interstitial cystitis. J Urol 2004;172:22615
Abrams P, Fenely R, Torrens M. Patient assessment. In: Abrams
P, Fenely R, Torrens M, eds. Urodynamics (1st edn). New York:
Springer, 1983: 627
Palns Hansen C, Klarskov P. The accuracy of the frequency
volume chart: comparison of self-reported and measured
volumes. Br J Urol 1998;81:70911
Mazurick CA, Landis JR. Evaluation of repeat daily voiding
measures in the National Interstitial Cystitis Data Base Study. J
Urol 2000;163:120811
Kirkemo A, Peabody M, Diokno AC, et al. Associations among
urodynamic findings and symptoms in women enrolled in the
Interstitial Cystitis Data Base (ICDB) Study. Urology 1997;49
(Suppl):7680
Diokno AC, Wells TJ, Brink CA. Comparison of self-reported
voided volume with cystometric bladder capacity. J Urol 1987;
137:698700
Parsons CL, Forrest J, Nickel JC, et al. Effect of pentosan
polysulphate on intravesical potassium sensitivity. Urology 2002;
59:32933
Parsons CL, Greenberger M, Gabal L, et al. The role of urinary
potassium in the pathogenesis and diagnosis of interstitial
cystitis. J Urol 1998;159:18627
Kuo HC. Urodynamic study and potassium sensitivity test
for women with frequencyurgency syndrome and interstitial
cystitis. Urol Int 2003;71:615
Nickel CJ. Controversies and trends in the diagnosis and
treatment of interstitial cystitis. Bladder master class, London,
23 June 2005
Tissot WD, Diokno AC, Peters KM. A referral centers
experience with transitional cell carcinoma misdiagnosed as
interstitial cystitis. J Urol 2004;172:47880
Nordling J, Anjum FH, Bade JJ, et al. Primary evaluation of
patients suspected of having interstitial cystitis (IC). Eur Urol
2004;45:6629
Rosamilia A, Igawa Y, Higashi S. Pathology of interstitial
cystitis. Int J Urol 2003;10 (Suppl):S1115
Erickson DR, Tomaszewski JE, Kunselman AR. Do the
National Institute of Diabetes and Digestive and Kidney
Diseases cystoscopic criteria associate with other clinical and
objective features of interstitial cystitis? J Urol 2005;173:937
Bade J, Ishizuka O, Yoshida M. Future research needs for the
definition/diagnosis of interstitial cystitis. Int J Urol 2003;10
(Suppl):S314
Nordling J, et al. European Copenhagen Workshop on
Interstitial Cystitis (IC). Eur Urol 2004;45:6629
Executive Committee Summary and Task Force Meeting Report.
Bethesda, 29 October 2003. See http://www.niddk.nih.gov/
fund/other/archived-conferences/2003/ic/TaskForce_Meeting_
Report.pdf (last accessed 21 October 2005)
Kahn BS, Stanford EJ, Mishell DR Jr, et al. Management of
patients with interstitial cystitis or chronic pelvic pain of bladder
origin: a consensus report. Curr Med Res Opin 2005;21:50916
Interstitial cystitis
40 Rovner E, Propert KJ, Brensinger C, et al. Treatments used in
women with interstitial cystitis: the interstitial cystitis data base
(ICDB) study experience. The Interstitial Cystitis Data Base
Study Group. Urology 2000;56:9405
41 Lukban JC, Whitmore KE, Sant GR. Current management of
interstitial cystitis. Urol Clin North Am 2002;29:64960
42 The Cystitis and Overactive Bladder Foundation. See http://
www.interstitialcystitis.co.uk/medicalconditions/IC/triggerfoods.
htm (last accessed 21 October 2005)
43 The Interstitial Cystitis Association. See http://www.ichelp.
org/TreatmentAndSelfHelp/ICAndDiet.html (last accessed 21
October 2005)
44 Ottem DP, Teichman JM. What is the value of cystoscopy with
hydrodistension for interstitial cystitis? Urology 2005;66:4949
45 Perez-Marrero R, Emerson LE, Feltis JT. A controlled study of
dimethyl sulfoxide in interstitial cystitis. J Urol 1988;140:369
46 Parsons CL, Housley T, Schmidt JD, Lebow D. Treatment
of interstitial cystitis with intravesical heparin. Br J Urol
1994;73:5047
47 Perez-Marrero R, Emerson LE, Maharajh DO, Juma S.
Prolongation of response to DMSO by heparin maintenance.
Urology 1993;41 (Suppl):646
48 Holm-Bentzen M, Jacobsen F, Nerstrom B, et al. A prospective
double-blind clinically controlled multicenter trial of sodium
pentosanpolysulfate in the treatment of interstitial cystitis and
related painful bladder disease. J Urol 1987;138:5037
49 Parsons CL, Benson G, Childs SJ, et al. A quantitatively
controlled method to study prospectively interstitial cystitis and
demonstrate the efficacy of pentosanpolysulfate. J Urol 1993;150:
8458
50 Nickel JC, Barkin J, Forrest J, et al. Randomized, double-blind,
dose-ranging study of pentosan polysulfate sodium for interstitial cystitis. Urology 2005;65:6548
51 Morales A, Emerson L, Nickel JC. Intravesical hyaluronic acid
in the treatment of refractory interstitial cystitis. Urology 1997;49
(Suppl 5A):11113
52 Nordling J, Jorgensen S, Kallestrup E. Cystistat for the
treatment of interstitial cystitis: a 3-year follow-up study. Urology
2001;57 (Suppl 1):123
53 Steinhoff G, Ittah B, Rowan S. The efficacy of chondroitin
sulfate 0.2% in treating interstitial cystitis. Can J Urol 2002;9:
14548
54 Mayer R, Propert KJ, Peters KM, et al. A randomized controlled
trial of intravesical bacillus CalmetteGuerin for treatment
refractory interstitial cystitis. J Urol 2005;173:118691
55 Peters K, Diokno A, Steinert B, et al. The efficacy of intravesical
Tice strain bacillus CalmetteGuerin in the treatment of interstitial cystitis: a double-blind, prospective, placebo controlled
trial. J Urol 1997;157:20904
56 de Seze M, Wiart L, Joseph PA, et al. Capsaicin and neurogenic
detrusor hyperreflexia: a double-blind placebo-controlled
study in 20 women with spinal cord lesions. Neurourol Urodyn
1998;17:51323
57 Maggi CA. The dual, sensory and efferent function of the
capsaicin-sensitive primary sensory neurons in the urinary
bladder and urethra. In: Maggi CA, ed. The Autonomic Nervous
System. Vol. 3: Nervous Control of the Urogenital System. Chur,
Switzerland: Harwood Academic Publishers, 1993: 383422
58 Payne CK, Mosbaugh PG, Forrest JB, et al. Intravesical
resiniferatoxin for the treatment of interstitial cystitis: a
randomized, double-blind, placebo controlled trial. J Urol 2005;
173:15904
59 Bryson HM, Wilde MI. Amitriptyline. A review of its pharmacological properties and therapeutic use in chronic pain states.
Drugs Aging 1996; 8:45976
60 Hanno PM. Amitriptyline in the treatment of interstitial cystitis.
Urol Clin North Am 1994;21:8991
61 van Ophoven A, Pokupic S, Heinecke A, et al. A prospective,
randomized, placebo controlled, double-blind study of
amitriptyline for the treatment of interstitial cystitis. J Urol
2004;172:5336
62 Minogiannis P, El-Mansoury M, Betances JA. Hydroxyzine
inhibits neurogenic bladder mast cell activation. Int J
Immunopharmacol 1998;20:55363
63 Theoharides TC, Sant GR. Hydroxyzine therapy for interstitial
cystitis. Urology 1997;49 (Suppl):10810
Journal of the British Menopause Society Vol. 11 No. 4 December 2005
139
Das könnte Ihnen auch gefallen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (399)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Medical Microbiology and Immunology Flash Cards - Sample ChapterDokument250 SeitenMedical Microbiology and Immunology Flash Cards - Sample Chapterleh.mo931580% (5)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (73)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Mccqe 1Dokument42 SeitenMccqe 1Vinil Saratchandran100% (4)
- CiullaDokument22 SeitenCiullaSalve Rachelle BillenaNoch keine Bewertungen
- Managing The Bladder and Bowel in Spina BifidaDokument77 SeitenManaging The Bladder and Bowel in Spina BifidaSeptinaAyuSamsiati100% (1)
- 2485 4888 1 SM PDFDokument10 Seiten2485 4888 1 SM PDFArif AshariNoch keine Bewertungen
- AfifahDokument2 SeitenAfifahArif AshariNoch keine Bewertungen
- Bhs Inggrs TransletDokument6 SeitenBhs Inggrs TransletArif AshariNoch keine Bewertungen
- Lamp IranDokument4 SeitenLamp IranArif AshariNoch keine Bewertungen
- Nursing ProcessDokument12 SeitenNursing ProcessArif AshariNoch keine Bewertungen
- Medical Standards in Armed Forces Gomilitary - inDokument48 SeitenMedical Standards in Armed Forces Gomilitary - inBharath Kumar100% (1)
- Microbiology - 18Dokument7 SeitenMicrobiology - 18karmylle andradeNoch keine Bewertungen
- Article For Patient EducationDokument4 SeitenArticle For Patient EducationZaveri Hemant GirishkumarNoch keine Bewertungen
- NCMA 219 RLE Course Practicum Unit 1Dokument3 SeitenNCMA 219 RLE Course Practicum Unit 1Nestor Cabacungan100% (1)
- News at NUST: ISAK Donates Measuring Equipment To Sports ScienceDokument8 SeitenNews at NUST: ISAK Donates Measuring Equipment To Sports ScienceRatanang Rj NokoNoch keine Bewertungen
- HS140 Unit 8 Quiz Study Guide-Oct2011Dokument3 SeitenHS140 Unit 8 Quiz Study Guide-Oct2011Crystal MendiolaNoch keine Bewertungen
- Recent Sensitivity Pattern of Escherichia Coli in Urinary Tract Infection - Open Access JournalsDokument3 SeitenRecent Sensitivity Pattern of Escherichia Coli in Urinary Tract Infection - Open Access JournalsWahyunita Fauzi CahyantiNoch keine Bewertungen
- 6 UTI in PediatricsDokument29 Seiten6 UTI in PediatricsYonas AwgichewNoch keine Bewertungen
- Preterm Labor Case Analysis For High Risk AntepartumDokument2 SeitenPreterm Labor Case Analysis For High Risk AntepartumJOPAR JOSE C. RAMOSNoch keine Bewertungen
- AMC Clinicals Sample ExamDokument36 SeitenAMC Clinicals Sample Examadrenaline_medico100% (2)
- Astro Diagnosis ScorpioDokument20 SeitenAstro Diagnosis ScorpioOvn MurthyNoch keine Bewertungen
- Zinnat - Suspension GDS27 IPI07Dokument7 SeitenZinnat - Suspension GDS27 IPI07Kim RNoch keine Bewertungen
- Urinary Tract Infection - FinalDokument86 SeitenUrinary Tract Infection - FinalShreyance Parakh100% (1)
- UTI in PregnancyDokument47 SeitenUTI in PregnancyAli MahmoudNoch keine Bewertungen
- Idsa GuidanceDokument92 SeitenIdsa GuidanceDodo NorNoch keine Bewertungen
- All Case Rate 3 PDFDokument2 SeitenAll Case Rate 3 PDFAmanda JonesNoch keine Bewertungen
- Diagnosis and Treatment of Urinary Tract Infections Across Age GroupsDokument12 SeitenDiagnosis and Treatment of Urinary Tract Infections Across Age GroupsBillyNicolasNoch keine Bewertungen
- MUST To KNOW in Clinical MicrosDokument43 SeitenMUST To KNOW in Clinical MicrosMerhella Amor Suerte MendozaNoch keine Bewertungen
- Activity 3 Part 2 in Microbiology and ParasitologyDokument9 SeitenActivity 3 Part 2 in Microbiology and ParasitologyYael EzraNoch keine Bewertungen
- Interpretations of Test Results in Routine Urine AnalysisDokument2 SeitenInterpretations of Test Results in Routine Urine AnalysisDevaughn Ray PacialNoch keine Bewertungen
- Vesicoureteral RefluxDokument5 SeitenVesicoureteral RefluxHUSAMNoch keine Bewertungen
- PED Concept MapDokument1 SeitePED Concept MapSamantha Smith PetiniotNoch keine Bewertungen
- Trauma InhalasiDokument10 SeitenTrauma InhalasiaziziNoch keine Bewertungen
- 18 - Urological Infections - LR PDFDokument106 Seiten18 - Urological Infections - LR PDFSandraYulianaNoch keine Bewertungen
- Renal Diseases PathophysiologyDokument6 SeitenRenal Diseases PathophysiologyBilly Gayados100% (1)
- What Is Bladder Exstrophy?Dokument6 SeitenWhat Is Bladder Exstrophy?preveennaNoch keine Bewertungen