Beruflich Dokumente
Kultur Dokumente
2 3 Useful Chemical Building Blocks
Hochgeladen von
griggans100%(1)100% fanden dieses Dokument nützlich (1 Abstimmung)
198 Ansichten1 SeiteThis document provides an overview of key chemical concepts including:
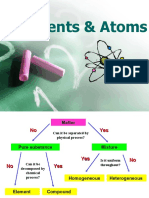
- Elements contain only one type of atom, while compounds contain two or more different types of atoms bonded together. Compounds are difficult to separate into their elements.
- Mixtures contain two or more substances that are not chemically bonded and are easy to separate.
- All matter is made up of atoms, which contain protons, neutrons, and electrons. The structure of the atom determines an element's identity.
Originalbeschreibung:
Originaltitel
2 3 useful chemical building blocks
Copyright
© Attribution Non-Commercial (BY-NC)
Verfügbare Formate
DOC, PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenThis document provides an overview of key chemical concepts including:
- Elements contain only one type of atom, while compounds contain two or more different types of atoms bonded together. Compounds are difficult to separate into their elements.
- Mixtures contain two or more substances that are not chemically bonded and are easy to separate.
- All matter is made up of atoms, which contain protons, neutrons, and electrons. The structure of the atom determines an element's identity.
Copyright:
Attribution Non-Commercial (BY-NC)
Verfügbare Formate
Als DOC, PDF, TXT herunterladen oder online auf Scribd lesen
100%(1)100% fanden dieses Dokument nützlich (1 Abstimmung)
198 Ansichten1 Seite2 3 Useful Chemical Building Blocks
Hochgeladen von
griggansThis document provides an overview of key chemical concepts including:
- Elements contain only one type of atom, while compounds contain two or more different types of atoms bonded together. Compounds are difficult to separate into their elements.
- Mixtures contain two or more substances that are not chemically bonded and are easy to separate.
- All matter is made up of atoms, which contain protons, neutrons, and electrons. The structure of the atom determines an element's identity.
Copyright:
Attribution Non-Commercial (BY-NC)
Verfügbare Formate
Als DOC, PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 1
2.
3 Useful Chemicals (Building Key Points Key terms
blocks)
Syllabus Summary (H) = higher only Elements, Compounds and Mixtures Element
You should be able to: • Elements contain only 1 type of atom, compounds contain at least 2 different atoms Product
Compound Balanced
• define an element, compound and mixture; joined together
Mixture Formula
• match up symbols and names of elements • Compounds are very difficult to separate into their elements
Atom Organic
• recall some chemical symbols • Mixtures are two substances that are not chemically combined
Nucleus Inorganic
• classify materials as elements, compounds • Mixtures are easy to separate
Proton
or mixtures using information provided
Neutron
• State that all matter is made up of atoms
Electron
• recall the structure of the atom
Reactant
• recall that all of the atoms that make up an
element have the same number of protons
and electrons in their atoms.
• Match up formulae and names of some
compounds
• Recognise reactants and products in a Element Compound Mixture
symbol equation
• name the some compounds listed given The Atom
• Atoms are made up of protons, neutrons and electrons. Protons and neutrons are
their formulae;
found in the nucleus, electrons orbit the nucleus in shells. The first shell can hold a
• write word and symbol equations to show
max of 2 electrons, the second and third shells hold a max of 8
how atoms are rearranged in the chemical
• Protons are positive and have a mass of 1, neutrons are neutral and have a mass of 1
reactions
and electrons are negative and have a mass of 0
• recall and use the formulae of some
compounds l to write balanced symbol Chemical Symbols and Formulae
equations (H)
• Most element symbols are the first letter of the name e.g. Carbon = C
• state that scientists sort out chemicals into C2H7N
• Some are the first two letters of the name e.g. Cobalt = Co
two groups, those that contain carbon and
those that do not; • A few are the first letter and another letter from the name e.g. Magnesium = Mg
• state that living things all contain the • Some are the first letter or letters of the latin name e.g. Sodium = Natrium = Na
element carbon and non-living usually don’t • The formula of a compound tells us which atoms and how many it contains e.g.
• recall the names of some chemicals that do CO2 = 1 x carbon atom and 2 x oxygen atoms
not contain the element carbon
• recognise inorganic and organic Equations
substances; • An equation can be written in words or symbols
• use the words organic and inorganic
correctly and explain the words origin E.G. Magnesium + Oxygen → Magnesium Oxide
• use chemical formulae to recognise a 2 Mg + O2 → 2MgO (must be balanced)
substance as organic or inorganic
(reactants) → (products)
• recall that there are many useful
substances that contain carbon atoms
Organic compounds
• explain why fossil fuels are a source of
• Organic compounds contain carbon, inorganic compound don’t
organic substances;
• Livings things all contain the element carbon
• state that organic chemicals are very • Many useful substances contain carbon e.g. plastics, petrol, products from oil
important in our lives and most are derived
from crude oil.
Das könnte Ihnen auch gefallen
- Chemistry of Cell Review: Quick Review Notes Chapter 2Von EverandChemistry of Cell Review: Quick Review Notes Chapter 2Noch keine Bewertungen
- Chapter 1: Atoms, Molecules and IonsDokument103 SeitenChapter 1: Atoms, Molecules and IonsSyahir HamidonNoch keine Bewertungen
- GRADE 8 CHEMISTRY Periodic Table Lesson 1Dokument20 SeitenGRADE 8 CHEMISTRY Periodic Table Lesson 1dodoNoch keine Bewertungen
- Topic 3 Atomic StructureDokument36 SeitenTopic 3 Atomic StructureKaixin HuangNoch keine Bewertungen
- BMED 105 Basic Chemistry of Life (NOTES)Dokument4 SeitenBMED 105 Basic Chemistry of Life (NOTES)Jobelle MalihanNoch keine Bewertungen
- Essentials Pearson 2016Dokument253 SeitenEssentials Pearson 2016Devansh SharmaNoch keine Bewertungen
- Elements Compounds and MixturesDokument24 SeitenElements Compounds and MixturesMegan SarahNoch keine Bewertungen
- 1 Lec - ChemDokument6 Seiten1 Lec - ChemAllen Rivera ReyesNoch keine Bewertungen
- CHM 092 CHAPTER 1 - Matter &stoichiometryDokument128 SeitenCHM 092 CHAPTER 1 - Matter &stoichiometryAisyah NadhirahNoch keine Bewertungen
- Chapter 2 Complete Lecture 4x4Dokument17 SeitenChapter 2 Complete Lecture 4x4michaelaNoch keine Bewertungen
- Non Metalic Substances and Covalent BondingDokument47 SeitenNon Metalic Substances and Covalent Bonding42h47n5zvrNoch keine Bewertungen
- Lecture 3 - Matter, Molecules and Periodic TableDokument40 SeitenLecture 3 - Matter, Molecules and Periodic TableNaff WariNoch keine Bewertungen
- Study Guides 2.1-2.3Dokument8 SeitenStudy Guides 2.1-2.3MA. ASUNCION BeroNoch keine Bewertungen
- Chapter 1 - Chemical FoundationDokument50 SeitenChapter 1 - Chemical Foundation杨致远Noch keine Bewertungen
- Engineering Chemistry: Course Code: 211502 Course NameDokument17 SeitenEngineering Chemistry: Course Code: 211502 Course NameSalama NaumanNoch keine Bewertungen
- Chapter 2 - Material Structure and Interatomic BondingDokument33 SeitenChapter 2 - Material Structure and Interatomic BondingamraqstnaNoch keine Bewertungen
- Intro Matter 2 PHDokument27 SeitenIntro Matter 2 PHCheick SANOUNoch keine Bewertungen
- Chapter 2 - Atoms and MoleculesDokument62 SeitenChapter 2 - Atoms and MoleculesRoselyn CastilloNoch keine Bewertungen
- BioM I Lectures 1 - 3 (SB) 2014Dokument69 SeitenBioM I Lectures 1 - 3 (SB) 2014nonhle nosiphoNoch keine Bewertungen
- Chemistry Workbook 1Dokument10 SeitenChemistry Workbook 1Dean KimNoch keine Bewertungen
- Atoms, Elements and Molecules & Compounds and MixturesDokument32 SeitenAtoms, Elements and Molecules & Compounds and MixturesTshanna RobertsNoch keine Bewertungen
- Lecture #9 Introduction To Organic Chemistry Organic ChemistryDokument10 SeitenLecture #9 Introduction To Organic Chemistry Organic ChemistryG8 ODL Mary Angeline M. GalmanNoch keine Bewertungen
- Atoms and Molecules: Larry Brown Tom HolmeDokument62 SeitenAtoms and Molecules: Larry Brown Tom Holmemuhammad ali shakeelNoch keine Bewertungen
- CHEMISTRYDokument3 SeitenCHEMISTRYSAN JOSE, KRIZZIA FAYE U.Noch keine Bewertungen
- Naming CompoundsDokument39 SeitenNaming CompoundsgallosaaronmatthewNoch keine Bewertungen
- Org Chem 5Dokument12 SeitenOrg Chem 5Bertille Marie ArdienteNoch keine Bewertungen
- Org Chem 3Dokument11 SeitenOrg Chem 3Bertille Marie ArdienteNoch keine Bewertungen
- Org Chem 2Dokument12 SeitenOrg Chem 2Bertille Marie ArdienteNoch keine Bewertungen
- Elements, Atoms, and The Atomic Theory: Type of AtomDokument7 SeitenElements, Atoms, and The Atomic Theory: Type of AtomSasikumar MadhavanNoch keine Bewertungen
- GEN CHEM CHEMICAL FORMULA and NAMING OF COMPOUNDSDokument35 SeitenGEN CHEM CHEMICAL FORMULA and NAMING OF COMPOUNDSKC KayeNoch keine Bewertungen
- Chemistry of LifeDokument11 SeitenChemistry of LifeNadiyah KarriemNoch keine Bewertungen
- Quarter 1 Module 2 Elements and CompoundsDokument30 SeitenQuarter 1 Module 2 Elements and CompoundsJeline MacallaNoch keine Bewertungen
- Science 9: The Variety of Carbon CompoundsDokument10 SeitenScience 9: The Variety of Carbon Compoundsrussel castilloNoch keine Bewertungen
- Principles of BiochemistryDokument68 SeitenPrinciples of Biochemistryblackss copsNoch keine Bewertungen
- Chapter 1: Fundamentals of Chemistry: Lesson 2: Atoms and MoleculesDokument9 SeitenChapter 1: Fundamentals of Chemistry: Lesson 2: Atoms and MoleculesKristine Cris VenusNoch keine Bewertungen
- Typeselements CompoundsDokument27 SeitenTypeselements CompoundsEverrome AsicoNoch keine Bewertungen
- 2.1 The Chemistry of Life EditedDokument68 Seiten2.1 The Chemistry of Life EditedPatricia Jayshree Samuel Jacob100% (1)
- Campbell Lecture Notes Chemistry of LifeDokument42 SeitenCampbell Lecture Notes Chemistry of LifeSophia Andrei VillalunaNoch keine Bewertungen
- CLASS 10 - ChemistryDokument38 SeitenCLASS 10 - ChemistryKavyansh GuptaNoch keine Bewertungen
- The Differences Between A Compound and A MixtureDokument27 SeitenThe Differences Between A Compound and A Mixturearies triwidajatiNoch keine Bewertungen
- Atom, Molecules & Ions (Ref: Dr. Khoa, Dr. Khánh, Dr. Thuận)Dokument22 SeitenAtom, Molecules & Ions (Ref: Dr. Khoa, Dr. Khánh, Dr. Thuận)HIEP PHAM HOANGNoch keine Bewertungen
- Basic Chemistry I: Gasal 2011/2012Dokument39 SeitenBasic Chemistry I: Gasal 2011/2012Mitch EspinasNoch keine Bewertungen
- Atoms PresentationDokument10 SeitenAtoms PresentationSu AlghNoch keine Bewertungen
- M1 Lec - HandoutDokument5 SeitenM1 Lec - HandoutAngel Fiona GañaNoch keine Bewertungen
- Study Questions Introduction To ChemistryDokument5 SeitenStudy Questions Introduction To ChemistryMsNoch keine Bewertungen
- Chemistry Functional Groups LECTUREDokument34 SeitenChemistry Functional Groups LECTUREkassy jayNoch keine Bewertungen
- Quarter1.Week2.Elements and CompoundDokument47 SeitenQuarter1.Week2.Elements and CompoundEvelyn ApostolNoch keine Bewertungen
- Elements and AtomsDokument28 SeitenElements and AtomsJulien Kristi HernandezNoch keine Bewertungen
- 3.3 Chemical FormulaDokument18 Seiten3.3 Chemical FormulaLIM CHEE BOON MoeNoch keine Bewertungen
- 2.1 Functional GroupsDokument4 Seiten2.1 Functional GroupsBasti SantiagoNoch keine Bewertungen
- ReactivityDokument42 SeitenReactivityDarryl WHDNoch keine Bewertungen
- Chapter 4-CarbonDokument23 SeitenChapter 4-Carbonjanardhan aghavNoch keine Bewertungen
- Lesson 4 General Chemistry 1Dokument16 SeitenLesson 4 General Chemistry 1Genevee Ryeleen DelfinNoch keine Bewertungen
- Chap 03Dokument22 SeitenChap 03AmandaNoch keine Bewertungen
- Edexcel AS Chemistry Note 1 Definitions of The TermsDokument3 SeitenEdexcel AS Chemistry Note 1 Definitions of The TermsSajaniNoch keine Bewertungen
- BASIC CHEMISTRY Lect 1Dokument23 SeitenBASIC CHEMISTRY Lect 1briosojoshua0Noch keine Bewertungen
- Lesson 2 and 3 ChemDokument3 SeitenLesson 2 and 3 ChemJazmine Lei PalomoNoch keine Bewertungen
- Schaum's Easy Outline of Organic Chemistry, Second EditionVon EverandSchaum's Easy Outline of Organic Chemistry, Second EditionBewertung: 3.5 von 5 Sternen3.5/5 (2)
- Biology FieldworkqsDokument9 SeitenBiology FieldworkqsgriggansNoch keine Bewertungen
- Biology b1 Core Practical QuestionsDokument17 SeitenBiology b1 Core Practical Questionsgriggans0% (1)
- Physics p1 Core PracticalsDokument19 SeitenPhysics p1 Core PracticalsgriggansNoch keine Bewertungen
- Exam-QuestionsQUADRATS AND TRANSECTSDokument8 SeitenExam-QuestionsQUADRATS AND TRANSECTSgriggansNoch keine Bewertungen
- Chemistry c1 Core PracticalsDokument18 SeitenChemistry c1 Core PracticalsgriggansNoch keine Bewertungen
- BTEC Science UNIT 5 NervesDokument4 SeitenBTEC Science UNIT 5 NervesgriggansNoch keine Bewertungen
- The Haber ProcessDokument1 SeiteThe Haber ProcessgriggansNoch keine Bewertungen
- Tomatosphere AssignmentDokument2 SeitenTomatosphere AssignmentgriggansNoch keine Bewertungen
- Heart Gcse Qs OnlyDokument6 SeitenHeart Gcse Qs OnlygriggansNoch keine Bewertungen
- Hormones: Part of Homeostasis and ControlDokument7 SeitenHormones: Part of Homeostasis and ControlgriggansNoch keine Bewertungen
- GSK AssignmentDokument4 SeitenGSK Assignmentgriggans100% (4)
- Chromatography StarterDokument1 SeiteChromatography StartergriggansNoch keine Bewertungen
- BTEC First Diploma in Applied Science Student Tracking SheetsDokument6 SeitenBTEC First Diploma in Applied Science Student Tracking Sheetsgriggans100% (6)
- Task and Mark Sheet 1)Dokument6 SeitenTask and Mark Sheet 1)griggans100% (1)
- Btec Front Sheet ParticlesDokument2 SeitenBtec Front Sheet ParticlesgriggansNoch keine Bewertungen
- Microscopes Assignment Brief and Task ListDokument2 SeitenMicroscopes Assignment Brief and Task Listgriggans100% (1)
- Btec Front Sheet RocketsDokument2 SeitenBtec Front Sheet Rocketsgriggans100% (1)
- Cats Milk Assignment Brief Sept 2006Dokument2 SeitenCats Milk Assignment Brief Sept 2006griggansNoch keine Bewertungen
- Assignment Title Chemical and Physical EvidenceDokument6 SeitenAssignment Title Chemical and Physical EvidencegriggansNoch keine Bewertungen
- Btec Front Sheet UnitsDokument1 SeiteBtec Front Sheet Unitsgriggans100% (2)
- 2 1 Living Organisms GeneticsDokument1 Seite2 1 Living Organisms GeneticsgriggansNoch keine Bewertungen
- Sats Revision QuizDokument126 SeitenSats Revision QuizgriggansNoch keine Bewertungen
- 2 2 Humans Moving Substances Around BodyDokument3 Seiten2 2 Humans Moving Substances Around Bodygriggans100% (1)
- Questions On Chrom Ato Graph yDokument5 SeitenQuestions On Chrom Ato Graph ygriggans100% (1)
- Chinese Paper Cutting Work SheetDokument4 SeitenChinese Paper Cutting Work Sheet黃梓Noch keine Bewertungen
- Bba VDokument2 SeitenBba VkunalbrabbitNoch keine Bewertungen
- CiscoDokument6 SeitenCiscoNatalia Kogan0% (2)
- Hand Out Fire SurpressDokument69 SeitenHand Out Fire SurpressSeptiawanWandaNoch keine Bewertungen
- Product Manual 26086 (Revision E) : EGCP-2 Engine Generator Control PackageDokument152 SeitenProduct Manual 26086 (Revision E) : EGCP-2 Engine Generator Control PackageErick KurodaNoch keine Bewertungen
- I I I I: Peroxid.Q!Dokument2 SeitenI I I I: Peroxid.Q!Diego PradelNoch keine Bewertungen
- Business-Communication Solved MCQs (Set-3)Dokument8 SeitenBusiness-Communication Solved MCQs (Set-3)Pavan Sai Krishna KottiNoch keine Bewertungen
- Dwnload Full Beckers World of The Cell 9th Edition Hardin Solutions Manual PDFDokument35 SeitenDwnload Full Beckers World of The Cell 9th Edition Hardin Solutions Manual PDFgebbielean1237100% (12)
- Bacacay South Hris1Dokument7.327 SeitenBacacay South Hris1Lito ObstaculoNoch keine Bewertungen
- SreenuDokument2 SeitenSreenuSubbareddy NvNoch keine Bewertungen
- Statistics and Probability: Quarter 4 - (Week 6)Dokument8 SeitenStatistics and Probability: Quarter 4 - (Week 6)Jessa May MarcosNoch keine Bewertungen
- Guidelines For Prescription Drug Marketing in India-OPPIDokument23 SeitenGuidelines For Prescription Drug Marketing in India-OPPINeelesh Bhandari100% (2)
- Building and Structural Construction N6 T1 2024 T2Dokument9 SeitenBuilding and Structural Construction N6 T1 2024 T2FranceNoch keine Bewertungen
- Chapter 1Dokument6 SeitenChapter 1Grandmaster MeowNoch keine Bewertungen
- 788 ManualDokument16 Seiten788 Manualn0rdNoch keine Bewertungen
- Futures Volume 1 Issue 3 1969 (Doi 10.1016/0016-3287 (69) 90026-3) Dennis Livingston - Science Fiction As A Source of Forecast MaterialDokument7 SeitenFutures Volume 1 Issue 3 1969 (Doi 10.1016/0016-3287 (69) 90026-3) Dennis Livingston - Science Fiction As A Source of Forecast MaterialManticora VenerabilisNoch keine Bewertungen
- Form Expense ClaimDokument2 SeitenForm Expense Claimviedelamonde_3868443Noch keine Bewertungen
- Practical Modern SCADA Protocols. DNP3, 60870.5 and Related SystemsDokument4 SeitenPractical Modern SCADA Protocols. DNP3, 60870.5 and Related Systemsalejogomez200Noch keine Bewertungen
- Marketing Channels: A Strategic Tool of Growing Importance For The Next MillenniumDokument59 SeitenMarketing Channels: A Strategic Tool of Growing Importance For The Next MillenniumAnonymous ibmeej9Noch keine Bewertungen
- A Hybrid Genetic-Neural Architecture For Stock Indexes ForecastingDokument31 SeitenA Hybrid Genetic-Neural Architecture For Stock Indexes ForecastingMaurizio IdiniNoch keine Bewertungen
- Walking in Space - Lyrics and Chord PatternDokument2 SeitenWalking in Space - Lyrics and Chord Patternjohn smithNoch keine Bewertungen
- Vemu Institute of Technology: Department of Computer Science & EngineeringDokument79 SeitenVemu Institute of Technology: Department of Computer Science & EngineeringSiva SankarNoch keine Bewertungen
- Case Study - Kelompok 2Dokument5 SeitenCase Study - Kelompok 2elida wenNoch keine Bewertungen
- Facebook: Daisy BuchananDokument5 SeitenFacebook: Daisy BuchananbelenrichardiNoch keine Bewertungen
- Retail Banking Black BookDokument95 SeitenRetail Banking Black Bookomprakash shindeNoch keine Bewertungen
- IcarosDesktop ManualDokument151 SeitenIcarosDesktop ManualAsztal TavoliNoch keine Bewertungen
- Adjective & VerbsDokument3 SeitenAdjective & VerbsDennis BerkNoch keine Bewertungen
- Aliping PDFDokument54 SeitenAliping PDFDirect LukeNoch keine Bewertungen
- Exotic DVM 11 3 CompleteDokument12 SeitenExotic DVM 11 3 CompleteLuc CardNoch keine Bewertungen
- FE CH 5 AnswerDokument12 SeitenFE CH 5 AnswerAntony ChanNoch keine Bewertungen