Beruflich Dokumente
Kultur Dokumente
An Analysis of Factors Affecting Particle-Size Distribution of Hydraulic Cements PDF
Hochgeladen von
saeedhoseiniOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
An Analysis of Factors Affecting Particle-Size Distribution of Hydraulic Cements PDF
Hochgeladen von
saeedhoseiniCopyright:
Verfügbare Formate
Subhas G. Malghan ~ and Lin-Sien H.
L u m ~
An Analysis of Factors Affecting Particle-Size Distribution of
Hydraulic Cements
REFERENCE: Malghan, S. G. and Lum, L.-S. H., "An Analysis of
Factors Affecting Particle-Size Distribution of Hydraulic Cements,"
Cement, Concrete, and Aggregates, CCAGDP, Vol. 13, No. 2, Winter
1991, pp. 115-120.
ABSTRACT: Particle-size distribution of hydraulic cements plays an
indispensable role in controlling the product quality and maintaining
reliable performance in end applications. Selection of particle-size
measurement methods is becoming increasingly complex due to the
availability of a variety of instruments based on different measurement principles. This paper discusses the primary criteria to be met
by a particle-size distribution analyzer for applications in hydraulic
cement characterization. Among the several methods available for
measuring the particle-size distribution, a light diffraction technique
was utilized to examine the effect of dispersion solvents on the resulting size distribution. Distilled water, distilled water containing a
polyacrylate surfactant, isopropyl alcohol, and a commercially available organic surfactant were used as dispersion solvents. The resulting
data are analyzed with respect to variations due to the dispersion
solvents and factors responsible for the observed variations.
KEY WORDS: particle-size distribution, hydraulic cements, dispersion in solvents, methods of size analysis, light scattering, instrument
selection, sample preparation
More than one property of hydraulic cement is relevant in
defining its characteristics. Many of these properties are related
to some of the more fundamental measurements of particles such
as mean size, size distribution, shape, concentration and form
of impurities, and surface chemical composition. The measurement of particle-size distribution constitutes one of the most
important aspects of hydraulic cement characterization because
particle size affects homogeneity, settling time, and strefigth of
the final product. Therefore, size distribution of the cement particulates can be obtained either for product quality evaluation
or for on-line measurement in process control.
In recent years, the availability of particle-size measurement
systems has increased markedly due to application of different
physical principles. The range of particle-size measurement covered by each instrument has also broadened. Some of the driving
forces for the availability of a large number of systems are: increased recognition of a need for measurement of particle-size
distribution, emphasis on quality control and reliability, and a
need for overall manufacturing cost reduction.
In response to these technical and market-driven forces, numerous particle-size measurement systems have been developed.
This paper discusses steps involved in the particle-size measurement, describes a classification of measurement methods, and
outlines criteria for selection of measurement systems. In addition, results are given for the measurement of particle-size distribution of a typical hydraulic cement in aqueous and nonaqueous liquid environment. A light diffraction technique was
used in these studies since it is not affected by density differences
of the particles.
Steps Involved in Particle-Size Analysis
Some of the requirements of the size analysis are to obtain
data in similar conditions to that existing in the process or to
meet technical specifications of the cement. This necessitates
obtaining a representative sample from a bulk lot. The second
requirement is the dispersion of particles in a chosen solvent so
that true particle size (as determined by scanning electron or
optical microscopy) is determined. Dry techniques do not require
the use of solvents for dispersion. However, as the mean particle
size of the hydraulic cement becomes smaller, dispersion-related
errors increase. For cements of particles sizes in the 1 to 100Ixm range, wet techniques are generally preferred. The third
requirement in the particle-size measurement is the appropriate
use of standard materials and procedures for material and instrument calibration. Each instrument should be checked for
calibration errors using the appropriate standard materials. The
steps involved [1] in the particle-size measurement are shown
schematically in Fig. 1. The overall measurement error is a function of the error associated with the individual steps. For hydraulic cements, errors due to chemical aspects of dispersion and
due to sampling are expected to dominate [].
Selection Criteria and Types of PSD Measurement Techniques
Selection of a particular technique for size distribution measurement is dependent on several criteria [-4]:
1. Accuracy of measurement (systematic error).
2. Reproducibility and repeatability of measurement (random
error).
3. Chemical reaction of particles with liquid.
4. Size range of application.
5. Time required for measurement.
6. Skill level of operator.
7. Cost of instrument.
Accurate measurement of particle-size distribution requires
the development of improved techniques of sample preparation
and associated measurement techniques. Systematic errors can
be minimized by enhanced precision (agreement within a set of
observations obtained as directed in a method) and repeatability
~Group leader, Ceramics Division, and chemical engineer, respectively, National Institute of Standards and Technology, Gaithersburg,
MD.
1991 by the American Society for Testing and Materials
115
This standard is for EDUCATIONAL USE ONLY.
116
CEMENT,CONCRETE, AND AGGREGATES
I Homogenization of Cement
I
I Sampllngfrom a LargeLot I
1
Preparationand
Dispersion ]
[ CalibratlonofInstrument]
l
1.-=-= I
I
Interpretationand
Conversion of Data
FIG. 1--Schematicofstepsinvolved m the measurement Qfpanic~size d~tribution.
(comparison of data obtained in a single laboratory and technique). However, reproducibility is totally dependent on precision involving comparison between laboratories and/or between
different techniques. Accuracy of the technique is enhanced when
the degree of agreement between the true and average of many
observed values increases.
Since hydraulic cements react with aqueous environment, other
solvents, especially organic solvents, can be used. However, for
ease of handling and cost effectiveness, the use of water-based
dispersants is desirable. Dispersion of cement particles in aqueous
environment can be carried out in the presence of low-molecularweight (5000 to 10 000) polyacrylates, which provide steric stabilization [5] at high pH. Examples of organic dispersants are
alcohols and commercially available mineral oil-based solvents.
The cost of the instrument, the time requirement for analysis,
and the skill level of the operator, etc, are some of the external
factors which influence the decision on the selection of instruments. The overall cost of analysis should also include those due
to the instruments, dispersion solvents, and cement samples.
When extensive cleaning between the runs is necessary, the overall cost increases. The time requirement for analysis can range
from 5 rain to several hours depending on the particle-size distribution of a specific material, instrument principle, and dispersion preparation. Instruments based on light scattering offer
high speed of operation. The selection of these instruments is
dependent on the skill level of the operator (e.g., production
environment versus research laboratory), In most cases, a reasonably well-trained operator is required.
Therefore, the question of which technique/instrument to select for a given application leads to the analysis of factors affecting particle-size distribution and specific requirements of the
process. The myriad of analysis techniques, measurement parameters, and the nominal range of application of these techniques are broadly grouped in Table 1.
Wet Versus Dry Methods
The potential for determining the particle-size distribution accurately depends on the ease with which the cement particles
can be dispersed. As the particles become smaller their tendency
to adhere to each other increases due to van der Waals and
coulombic attractive (between chemically dissimilar particles)
forces. Hence, in a dry mode of particle-size measurement, agglomeration or the tendency of fine particles to stick to large
ones cannot be easily controlled. Therefore, accurate and reproducible size distribution measurement may not be obtained
by methods based on dry particulates. In addition, accuracy of
quantitative determination of the finest size fraction is diminished
in the case of dry powders.
Though wet techniques are preferred over dry techniques, they
suffer from interactions between the particles and surrounding
solvent. When the hydraulic cement comes in contact with a
solvent, the silicates and aluminates of lime are expected to
undergo surface dissolution and thereby contributes ionic species
to the adjoining bulk solution. The extent of surface dissolution
depends on a number of characteristics of which the chemical
composition of the cement and that of the solvent are the most
important, Water, being a polar solvent, induces higher surface
dissolution than organic solvents. The resulting electrical double
layer properties of a given cement particle are expected to be
different in water than in organic solvents. The electrical double
layer formation and the resulting dispersion properties are described elsewhere [6].
Size Distribution Measurement in Wet Dispersions
The primary goal of the measurement of particle-size distribution is to determine the absolute size distribution of the particles in the cement sample. Therefore, the formation of agglomerates should be prevented. The major steps involved in the
preparation of highly deagglomerated dispersions in aqueous and
nonaqueous solvents are shown in Fig. 2.
In the case of hydraulic cement, obtaining a representative
sample for size distribution measurement involves the use of a
spinning riffler [2]. Since cement is a highly heterogeneous maTABLE 1--Techniques of particle-size analysis and a typical
measurement parameter 1~-41
Techniques
Measurement
Diameter
Nominal Range,
Ixm
Image
Optical
Electron
Image
Mechanical sieving
Dry
Wet
Sieve/mechnical
Sedimentation
Elutriation
Gravity
Centrifugal
Dynamic/
Stoke's
1-100
0.1-130
0.05-100
Zone sensing
Optical
Resistivity
Geometric
0.01 - 500
0,05-500
Hydrodynamic
chromatography
Geometric
0.0t-1.0
Brownian motion
Geometric
Equivalent
spherical
0.01-I
Other
Gas adsorption
Gas permeability
Mercury intrusion
This standard is for EDUCATIONAL USE ONLY.
0,5-1000
0.01 - 10
>10
>2
0.05-50
O.1-40
O.1- 200
MALGHAN AND LUM ON HYDRAULIC CEMENTS
Dry Sample Preparation ]
[ Wetting and Mixingwlth a Surfaetant ]
!
-r.=-.-oo
I Measurementof Slze Distributlon 1
I
[ ,,Presentationof Data [
FIG. 2--Sequence of steps involved in the measurement of particlesize distribution by wetting the cement.
terial consisting of chemical components of various sizes and
densities, segregation is a serious issue. Therefore, care should
be exercised in the division of a bulk lot of cement sample into
subsamples. Each type of instrument requires a different weight
of the sample. Errors in sampling can be minimized by using the
largest possible subsample for analysis consistent with the sample
requirement. For fine-size cements, wet samples of higher than
the required particle concentration can be prepared, and the final
sample for size determination can be taken from this parent
sample. When dilution of the parent sample is involved, the
surfactant concentration in the final dispersion should be maintained at the same level as in the parent sample.
Wetting and mixing of the powder requires the selection of a
proper combination of the solvent and a surfactant. Organic or
inorganic surfactants [6] can be used; however, organic dispersants provide higher flexibility and more definite dispersion in
complex chemical systems. Water is a preferred solvent, though
it has certain disadvantages, such as surface dissolution, swelling,
and setting. In dilute suspensions (less than 1% by weight), the
effects of these interactions are considered to be minimum.
The selection of an appropriate dispersant requires the knowledge of surface charge and isoelectric point, pH~ep (the pH at
which the particles carry a net zero charge) of the cement particles in a given solvent. Since cement has a highly complex
chemical composition, accurate determination of pHie p by traditional electrokinetic techniques is somewhat tedious and difficult. However, based on relative weight ratios of silicates, aluminates, and other components, the pHiep can be estimated.
Also, experimental determination of the pH~ep is possible by the
application of a relatively new technique in which electrokinetic
sonic amplitude is measured [7] as a function of pH.
Ultrasonication of the slurry containing the cement particles
is an essential step in the preparation of well-dispersed suspensions. During ultrasonication, the particles are subjected to cavitation which dislodges loosely held particles [8]. Two primary
devices of ultrasonication are baths and probes. The probe is a
preferred device because it allows the application of a specific
quantity of ultrasonic power localized to the sample. Ultrasonication by the probe is completed in just a few minutes since the
power intensity is high.
The size distribution of a well-dispersed slurry is determined
by following the recommended procedure of a particular equipment. If the suspension has to be diluted, the solvent chemistry
117
should be the same as that used in the preparation of the parent
suspension. Care should be exercised to avoid settling of coarse
particles during sampling of the parent suspension.
Presentation of the data is dependent on the equipment. Most
often, the data are provided in the form of number or weight/
volume fraction. Direct conversion of data from the number to
weight/volume fraction is not possible without taking into account densities of different chemical components of the hydraulic
cement. Similarly, the size distribution data from gravitational
and centrifugal sedimentation devices is affected by different
densities of the constituting chemical components of the cement.
This is probably one of the main reasons for using the light
diffraction-based device in this study for the size measurement
of cement. Irrespective of the method of size measurement, the
resulting data can be analyzed by using a number of mathematical
models. The parameters of these models can serve the purpose
of comparing the characteristics of various cements.
Application of the Light-Scattering Method
The particle-size distribution of Portland Cement Proficiency
Sample 95 [9], distributed by the Cement and Concrete Reference Laboratory (CCRL), was determined using a light diffraction-based particle-size measurement system (Horiba LA-500)
[10]. CCRL is a research associate program at NIST sponsored
by ASTM [11]. The primary focus of this study was to examine
the influence of solvents in which the cement is dispersed. The
solvents examined were distilled water, distilled water containing
500 ppm polyacrylate (Darvan C-Vanderbilt Chemical Company) as a dispersant, isopropyl alcohol, and Sedisperse A-12 (a
surfactant manufactured by Micromeritics Corporation). 2
Dispersions of the cement were prepared by mixing cement
and the solvent in the weight ratio of 1 to 20, followed by ultrasonication (ultrasonic probe of 19 mm diameter, 30 W, total of
3 rain). The dispersion was ultrasonicated stepwise by applying
power for 1 min followed by 1 rain of cooling. This process was
repeated to give a total uttrasonication time of 3 rain. The dispersed powders were size analyzed using a procedure developed
for ceramic powders. Each measurement is an average of 25
scans, and triplicate data were obtained using three separate
subsamples from the primary dispersion. The data presented are
an average of the three measurements.
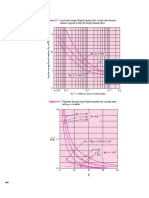
The size distribution data of the cement sample are presented
in Fig. 3. In general, all four dispersions show a bimodal size
distribution in which there is a small quantity of submicrometre
fraction and a large quantity of fraction whose ds,, is in the range
of 10 to 20 p.m. The dso is the size in micrometres through which
50% of the particles pass. The following general guideline was
used in the evaluation of the effectiveness of solvents and dispersants: the most suitable solvent-dispersant combination is one
that gives the finest size distribution. Based on this criterion,
isopropyl alcohol (IPA) appears to be the most suitable solvent.
On the other hand, Sedisperse A-12 is the least effective dispersant. Both water and water containing 500 ppm Darvan C
give similar size distributions in the fine-size range and a slightly
2Certain commercial equipment, vendors, instruments, or materials
are identified in this report in order to describe adequately experimental
requirements. In no case does such identification imply recommendation
or endorsement by the National Institute of Standards and Technology,
nor does it imply that the vendor, material, or equipment identified is
necessarily the best available for the purpose.
This standard is for EDUCATIONAL USE ONLY.
118
CEMENT,CONCRETE, AND AGGREGATES
11
10
Cement in H20
- - - Cement in H20 w / 5 0 0 ppm Darvan
9
rItc(3n
,m
0)
>
-)
c
Cement in IPA
-.-
Cement in A - 1 2
6
5
2
1
.'
.,;s"
0 ,-
1.0
0.1
10.0
100.0
Particle Diameter, microns
FIG. 3--Size distribution data of CCRL portland cement proficiency sample by using Horiba LA-500
device. The effect of four solvents is shown: (1) water; (2) water plus 500 ppm Darvan C; (3) 1PA; and
(4) Sedisperse A-12.
Another important feature of these data is that 90% of the
weight is finer than 19 ~m in the case of IPA. When using other
solvents, a coarser size distribution is observed. In addition, in
the case of IPA, more fines are observed at the finer end of size
distribution. A possible reason for IPA's superiority in this particular case is its inertness to surface reactivity with cement particles.
different dgo. The overall differences in the entire size range are
presented in Fig. 4. These data, in terms of cumulative size
fraction, show that IPA is by far a superior solvent. The dg0, ds0,
and d25 data summarized in Table 2 show specific differences in
the size distributions. The size distribution data are presented in
terms of weight fraction by assuming that density variation between the particles is negligible.
tO0
Cement in H20
90
-
Q)
.E
ta_
.Cn
, m
Q)
80
70
"'"
Cement in H20 w / 5 0 0 ppm Darvan
Cement in IPA
." t/~/t
b,'
---- Cement in A - 1 2
." t~,/
60
." .i/1P
50
:;7
>
-.a,.,a
/ ~
/ Y
..' / /
20
- f~
jQ
1"
10
O,
0.1
1
1
1.0
10.0
Porticle Diometer, microns
I
100.0
FIG. 4--Size distribution data of Fig. 3 presented in the form of cumulative weight fraction to highlight
differences in the entire size distribution,
This standard is for EDUCATIONAL USE ONLY.
MALGHAN AND LUM ON HYDRAULIC CEMENTS
TABLE 2--Effect of solvent on the dispersion of a cement sample
(CCRL Portland Cement Proficiency Sample 95) during particle-size
determination by Horiba LA-500.
Size in Micrometres Through
Which the Specific Weight
Percent Passes
IPA
d9o
dso
dz5
19.0
9.0
3.6
Water
26.0
12.0
7.5
Water + Darvan C
30.0
13.0
7.5
A-12
30.0
it.0
5.0
Bimodal distribution obtained for this sample could be the
result of the application of both ultrasonication energy and solvents for dispersion. Fine, submicrometre particles loosely adhered to coarse particles tend to dislodge by the application of
a combination of mechanical (ultrasonication promoting cavitation) and surface chemical (steric in the case of IPA and A12) dispersive forces. During ultrasonication, the particles are
subjected to cavitation which separates loosely held particles.
Once the particles are separated, they are kept dispersed by the
steric forces imparted by the adsorbed hydrocarbon molecules
of IPA. If the size distribution is carried out by dry sieving, the
bimodal distribution may not be observed in such detail.
Repeatability of the size distribution data is shown in Figs. 5
and 6 for IPA and Sedisperse A-12, respectively. These data
show that, irrespective of the solvent used, the repeatability is
excellent in the case of fine-size fraction (<1.0 ~m). However,
there is a slightly larger variation at the coarse-size fraction when
using IPA; whereas, for Sedisperse A-12, the variation is neg-
ligible. Since the final selection of a solvent-dispersant should
include the evaluation of both accuracy and repeatability, a thorough evaluation of those factors is required in a round-robin
characterization program.
Summary and Conclusions
Major steps involved in the measurement of particle-size distribution are sampling and sample preparation (including dispersion). Errors in these steps are often responsible for lack of
repeatability and reproducibility. A large number of systems are
available that provide data over a wide range of size distributions.
The selection of instruments for a given application depends on
a number of criteria. In recent years, systems based on lightscattering principles are widely used for particulate systems.
Dispersion of hydraulic cement particles in a suitable solvent
is an excellent way to ensure the determination of accurate size
distribution. Selection of an appropriate level and type of ultrasonication and dispersion parameters is the key factor in obtaining near-true size distribution data. The selection of solvents and
dispersants for hydraulic cements can be systematized if surface
charge and pHi~p data are available. In the absence of such data,
four solvents were evaluated by using the CCRL portland cement
proficiency sample. The size distribution data from a light-scattering device indicated that the cement sample has a bimodal
size distribution irrespective of the sotvent-dispersant composition. However, the data from isopropyl alcohol showed that the
overall size distribution was finer. Independent verification of
these size distribution data with those from scanning electron
microscopy is required to test their validity. A slight variation
was observed at the peak of the coarse end of the PSD in the
replicate tests using IPA. In the case of Sedisperse A-12, this
type of variation was negligible.
CEMENT IN IPA
1
0
Sample I
9
C
cD
I,
cljn
{D
Sample
--'- S a m p l e 3
6
,5
.//
>
-
0
O.
1.0
119
'ii,
10.0
100.0
Particle Diameter, microns
FIG. 5--Repeatability of size distribution data by using Horiba LA-500 for cement in 1PA.
This standard is for EDUCATIONAL USE ONLY.
120
CEMENT, CONCRETE, AND AGGREGATES
CEMENT IN A - 1 2
11
10
Sample
- Sample
c-
- - . - S a m p l e .3
b..
o~
,!
f.
/Z
3~
"
>
-{3
c"
~
~
O.1
1.0
10.0
100.0
Porticle Diometer, microns
FIG. 6--Repeatability of size distribution data by using Horiba LA-500 for cement in Sedisperse A-12.
References
[1] Allen, T. and Davies, R., "Modern Aspects of Particle Size Analysis," Advances in Ceramics, Vol. 21: Ceramic Powder Science,
G. L. Messing et at., Eds., American Ceramic Society, pp. 721746.
[2] Allen, T., Particle Size Measurement, 3rd ed., Chapman and Hall,
New York, 1981.
[3] Modern Methods of Particle Size Analysis, H. G. Barth, Ed., Wiley
Interscience, New York, 1984.
[4] Pohl, M., "Selecting a Particle Size Analyzer: Factors to Consider,"
Powder and Bulk Engineering, Vol. 4, No. 2, February 1990, pp.
26-29.
[5] Napper, D. H., Polymeric Stabilization of Colloidal Dispersions,
Academic Press, Inc., London, 1983, pp. 18-30.
[6] Parfitt, G. D., Dispersion of Powders in Liquids, Chapters 1 and
2, Applied Science Publishers, 198t.
[7] Malghan, S. G. and Lum, L.-S., "'Factors Affecting Interface Properties of Silicon Nitride Powders in Aqueous Environment," in
Ceramic Powder Science, G. L. Messing, et al., Eds., American
Ceramic Society, September 1990.
[8] Anonymous, "Branson Sonifer," Bulletin 200-214-082, Branson Ultrasonics Corp., Danbury, CT, 1989.
[9] Samples obtained from Pielert, J. H., Cement and Concrete Reference Laboratory, Building Materials Division, NIST, July 1990.
[10] Anonymous, "Horiba LA-500 Laser Diffraction Particle Size Analyzer," Bulletin HRE-3610B, Kyoto, Japan, January 1990.
[11] Pielert, J. H., "Construction Materials Reference Laboratories at
NIST," ASTM Standardization News, December 1989, pp. 40-44.
This standard is for EDUCATIONAL USE ONLY.
Das könnte Ihnen auch gefallen
- Cirsys 19Dokument11 SeitenCirsys 19saeedhoseiniNoch keine Bewertungen
- Application of Variography To The Control of Species in Material Process Streams: in An Iron Ore ProductDokument14 SeitenApplication of Variography To The Control of Species in Material Process Streams: in An Iron Ore ProductsaeedhoseiniNoch keine Bewertungen
- Application of Variography To The Control of Species in Material Process Streams: in An Iron Ore ProductDokument14 SeitenApplication of Variography To The Control of Species in Material Process Streams: in An Iron Ore ProductsaeedhoseiniNoch keine Bewertungen
- (Mathematics and Its Applications 38) L. Keviczky, M. Hilger, J. Kolostori (Auth.) - Mathematics and Control Engineering of Grinding Technology - Ball Mill Grinding-Springer Netherlands (1989) PDFDokument185 Seiten(Mathematics and Its Applications 38) L. Keviczky, M. Hilger, J. Kolostori (Auth.) - Mathematics and Control Engineering of Grinding Technology - Ball Mill Grinding-Springer Netherlands (1989) PDFsaeedhoseiniNoch keine Bewertungen
- PHD Thesis Michal Hlobil 2016Dokument227 SeitenPHD Thesis Michal Hlobil 2016saeedhoseiniNoch keine Bewertungen
- Non-Portland Cement Activation of Blast Furnace SlagDokument69 SeitenNon-Portland Cement Activation of Blast Furnace SlagsaeedhoseiniNoch keine Bewertungen
- Reid, Henry - The Science and Art of The Manufacture of Portland Cement - With Observations On Some of Its Constructive applications-ICE Publishing (2011) PDFDokument448 SeitenReid, Henry - The Science and Art of The Manufacture of Portland Cement - With Observations On Some of Its Constructive applications-ICE Publishing (2011) PDFsaeedhoseiniNoch keine Bewertungen
- ASTM C-1356, Quantitative Determination of Phases in Portland Cement PDFDokument5 SeitenASTM C-1356, Quantitative Determination of Phases in Portland Cement PDFsaeedhoseini100% (1)
- Index 12Dokument103 SeitenIndex 12saeedhoseiniNoch keine Bewertungen
- Early-Age Hydration Studies of Portland Cement PDFDokument172 SeitenEarly-Age Hydration Studies of Portland Cement PDFsaeedhoseiniNoch keine Bewertungen
- Performance and Mechanism of A Multi-Functional Superplasticizer For ConcreteDokument6 SeitenPerformance and Mechanism of A Multi-Functional Superplasticizer For ConcretesaeedhoseiniNoch keine Bewertungen
- Zhang ThesisDokument43 SeitenZhang ThesissaeedhoseiniNoch keine Bewertungen
- Early-Age Hydration Studies of Portland Cement PDFDokument172 SeitenEarly-Age Hydration Studies of Portland Cement PDFsaeedhoseiniNoch keine Bewertungen
- Compare Sizing Methods PDFDokument14 SeitenCompare Sizing Methods PDFAnonymous NxpnI6jCNoch keine Bewertungen
- Blast Furnace Slag As CementDokument5 SeitenBlast Furnace Slag As CementsaeedhoseiniNoch keine Bewertungen
- Optimal Fluorite/gypsum Mineralizer Ratio in Portland Cement ClinkeringDokument12 SeitenOptimal Fluorite/gypsum Mineralizer Ratio in Portland Cement ClinkeringsaeedhoseiniNoch keine Bewertungen
- FineGrindingasEnablingTechnology TheIsaMillDokument21 SeitenFineGrindingasEnablingTechnology TheIsaMillPVENARDNoch keine Bewertungen
- Cao Inv Low CemDokument79 SeitenCao Inv Low CemTarek ChikerNoch keine Bewertungen
- Rebelo Jonathan 201406 MASc Thesis PDFDokument149 SeitenRebelo Jonathan 201406 MASc Thesis PDFBartoFreitasNoch keine Bewertungen
- Concrete Strength DevelopmentDokument14 SeitenConcrete Strength DevelopmentGabriel LimNoch keine Bewertungen
- Optimal Fluorite/gypsum Mineralizer Ratio in Portland Cement ClinkeringDokument12 SeitenOptimal Fluorite/gypsum Mineralizer Ratio in Portland Cement ClinkeringsaeedhoseiniNoch keine Bewertungen
- Rebelo Jonathan 201406 MASc Thesis PDFDokument149 SeitenRebelo Jonathan 201406 MASc Thesis PDFBartoFreitasNoch keine Bewertungen
- Sayesh Nahaee PDFDokument4 SeitenSayesh Nahaee PDFsaeedhoseiniNoch keine Bewertungen
- 5.ISCA-RJRS-2012-078 DoneDokument14 Seiten5.ISCA-RJRS-2012-078 DonesaeedhoseiniNoch keine Bewertungen
- Jumate Jaes Issue2 2011Dokument8 SeitenJumate Jaes Issue2 2011saeedhoseiniNoch keine Bewertungen
- Optimization of The Preheater Cyclone Separators Used in The Cement IndustryDokument16 SeitenOptimization of The Preheater Cyclone Separators Used in The Cement IndustrysaeedhoseiniNoch keine Bewertungen
- 90 003Dokument8 Seiten90 003saeedhoseiniNoch keine Bewertungen
- 908667Dokument7 Seiten908667saeedhoseiniNoch keine Bewertungen
- Jeas 1017 6416Dokument8 SeitenJeas 1017 6416saeedhoseiniNoch keine Bewertungen
- Circulation Phenomena in The Clinkerization ProcessDokument101 SeitenCirculation Phenomena in The Clinkerization ProcessRaden Ayu Wilda AnggrainiNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Royal Ultra 865MDokument9 SeitenRoyal Ultra 865Mjuanfdez42368Noch keine Bewertungen
- H2 Inorganic ChemistryDokument7 SeitenH2 Inorganic ChemistrykitoniumNoch keine Bewertungen
- Vapor-Liquid Equilibria Measurements of Methane + 2-Methylpropane PDFDokument7 SeitenVapor-Liquid Equilibria Measurements of Methane + 2-Methylpropane PDFTino KhamphasithNoch keine Bewertungen
- Physics Q PaperDokument2 SeitenPhysics Q Papermaulik2323Noch keine Bewertungen
- QUEST 3 Product SearchDokument2 SeitenQUEST 3 Product Searchbegone thotNoch keine Bewertungen
- Andreas Penckwitt - Rotating Bose-Einstein Condensates: Vortex Lattices and ExcitationsDokument223 SeitenAndreas Penckwitt - Rotating Bose-Einstein Condensates: Vortex Lattices and ExcitationsLomewcxNoch keine Bewertungen
- NMR and EsrDokument52 SeitenNMR and EsrSenthil Sethu FvfcNoch keine Bewertungen
- Nature of MatterDokument39 SeitenNature of MatterChaela GonzagaNoch keine Bewertungen
- Notes of ClassDokument9 SeitenNotes of ClassRishi PrakashNoch keine Bewertungen
- Chemicals Zetag DATA Powder Zetag 7650 - 0410Dokument2 SeitenChemicals Zetag DATA Powder Zetag 7650 - 0410PromagEnviro.comNoch keine Bewertungen
- Ureas and Thioureas As Asymmetric OrganocatalystsDokument60 SeitenUreas and Thioureas As Asymmetric OrganocatalystsAlex FNoch keine Bewertungen
- Types of MicroscopeDokument3 SeitenTypes of MicroscopeRommel Paz AlbertoNoch keine Bewertungen
- Molecular Sieve Type 3ADokument2 SeitenMolecular Sieve Type 3Akanu PatelNoch keine Bewertungen
- Heat Transfer TableDokument1 SeiteHeat Transfer TablehydarNoch keine Bewertungen
- Intro To Cloud PhysicsDokument10 SeitenIntro To Cloud PhysicsShowna Lee100% (1)
- Class 11 Chemistry Revision Notes Classification of Elements and Periodicity in PropertiesDokument23 SeitenClass 11 Chemistry Revision Notes Classification of Elements and Periodicity in PropertiesPriyanshuNoch keine Bewertungen
- Tunnel DryerDokument15 SeitenTunnel Dryerمحمد العراقيNoch keine Bewertungen
- Locanit SDokument2 SeitenLocanit SMuhammad Aasim HassanNoch keine Bewertungen
- Electroless Plating28266Dokument17 SeitenElectroless Plating28266fsarfrazNoch keine Bewertungen
- University of Cambridge International Examinations General Certifi Cate of Education Advanced Subsidiary Level and Advanced LevelDokument12 SeitenUniversity of Cambridge International Examinations General Certifi Cate of Education Advanced Subsidiary Level and Advanced LevelHubbak KhanNoch keine Bewertungen
- Abaqus Analysis User's Guide (6Dokument11 SeitenAbaqus Analysis User's Guide (6vinhtungbkNoch keine Bewertungen
- Module 3 and 4Dokument8 SeitenModule 3 and 4Thea Louise GarciaNoch keine Bewertungen
- Kenneth Denbigh - The Principles of Chemical Equilibrium - With Applications in Chemistry and Chemical Engineering (1981, Cambridge University Press) PDFDokument514 SeitenKenneth Denbigh - The Principles of Chemical Equilibrium - With Applications in Chemistry and Chemical Engineering (1981, Cambridge University Press) PDFNilton Rosenbach Jr100% (1)
- Two Way 4D Printing - A Review On The Reversibility of 3D Printed Shape Memory MaterialsDokument12 SeitenTwo Way 4D Printing - A Review On The Reversibility of 3D Printed Shape Memory MaterialsEdgar ApitanaNoch keine Bewertungen
- Convection Heat TransferDokument29 SeitenConvection Heat TransferAjay BansalNoch keine Bewertungen
- EsterificationDokument4 SeitenEsterificationMichael Guinita100% (2)
- Apresentação - Oil and Water SeparationDokument55 SeitenApresentação - Oil and Water SeparationAlisson AlmeidaNoch keine Bewertungen
- Anions 1Dokument8 SeitenAnions 1Nitty MeYaNoch keine Bewertungen
- Problem PDFDokument6 SeitenProblem PDFJack WillNoch keine Bewertungen
- Organic Chemistry 1Dokument324 SeitenOrganic Chemistry 1Bellony Sanders100% (7)