Beruflich Dokumente
Kultur Dokumente
2016 Healthcare Outlook
Hochgeladen von
eliforuCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
2016 Healthcare Outlook
Hochgeladen von
eliforuCopyright:
Verfügbare Formate
December
December 17,
17, 2015
2015
Americas: Healthcare
2016 Healthcare Outlook: Shifting
Sands
Equity Research
Becoming more selective after five years of outperformance
Getting more selective
Drug pricing debate to intensify
As a confluence of macro, micro and political
vectors shifts the sands under the investment
case for HC after an unprecedented five-year bout
of outperformance, we see the need to get more
selective. Specifically, we see (1) a tougher
backdrop for sector level multiple expansion into
both the election and a rising rates cycle, and as
the sectors relative growth vs. the S&P moderates,
and (2) an increased need to focus on single stock
selection rather than overarching sector themes
(product cycles, utilization, ACA and M&A).
We do not expect Congressional action on drug
pricing but headline risk will persist. Hence, we
prefer a barbell approach, recommending truly
innovative names (BMY, BMRN, REGN) and
generic drug companies which we view as more
defensive (MYL, TEVA, ENDP).
Whats changed?
Going into 2016 we (1) lower exposure to ACA
exchange verticals; we downgrade Providers to
Neutral (remove HCA from CL and downgrade to
Neutral); (2) shift allocations within the Supply
Chain to names with clearer earnings/balance
sheet support as industry tailwinds moderate we
upgrade CAH to Buy and downgrade CVS to
Neutral; (3) add Q to the CL and upgrade the CROs
to Attractive to leverage biopharma innovation;
(4) add MYL to CL as generics will be defensive.
Utilization now a mixed bag
While we expect core HC volumes to improve,
uncertainty over ACA will likely result in investors
paying a lower multiple across HC services
(hospitals, labs). Therefore, we prefer sectors with
more idiosyncratic growth drivers (i.e., product
cycles like MedTech (top-pick: MDT/CL-Buy).
Jami Rubin
(212) 357-7536 jami.rubin@gs.com
Goldman, Sachs & Co.
Asad Haider, CFA
(212) 902-0691 asad.haider@gs.com
Goldman, Sachs & Co.
David H. Roman
(212) 902-7839 david.roman@gs.com
Goldman, Sachs & Co.
Matthew Borsch, CFA
(212) 902-6784 matthew.borsch@gs.com
Goldman, Sachs & Co.
M&A less of a stock driver
Despite substantial firepower, we expect the pace
of M&A to slow after a record year in 2015. That
said, with ballooning cash stockpiles on
Biopharma balance sheets we expect more
strategic (over financial) deals. We update the GS
HC Strategic Assets Basket (GSGLGHSA) as a way
to monitor and implement the M&A theme.
Robert P. Jones
(212) 357-3336 robert.p.jones@gs.com
Goldman, Sachs & Co.
Goldman Sachs does and seeks to do business with companies covered in its research reports. As a result, investors should be aware that the firm may have a
conflict of interest that could affect the objectivity of this report. Investors should consider this report as only a single factor in making their investment decision.
For Reg AC certification and other important disclosures, see the Disclosure Appendix, or go to www.gs.com/research/hedge.html. Analysts employed by non-US
affiliates are not registered/qualified as research analysts with FINRA in the U.S.
The Goldman Sachs Group, Inc.
Goldman Sachs Global Investment Research
Global Investment Research
December 17, 2015
Americas: Healthcare
Table of Contents
GIR Americas Healthcare Team
PM Summary: Shifting Sands
2016 View: Whats Changed
Highest Conviction Single Stock Ideas for 2016
GS Healthcare Team 2016 Subsector Coverage Views + Key Stock Picks
A View from Washington: Marking Time Until the Election
87
Emerging Uncertainties: Key Themes Facing New Challenges
10
Stress Testing the 2016 Pricing Overhang: 6 Key Variables
12
Biopharma Big Picture: 2016 Investable Themes
16
From Launch to Margin: Focus on Expanders
21
Utilization Tug of War: Need For A Product Cycle Overlay
23
The M&A March: Slower, Selective, More Strategic
26
Disclosure Appendix
35
Goldman Sachs Global Investment Research
December 17, 2015
Americas: Healthcare
GIR Americas Healthcare Team
Pharmaceuticals
Jami Rubin
1-212-357-7536
jami.rubin@gs.com
1-212-902-0691
asad.haider@gs.com
1-212-902-7839
david.roman@gs.com
1-212-357-3336
1-212-934-4218
robert.p.jones@gs.com
stephan.stewart@gs.com
1-212-902-6784
matthew.borsch@gs.com
Sector Specialist
Asad Haider, CFA
Medical Technology
David H. Roman
Healthcare Supply Chain
Robert P. Jones
Stephan Stewart, CFA
Managed Care & Facilities
Matthew Borsch, CFA
Life Science Tools & Diagnostics
Isaac Ro
1-212-902-6393
isaac.ro@gs.com
1-212-357-5057
1-212-934-4204
terence.flynn@gs.com
salveen.richter@gs.com
Biotechnology
Terence Flynn
Salveen Richter
Specialty & SMID Pharmaceuticals
Gary Nachman
1-212-855-7725
gary.nachman@gs.com
1-202-637-3746
alec.phillips@gs.com
Washington Research
Alec Phillips
Goldman Sachs Global Investment Research
December 17, 2015
Americas: Healthcare
PM Summary: Shifting Sands
While it may not feel like it given the recent rollover in the sector, HC is on track to clock its fifth consecutive year of outperformance
in 2015 (XLV +3% ytd vs. S&P down -2%; HC third best S&P sector ytd), a feat achieved for the first time in the last 25 years. In
subsectors, managed care fared best, partly on the M&A/consolidation theme, while laggards were spec pharma (on business
model risk) and hospitals (as ACA trades reversed, and the high leverage factor theme hurt). Five of the sectors 10 best performers
(BSX, REGN, EW, LLY and BMY) were product cycle / pipeline stories, an investable theme that we think will be increasingly
important for the forward outlook, irrespective of the direction of the overall HC tape. As we look toward 2016, we see a confluence
of macro, political and micro vectors shifting the sands beneath the bull case. On the macro, three factors will be top of mind.
1.
The election. Performance data going back to 1976 shows that, in election years, (i) HC has outperformed less than 40% of
the time and (ii) the median HC stock delivers 200 bp of underperformance. The overlay of the tough pricing rhetoric
coming out of DC could keep the bar high for multiple expansion.
2.
Interest rates. Analogs show HC lags with other defensive/dividend yielding areas a rising rates environment. While we
believe the yield component has been only a partial factor in HCs 5 year bout of outperformance micro themes have
mattered equally, if not more we see rotational risk as generalist PMs re-evaluate sector level allocations as we move into
an elongated period of potential interest rate hikes from the Fed for the first time in nearly a decade.
3.
Relative growth. Part of the bull angle on HC has been its superior sales growth relative to the S&P (11% in 2015 vs. S&P
3%) though it is worth noting that this gap narrows in 2016 to 7% for HC vs. 6% for the S&P, per GS equity strategy
which could make the sector relatively less compelling. FX is likely to also remain a key swing factor continuing USD
strength could be a headwind. GS currency strategists see USD/EUR rates of 0.95 by year-end 2016 (vs. current 1.1).
Further, the following sector-level/micro themes are likely to remain areas of debate.
4.
Drug pricing. The risk of legislative changes in 2016 is low, though our Washington DC strategist notes that at some point
down the road, discounts/rebates for Medicare (particularly Part B) could get traction even under a Republican Congress. As
we enter what could be a multi-year debate on branded pricing vulnerabilities, we believe that market should and will
place a premium on true innovation (cancer and orphan disease). With regards to generic pricing, we expect the
moderation in generic prices experienced in 2H15 could continue into next year, potentially driving a return to single-digit
price deflation, driven by a declining GRx FDA backlog and greater government scrutiny on select outsized price increases.
5.
Utilization. As the ACA-fueled components of recent utilization strength anniversary in 2016, the onus will be on macro /
cyclical factors to carry HC utilization higher. While we expect core HC volumes to improve in 2016, we believe uncertainty
over the outlook for ACA coverage will result in investors paying a lower multiple across HC services (hospitals, drugstores,
labs) for core utilization growth vs. the ACA bonanza of the last few years.
6.
M&A. Despite substantial balance sheet firepower across the sector, we expect the pace of M&A going forward to slow
after a record year in 2015. Notably, acquirer stocks have now started to underperform after deals are announced.
The net effect of these moving parts create a tougher backdrop for further sector level outperformance, in our view, and an
environment in which stock selection will become ever more important. With this outlook piece, we make several changes
that now reflect a more Neutral view on Healthcare.
Goldman Sachs Global Investment Research
December 17, 2015
Americas: Healthcare
2016 View: Whats Changed
We maintain our Attractive views on Medical Devices, Life Science Tools, Large Cap Pharma, Generics, Specialty pharma (due to
low valuations), but maintain Neutral views on Biotech (Large and Smid-cap), and Drug Chains. The following changes that drive
our views for 2016.
1.
2.
Lowering exposure to ACA exchange verticals, as we expect uncertainty to persist.
We lower our Providers coverage view to Neutral from Attractive, as we believe uncertainty over the sustainability of ACA
exchanges may persist through 2016. We remove HCA from the CL and downgrade it to Neutral. That being said, we favor
ambulatory surgery centers ASC`s as they have very low Medicaid/uninsured/self-pay exposure and are continuing to
benefit from secular trend of surgeries moving from inpatient to outpatient setting with that, we upgrade SCAI from
Neutral to Buy (AMSG already Buy rated) We also recently downgraded Labs to Neutral from Attractive, as we believe
consensus now captures a reasonable base case for underlying volumes and the negative shift in tone around ACA presents
an overhang that could weigh on multiples.
We maintain our Neutral coverage view of Managed Care, as we do not have a basis to predict the outcome of what we
think will be key sector catalysts in the outcomes for regulatory review of the pending mergers (i.e., Big 5 to Big 3) nor
how evolution of the Presidential election will influence the market view of health policy and regulation. Cigna and WellCare
remain our favorites within the managed care group.
Shifting allocations within the HC Supply Chain as industry tailwinds moderate.
3.
Gaining more exposure to verticals leveraged to accelerating biopharma R&D spend
4.
In the Supply Chain/Drug Retail, we upgrade CAH to Buy and downgrade CVS to Neutral. We believe recent Supply Chain
industry tailwinds are moderating (new generic launches, GRx inflation), reimbursement/margin pressure is becoming more
acute for the drug retailers, and though we expect a modest improvement in drug volumes off an easy compare, Rx volumes
will not in our opinion drive sector performance. Against this backdrop we prefer names with (1) idiosyncratic EPS upside, (2)
proven returns discipline, and (3) balance sheet flexibility. As a result, we upgrade CAH to Buy and remain Buy on MCK.
We raise our CRO coverage view to Attractive from Neutral as pharma pipeline growth and strong biotech funding have
driven above-trend bookings ytd, which we expect will drive accelerating sales growth across the group. Also, despite the solid
fundamental outlook, the group trades at an attractive relative valuation. In addition, we add Q to the CL given recent
underperformance since 3Q, and note its group leading 12-month book-to-bill and growing balance sheet flexibility. Our CRO
coverage view change comes on the back of our recent upgrade of Tools which was partly driven by our constructive view on
accelerating biopharma end markets, better outlook for NIH funding (TMO top pick).
Adding MYL to CL, as we see generic drug makers as more insulated from threats of pricing pressure.
We see generic drug makers as more insulated from threats of pricing pressure as their generic portfolios have been less
exposed to outsized price increases. We add MYL to the CL given its strong 2016-2018 outlook and dislocated share price in
the aftermath of the unsuccessful PRGO bid. Our view is that management is likely to be incentivized to repair its bruised
relationship with shareholders, creating a positive set up for the stock.
The following page summarizes our 2016 subsector coverage views and top stock picks.
Goldman Sachs Global Investment Research
December 17, 2015
Americas: Healthcare
Highest Conviction Single Stock Ideas for 2016
Exhibit 1: Our highest-conviction ideas for 2016
GS Healthcare stocks on the Americas Conviction Buy list, bolded companies are recent additions
Price
Target
Price
(12/15/15)
Upside
Major Pharmaceuticals
$80
$56.39
42%
Terence Flynn, PhD
Large-Cap Biotech
$213
$162.62
31%
BMY
Jami Rubin
Major Pharmaceuticals
$80
$70.22
14%
Bullish on BMY's positioning for Opdivo and larger I-O franchise
HOLX
Isaac Ro
Diagnostics
$49
$38.50
27%
Tomo product cycle underappreciated, with execution from management on de-leveraging
and operating improvements
MDT
David H. Roman
Medical Devices
$90
$77.10
17%
Strong organic growth with multiple growth drivers for future organic growth and cash
optionality
MYL
Jami Rubin
Generic Pharmaceuticals
$65
$53.99
20%
Robert P. Jones
CROs
$86
$68.93
25%
TMO
Isaac Ro
Life Science Tools
$158
$137.49
15%
ZLTQ
David H. Roman
Medical Devices
$44
$27.60
59%
Ticker
Analyst
Coverage Group
ABBV
Jami Rubin
AMGN
GS View
Bullish on stability and growth of Humira, growth from the pipeline, and the potential to
increase margins
New product cycles (PCSK9), pipeline readouts (Romo) and internal biosimilars will allow it
to replace the potential revenue that could be lost to competition and drive outer-year
growth
Strong 2016-2018 outlook, dislocated share price and insulation from pricing
pressure
Attractive entery point in light of: (1) Sector leading book-to-bill, supporting
accelerating revenue growth, (2) limited client concentration, and (3) growing balance
sheet flexibility.
Underappreciated growth story with an improving balance sheet which stands to benefit
from improving fundamentals in Tools' end markets
Differentiated asset avoiding negative controversies (i.e., drug pricing, utilization
uncertainty) with exposure to new product cycles and consumer spending patterns
Source: Factset, Goldman Sachs Global Investment Research.
Goldman Sachs Global Investment Research
December 17, 2015
Americas: Healthcare
GS Healthcare Team 2016 Subsector Coverage Views + Key Stock Picks
Exhibit 2: Key Themes Across our Coverages
Coverage Group
Analyst
Coverage View
Buys
Key Themes
Major Pharmaceuticals
Jami Rubin
Attractive
BMY (CL), ABBV (CL)
New product cycles, margin expansion, pricing dynamics, and capital allocation,
Generic Pharmaceuticals
Jami Rubin
Attractive
MYL (CL), TEVA
Specialty & SMID Pharmaceuticals
Gary Nachman
Attractive
ANAC, ENDP, SAGE, HZNP,
PCRX
Execution on recent deals, driving growth in niche specialty areas, new product
launches and data, selective and strategic consolidation
Medical Devices
David H. Roman
Attractive
MDT (CL), ZLTQ (CL), BSX,
EW, ISRG, SYK
New product cycles, margin expansion, stabilizing utilization
Tools
Isaac Ro
Attractive
TMO (CL), A, FEIC, VWR
CROs
Bob Jones
Attractive
Q (CL), INCR
Large Cap Biotech
Terence Flynn
Neutral
AMGN (CL), DMTX, FGEN,
REGN, VRTX
New product cycles, pricing dynamics, pipelines and capital allocation
SMID Biotech
Salveen Richter
Neutral
BMRN, BLUE, INCY, KITE
R&D innovation in key areas (e.g. oncology, gene therapy, rare disease),
Pipeline catalysts, M&A (acquisition targets for Pharma and Large-Cap Biotech)
Medical Supplies
David H. Roman
Neutral
BAX, BCR
Labs and Services
Isaac Ro
Neutral
Improving utlization, capital allocation, pricing pressure and reimbursement
reforms
Drug Chains
Robert P. Jones
Neutral
Reimbursement/margin pressures, solid Rx volumes, implications of
consolidation, remaining purchasing synergies
Supply Chain
Bob Jones
Neutral
BKD, MCK, CAH
HCIT
Bob Jones
Neutral
CERN, EVH
Diagnostics
Isaac Ro
Neutral
HOLX (CL), ALR
Facilities
Matt Borsch
Neutral
LPNT, AMSG, ADPT, SCAI,
EVHC
Healthcare reform impact on payer mix, volumes, bad debt reduction, industry
consolidation, outpatient vs. inpatient
Managed Care
Matt Borsch
Neutral
CI, WCG
Pace of cyclical utilization recovery, ACA exchange sustainability, final stages
in large-scale M&A, pricing, "margin over membership"
Dental
Bob Jones
Neutral
ALGN
Industry consolidation, specialty generics, defensive
Improving fundamentals across biopharma R&D and academic end markets,
muted implications from energy end market volality, consolidation
Solid bookings momentum, robust biotech funding trends. further margin
expansion potential, growing balance sheet flexibility
Leverage to macro healthcare growth and utilization, restructuring, capital
allocation
Moderating generic inflation and generic launch schedule, inorganic wholesaler
earnings support, PBM pricing, large MCO relationship overhangs
Duration of the EMR replacement cycle, accelerating Population Health
adoption, increasing outsourced services penetration
Reimbursement overhangs, product cycles, turn-around stories
Implications of proposed XRAY/SIRO merger on competitive landscape,
continued solid dental/vet consumables growth, uneven equipment growth
Source: Goldman Sachs Global Investment Research.
Goldman Sachs Global Investment Research
December 17, 2015
Americas: Healthcare
A View from Washington: Marking Time Until the Election
2016 election likely to overshadow near-term policy discussions
This section written
by GS Washington
analyst Alec Phillips
Health-related legislative activity in 2016 should be minimal. In our view, there is a low likelihood that significant changes to health
laws will be made in 2016, as the election campaign crowds out policy debates. That said, we will be following two sets of legislative
issues.
1.
Any further changes to the Affordable Care Act. Congressional Democrats have opposed efforts to make significant
changes to the ACA since it was enacted in 2010, but have now embraced changes to certain aspects of the law. The
recently announced omnibus spending and tax bill, which we expect to become law shortly, includes several notable
changes: (1) it delays implementation of the Cadillac tax from 2018 to 2020, suspends the 2.3% tax on medical device
sales for 2016 and 2017, and suspends the tax on health insurance premiums for 2017. That bill will also extend last years
requirement that CMS implement the risk corridor for plans in ACA exchanges on a budget-neutral basis, which will limit
CMSs ability to make the full payments. Since these changes look likely to be enacted in the near term, we would expect
little additional action on them over the coming year, though we would expect these and other ACA provisions to become a
focus again following the presidential election.
2.
Drug pricing-related policies. In the near-term, we do not envision Congress taking action on drug pricing-related issues,
mainly because there is little consensus on what should be done and because there is no clear catalyst/deadline for action.
That said, it seems likely that pressure can also be brought to bear by investigating specific practices deemed to be
problematic. Congressional committees have begun to hold hearings looking into drug pricing practices, and more are
likely. That said, the committees most focused on the issue thus far do not actually have jurisdiction over health laws and
such investigations are more likely to result in headlines than systematic reforms.
The major focus in 2016, however, is likely to be the presidential election. While former Secretary of State Hillary Clinton has a clear
lead for the Democratic nomination, we expect the outlook for the Republican nomination to be uncertain for at least several months,
and possibly all the way until the Republican convention in July. Control of Congress will be an important consideration, since
nearly all major health-related proposals would need to go through Congress to become law. In light of the seemingly high
probability that Republicans will maintain their House majority and a roughly evenly matched contest for Senate control, we are
focused primarily on general election outcomes involving a divided government similar to the current situation (though perhaps
with slightly greater influence via a slim Democratic majority in the Senate) or the possibility of an all-Republican government.
Pricing debate in focus low likelihood of changes without bipartisan support
The price of health care has become a more salient political issue, displacing Obamacare as the top health-related voter concern in
several recent public opinion polls. Recognizing this, the issue has become a focus on the campaign trail. A variety of proposals
have been discussed, which we think of in three categories.
1.
Goldman Sachs Global Investment Research
Regulatory efforts. Several Republican candidates have suggested that reforming the FDA approval process and reducing
regulatory burdens could lower drug prices. By contrast, Clinton has proposed to increase competition for specialty
biologics by lowering the exclusivity period from 12 years to 7 years and giving priority review to biosimilar applications.
8
December 17, 2015
Americas: Healthcare
While we would not expect congressional Republicans to support changes to exclusivity periods, it is possible that they
would support an incremental increase in funding to ease the approval process.
2.
Reforming public-sector purchasing. Clinton, like President Obama and most congressional Democrats, supports allowing
Medicare to negotiate prices for drugs and biologics and applying the Medicaid rebate to Medicare beneficiaries eligible for
both programs. Republicans generally oppose plans such as these that rely on the governments purchasing power to set
prices. That said, they have generally supported efforts to reduce Medicare spending, so incremental changes to Medicare
drug pricing that reduce federal spending cannot be ruled out over the longer run.
3.
Intervening in market pricing and practices. While Republicans have generally opposed intervening in private-sector pricing
decisions, Clinton has proposed capping out-of-pocket costs for drugs to treat chronic or serious health conditions to
$250/month. She has also proposed to spend a minimum level on R&D or pay tax penalties, essentially representing a
maximum allowable profit margin, similar to the minimum medical loss ratio established for health insurers under the ACA.
However, most of these changes would be impossible to implement without bipartisan support.
1.
Major policy changes generally require legislation. Nearly all of the changes discussed above would require legislative
approval in Congress. In some cases, the federal government lacks the authority to require the changes, while in other
cases (such as Medicare price negotiation) the law specifically prohibits the proposed policy and would need to be changed.
Most observers expect Republicans to maintain their control of the House of Representatives, so even in a scenario in which
Clinton wins the White House with a Democratic-majority Senate, the House would be likely to block most of her proposals.
2.
More incremental changes can be accomplished via regulation, which does not require congressional approval. The
candidates have not proposed much in this area, but there has nevertheless been some discussion among market
participants of what CMS could do unilaterally. While CMS is prohibited from interfering in prices for drugs covered under
Medicare Part D and drugs under Part B are paid based on average sales price (ASP), the Center for Medicare and Medicare
Innovation (CMMI) has authority to establish pilot projects to develop alternative reimbursement systems, and plans do so
for oncology drugs. If successful (measured in this case as generating savings without any deterioration in quality, or
increasing quality without increasing spending) CMMI could expand its system more broadly over time. While this could
over the long run create some pressure on high cost medications, this would probably take several years and the actual
savings could turn out to be modest.
Goldman Sachs Global Investment Research
December 17, 2015
Americas: Healthcare
Emerging Uncertainties: Key Themes Facing New Challenges
The healthcare sector has been a huge beneficiary of several important key drivers that have powered the groups upward and long
lasting momentum. Strong new product cycles, with higher quality products versus past cycles, volume tailwinds from healthcare
reform and strengthening utilization, and a favorable M&A environment have supported tremendous optimism among investors.
But headwinds are lurking in the distance, some more obvious than others that we think could represent new challenges.
1.
Pricing debate likely to grow during election year, taking some wind out of new product sails. Our expectation is that little
if anything will happen to branded drug pricing over the next couple of years, given that modifications for Medicare (dual
eligible) and other strategies that would curb drug pricing would require Congressional legislation, and even under a
potential Clinton administration, Congressional gridlock is the most likely outcome. But headline risk is likely to persist as
specialty drug pricing has led to higher co-pays where consumers bear a larger amount out of pocket and the cost of cancer
drugs and other specialty drugs (hepC) has garnered a lot of attention. Moreover, we see a potential black swan scenario
where the administration has the ability to enact broad restrictions on drug pricing through either the Independent Payment
Advisory Board (IPAB) and/or the Center for Medicare and Medicaid Innovation (CMMI). Offsetting the headline risk is
continued new product momentum, which remains very strong. We prefer a barbell approach, recommending truly
innovative companies that deliver drugs that justify their price tag (BMY, AMGN, ABBV, BMRN and REGN) and generic drug
companies which we view as more defensive (MYL, TEVA, ENDP).
2.
Utilization now more mixed; ACA has become a headwind. We expect further cyclical recovery in core (non-ACA)
utilization in 2016, consistent with our modeling of trend relative to the timing of prior economic cycles. We believe the
balance of 2014-2015 results show an upswing in utilization trend. While so far more gradual as compared to what we have
seen in prior economic cycles, our model favors some acceleration in 2016. At the same time, we are unsure if there will be
any meaningful contribution to volumes from the ACA next year, given the uncertainty over the outlook for and
sustainability of the ACA exchanges. Also, we do not have visibility on further ACA Medicaid expansion, which seems likely
to remain uncertain for most or all of next year. The implications of this mix of factors (cyclical recovery but without
contribution from the ACA) differ by sector. We think the net impact of this mix of factors will still be positive for most
volume-sensitive healthcare names (as cyclical recovery offsets lack of contribution from the ACA, at least relative to
expectations) but net negative for hospitals (given their bottom-line leverage to coverage expansion) unless and until
uncertainties on the outlook for ACA coverage are resolved. More immediately, we continue to expect strong volumes in
4Q2015 driven by an acceleration in the seasonal trend towards year-end loaded utilization that has come with the
increased penetration of high deductible plans), which we think bodes well for a number of sectors in 4Q earnings,
including Providers, MedTech, Diagnostics, and Labs.
For MedTech the presence of a stable macro backdrop should allow for new products to contribute incremental growth and
yield accelerating EPS growth in 2016. Specifically, we see the sector generating organic revenue growth in the 4%-7%
largely on the heels of new market growth with the biggest opportunities in cardiology and neurology. On the earnings line,
abating FX headwinds, improving price (i.e., less negative), and favorable mix should all contribute to better EPS growth in
2016 when compared to 2015. These factors combined with favorable valuation make the setup in MedTech compelling, in
our view.
3.
Goldman Sachs Global Investment Research
M&A: Waning appetite for deals (esp. financial), but companies still have firepower. We expect the pace of M&A going
forward to slow given that acquirers stocks have been starting to underperform again after deals are announced. Since
10
December 17, 2015
Americas: Healthcare
2012, we have seen acquirers across all sectors (except Energy) being rewarded for M&A with aggregate stock
outperformance post deal announcements. However, the magnitude of outperformance has steadily declined over the last
several years with 2015 just barely positive, and we are seeing a gradual reversion to the mean when considering that
acquirers stocks underperformed in almost every year in the period 1996-2011. Specifically in healthcare, when comparing
stock performance for acquirers in 2015 vs. 2014 it is clear that 2015 has been a lot more challenging (in biopharma and
managed care acquirers actually underperformed significantly this year).
When considering healthcare M&A overall, we believe investors have most soured on deals that have been primarily for
financial engineering purposes (e.g., acquiring mature assets and driving accretion largely through price increases and/or
cost synergies, tax inversions, etc.). Where we expect companies to be more focused with their M&A going forward is on
strategic deals that add new products or technologies that have very good durability, and that will be significant
contributors to long-term organic growth even if they are less accretive in the very near term.
Many companies within healthcare continue to have significant firepower on their balance sheets despite the substantial
amount of deal making over the last few years. Average net leverage ratios across all of healthcare have been continuing to
climb, but are still at a reasonable level in our view at roughly 1.4x. There are some concerns that a higher interest rate
environment may impede future deals, but what we have seen in the past is that higher rates are typically associated with a
growing economy and with higher consumer and business confidence that ultimately drives greater M&A activity. As such,
we do not believe a higher interest rate environment in and of itself would necessarily change the level of M&A in
healthcare. If borrowing costs do end up going higher, stock deals could also potentially be a trend that continues to
increase going forward.
Goldman Sachs Global Investment Research
11
December 17, 2015
Americas: Healthcare
Stress Testing the 2016 Pricing Overhang: 6 Key Variables
US drug prices have moved to the forefront as a result of political posturing ahead of elections. While we do not expect
broad changes to pricing dynamics in the US near-term, we wanted to use sensitivity analyses to gauge the impact of
several potential scenarios to provide a framework. The three scenarios we considered are (1) if rebates on dual eligibles
(beneficiaries eligible for both Medicare and Medicaid) are stepped up to Medicaids rebate (minimum rebate of 23.1%), (2) apply a
rebate across all Medicare drugs, which could start low and grow over time or (3) if the ability to take price increases in the US
became nearly impossible (similar to the current situation in Europe). We walk through each of these below.
Cancer drugs, orphan
disease, and generic
drugs are likely to be
more insulated from
political pressure
With pricing concerns as the backdrop for 2016 we identify a few areas we believe will be more insulated. First we expect less price
pushback on cancer drugs due to the recent innovation, life-saving technology and often favorable distribution channels through
hospitals. We believe BMY (CL-Buy) is well positioned to benefit from strong volume growth as its key drug Opdivo expands into
additional indications. Secondly, ALXN (Neutral), BMRN (Buy) and VRTX (Buy) are well position, as orphan drug pricing is likely to
be more insulated given it represents a smaller proportion of total drug spend and often deliver significant benefits to patients.
Thirdly, we see generic drug makers, MYL (CL-Buy) and TEVA (Buy) as more insulated from threats of pricing pressure as their
generic portfolios have been less exposed to outsized price increases.
1.
Sizing the potential impact from Dual Eligible changes. Drug manufacturers enjoyed large windfalls under the creation of
Medicare Part D as dual eligible beneficiaries (eligible for both Medicare and Medicaid) were stepped up to the higher Part
D prices. The Committee on Oversight and Government Reform estimates the industry received a gain of almost $7bn in the
first two years post the enactment of Part D. The debate is whether or not the rebates on dual eligibles be stepped up to
Medicaids rebate (minimum rebate of 23.1%). This proposal has been discussed in almost every broad healthcare cost
cutting debate, but has never been acted on. Nevertheless, with the potential for changing political winds, we revisit the
exposures of each of our biopharma companies (Exhibit 3).
2.
Broad rebate applied to all Medicare drugs. Beyond dual eligibles another potential way in which the US government could
look to control drug costs could be via a broad rebate applied to all Medicare drugs. This could start low and grow over
time. However, this would likely be much more complicated than making changes to dual eligible rebates as there is no
framework/policy in place. Below we provide estimates for Medicare exposure across our coverage universe (Exhibits 4-5).
3.
What if US pricing power disappears? Finally we wanted to explore the scenario where the ability to take price increases in
the US becomes nearly impossible (similar to the current situation in Europe). While we think this is an extremely low
likelihood scenario, we ran an analysis on two of our companies for illustrative purposes. Clearly the bottom-line impact is
more significant than the top line (Exhibit 6).
Goldman Sachs Global Investment Research
12
December 17, 2015
Americas: Healthcare
Exhibit 3: Assessing the impact of a dual eligible step up to Biopharma 2015 EPS
Impact on EPS evaluated based on a 23% rebate to estimated Medicare Part D Exposure
$18.00
2015E EPS
EPS Impact
-20%
EPS Impact (%)
-18%
$16.00
-16%
$14.00
-14%
$12.00
-12%
$10.00
-10%
$8.00
-8%
$6.00
6.8%
-6%
4.9%
$4.00
-4%
$2.00
2.2%
2.5%
1.9%
1.5%
1.8%
1.2%
-2%
1.6%
1.2%
0.3%
$0.00
BMY
LLY
ABBV
MRK
JNJ
PFE
BXLT
GILD
CELG
Pharma
BIIB
AMGN
0.0%
0.0%
ALXN
REGN
-0%
Biotech*
*Non-GAAP EPS for ALXN, AMGN, BIIB, CELG, GILD, and REGN
Source: Goldman Sachs Global Investment Research
Exhibit 4: Government exposure as a % of 2014 revenue in Pharma
Exhibit 5: Government exposure as a % of 2014 revenue in Biotech
Based on GS estimates if company data not available
Based on GS estimates if company data not available
Company
Total
Medicare/Medicaid
Exposure
ABBV
BMY
BXLT
JNJ
LLY
MRK
PFE
AGN
12%
36%
24%
12%
26%
16%
23%
25%
ABBV: based on Humira sales
Exposure as a % of Revenue
Medicare
Part B
Medicare
Part D
1%
10%
22%
5%
9%
5%
15%
1%
11%
26%
1%
7%
17%
11%
8%
24%
Exposure (as a % of 2014 Revenue)
Total Government
Exposure
Medicaid
Medicare
Medicare
Part D
Medicare
Part B
ALXN
7%
6%
1%
0%
1%
AMGN
30%
4%
27%
5%
22%
BIIB
18%
2%
16%
8%
9%
CELG
35%
3%
32%
32%
0%
GILD
44%
26%
18%
18%
0%
REGN
44%
2%
42%
0%
42%
VRTX
9%
9%
0%
0%
0%
Medicare Part B/D breakdown estimated based on molecule type
AGN: based on proforma estimates (excluding generic sales)
LLY and MRK: based on 2013 figures
Source: Company data, Goldman Sachs Global Investment Research.
Goldman Sachs Global Investment Research
Source: Company data, Goldman Sachs Global Investment Research.
13
December 17, 2015
Americas: Healthcare
Exhibit 6: Pricing Sensitivity Lilly and Celgene
Impact of the removal of price growth on Sales and EPS for LLY and CELG
$30,000
$10,000
8% Impact
$8,000
$6,000
$4,000
Current Estimates (CAGR = 15%)
Flat Pricing Estimates (CAGR = 12%)
$2,000
$0
CELG EPS Estimates
FY14
FY15
FY16
FY17
LLY Sales Estimates
$12,000
$25,000
$20,000
$10,000
$0
FY14
$7.00
$7.00
$6.00
$5.00
15% Impact
$4.00
$3.00
$2.00
Current Estimates (CAGR = 19%)
Flat Pricing Estimates (CAGR = 14%)
$1.00
$0.00
Current Estimates (CAGR = 5%)
Flat Pricing Estimates (CAGR = 4%)
$5,000
$8.00
$6.00
9% Impact
$15,000
FY18
LLY EPS Estimates
CELG Sales Estimates
$14,000
FY15
FY16
FY17
FY18
FY19
FY20
$5.00
$4.00
30% Impact
$3.00
$2.00
Current Estimates (CAGR = 15%)
Flat Pricing Estimates (CAGR = 8%)
$1.00
$0.00
FY14
FY15
FY16
FY17
FY18
FY14
FY15
FY16
FY17
FY18
FY19
FY20
Source: Company data, FactSet, Goldman Sachs Global Investment Research.
4.
Cancer combination pricing: the white elephant in the room. Affordability of new cancer drugs has become a growing
societal issue given the high cost of new therapies (about $10,000 per month). But more pressing is the growing realization
that oncology treatment is moving to combinations of very expensive drugs and the potential for more chronic usage of
drugs. There are numerous IO + IO and IO + target therapy combinations that are in clinical development each of which
costs more than $100K/year. Opdivo and Yervoy were recently approved in 1L melanoma (October 2015) and as the drugs
are not co-formulated the pricing is based on existing prices of the single agent. Opdivo is priced at $12.5K/month or
$150K/year in the US and Yervoy is $132K for four doses. Based on the dosing regimens the combination of the two for a
full year is $256K. Additionally there are now numerous triple combinations in multiple myeloma being analyzed. Darazlex
(daratumumab) is priced at $134K for the initial year of therapy and $70K for subsequent years. While it will be used as
monotherapy initially JNJ/Genmab are running trials in combination with Revlimid which is priced at $127K.
5.
The Black Swan Potential for broad price restrictions. Under the Affordable Care Act (ACA), a long shot scenario could
materialize where the president has the ability to enact broad restrictions on drug pricing through either the Independent
Payment Advisory Board (IPAB) and/or the Center for Medicare and Medicaid Innovation (CMMI). IPAB is triggered when
the rate of Medicares rate of spending growth exceeds inflation (potential impact in 2017+). While IPAB has not officially
Goldman Sachs Global Investment Research
14
December 17, 2015
Americas: Healthcare
been seated, its powers are held with the Secretary of HHS. If IPAB is triggered, theoretically it could give CMS the authority
to target spending outliers that could focus on specific categories rather than specific drugs. CMMI has the authority to
institute broad pilot programs designed to save costs. This could include paths such as least costly alternative or extending
drug spend into ACO models (possibly pulling part D drugs into bundles). However, we note its possible that movement on
any of the above fronts would likely result in legal challenges that could delay the implementation of these policies. In
totality, it appears that these debates may mostly serve as overhangs and headline risk in 2016.
6.
A word on biosimilars. While 2015 marked FDA approval of the first true biosimilar in the US, Sandozs Zarxio, it also
featured a number of delays (Celltrions biosimilar Remicade, Apotexs biosimilar G-CSF drugs and PFEs biosimilar
Epogen). Hence in 2016 our focus will be three-fold, assessing the uptake and impact of Zarxio on AMGNs Neupogen
(consensus is modelling a 12% yoy decline) and monitoring regulatory progress for biosimilars which experienced delays in
2015. We also expect focus on ABBVs Humira and any potential updates on competition from AMGNs biosimilar. ABBV
recently outline significant IP that they believe will protect Humira until into the next decade in the US. Additionally AMGN
management comments shifted on the 3Q earnings call acknowledging Humiras IP.
Like any drug market, in our view the biosimilars market will be defined by the competitive landscape (number of
options), doctor/patient acceptance and pricing/reimbursement considerations. As we have previously written the
technical challenges of developing and commercializing biosimilars could ultimately limit the number of players and
potential share/price erosion to less than that seen with small molecule generics. While this story is still playing out, we did
see some evidence of this in 2015.
Exhibit 7: Status of select US biosimilars
Reference drug
Biosimilar
Company
Filing status
Next steps
Epogen
Retacrit
Hospira (PFE)
Received complete response letter (CRL)
Submit response to CRL in 1H16
Humira
ABP 501
AMGN
Filed with FDA and EMA (11/25/15, 12/3/15)
FDA filing acceptance
Neupogen
Zarxio
Sandoz (Novartis)
Approved on 3/6/15
Launched September 2015
Neupogen
Grastofil
Apotex
FDA accepted BLA on 02/17/2015
PDUFA likely passed
Neulasta
Pegfilgrastim Apotex
Apotex
FDA accepted BLA on 12/17/2014
PDUFA likely passed
Neulasta
Pegfilgrastim Sandoz
Sandoz (Novartis)
FDA accepted BLA on 11/18/2015
PDUFA 3Q16
Remicade
Remsima
Celltrion
Filed with FDA (8/11/14)
PDUFA likely passed
Source: Company data, Goldman Sachs Global Investment Research.
Goldman Sachs Global Investment Research
15
December 17, 2015
Americas: Healthcare
Biopharma Big Picture: 2016 Investable Themes
As we look toward 2016 our focus is on (1) Immuno-oncology, which we believe remains an investible theme that is still in
its early stages, (2) the Alzheimers class which we believe will attract increased investor attention, though we remain
Neutral-rated on globally leveraged names BIIB and LLY, (3) key launches in the sector, including PCSK9 cholesterol drugs
from AMGN and REGN/SNY, (4) growth in late-stage CRO book-to-bills, which have accelerated in 2015 amid record biotech
funding and strong late-stage pharma pipeline growth, and (5) smid biotech catalysts where we have high conviction.
1.
2.
Early innings of the Immuno-oncology story. We expect first full year PD-1 sales of around $1.5 bn for Opdivo and Keytruda
driven by the rollout of new indications in lung, melanoma and renal. While appreciation for the IO class has certainly
grown over the past couple of years, from a commercial perspective we believe we are in the early innings of the story. We
believe the class will generate 2025 sales of approximately $30-40 bn (with half of that coming from lung, renal, and
melanoma), which could include potential for combinations. IO will continue to dominate the news flow across the sector,
with multiple new tumor studies in 2016 potentially stopped early for registration, including 1L lung, bladder, head and neck
and blood cancers. Key catalysts in 2016:
Will Roches lung filing for atezolizumab based on BIRCH trial be accepted for registration in 1Q16 or will the FDA
request the company to submit its application until after results from OAK (phase 3 all comers randomized OS trial).
If Roche is delayed by a couple of years, we see substantial upside to Opdivo and Keytruda.
IO combinations will be brought to the forefront in 2016 likely at ASCO. BMY will have data readouts for their LAG3,
anti-KIR, and CD-137 in 2016 and will enter into new combination trials for their IDO, OX-40, CD-73, and GITR. We
believe MRK may have potential data readouts for Keytruda in combination with the companys own GITR, LLYs
Alimta, PFEs 4-1BB, Novartiss BRAF/MEK and possibly other combos. We see new IO partnerships announced
almost weekly to look at different combinations across tumor types.
We expect early proof-of-concept from INCYs IDO in combination with PD1/PDL1s from Pharma partners MRK,
BMY, AZN and Roche in multiple indications throughout 2016, with potential for progress into pivotal trials. In
addition, INCY and MRK are slated to initiate a pivotal IDO/Keytruda combination study in frontline melanoma in
H1/16.
Engineered T-cell platforms (CAR Ts/TCRs) from KITE, JUNO and NVS will also yield data throughout 2016 including
pivotal data in leukemia and lymphoma by YE16 (KITE in DLBCL, JUNO in adult ALL and NVS in pediatric ALL) and
potential proof of concept in solid tumors (KITE in cervical and head and neck cancer and potentially glioblastoma
and other solid tumors by ASCO, JUNO in neuroblastoma and potentially other solid tumors by YE16). We see
proof-of-concept for CAR-Ts/TCRs in solid tumors as a major driver of upside for players in the engineered T-cell
space.
Alzheimers disease (AD) catalysts will also be front and center in 2016, particularly for LLY and BIIBs assets.
Goldman Sachs Global Investment Research
LLY demonstrated a deep commitment to Alzheimers at its recent investor event. Management outlined multiple
platforms for targeting various points in the AD disease cascaded. Sola EXPEDITION 3 results could potentially topline at the very end of 2016 and the full data is likely to be presented in 2017. We model a 25% probability of success,
or $1.0 bn by 2020. (Our un-risk adjusted peak sales estimate is $8.0 bn).
16
December 17, 2015
Americas: Healthcare
BIIBs Aducanumab (a-beta antibody) Ph3 AD program is under way and we expect data in 2019/2020. But in 2016
we could get two updates from the initial Ph1b trial data from the titration arm as well as from the rollover, openlabel extension portion (beyond one year). However, we do not view these as upside drivers given BIIB has already
designed the Aducanumab Ph3 program to incorporate dose titration and the Ph1b open-label extension lacks a
control arm. With respect to additional assets in BIIBs AD portfolio, we expect updates on Eisais BAN2401 (a-beta
antibody) and E2609 (BACE inhibitor) in 2016. For BAN2401 the Ph2 data will be fairly robust (N=650 patients over a
range of doses) and will include cognition data (novel composite endpoint called ADCOMS), 65% of the components
come from CDR-sb and hence might be generally comparable to Aducanumab Ph1b. Whereas the E2609 data will be
primarily focused on safety/tolerability.
Exhibit 8: Timeline for AD catalysts
2016 is a major year for Alzheimers
2016
Eli Lilly
2017
2018
Primary
Completion
solanezumab EXPEDITION 3
2019
Eli Lilly
2021
Primary
Completion
BACE Inhibitor (LY3314814) AMARANTH
Eisai / Biogen
2020
Study
Complete
2015
Study
Complete
Data
Release
Eisai / Biogen
BACE Inhibitor (BAN2401) Phase 2
Eisai / Biogen
aducanumab (BIIB037)
Phase 1b
Data
Release
Study
Complete
BACE Inhibitor Phase 1/2
Eisai / Biogen
Primary
Completion
Merck
BACE inhibitor verubecestat (MK-8931) EPOCH
Primary
Completion
Mild to Mod, no PET scan
Merck
BACE inhibitor verubecestat (MK-8931) APECS, Prodromal, w/PET scan
Roche
gantenerumab, Mild w/PET scan
Study
Complete
BIIB037 Phase 3 Alzheimer's Data
Primary
Completion
Study
Complete
2014
Primary
Completion
Source: clinicaltrials.gov
Goldman Sachs Global Investment Research
17
December 17, 2015
Americas: Healthcare
Beyond AD, we expect a high degree of focus on BIIBs anti-Lingo Ph2 MS data. While not a pivotal trial, it has the potential
to significantly impact BIIBs multiple give the target profile of this drug (remyelination for MS). Earlier this year reported
data from the first Ph2 trial of the drug in AON. In our view the data were mixed as while there appears to be a signal of
activity on the primary endpoint recovery of optic nerve latency (time for a signal to travel from the retina to the visual
cortex), as measured by full field visual evoked potential (FF-VEP), relative to placebo the study showed no effect on
secondary endpoints. We were looking for a signal of activity across multiple measures. Hence, we expect definitive proofof-concept for anti-Lingo to remain an outstanding question until we see data in a second Ph2 trial in MS in 2016.
3.
Outside of cancer and CNS within large-cap biotech we see several additional important pipeline catalysts including
AMGN/REGN PCSK9 Ph3 CV outcomes data, AMGNs Romo Ph3 osteoporosis data, REGNs Dupi Ph3 atopic dermatitis data,
CELGs Revlimid Ph3 REMARC trial and GILDs Simtuzumab Ph2 IPF and NASH data. In our view expectations are highest
for PCSK9 outcomes data (given robust cholesterol reductions and genetic support) and lowest for Simtuzumab (given prior
failures in cancer).
4.
Gaining leverage to the overall biopharma innovation cycle through the CROs. The CRO group has experienced high-single
digit backlog growth on average through 2015, which has contributed to an acceleration of trailing 12-month book-to-bills
well above normalized levels. In our view, record biotech funding and a continued focus by large pharma in investing in
their late-stage pipelines (which grew 8% on an annualized basis through the first nine months of the year) have been the
key drivers of this recent backlog strength we expect these trends to continue. As a result, we expect 2016 to deliver
accelerating sales growth across the group, helping drive margin expansion and generate greater levels of cash flow for
capital allocation.
Goldman Sachs Global Investment Research
18
December 17, 2015
Americas: Healthcare
Exhibit 9: Risk/Reward for select Ph3 pipeline catalysts
LLY/Solanezumab
Alzheimers
HigherReward
ALKS/5461
Depression
OPHT/Fovista-Wet AMD
BMY/Opdivo-New indications
INCY/Baricitinib-RA
KITE/KTE-C19
DLBCL
JUNO/JCAR015-ALL
BMRN/BMN 190-Batten
MRK/Keytruda
New indications
REGN/Dupi
Atopic derm/asthma
TEVA/Auspex
Tardive dyskinesia
ABBV/Elagolix
Endometriosis
AMGN/Repatha
CV outcomes
AMGN/Romo
osteoporosis
LowerReward
FGEN/Roxa-CKD China
BMRN/PEG-PAL
PKU
INCY/Jakafi
Pancreatic cancer
ALXN/Soliris-MG
BLUE/Lenti-D-CCALD
SGEN/Adcetris
CTCL-frontline
JNJ/Sirukumab-RA
LowerRisk
Pharma
HigherRisk
Largecap Biotech
Smidcap Biotech
Source: Company data, Goldman Sachs Global Investment Research
Goldman Sachs Global Investment Research
19
December 17, 2015
Americas: Healthcare
5.
Goldman Sachs Global Investment Research
Smid-biotech catalysts: Therapeutic modalities have evolved rapidly from small molecules and large molecules, now
proven platforms, to antibody-drug conjugates, antisense/RNAi, epigenetics, engineered T cells, gene therapy/editing and
cancer vaccines among others. We look for these novel platforms to yield transformative therapies, with pipeline progress
and pivotal readouts in 2016.
BMRN leading diversified rare disease play: We expect a multitude of 2016 catalysts. We look towards BMN 701
data (n=20, old manufacturing process in pivotal study) in Pompe disease in January, BMN 190 Batten disease final
data (likely at WORLD conference from Feb 29-Mar 4) and Phase III PEG-PAL for PKU results in Q1. BMN 250 Phase
I/II in MPS IIIB will enter the clinic in Q1. At the April 20, 2016 R&D day, we expect first BMN 270 data for hemophilia
A (upside to model), BMN 111 (Phase I/II1-year data at Phase III dose) and a potential new program disclosure. We
also look to the Kyndrisa in Duchennes muscular dystrophy CHMP decision in E.U. in 2Q.
BLUE lead gene therapy company: In terms of catalysts for 2016, we expect Lenti-D pivotal data in CCALD (rare
neurological disorder) to serve as proof-of-concept in terms of efficacy/durability, clarity on second-generation
LentiGlobin post FDA discussions (whether an IND is required), LentiGlobin data in Beta-thal and SCD at EHA (June
9-12) and ASH (December 3-6), and the immuno-oncology platform to progress with bb2121 for relapsed multiple
myeloma to enter the clinic in 1Q16, with data by YE16/2017.
KITE A CAR-T/TCR immuno-oncology leader: In 2016, we expect to see NCI Phase I/II data from the TCR targeting
HPV-16 E6 in cervical and head and neck cancer and possibly from the CAR targeting EGFRvIII in glioblastoma (an
aggressive brain tumor) by the American Society of Clinical Oncology (ASCO, June 3-7) meeting, which should
serve as major catalysts. The lead CAR-T program, KTE-C19, Phase I/II interim pivotal data is expected in 2H
followed by a BLA filing by YE16.
INCY immuno-oncology biotech leader: Positioned for abundant newsflow in 2016 from the lead immunooncology drug IDO, with Jakafi pivotal data and multiple readouts from the early stage pipeline. We look towards
additional readouts for IDO combinations with e BMYs PD-1 antibody Opdivo, RHHBYs PD-L1 antibody
atezolizumab, and AZNs PD-L1 antibody durvalumab likely through 1H and at ASCO with likely progress into
pivotal programs. We look towards a potential baricitinib approval in RA in the US in late-2016 (pending regulatory
submission by LLY in late-2015). Phase III JANUS-1 study of Jakafi in pancreatic cancer and Phase II results in breast
and colon cancer will read out in 2016. Initial Phase I/II data for novel FGFR, PIM, and BET inhibitors in multiple
malignancies should also read out in 2016.
20
December 17, 2015
Americas: Healthcare
From Launch to Margin: Focus on Expanders
Top margin inflection
picks: ABBV, AMGN,
BMY, VRTX
Strong new product momentum has led to continued improvements in margins and the converging of biotech and pharma (blurred
lines) into one bio-pharma sector. Successful new product launches have blurred the distinctions between the traditional pharma
companies and biotech companies. Indeed, BMY and LLY trade in line with higher-multiple biotechs while GILD, AMGN and now
BIIB sport multiples that are more typical of traditional pharma. This is a trend we expect to continue, with pipeline progress from
pharma looking more like biotech. Our call on large-cap pharma continues to be constructive (Attractive view) as we see the sector
entering a period of improved operating margin expansion after several years of flat to down margins driven by the patent cliff. We
see 600 basis points of margin expansion for the large cap pharma sector, with BMY showing the most leverage among its peer
group diven by Opdivo sales. Biotech saw a significant amount of margin expansion over the past several years driven by major
new product launches, and while we still expect improvement going forward, it is likely to be at a slower pace.
Our coverage view on pharma remain Attractive: On a composite basis, we model 600 basis points of operating margin
improvement for the large cap pharma sector. No company is likely to exhibit as much margin leverage as BMY over the
next five years, with EBIT margins growing from 25% to 43% by 2020. As Opdivo, with an operating margin estimated in the
75% range ramps to its peak potential of >$10 bn driven by numerous cancer indications. We also see margin improvement
from ABBV (42.5% to 49% by 2020) from growth in Humira, pipeline launches, and operational efficacy. With the stock
trading at 11x our 2016 EPS estimate (versus 17x for the sector) we believe the market is grossly undervaluing ABBVs
future cash flows due to continued concerns about biosimilars, despite our continued view that biosimilar competition will
likely behave more like proprietary drugs than AB-rated generics and the increased likelihood that prolonged patent
litigation will delay the launch of biosimilars into the next decade. On the other hand, we see flat operating margins for JNJ
(31% unchanged through 2020) driven by continued pressure from MD&D and Consumer.
Our coverage view on Biotech remains Neutral and we continue to see a need to remain selective heading into 2016 as we
expect the year to be similar to 2015. We expect margins among the core four (AMGN, BIIB, CELG and GILD; Exhibit 10) to
expand slightly (from 54.6% to 55.3% over 2015-18E), while margins for ALXN, REGN and VRTX should continue to expand
more significantly (VRTX should see the most significant swing as it transitions to profitability on the back of the Orkambi
launch).
When looking at expected margin expansion versus P/E, we highlight ABBV, which has one of the highest expected margin
expansion profiles in our biopharma coverage (6.5% from 2015-18E) but is trading at the second lowest multiple (10.6x 2016 EPS)
compared to peers. BMY has the highest expected margin expansion in our biopharma coverage (10% from 2015-18E), but is also
trading at the highest multiple (28x). GILD is notable because it is the only company in our coverage we forecast to see material
margin degradation over the next three years (-5% from 2015-18E), and is also trading at the lowest multiple in our coverage (9x).
Goldman Sachs Global Investment Research
21
December 17, 2015
Americas: Healthcare
Exhibit 10: Margin expansion expected to continue through pharma and biotech names
2015-18E margin expansion vs. 2016 P/E
35x
30x
BMY
2016 P/E
25x
LLY
20x
CELG
BIIB
PFE
15x
10x
JNJ
MRK
AMGN
ABBV
GILD
5x
0x
-10%
-5%
0%
5%
10%
15%
2015E - 2018E Margin Expansion
Source: Company data, FactSet, Goldman Sachs Global Investment Research
Goldman Sachs Global Investment Research
22
December 17, 2015
Americas: Healthcare
Utilization Tug of War: Need For A Product Cycle Overlay
As we look toward 2016 we expect (1) a waning benefit from the ACA tailwinds of the last 12-18 months and (2) a pickup in cyclical
/core utilization growth. With these two potentially opposing forces tugging at the utilization theme, we prefer an overlay of
product cycles and margin expansion opportunities, which we express via our preference for MedTech.
Diving into a little more detail, we see stable utilization as important backdrop under which companies can execute idiosyncratic
growth drives. Importantly, for MedTech, our analysis suggests that very little of the top-line acceleration seen over the past two
years has come from core business improvement. In fact, we have observed no incremental contribution from underlying trends
with the bulk of the uptick from new product launches. In other sectors like Providers, however, we would argue that a large
percentage of the volume acceleration seen in 2014 and into 1H15 came from ACA and other external factors. Given some of the
gives and takes involved with the forward utilization view, we think that multiple expansion needs to come from more companyspecific drivers, further supporting our MedTech view.
Putting the utilization theme in context
Utilization in isolation has not proven to be the sole barometer of stock performance, evidenced by the lack of an observed
correlation between volume-exposed stocks and relative share price movement over the ytd period or last 24 months. Over these
representative time frames, outperformance has stemmed from idiosyncratic drivers (product cycles, capital allocation,
restructuring) in the context of stable utilization; while, underperformance has resulted from both company-specific factors
(competition, P&L pressure, high expectations) as well as external variables (concerns over ACA exchanges). Looking to 2016, our
long standing macro utilization model continues to suggest an uptick in same store volumes, which we expect to be a tailwind to
a number of industry verticals. Further, as we dissect the conflicting factors related to utilization (macro uptick vs. ACA risk), we
think that the cyclical will win out over exogenous drivers.
From a stock perspective, however, we do not see any reason for a departure from performance drivers since 2014 and, while we
are constructive on utilization, this one input is not sufficient to be the sole basis for any of our stock or sector recommendations. To
support our forward outlook, we think a stable utilization environment is critical such that companies can engage in business
investment and drive top-line growth, margin expansion, deploy capital, etc. In turn, these activities should form the basis for our
stock picking framework. Within this context, we retain our Attractive view on Medical Devices and Neutral coverage view on
Diagnostics, the Supply Chain/Drug Chains, Providers, and Managed Care.
Revisiting our macro utilization model
Five years ago, we introduced our top-down healthcare consumption model that called for a multi-year slowdown in utilization
(defined as per capita spending, or same store sales growth). Specifically, we established a view that utilization would decelerate
and remained subdued until the 2014 time frame with improvements thereafter (see our note Weak volumes likely to persist;
positioning ahead of 3Q dated October 6, 2010). Over this economic cycle, we point out that the rate of job growth and
improvement in real wages has been somewhat more moderate than in past recoveries this has somewhat pushed out the timing
for a volume inflection; however, we do not think a slower recovery is reflective of a lack of recovery, and we expect 2016 to show
more significant growth in medical cost trend.
Goldman Sachs Global Investment Research
23
December 17, 2015
Americas: Healthcare
Exhibit 11: The economic cycle and healthcare spending trends
Commercial medical cost trend relative to the timing of the economic cycle since 1982
16.0%
Recession
Recession
Recession
Recession
14.0%
12.0%
10.0%
8.0%
?
6.0%
4.0%
2.0%
1982
1983
1984
1985
1986
1987
1988
1989
1990
1991
1992
1993
1994
1995
1996
1997
1998
1999
2000
2001
2002
2003
2004
2005
2006
2007
2008
2009
2010
2011
2012
2013
2014
2015E
0.0%
Source: Company data, industry surveys, CMS, Goldman Sachs Global Investment Research.
Goldman Sachs Global Investment Research
24
December 17, 2015
Americas: Healthcare
Lateral read and cross sector data points
Hospitals: Same-store admissions growth peaked in 1Q15 and has decelerated sequentially in both 2Q and 3Q15, reflecting
diminished contribution from ACA coverage expansion. That said, the most recent (3Q) volume growth was still strong for
most companies and we expect sequential acceleration into 4Q. In 2016, we are assuming no contribution from the ACA to
volumes beyond roughly maintaining the level of coverage expansion achieved through this year. However, we are
expecting core (non-ACA) volumes will continue to be a key driver.
Labs: After 3 quarters of sequential volume acceleration (4Q14-3Q15), volume growth decelerated in 3Q15. On a two-year
basis volume growth, the rate of acceleration slowed in 3Q15 following a robust 2Q15 and trend appears to be normalizing
at a low-single-digit rate.
Medical Devices: Since 4Q14 organic growth has stabilized across various MedTech end markets (orthopaedics, supplies).
That said, the rate of acceleration has moderated as the industry laps tough comps from 2H14. Overall, multi-year trend
appears to be improving albeit at a slower pace.
Prescription drug volumes: We expect a gradual acceleration in Rx growth for 2016 on modest improvement in same-store
volumes against easy compares. While the deceleration in Rx volumes in 2015 is worth noting, we think it was largely due
to cycling the uplift from ACA the prior year. The two-year stack has remained relatively stable throughout 2015, and we
expect that once ACA comparisons are cycled, we should see y/y Rx growth accelerate.
Goldman Sachs Global Investment Research
25
December 17, 2015
Americas: Healthcare
The M&A March: Slower, Selective, More Strategic
1.
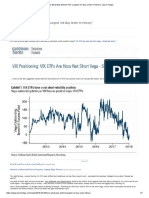
We expect the pace of M&A going forward to slow given that acquirers stocks have been starting to underperform
again after deals are announced. Since 2012, we have seen acquirers across all sectors (with the exception of Energy)
being rewarded for M&A with aggregate stock outperformance two days post deal announcements (Exhibit 12). However,
the magnitude of outperformance has steadily declined over the last several years with 2015 just barely positive, and we are
seeing a gradual reversion to the mean when considering that acquirers stocks underperformed in almost every year in the
period 1996-2011. Specifically in healthcare (Exhibits 13-14), when comparing stock performance for acquirers in 2015 vs.
2014 it is clear that 2015 has been a lot more challenging. In 2015, acquirers have outperformed to a much lesser extent
than in 2014 in the two days following deal announcements across all subsectors of healthcare, and when looking at
performance through the end of the year the difference was even more dramatic (in biopharma and managed care
acquirers actually underperformed significantly this year). With M&A losing some of its appeal and acquirers getting much
less credit by investors for deals, we expect the pace of M&A to slow down somewhat going forward.
Acquirer stocks
starting to
underperform
Exhibit 12: Acquirers have been outperforming around large ($1.5bn+) M&A deal announcements, except in Energy
Across these sectors, acquirers have outperformed for twothirds of the $1.5bn+ deal announcements since 2012
6%
4.6%
8%
4%
3.3%
6%
1.8%
2%
0.4%
1.3%
0.7%
0.2%
The hit rate of
outperformance is
lower for Utilities (50%)
and Energy (30%)
4%
2%
0%
(0.5%)
(0.9%)
(1.0%)
-2%
0%
(1.6%)
(2.0%)
(2.0%)
-2%
(2.5%)
(3.0%)
(3.3%)
(3.5%)
-4%
-4%
(4.4%)
-6%
(5.2%)
-6%
(6.3%)
2015
2014
2013
2012
2011
2010
2009
2008
2007
2006
2005
2004
2003
2002
2001
2000
1999
1998
1997
1996
-8%
Source: Company data, Goldman Sachs Global Investment Research.
Goldman Sachs Global Investment Research
26
December 17, 2015
Americas: Healthcare
Exhibit 13: Performance from 1 day before to 2 days post announcement of
the deal
Exhibit 14: Performance from 1 day before announcement of the deal to end
of the year
7%
6%
40%
6%
30%
5%
30%
4%
3%
3%
2%
2%
1%
1%
1%
0.4%
0%
Biopharma
Medtech
Managed Care
Price performance
Price performance
5%
15%
10%
10%
4%
0.5%
0%
Biopharma
Medtech
Managed Care
-20%
-15%
-1%
-2%
2014 Average
2015 Average
Source: Company data, Goldman Sachs Global Investment Research.
2.
Biopharma firepower
could rise to $168bn186bn in 2018 from
$88bn-96bn today
Goldman Sachs Global Investment Research
HC Services
-10%
HC Services
-1%
19%
20%
-22%
-30%
2014 Average
2015 Average
Source: Company data, Goldman Sachs Global Investment Research.
Still a lot of balance sheet firepower for M&A, so some level of deals could continue but likely strategic over
financial. Despite the substantial amount of deal making over the last few years within healthcare, companies balance
sheets across all the subsectors in healthcare continue to have significant firepower. Exhibit x shows the estimated
firepower to do deals when balance sheets are levered up to 2x and 4x, and compares the levels in 2015 with 2012 (when a
lot of the M&A started). Interestingly, those levels have not come down that much with Pharma and Biotech actually
showing greater firepower in 2015 than in 2012. Similarly, a bottoms up analysis of large-cap pharma and biotech shows
that these companies could have up to $88-96bn of firepower currently and up to $168-186bn of firepower in 2018 if those
companies are levered up to 2x. Average net leverage ratios across all healthcare have been continuing to climb over time,
but is still at a reasonable level in our view at roughly 1.4x. When looking at the net leverage across the Specialty & SMID
pharma (one of the more active participants in recent M&A), we expect the larger companies that are more highly levered at
around 4x (e.g., VRX, ENDP, MNK) to focus more on execution and paying down debt from recent deals in the near term.
However, we think some of the SMID pharma companies that are much less levered (e.g., JAZZ, IPXL, AKRX) could
potentially drive more of the M&A going forward as they look to get bigger and create more efficiencies. When considering
healthcare M&A overall, we believe investors have most soured on deals that have been primarily for financial engineering
purposes (e.g., acquiring mature assets and driving accretion largely through price increases and/or cost synergies, tax
inversions, etc.). Where we expect companies to be more focused with their M&A going forward is on strategic deals that
add new products or technologies, that have very good durability, and that will be significant contributors to long-term
organic growth even if they are less accretive in the very near term.
27
December 17, 2015
Americas: Healthcare
Exhibit 15: Aggregate balance sheet firepower by Health Care subsector
300
300
2012 - Firepower at 4x
2012 - Firepower at 2x
250
2015 - Firepower at 4x
2015 - Firepower at 2x
200
200
150
150
100
100
50
Aggregate Firepower ($, billions)
Aggregate Firepower ($, billions)
250
50
2012
2015
2012
Pharma
2015
Biotech
2012
2015
HC Providers & Svcs
2012
2015
HC Tech & Equip
2012
2015
Life Sc Tools & Dx
Source: Company data, Goldman Sachs Global Investment Research.
3.
Rising interest rates
unlikely to be a
headwind
Goldman Sachs Global Investment Research
Higher interest rates probably will not change the dynamics for M&A that much, including a trend towards more
stock deals. Interestingly, there is a very strong correlation between the Federal funds rate and the 10 year Treasury rates
with the US M&A volumes over the last ten years (Exhibit 16). What we have seen in the past is that higher rates are
typically associated with a growing economy with higher consumer and business confidence that ultimately drives greater
M&A activity. As such, we do not believe a higher interest rate environment in and of itself would necessarily change the
level of M&A in healthcare. However, the level of accretion from such deals could certainly decrease with a higher cost of
debt. As an example in Specialty Pharma with ENDPs deal for Par that closed on September 28, 2015, based on our
estimates the deal is likely to be accretive to EPS by 18-21% in 2016. We ran a sensitivity analysis and for every incremental
100bps in interest rate (actual rate for deal was 5.25-6%) that would have reduced the accretion by only ~1-2% (with ~$25mn
of additional synergies needed to offset that). So even if interest rates went up 200-300bp we do not think that would have
been a deal-breaker for ENDP. A noticeable trend within healthcare has been the increased use of stock in deals which has
gone up significantly over the last two years (Exhibit 17). All cash deals have represented only 25% of the overall healthcare
M&A volume YTD in 2015, well below the 20-year average of 47%. If borrowing costs end up going higher, stock deals
could potentially be a trend that continues to increase going forward.
28
December 17, 2015
Americas: Healthcare
Exhibit 16: Correlation with US M&A Volumes as percentage of market cap (1995 2014)
78%
72%
64%
61%
59%
53%
51%
-7%
-17%
-22%
-23%
-55%
Source: Federal Reserve, Dealogic, Bloomberg, FactSet, iBoxx, Goldman Sachs Global Investment Research
Goldman Sachs Global Investment Research
29
December 17, 2015
Americas: Healthcare
Exhibit 17: Health Care announced M&A volumes by payment type
600
100%
90%
500
80%
70%
400
60%
300
50%
40%
200
30%
20%
100
2015TD, All-Cash
deals represent just
25% of total
volumes, well below
20-year average
levels (47%).
10%
0%
Stock Only
Cash / Stock & Other
Cash Only
Cash Only (%, RHS)
20yr Avg. ('95-'14)
Source: Dealogic, Goldman Sachs Global Investment Research. As of November 30, 2015.
Goldman Sachs Global Investment Research
30
December 17, 2015
Americas: Healthcare
M&A Implementation: GS HC Strategic Assets Basket Refresh + LBO Screen
We refresh our Healthcare Strategic Assets basket (Bloomberg ticker: GSGLGHSA) and add 15 new names due to expansion of coverage
and reassessment of strategic potential. The GS Healthcare Strategic Assets Basket now offers 40 names where our analysts see 15% or
greater probability of strategic M&A in the next 12 months. Year to date, the basket the basket is up 16 % vs. the S&P HC Index up 4% and
the S&P 500 down 1%. Since inception on September 20, 2010, the basket is up 204% vs. the S&P HC Index up 79% and the S&P 500 up
79%.
We offer this basket as one way to implement a sector M&A theme and to identify where there could be M&A risk.
We also highlight six companies within our Europe Coverage where an M&A component is factored into our analysts valuation
methodology and target. Buy-rated names on the list include: GLPG.AS, IPH.PA, SN.L, and ZELA.CO (Exhibit 19).
M&A framework
Across our coverage universe, we examine stocks using an M&A framework, considering both quantitative factors (IRR and
theoretical returns following a buyout, using our standardized departmental LBO model) and qualitative factors (market opportunity
for key products, pipeline, intellectual property and competitive landscape) to incorporate the potential that certain companies could
be acquired at a premium to current share prices.
We then calculate an M&A score as a means of ranking companies under coverage from 1 to 4, with 1 representing high (30%-50%)
probability of M&A activity, 2 representing medium (15%-30%) probability, 3 representing low (10%-15%) probability and 4
representing minimal to no probability (0%-10%). For companies ranked 1 or 2, in line with our departmental guidelines, we
incorporate an M&A component into our target price.
Exhibit 18: M&A Basket sector composition
MedTech
20%
Specialty &
SMID Pharma
17%
Biotech
22%
Managed Care
10%
Exhibit 19: EU Healthcare companies with M&A included in price
Ticker
SN.L
ATLN.VX
GEN.CO
IPH.PA
ZELA.CO
EKTAb.ST
GN.CO
GXIG.DE
SOON.VX
GLPG.AS
MORG.DE
Sector
MedTech
Biotech
Biotech
Biotech
Biotech
MedTech
MedTech
MedTech
MedTech
Biotech
Biotech
Rating
Buy
Neutral
Neutral
Buy*
Buy
Neutral
Neutral
Sell
Neutral
Buy
Neutral
M&A
Weighting
30%
30%
30%
30%
30%
15%
15%
15%
15%
15%
12%
Pricing
Currency
GBP
CHF
DKK
EUR
DKK
SEK
DKK
EUR
CHF
EUR
EUR
M&A
Value
1,510
197
1,500
19
218
100
200
60
144
80
87
Target Price
1,300
121
1,000
22
200
55
145
44
121
26
75
Target
Period
12 months
12 months
12 months
12 months
12 months
12 months
12 months
12 months
12 months
12 months
12 months
Current
Price
1,086
135
920
13
142
73
120
70
124
52
56
*IPH.PA is on the European Conviction List.
Other
15%
Life
Science
Tools
8%
Facilities
8%
*Other includes Diagnostics, Supply Chain, Major Pharma each 5% of the basket and Dental
which represents 2% of the basket
Source: Goldman Sachs Global Investment Research, FactSet
Goldman Sachs Global Investment Research
For EU Biotech M&A framework see Europe: Healthcare: Biotechnology: Innovation drives value
creation (2): EU biotech acquisition target, Published February 2, 2015
For EU MedTech M&A framework see Europe: Healthcare: Medical Technology: Adjusting to a new
normal: Less growth and more buyer consolidation drive focus on cash, Published June 23, 2014
Source: Goldman Sachs Global Investment Research, FactSet, Pricing as of November 26, 2014.
31
December 17, 2015
Americas: Healthcare
Exhibit 20: Potential Healthcare candidates for M&A activity under GS coverage
Checked () names are included in the GSGLGHSA basket; BOLD names are new additions
Ticker
Company Name
A
ALGN
ALKS
ANAC
ARIA
BCR
BKD
BLUE
BMRN
CNC
COO
EVH
EW
EXAS
FEIC
GBT
GKOS
GMED
HAE
HOLX
HTWR
INCY
IPXL
IRWD
JAZZ
KITE
LPNT
MCRB
MGLN
MNTA
MOH
NUVA
PCRX
PMC
SAGE
SCAI
TMH
TTOO
VRTX
WAT
WCG
ZTS
Agilent Technologies, Inc
Align Technology Inc.
Alkermes Plc
Anacor Pharmaceuticals Inc.
Ariad Pharmaceuticals Inc.
C.R. Bard Inc.
Brookdale Senior Living Inc.
bluebird bio
BioMarin Pharmaceutical Inc.
Centene Corp.
The Cooper Companies Inc.
Evolent Health Inc.
Edwards Lifesciences Corp.
Exact Sciences Corp.
FEI Co.
Global Blood Therapeutics Inc.
Glaukos Corp.
Globus Medical Inc.
Haemonetics Corp.
Hologic Inc.
HeartWare International Inc.
Incyte Corp.
Impax Laboratories Inc.
Ironwood Pharmaceuticals Inc.
Jazz Pharmaceuticals
Kite Pharma Inc.
LifePoint Health Inc.
Seres Therapeutics Inc.
Magellan Health Services Inc.
Momenta Pharmaceuticals Inc.
Molina Healthcare Inc.
NuVasive Inc.
Pacira Pharmaceuticals Inc.
PharMerica Corp.
Sage Therapeutics Inc.
Surgical Care Affiliates Inc.
Team Health Holdings
T2 Biosystems Inc.
Vertex Pharmaceuticals Inc.
Waters Corp.
WellCare Health Plans Inc.
Zoetis Inc.
Analyst
Isaac Ro
Robert P. Jones
Terence Flynn, PhD
Gary Nachman
Terence Flynn, PhD
David H. Roman
Stephan Stewart, CFA
Salveen Richter
Salveen Richter
Matthew Borsch, CFA
David H. Roman
Robert P. Jones
David H. Roman
Isaac Ro
Isaac Ro
Terence Flynn, PhD
David H. Roman
David H. Roman
David H. Roman
Isaac Ro
David H. Roman
Salveen Richter
Gary Nachman
Gary Nachman
Gary Nachman
Salveen Richter
Matthew Borsch, CFA
Terence Flynn, PhD
Matthew Borsch, CFA
Gary Nachman
Matthew Borsch, CFA
David H. Roman
Gary Nachman
Stephan Stewart, CFA
Gary Nachman
Matthew Borsch, CFA
Matthew Borsch, CFA
Isaac Ro
Terence Flynn, PhD
Isaac Ro
Matthew Borsch, CFA
Jami Rubin
Sector
Life Science Tools
Dental
SMID Biotech
Specialty & SMID Pharma
SMID Biotech
Medical Supplies
Healthcare Supply Chain
Emerging Biotech
Emerging Biotech
Managed Care
Medical Devices
Healthcare IT
Medical Devices
Diagnostics
Life Science Tools
SMID Biotech
Medical Devices
Medical Devices
Medical Supplies
Diagnostics
Medical Devices
Emerging Biotech
Specialty & SMID Pharma
Specialty & SMID Pharma
Specialty & SMID Pharma
Emerging Biotech
Facilities
SMID Biotech
Managed Care
Specialty & SMID Pharma
Managed Care
Medical Devices
Specialty & SMID Pharma
Healthcare Supply Chain
Specialty & SMID Pharma
Facilities
Facilities
Diagnostics
Large/Mid-Cap Biotech
Life Science Tools
Managed Care
Major Pharmaceuticals
M&A Rank
Analyst
Rating
Pricing
Currency
Market Cap
($, mn)
Last Close
($)
M&A Value
($)
GSGLGHSA
Basket
1
2
1
1
2
2
2
2
1
2
2
2
2
2
2
2
1
1
2
2
2
1
1
2
2
2
2
1
2
2
2
1
1
2
2
2
2
2
1
2
1
2
Buy
Buy
Neutral
Buy
Neutral
Buy
Buy
Buy
Buy
Sell
Neutral
Buy
Buy
Neutral
Buy
Neutral
Neutral
Neutral
Sell
Buy*
Neutral
Buy
Neutral
Sell
Neutral
Buy
Buy
Neutral
Neutral
Neutral
Neutral
Neutral
Buy
Neutral
Buy
Buy
Neutral
Neutral
Buy
Neutral
Buy
Neutral
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
USD
13,525
5,170
10,749
5,150
1,094
13,870
3,376
2,132
15,195
7,000
6,636
762
17,263
771
3,221
971
743
2,523
1,602
10,606
797
20,350
2,898
1,570
8,315
2,838
2,952
1,481
1,332
1,076
3,280
2,472
2,295
955
1,619
1,447
3,256
191
28,429
10,748
3,517
23,387
40.81
65.84
72.74
112.98
6.15
187.72
18.27
59.97
99.14
58.72
136.51
12.92
80.03
8.34
79.51
33.02
23.15
26.50
31.59
38.50
46.38
114.73
41.82
11.00
138.47
64.24
69.28
40.90
56.30
15.94
58.48
51.55
70.89
31.36
56.10
37.33
44.94
9.70
117.28
131.61
79.74
46.97
52.00
76.00
68.00
182.00
20.00
241.00
46.00
197.00
170.00
71.00
161.00
37.00
184.00
13.00
100.00
108.00
37.00
30.00
46.00
54.00
85.00
180.00
53.00
18.00
173.00
132.00
105.00
66.00
80.00
20.00
105.00
65.00
80.00
45.00
93.00
46.00
73.00
25.00
181.00
168.00
135.00
51.00
*HOLX is on the Americas Conviction List.
We have excluded EVH and TTOO from the basket because of trading liquidity constraints. M&A rank definitions are provided in the M&A framework section on the previous page.
Source: Goldman Sachs Global Investment Research, FactSet. Pricing as of December 15, 2015.
Goldman Sachs Global Investment Research
32
December 17, 2015
Americas: Healthcare
Financial buyer list: Companies with the highest IRRs
In addition to our analyst-driven Healthcare Strategic Assets basket, we provide a list of companies in our US Healthcare coverage
universe with IRRs over 15% based on our quantitative standardized departmental LBO model.
Assumptions: As a base case, this purely quantitative model (applicable to our non-financial coverage) assumes a five-year holding
period, a 20% premium, initial leverage based on S&P GICS Level 1 sector-specific debt/EBITDA multiples (median plus 1 standard
deviation; and rent-adjusted for Retail companies), a 7% weighted average cost of debt, and an exit multiple based on a companys
three-year historical EV/EBITDA. 2016 is year 1. Operational assumptions for sales growth, EBITDA margin and capex growth are
taken from our analyst forecasts, extrapolated when required.
Taking size into consideration, we exclude any companies with a market cap of over $15 bn (and under $200 mn), those who already
operate with high leverage and Managed Care names. In addition, we exclude relatively recent IPOs (those priced since January 1,
2013).
Given the variety of qualitative and industry-specific factors that go into a leveraged buyout, we do not necessarily believe that each
of the companies with IRRs greater than 15%, is a viable buyout candidate. However, a high IRR, at the very least, serves as a
positive valuation signpost.
This list includes all stocks under our coverage irrespective of our analysts ratings.
Exhibit 21: Healthcare companies with IRRs of 15% or more
IRRs based on our standardized departmental LBO model (IRRs and pricing as of the market close of December 15, 2015
Ticker
Company name
IRR
Market Cap Sector
($, mn)
TMH
Team Health Holdings
21.7%
3,256
Providers
JAZZ
Jazz Pharmaceuticals
19.9%
8,516
Specialty & SMID Pharma
AMSG Amsurg Corp.
18.7%
4,048
Providers
IPXL
Impax Laboratories Inc.
16.1%
2,949
Specialty & SMID Pharma
LPNT
LifePoint Health Inc.
15.4%
3,015
Hospitals
M&A rank definitions are provided in the M&A framework section on page 3.
Rating
Neutral
Neutral
Buy
Neutral
Buy
Last Price
($)
44.94
138.47
83.55
41.82
69.28
Target
Price
($)
63.00
154.00
101.00
40.00
84.00
Upside To
Price
Target
40%
11%
21%
-4%
21%
Target
Period
12 months
12 months
12 months
12 months
12 months
Source: Goldman Sachs Global Investment Research.
Goldman Sachs Global Investment Research
33
December 17, 2015
Americas: Healthcare
Rating and pricing information
AbbVie Inc. (B/A, $57.63), Adeptus Health Inc. (B/A, $52.01), Alere Inc. (B/N, $39.70), Alexion Pharmaceuticals Inc. (N/N, $188.91),
Amgen Inc. (B/N, $164.58), Baxter International Inc. (B/N, $37.83), Boston Scientific Corp. (B/A, $18.88), Bristol-Myers Squibb Co. (B/A,
$70.71), Cardinal Health Inc. (B/N, $88.22), Cerner Corp. (B/N, $61.16), Cigna Corp. (B/N, $141.19), Dimension Therapeutics (B/N,
$9.98), Endo International Plc (B/A, $62.03), Envision Healthcare Holdings (B/N, $24.02), Evolent Health Inc. (B/N, $13.39), FibroGen
Inc. (B/N, $30.78), Hologic Inc. (B/N, $38.99), Horizon Pharma Plc (B/A, $21.15), INC Research Holdings (B/A, $48.00), Intuitive Surgical
Inc. (B/A, $545.13), McKesson Corp. (B/N, $190.72), Medtronic plc (B/A, $78.57), Mylan NV (B/A, $54.39), Quintiles Transnational
Holdings (B/A, $68.73), Regeneron Pharmaceuticals Inc. (B/N, $559.67), Stryker Corp. (B/A, $93.93), Teva Pharmaceuticals (B/A,
$65.87), Thermo Fisher Scientific Inc. (B/A, $140.45), VWR Corp. (B/A, $26.40) and ZELTIQ Aesthetics Inc. (B/A, $28.25).
Equity basket disclosure
The ability to trade the basket(s) discussed in this research will depend upon market conditions, including liquidity and borrow
constraints at the time of trade.
Goldman Sachs Global Investment Research
34
December 17, 2015
Americas: Healthcare
Disclosure Appendix
Reg AC
We, Jami Rubin, David H. Roman, Matthew Borsch, CFA, Robert P. Jones, Salveen Richter, Terence Flynn, PhD, Isaac Ro, Gary Nachman, Stephan Stewart, CFA and Adam Noble, hereby certify that all
of the views expressed in this report accurately reflect our personal views about the subject company or companies and its or their securities. We also certify that no part of our compensation was, is
or will be, directly or indirectly, related to the specific recommendations or views expressed in this report.
We, Asad Haider, CFA and Alec Phillips, hereby certify that all of the views expressed in this report accurately reflect our personal views, which have not been influenced by considerations of the firm's
business or client relationships.
Unless otherwise stated, the individuals listed on the cover page of this report are analysts in Goldman Sachs' Global Investment Research division.
Investment Profile
The Goldman Sachs Investment Profile provides investment context for a security by comparing key attributes of that security to its peer group and market. The four key attributes depicted are: growth,
returns, multiple and volatility. Growth, returns and multiple are indexed based on composites of several methodologies to determine the stocks percentile ranking within the region's coverage
universe.
The precise calculation of each metric may vary depending on the fiscal year, industry and region but the standard approach is as follows:
Growth is a composite of next year's estimate over current year's estimate, e.g. EPS, EBITDA, Revenue. Return is a year one prospective aggregate of various return on capital measures, e.g. CROCI,
ROACE, and ROE. Multiple is a composite of one-year forward valuation ratios, e.g. P/E, dividend yield, EV/FCF, EV/EBITDA, EV/DACF, Price/Book. Volatility is measured as trailing twelve-month
volatility adjusted for dividends.
Quantum
Quantum is Goldman Sachs' proprietary database providing access to detailed financial statement histories, forecasts and ratios. It can be used for in-depth analysis of a single company, or to make
comparisons between companies in different sectors and markets.
GS SUSTAIN
GS SUSTAIN is a global investment strategy aimed at long-term, long-only performance with a low turnover of ideas. The GS SUSTAIN focus list includes leaders our analysis shows to be well
positioned to deliver long term outperformance through sustained competitive advantage and superior returns on capital relative to their global industry peers. Leaders are identified based on
quantifiable analysis of three aspects of corporate performance: cash return on cash invested, industry positioning and management quality (the effectiveness of companies' management of the
environmental, social and governance issues facing their industry).
Disclosures
Coverage group(s) of stocks by primary analyst(s)
Jami Rubin: America-Major Pharmaceuticals, America-Pharmaceuticals Generics. David H. Roman: America-Medical Devices, America-Medical Supplies. Matthew Borsch, CFA: America-HCManaged,
America-Healthcare Services:Facilities. Robert P. Jones: America-Dental services and Equipment, America-Drug Chains, America-Healthcare IT, America-Healthcare Services: CROs, America-Healthcare
Supply Chain. Salveen Richter: America-Emerging Biotech. Terence Flynn, PhD: America-Large Biotech, America-SMID Biotech. Isaac Ro: America-Diagnostics, America-Labs and Services, AmericaLife Science Tools. Gary Nachman: America-Specialty & SMID Pharmaceuticals. Stephan Stewart, CFA: America-Healthcare Supply Chain.
America-Dental services and Equipment: Align Technology Inc., Dentsply International Inc., Henry Schein Inc., Patterson Cos., Sirona Dental Systems Inc..
America-Diagnostics: Alere Inc., Cepheid, Exact Sciences Corp., Foundation Medicine Inc., Hologic Inc., Myriad Genetics Inc., Qiagen NV, T2 Biosystems Inc..
America-Drug Chains: CVS Health Corp., Rite Aid Corp., Walgreens Boots Alliance Inc..
America-Emerging Biotech: Atara Biotherapeutics Inc., BioMarin Pharmaceutical Inc., bluebird bio, Incyte Corp., Isis Pharmaceuticals Inc., Juno Therapeutics Inc., Kite Pharma Inc., Seattle Genetics Inc..
America-HCManaged: Aetna Inc., Anthem Inc., Centene Corp., Cigna Corp., Health Net Inc., Humana Inc., Magellan Health Services Inc., Molina Healthcare Inc., UnitedHealth Group, WellCare Health
Plans Inc..
America-Healthcare IT: Allscripts Healthcare Solutions, Athenahealth Inc., Castlight Health Inc., Cerner Corp., Evolent Health Inc..
America-Healthcare Services: CROs: Charles River Laboratories, ICON Plc, INC Research Holdings, Parexel International Corp., Quintiles Transnational Holdings.
America-Healthcare Services:Facilities: Adeptus Health Inc., Amsurg Corp., Community Health Systems Inc., DaVita Inc., Envision Healthcare Holdings, ExamWorks Group, HCA Holdings, LifePoint
Health Inc., Surgery Partners Inc., Surgical Care Affiliates Inc., Team Health Holdings, Tenet Healthcare Corp., Universal Health Services Inc..
America-Healthcare Supply Chain: AmerisourceBergen Corp., Bright Horizons Family, Brookdale Senior Living Inc., Cardinal Health Inc., Express Scripts Holding, McKesson Corp., Owens & Minor Inc.,
PharMerica Corp..
America-Labs and Services: Laboratory Corp. of America Holdings, Quest Diagnostics Inc., Stericycle Inc..
Goldman Sachs Global Investment Research
35
December 17, 2015
Americas: Healthcare
America-Large Biotech: Alexion Pharmaceuticals Inc., Amgen Inc., Biogen Inc., Celgene Corp., Gilead Sciences Inc., Regeneron Pharmaceuticals Inc., Vertex Pharmaceuticals Inc..
America-Life Science Tools: Agilent Technologies, Bruker Corp., Danaher Corp., FEI Co., Illumina Inc., Mettler-Toledo International Inc., PerkinElmer Inc., Thermo Fisher Scientific Inc., VWR Corp.,
Waters Corp..
America-Major Pharmaceuticals: AbbVie Inc., Baxalta Inc., Bristol-Myers Squibb Co., Eli Lilly & Co., Johnson & Johnson, MannKind Corp., Merck & Co., Pfizer Inc., Zoetis Inc..
America-Medical Devices: Boston Scientific Corp., Edwards Lifesciences Corp., Glaukos Corp., Globus Medical Inc., HeartWare International Inc., Hill-Rom Holdings, Intuitive Surgical Inc., Medtronic plc,
NuVasive Inc., St. Jude Medical Inc., Stryker Corp., The Cooper Companies Inc., Varian Medical Systems Inc., ZELTIQ Aesthetics Inc., Zimmer Biomet Holdings.
America-Medical Supplies: Abbott Laboratories, Baxter International Inc., Becton Dickinson & Co., C.R. Bard Inc., Haemonetics Corp..
America-Pharmaceuticals Generics: Allergan Plc, Mylan NV, Perrigo Co., Teva Pharmaceuticals.
America-SMID Biotech: Agios Pharmaceuticals Inc., Alkermes Plc, Alnylam Pharmaceuticals Inc., Ariad Pharmaceuticals Inc., Blueprint Medicines Corp., Clovis Oncology Inc., Dimension Therapeutics,
FibroGen Inc., Global Blood Therapeutics Inc., Ophthotech Corp., Seres Therapeutics Inc., United Therapeutics Corp..
America-Specialty & SMID Pharmaceuticals: Akorn Inc., Anacor Pharmaceuticals Inc., Catalent Inc., Endo International Plc, Horizon Pharma Plc, Impax Laboratories Inc., Ironwood Pharmaceuticals Inc.,
Jazz Pharmaceuticals, Mallinckrodt Plc, Momenta Pharmaceuticals Inc., Pacira Pharmaceuticals Inc., Sage Therapeutics Inc., Valeant Pharmaceuticals, Valeant Pharmaceuticals.
Distribution of ratings/investment banking relationships
Goldman Sachs Investment Research global coverage universe
Rating Distribution
Buy
Hold
Investment Banking Relationships
Sell
Buy
Hold
Sell
Global
32%
53%
15%
63%
57%
52%
As of October 1, 2015, Goldman Sachs Global Investment Research had investment ratings on 3,221 equity securities. Goldman Sachs assigns stocks as Buys and Sells on various regional Investment
Lists; stocks not so assigned are deemed Neutral. Such assignments equate to Buy, Hold and Sell for the purposes of the above disclosure required by NASD/NYSE rules. See 'Ratings, Coverage
groups and views and related definitions' below.
Regulatory disclosures
Disclosures required by United States laws and regulations
See company-specific regulatory disclosures above for any of the following disclosures required as to companies referred to in this report: manager or co-manager in a pending transaction; 1% or
other ownership; compensation for certain services; types of client relationships; managed/co-managed public offerings in prior periods; directorships; for equity securities, market making and/or
specialist role. Goldman Sachs usually makes a market in fixed income securities of issuers discussed in this report and usually deals as a principal in these securities.
The following are additional required disclosures: Ownership and material conflicts of interest: Goldman Sachs policy prohibits its analysts, professionals reporting to analysts and members of their
households from owning securities of any company in the analyst's area of coverage. Analyst compensation: Analysts are paid in part based on the profitability of Goldman Sachs, which includes
investment banking revenues. Analyst as officer or director: Goldman Sachs policy prohibits its analysts, persons reporting to analysts or members of their households from serving as an officer,
director, advisory board member or employee of any company in the analyst's area of coverage. Non-U.S. Analysts: Non-U.S. analysts may not be associated persons of Goldman, Sachs & Co. and
therefore may not be subject to NASD Rule 2711/NYSE Rules 472 restrictions on communications with subject company, public appearances and trading securities held by the analysts.
Distribution of ratings: See the distribution of ratings disclosure above. Price chart: See the price chart, with changes of ratings and price targets in prior periods, above, or, if electronic format or if
with respect to multiple companies which are the subject of this report, on the Goldman Sachs website at http://www.gs.com/research/hedge.html.
Additional disclosures required under the laws and regulations of jurisdictions other than the United States
The following disclosures are those required by the jurisdiction indicated, except to the extent already made above pursuant to United States laws and regulations. Australia: Goldman Sachs Australia
Pty Ltd and its affiliates are not authorised deposit-taking institutions (as that term is defined in the Banking Act 1959 (Cth)) in Australia and do not provide banking services, nor carry on a banking
business, in Australia. This research, and any access to it, is intended only for "wholesale clients" within the meaning of the Australian Corporations Act, unless otherwise agreed by Goldman Sachs. In
producing research reports, members of the Global Investment Research Division of Goldman Sachs Australia may attend site visits and other meetings hosted by the issuers the subject of its research
reports. In some instances the costs of such site visits or meetings may be met in part or in whole by the issuers concerned if Goldman Sachs Australia considers it is appropriate and reasonable in the
specific circumstances relating to the site visit or meeting. Brazil: Disclosure information in relation to CVM Instruction 483 is available at http://www.gs.com/worldwide/brazil/area/gir/index.html.
Where applicable, the Brazil-registered analyst primarily responsible for the content of this research report, as defined in Article 16 of CVM Instruction 483, is the first author named at the beginning of
this report, unless indicated otherwise at the end of the text. Canada: Goldman Sachs Canada Inc. is an affiliate of The Goldman Sachs Group Inc. and therefore is included in the company specific
disclosures relating to Goldman Sachs (as defined above). Goldman Sachs Canada Inc. has approved of, and agreed to take responsibility for, this research report in Canada if and to the extent that
Goldman Sachs Canada Inc. disseminates this research report to its clients. Hong Kong: Further information on the securities of covered companies referred to in this research may be obtained on
request from Goldman Sachs (Asia) L.L.C. India: Further information on the subject company or companies referred to in this research may be obtained from Goldman Sachs (India) Securities Private
Limited. Goldman Sachs may beneficially own 1% or more of the securities (as such term is defined in clause 2 (h) the Indian Securities Contracts (Regulation) Act, 1956) of the subject company or
companies referred to in this research report. Japan: See below. Korea: Further information on the subject company or companies referred to in this research may be obtained from Goldman Sachs
(Asia) L.L.C., Seoul Branch. New Zealand: Goldman Sachs New Zealand Limited and its affiliates are neither "registered banks" nor "deposit takers" (as defined in the Reserve Bank of New Zealand Act
1989) in New Zealand. This research, and any access to it, is intended for "wholesale clients" (as defined in the Financial Advisers Act 2008) unless otherwise agreed by Goldman
Goldman Sachs Global Investment Research
36
December 17, 2015
Americas: Healthcare
Sachs. Russia: Research reports distributed in the Russian Federation are not advertising as defined in the Russian legislation, but are information and analysis not having product promotion as their
main purpose and do not provide appraisal within the meaning of the Russian legislation on appraisal activity. Singapore: Further information on the covered companies referred to in this research
may be obtained from Goldman Sachs (Singapore) Pte. (Company Number: 198602165W). Taiwan: This material is for reference only and must not be reprinted without permission. Investors should
carefully consider their own investment risk. Investment results are the responsibility of the individual investor. United Kingdom: Persons who would be categorized as retail clients in the United
Kingdom, as such term is defined in the rules of the Financial Conduct Authority, should read this research in conjunction with prior Goldman Sachs research on the covered companies referred to
herein and should refer to the risk warnings that have been sent to them by Goldman Sachs International. A copy of these risks warnings, and a glossary of certain financial terms used in this report,
are available from Goldman Sachs International on request.
European Union: Disclosure information in relation to Article 4 (1) (d) and Article 6 (2) of the European Commission Directive 2003/126/EC is available at
http://www.gs.com/disclosures/europeanpolicy.html which states the European Policy for Managing Conflicts of Interest in Connection with Investment Research.
Japan: Goldman Sachs Japan Co., Ltd. is a Financial Instrument Dealer registered with the Kanto Financial Bureau under registration number Kinsho 69, and a member of Japan Securities Dealers
Association, Financial Futures Association of Japan and Type II Financial Instruments Firms Association. Sales and purchase of equities are subject to commission pre-determined with clients plus
consumption tax. See company-specific disclosures as to any applicable disclosures required by Japanese stock exchanges, the Japanese Securities Dealers Association or the Japanese Securities
Finance Company.
Ratings, coverage groups and views and related definitions
Buy (B), Neutral (N), Sell (S) -Analysts recommend stocks as Buys or Sells for inclusion on various regional Investment Lists. Being assigned a Buy or Sell on an Investment List is determined by a
stock's return potential relative to its coverage group as described below. Any stock not assigned as a Buy or a Sell on an Investment List is deemed Neutral. Each regional Investment Review
Committee manages various regional Investment Lists to a global guideline of 25%-35% of stocks as Buy and 10%-15% of stocks as Sell; however, the distribution of Buys and Sells in any particular
coverage group may vary as determined by the regional Investment Review Committee. Regional Conviction Buy and Sell lists represent investment recommendations focused on either the size of the
potential return or the likelihood of the realization of the return.
Return potential represents the price differential between the current share price and the price target expected during the time horizon associated with the price target. Price targets are required for all
covered stocks. The return potential, price target and associated time horizon are stated in each report adding or reiterating an Investment List membership.
Coverage groups and views: A list of all stocks in each coverage group is available by primary analyst, stock and coverage group at http://www.gs.com/research/hedge.html. The analyst assigns one
of the following coverage views which represents the analyst's investment outlook on the coverage group relative to the group's historical fundamentals and/or valuation. Attractive (A). The
investment outlook over the following 12 months is favorable relative to the coverage group's historical fundamentals and/or valuation. Neutral (N). The investment outlook over the following 12
months is neutral relative to the coverage group's historical fundamentals and/or valuation. Cautious (C). The investment outlook over the following 12 months is unfavorable relative to the coverage
group's historical fundamentals and/or valuation.
Not Rated (NR). The investment rating and target price have been removed pursuant to Goldman Sachs policy when Goldman Sachs is acting in an advisory capacity in a merger or strategic
transaction involving this company and in certain other circumstances. Rating Suspended (RS). Goldman Sachs Research has suspended the investment rating and price target for this stock, because
there is not a sufficient fundamental basis for determining, or there are legal, regulatory or policy constraints around publishing, an investment rating or target. The previous investment rating and
price target, if any, are no longer in effect for this stock and should not be relied upon. Coverage Suspended (CS). Goldman Sachs has suspended coverage of this company. Not
Covered (NC). Goldman Sachs does not cover this company. Not Available or Not Applicable (NA). The information is not available for display or is not applicable. Not Meaningful (NM). The
information is not meaningful and is therefore excluded.
Global product; distributing entities
The Global Investment Research Division of Goldman Sachs produces and distributes research products for clients of Goldman Sachs on a global basis. Analysts based in Goldman Sachs offices
around the world produce equity research on industries and companies, and research on macroeconomics, currencies, commodities and portfolio strategy. This research is disseminated in Australia
by Goldman Sachs Australia Pty Ltd (ABN 21 006 797 897); in Brazil by Goldman Sachs do Brasil Corretora de Ttulos e Valores Mobilirios S.A.; in Canada by either Goldman Sachs Canada Inc. or
Goldman, Sachs & Co.; in Hong Kong by Goldman Sachs (Asia) L.L.C.; in India by Goldman Sachs (India) Securities Private Ltd.; in Japan by Goldman Sachs Japan Co., Ltd.; in the Republic of Korea by
Goldman Sachs (Asia) L.L.C., Seoul Branch; in New Zealand by Goldman Sachs New Zealand Limited; in Russia by OOO Goldman Sachs; in Singapore by Goldman Sachs (Singapore) Pte. (Company
Number: 198602165W); and in the United States of America by Goldman, Sachs & Co. Goldman Sachs International has approved this research in connection with its distribution in the United
Kingdom and European Union.
European Union: Goldman Sachs International authorised by the Prudential Regulation Authority and regulated by the Financial Conduct Authority and the Prudential Regulation Authority, has
approved this research in connection with its distribution in the European Union and United Kingdom; Goldman Sachs AG and Goldman Sachs International Zweigniederlassung Frankfurt, regulated
by the Bundesanstalt fr Finanzdienstleistungsaufsicht, may also distribute research in Germany.
General disclosures
This research is for our clients only. Other than disclosures relating to Goldman Sachs, this research is based on current public information that we consider reliable, but we do not represent it is
accurate or complete, and it should not be relied on as such. We seek to update our research as appropriate, but various regulations may prevent us from doing so. Other than certain industry reports
published on a periodic basis, the large majority of reports are published at irregular intervals as appropriate in the analyst's judgment.
Goldman Sachs conducts a global full-service, integrated investment banking, investment management, and brokerage business. We have investment banking and other business relationships with a
substantial percentage of the companies covered by our Global Investment Research Division. Goldman, Sachs & Co., the United States broker dealer, is a member of SIPC (http://www.sipc.org).
Goldman Sachs Global Investment Research
37
December 17, 2015
Americas: Healthcare
Our salespeople, traders, and other professionals may provide oral or written market commentary or trading strategies to our clients and principal trading desks that reflect opinions that are contrary
to the opinions expressed in this research. Our asset management area, principal trading desks and investing businesses may make investment decisions that are inconsistent with the
recommendations or views expressed in this research.
The analysts named in this report may have from time to time discussed with our clients, including Goldman Sachs salespersons and traders, or may discuss in this report, trading strategies that
reference catalysts or events that may have a near-term impact on the market price of the equity securities discussed in this report, which impact may be directionally counter to the analyst's published
price target expectations for such stocks. Any such trading strategies are distinct from and do not affect the analyst's fundamental equity rating for such stocks, which rating reflects a stock's return
potential relative to its coverage group as described herein.
We and our affiliates, officers, directors, and employees, excluding equity and credit analysts, will from time to time have long or short positions in, act as principal in, and buy or sell, the securities or
derivatives, if any, referred to in this research.
The views attributed to third party presenters at Goldman Sachs arranged conferences, including individuals from other parts of Goldman Sachs, do not necessarily reflect those of Global Investment
Research and are not an official view of Goldman Sachs.
Any third party referenced herein, including any salespeople, traders and other professionals or members of their household, may have positions in the products mentioned that are inconsistent with
the views expressed by analysts named in this report.
This research is not an offer to sell or the solicitation of an offer to buy any security in any jurisdiction where such an offer or solicitation would be illegal. It does not constitute a personal
recommendation or take into account the particular investment objectives, financial situations, or needs of individual clients. Clients should consider whether any advice or recommendation in this
research is suitable for their particular circumstances and, if appropriate, seek professional advice, including tax advice. The price and value of investments referred to in this research and the income
from them may fluctuate. Past performance is not a guide to future performance, future returns are not guaranteed, and a loss of original capital may occur. Fluctuations in exchange rates could have
adverse effects on the value or price of, or income derived from, certain investments.
Certain transactions, including those involving futures, options, and other derivatives, give rise to substantial risk and are not suitable for all investors. Investors should review current options
disclosure documents which are available from Goldman Sachs sales representatives or at http://www.theocc.com/about/publications/character-risks.jsp. Transaction costs may be significant in option
strategies calling for multiple purchase and sales of options such as spreads. Supporting documentation will be supplied upon request.
All research reports are disseminated and available to all clients simultaneously through electronic publication to our internal client websites. Not all research content is redistributed to our clients or
available to third-party aggregators, nor is Goldman Sachs responsible for the redistribution of our research by third party aggregators. For research, models or other data available on a particular
security, please contact your sales representative or go to http://360.gs.com.
Disclosure information is also available at http://www.gs.com/research/hedge.html or from Research Compliance, 200 West Street, New York, NY 10282.
2015 Goldman Sachs.
No part of this material may be (i) copied, photocopied or duplicated in any form by any means or (ii) redistributed without the prior written consent of The Goldman Sachs
Group, Inc.
Goldman Sachs Global Investment Research
38
Das könnte Ihnen auch gefallen
- Loan Workouts and Debt for Equity Swaps: A Framework for Successful Corporate RescuesVon EverandLoan Workouts and Debt for Equity Swaps: A Framework for Successful Corporate RescuesBewertung: 5 von 5 Sternen5/5 (1)
- The GIC Weekly Update November 2015Dokument12 SeitenThe GIC Weekly Update November 2015John MathiasNoch keine Bewertungen
- M & A ActivitiesDokument6 SeitenM & A ActivitieschengadNoch keine Bewertungen
- Reshuffling The Debt: The Credit Implications of The New Wave of LbosDokument10 SeitenReshuffling The Debt: The Credit Implications of The New Wave of LbosDavid TollNoch keine Bewertungen
- CS - Investment Outlook 2023 A Fundamental Reset - 20221124Dokument35 SeitenCS - Investment Outlook 2023 A Fundamental Reset - 20221124Alex ZumwinkelNoch keine Bewertungen
- Best Ideas 10.3Dokument65 SeitenBest Ideas 10.3Mallory WeilNoch keine Bewertungen
- 2014-01-28 From JPM Squaring The Circle - A Comprehensive Review of AT1 Valuation DriversDokument72 Seiten2014-01-28 From JPM Squaring The Circle - A Comprehensive Review of AT1 Valuation DriverserereretzutzutzuNoch keine Bewertungen
- Initiating Coverage of Single-Family Rental Reits With Favorable View - Buy SFRDokument50 SeitenInitiating Coverage of Single-Family Rental Reits With Favorable View - Buy SFRtempvjNoch keine Bewertungen
- The Effects of Leveraged Buyouts On ProdDokument55 SeitenThe Effects of Leveraged Buyouts On ProdsayyedtarannumNoch keine Bewertungen
- JPM Registry Provider Survey Report March 2012Dokument33 SeitenJPM Registry Provider Survey Report March 2012Nicholas AngNoch keine Bewertungen
- Moody's Rating SystemDokument2 SeitenMoody's Rating SystemTL Ngoc ThaoNoch keine Bewertungen
- Lbo Model Long FormDokument5 SeitenLbo Model Long FormGabriella RicardoNoch keine Bewertungen
- CampelloSaffi15 PDFDokument39 SeitenCampelloSaffi15 PDFdreamjongenNoch keine Bewertungen
- RBC - ARCC - Initiation - Research at A Glance - 17 PagesDokument17 SeitenRBC - ARCC - Initiation - Research at A Glance - 17 PagesSagar PatelNoch keine Bewertungen
- Moodys Analytics Riskcalc Transfer Pricing SolutionDokument9 SeitenMoodys Analytics Riskcalc Transfer Pricing SolutionManuel CámacNoch keine Bewertungen
- Evercore - 2022 Year in ReviewDokument35 SeitenEvercore - 2022 Year in ReviewMehul GargNoch keine Bewertungen
- Assignment 2 - Islamic FinanceDokument10 SeitenAssignment 2 - Islamic FinanceAina SaffiyahNoch keine Bewertungen
- U.S. Large-Cap & Mid-Cap Banks - Drilling Into Energy ExposureDokument39 SeitenU.S. Large-Cap & Mid-Cap Banks - Drilling Into Energy ExposureMartin TsankovNoch keine Bewertungen
- Starboard Value Activist - CalgonDokument53 SeitenStarboard Value Activist - CalgonCanadianValueNoch keine Bewertungen
- Cole Park CLO LimitedDokument22 SeitenCole Park CLO Limitedeimg20041333Noch keine Bewertungen
- Fitch C Do RatingDokument29 SeitenFitch C Do RatingMakarand LonkarNoch keine Bewertungen
- Jpmorgan Chase & Co.: Company OverviewDokument4 SeitenJpmorgan Chase & Co.: Company OverviewIsh SelinNoch keine Bewertungen
- How Sovereign Credit Quality May Affect Other RatingsDokument9 SeitenHow Sovereign Credit Quality May Affect Other RatingsxanasadjoNoch keine Bewertungen
- GMT Research On Chinese SportswearDokument43 SeitenGMT Research On Chinese SportswearJimmy C K ChoiNoch keine Bewertungen
- S&P Default Rates and Recovery Jan07Dokument9 SeitenS&P Default Rates and Recovery Jan07gimy2010Noch keine Bewertungen
- 2Q14 Roadmap To ValuationDokument24 Seiten2Q14 Roadmap To ValuationRyan BrookmanNoch keine Bewertungen
- Stern Stewart - Evaluation PDFDokument8 SeitenStern Stewart - Evaluation PDFAntoanela MihăilăNoch keine Bewertungen
- q2 Valuation Insights Second 2020 PDFDokument20 Seitenq2 Valuation Insights Second 2020 PDFKojiro FuumaNoch keine Bewertungen
- GICS MapbookDokument52 SeitenGICS MapbookVivek AryaNoch keine Bewertungen
- JPM 2018 Biotech OutlookDokument92 SeitenJPM 2018 Biotech Outlookaaquibnasir100% (1)
- FADMDokument27 SeitenFADMManju TripathiNoch keine Bewertungen
- JPM Software Technology 2013-07-12 1161609Dokument24 SeitenJPM Software Technology 2013-07-12 1161609Dinesh MahajanNoch keine Bewertungen
- Leveraged Buyout Bankrupties, The Problem of HindsightDokument104 SeitenLeveraged Buyout Bankrupties, The Problem of Hindsight83jjmackNoch keine Bewertungen
- Premier Hedge Fund Conference Agenda 2017 PDFDokument4 SeitenPremier Hedge Fund Conference Agenda 2017 PDFLuwei ShenNoch keine Bewertungen
- Lbo ModelDokument4 SeitenLbo Modelharshalp1212Noch keine Bewertungen
- 2019 q1 Earnings Results PresentationDokument23 Seiten2019 q1 Earnings Results PresentationValter SilveiraNoch keine Bewertungen
- Oil Price Benchmarks: Legal DisclaimerDokument53 SeitenOil Price Benchmarks: Legal Disclaimersushilk28Noch keine Bewertungen
- Convertibles Primer: Convertible SecuritiesDokument6 SeitenConvertibles Primer: Convertible SecuritiesIsaac GoldNoch keine Bewertungen
- CRE Chartbook 4Q 2012Dokument11 SeitenCRE Chartbook 4Q 2012subwayguyNoch keine Bewertungen
- 4 - Tech Hardware SectorDokument126 Seiten4 - Tech Hardware SectorgirishrajsNoch keine Bewertungen
- BM410-16 Equity Valuation 2 - Intrinsic Value 19oct05Dokument35 SeitenBM410-16 Equity Valuation 2 - Intrinsic Value 19oct05muhammadanasmustafaNoch keine Bewertungen
- GS 1674363437Dokument18 SeitenGS 1674363437Piyush GujarNoch keine Bewertungen
- JPM Mortgage ResearchDokument30 SeitenJPM Mortgage ResearchflhaliliNoch keine Bewertungen
- Barcap India - Capital - Goods 7 Dec 2011Dokument159 SeitenBarcap India - Capital - Goods 7 Dec 2011Sunayan PalNoch keine Bewertungen
- Nordea TaaDokument25 SeitenNordea Taaintel74Noch keine Bewertungen
- JP Morgan Best Equity Ideas 2014Dokument61 SeitenJP Morgan Best Equity Ideas 2014Sara Lim100% (1)
- The GIC WeeklyDokument12 SeitenThe GIC WeeklyedgarmerchanNoch keine Bewertungen
- Gic WeeklyDokument12 SeitenGic WeeklycampiyyyyoNoch keine Bewertungen
- Rates, Rotation, and Re Ation: Taking Stock of 1Q 2021: US Quarterly ChartbookDokument51 SeitenRates, Rotation, and Re Ation: Taking Stock of 1Q 2021: US Quarterly ChartbookShivam GuptaNoch keine Bewertungen
- Cio Spotlight ReportDokument9 SeitenCio Spotlight Reporthkm_gmat4849Noch keine Bewertungen
- Facebook Analysis BernsteinDokument36 SeitenFacebook Analysis BernsteinRama RajuNoch keine Bewertungen
- GS OutlookDokument30 SeitenGS OutlookMrittunjayNoch keine Bewertungen
- WTW 3Q12 Earnings TranscriptDokument14 SeitenWTW 3Q12 Earnings Transcriptrtao719Noch keine Bewertungen
- Asean Strategy - JPM PDFDokument19 SeitenAsean Strategy - JPM PDFRizkyAufaSNoch keine Bewertungen
- Seminar 3 Discount Rates & Yield Curves (Lecture Notes Recap)Dokument6 SeitenSeminar 3 Discount Rates & Yield Curves (Lecture Notes Recap)api-3695734100% (1)
- Leverage Buyout and Junk KbondsDokument31 SeitenLeverage Buyout and Junk KbondsChetan VenuNoch keine Bewertungen
- Core ConceptsDokument35 SeitenCore ConceptsMin ChenNoch keine Bewertungen
- CLO Investing: With an Emphasis on CLO Equity & BB NotesVon EverandCLO Investing: With an Emphasis on CLO Equity & BB NotesNoch keine Bewertungen
- Canary or Phoenix Balancing The Key Drivers of Credit in 2016 Mar 2016Dokument10 SeitenCanary or Phoenix Balancing The Key Drivers of Credit in 2016 Mar 2016satish sNoch keine Bewertungen
- The VIX Finally Went Bananas - Morgan Stanley's Post Mortem of Everything That Happened - Zero HedgeDokument12 SeitenThe VIX Finally Went Bananas - Morgan Stanley's Post Mortem of Everything That Happened - Zero HedgeeliforuNoch keine Bewertungen
- XIV Trader Loses $4 Million and 3 Years of Work Overnight - Here Is His Story - Zero HedgeDokument9 SeitenXIV Trader Loses $4 Million and 3 Years of Work Overnight - Here Is His Story - Zero HedgeeliforuNoch keine Bewertungen
- 1987 Zero HedgeDokument9 Seiten1987 Zero Hedgeeliforu100% (1)
- Here Is What Was Behind The - Largest VIX Buy Order in History - Zero HedgeDokument7 SeitenHere Is What Was Behind The - Largest VIX Buy Order in History - Zero HedgeeliforuNoch keine Bewertungen
- Why For Stock Markets Bulls, Monday Could Be - The Most Important Day - Zero HedgeDokument4 SeitenWhy For Stock Markets Bulls, Monday Could Be - The Most Important Day - Zero HedgeeliforuNoch keine Bewertungen
- Revisiting Bob Farrell's 10-Rules of Investing - Zero HedgeDokument13 SeitenRevisiting Bob Farrell's 10-Rules of Investing - Zero HedgeeliforuNoch keine Bewertungen
- Was Coinbase's Bitcoin Cash Rollout A Designed HitDokument5 SeitenWas Coinbase's Bitcoin Cash Rollout A Designed HiteliforuNoch keine Bewertungen
- The US Government Has Experimented With Controlling HurricanesDokument6 SeitenThe US Government Has Experimented With Controlling HurricaneseliforuNoch keine Bewertungen
- Lead From BehindDokument4 SeitenLead From BehindeliforuNoch keine Bewertungen
- How To Forecast Markets - A Departing Top JPMorgan Strategist Reveals What He Learned After 30 YearsDokument7 SeitenHow To Forecast Markets - A Departing Top JPMorgan Strategist Reveals What He Learned After 30 Yearseliforu100% (1)
- When You Kill Ten Million Africans You Aren't Called Hitler' - Diary of A Walking Butterfly - Diary of A Walking ButterflyDokument3 SeitenWhen You Kill Ten Million Africans You Aren't Called Hitler' - Diary of A Walking Butterfly - Diary of A Walking Butterflyeliforu100% (1)
- Crafting A StrategyDokument5 SeitenCrafting A StrategyJanakiRamanNoch keine Bewertungen
- IBPS PO Model Paper - Bank of Maharashtra Probationary Officer 2007 Solved Question PaperDokument38 SeitenIBPS PO Model Paper - Bank of Maharashtra Probationary Officer 2007 Solved Question PaperMurali KhanNoch keine Bewertungen
- Inbound Foreign Direct Investment in Japan A Typology PDFDokument12 SeitenInbound Foreign Direct Investment in Japan A Typology PDFThu LeNoch keine Bewertungen
- Fixed AssetsDokument188 SeitenFixed AssetsPaul PulancoNoch keine Bewertungen
- Dow Completes Merger With Rohm and HaasDokument2 SeitenDow Completes Merger With Rohm and HaasLh KooNoch keine Bewertungen
- Ebook PDF Commentaries and Cases On The Law of Business Organization 5th PDFDokument41 SeitenEbook PDF Commentaries and Cases On The Law of Business Organization 5th PDFrobert.gonzales146100% (38)
- SBI Merger With His Associate BanksDokument38 SeitenSBI Merger With His Associate BanksVipulNoch keine Bewertungen
- Chap 016Dokument8 SeitenChap 016Bobby MarionNoch keine Bewertungen
- M&A ProjectDokument20 SeitenM&A ProjectPUNYASHLOK PANDANoch keine Bewertungen
- Strategic Management MethodologyDokument203 SeitenStrategic Management MethodologyChristophe LacourNoch keine Bewertungen
- Compilation OF Suggested Answers: QuestionsDokument8 SeitenCompilation OF Suggested Answers: QuestionsRaul KarkyNoch keine Bewertungen
- Danta Sowmya FinanceDokument1 SeiteDanta Sowmya FinanceAbhishekccNoch keine Bewertungen
- Ch14 HRM LenovoDokument20 SeitenCh14 HRM Lenovohiennguyen.31211020907Noch keine Bewertungen
- Types of FDIDokument2 SeitenTypes of FDIhiwale_sonalNoch keine Bewertungen
- RECRUITMENT AND SELECTION McqsDokument6 SeitenRECRUITMENT AND SELECTION Mcqsalostguy1100% (1)
- Golden Age of China's Education Industry: Seize The MomentumDokument42 SeitenGolden Age of China's Education Industry: Seize The MomentumJ. BangjakNoch keine Bewertungen
- Fall 2020 Barzuza CorporationsDokument12 SeitenFall 2020 Barzuza Corporationsa thaynNoch keine Bewertungen
- Ingenious ExecutiveOverview 30sep2019Dokument52 SeitenIngenious ExecutiveOverview 30sep2019William DuNoch keine Bewertungen
- Philippines Land Ownership and AcquisitionDokument5 SeitenPhilippines Land Ownership and AcquisitionLRMNoch keine Bewertungen
- Group Accounts (IFRS)Dokument10 SeitenGroup Accounts (IFRS)Yaz CarlomanNoch keine Bewertungen
- Marketing Research TemplateDokument3 SeitenMarketing Research TemplateSonakshi BehlNoch keine Bewertungen
- Work of Investment Banking (Repaired)Dokument12 SeitenWork of Investment Banking (Repaired)Shivani Singh ChandelNoch keine Bewertungen
- Corporate RestructuringDokument20 SeitenCorporate RestructuringHardik UdaniNoch keine Bewertungen
- 2018 Elf DirectoryDokument20 Seiten2018 Elf DirectoryAkanksha GehlotNoch keine Bewertungen
- Price List Principal - 02 Okt 19Dokument439 SeitenPrice List Principal - 02 Okt 19Stephen SatigiNoch keine Bewertungen
- Strategic Approaches in M&ADokument39 SeitenStrategic Approaches in M&AbrekhaaNoch keine Bewertungen
- Managerial Notes For All ChaptersDokument479 SeitenManagerial Notes For All Chaptershenryuab100% (1)
- CV 1e - PPT - Ch01 - 051115Dokument74 SeitenCV 1e - PPT - Ch01 - 051115Yu FengNoch keine Bewertungen
- Dissertation Topics For Masters in FinanceDokument6 SeitenDissertation Topics For Masters in FinancePayForPaperUK100% (1)
- Reasons Behind Non Ethical BehaviourDokument15 SeitenReasons Behind Non Ethical BehaviourAndreea JustNoch keine Bewertungen