Beruflich Dokumente
Kultur Dokumente
SAT Chem 04 Solids, Liquids, Gases
Hochgeladen von
BiologyCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
SAT Chem 04 Solids, Liquids, Gases
Hochgeladen von
BiologyCopyright:
Verfügbare Formate
SAT II-Review Questions
For 1-4
For 5-7
a.
b.
c.
d.
e.
Boyles law
Charles law
Avogardros law
Ideal gas law
Daltons law
1.
The total pressure of a gaseous mixture is equal
to the sum of the partial pressures is
2.
Volume is inversely proportional to pressure is
3.
Volume is directly proportional to temperature is
4.
All gases have the same number of moles in the
same volume at constant T and P is
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
a.
b.
c.
d.
e.
Gas solid is called
6.
Gas liquid is called
7.
Solid gas is called
For 8-10
a.
b.
c.
Which of the following is responsible for the
abnormally high boiling point of water?
a.
Covalent bonding
b.
Hydrogen bonding
c.
High polarity
d.
Large dielectric constant
e.
Low molecular weight
2.0
24.
If one mole of H2 is compressed from 10 L to 7.5
L at constant temperature, what happens to the
gas pressure?
a.
It increases by 25%
b.
It decreases by 25%
c.
It increases by 33%
d.
It increases by 50%
e.
None of the above
Which shows melting?
9.
Which shows increasing the kinetic energy of a
liquid?
10. Which shows boiling?
Statement II
Interactions between particles cannot be neglected under these conditions
Because
Its liquid form is impossible to produce
Temperature and volume are directly proportional
Gas molecules are considered volume-less particles, with no intermolecular
forces, in constant random motion
Changes of state bring about changes in a substances potential energy, not
in its kinetic energy
The vapor pressure of water is lower at higher altitude
Because
Because
Pressure and volume are inversely related
Temperature and volume are inversely proportional
Because
Decreasing temperature and increasing pressure will cause the volume of a
gas to decrease
Because
30.
Five liters of gas at STP have a mass of 12.5 g.
What is the molecular mass of the gas?
a.
12.5 g/mol
b.
25.0 g/mol
c.
47.5 g/mol
d.
56.0 g/mol
e.
125 g/mol
31.
Equal molar quantities of hydrogen gas and
oxygen gas are present in a closed container at a
constant pressure. Which of the following
quantities will be the same for the two gases?
a.
Partial pressure
b.
Partial pressure & average KE
c.
Partial pressure & average molecular
velocity
d.
Average KE & average molecular velocity
e.
Partial pressure, average KE, average
molecular velocity
1.5
1.0
0.5
a
100
26.
28.
An ideal gas in a closed inflexible container has a
pressure of 6 atm and a temperature of 27 deg
C. What will be the new pressure at -73 deg C?
a.
2 atm
b.
3 atm
c.
4 atm
d.
8 atm
e.
9 atm
Because
Because
Because
29.
DE
EF
8.
27.
What is the volume at STP of 10 L of gas initially
at 546 K, 2 atm?
a.
5L
b.
10 L
c.
15 L
d.
20 L
e.
25 L
Because
Because
For the next few questions, refer to the diagram below,
regarding substance Z.
Which of the following is (are) the weakest
attractive forces?
a.
Van der Waals
b.
Coordinate covalent bonding
c.
Covalent bonding
d.
Polar covalent bonding
e.
Ionic bonding
23.
25.
AB
BC
CD
Statement I
The ideal gas law does not hold under low temperatures and high
pressure
CO2 is able to sublimate at atmospheric pressure
When an ideal gas is cooled its volume will increase
According to the KMT, collisions between gas particles and the
walls of the container are elastic
As ice absorbs heat and begins to melt, its temperature remains
constant
Water boils at a lower temperature at high altitudes compared to
low altitudes
Decreasing the volume of a system decreases pressure
At constant pressure, a certain amount of gas will double in
volume as the temperature is halved
The volume of a gas at 100 deg C and 600 mmHg will be lower at
STP
What volume would 16 g of molecular oxygen
gas occupy at STP?
a.
5.6 L
b.
11.2 L
c.
22.4 L
d.
33.6 L
e.
44.8 L
d.
e.
Sublimation
Condensation
Evaporation
Deposition
melting
5.
P (atm)
Q
11.
OMalley
Solids, Liquids, Gases
b
200 300 400 500
600
T (K)
Substance Z is at 0.5 atm and 200 K. If the
pressure on substance Z is steadily increased
and its temperature is kept constant, what phase
change will eventually occur?
a.
condensation
b.
freezing
c.
melting
d.
sublimation
e.
vaporization
The normal boiling point of substance Z is
approximately
a.
100 K
b.
200 K
c.
300 K
d.
400 K
e.
500 K
In what pressure range will the compound
sublime?
a.
Less than 0.5 atm
b.
Between 0.5 and 1.0
c.
Between 1.0 and 2.0
d.
Between 0.5 and 2.0
e.
This compound wont sublime
Crossing line bd is:
a.
condensation
b.
melting
c.
evaporation
d.
sublimation
e.
boiling
For the next few questions: A closed 5.0 L vessel
contains a sample of neon. The temperature inside
the container is 25 oC and the pressure is 1.5 atm.
32.
Which of the following expressions is equal to the
moles of gas in the sample?
a.
(1.5 x 5.0) / (0.08 x 25)
b.
(0.08 x 250 / (1.5 x 5.0)
c.
(1.5 x 25) / (0.08 x 5.0)
d.
(0.08 x 298) / (1.5 x 5.0)
e.
(1.5 x 5.0) / (0.08 x 298)
33.
If the neon gas in the vessel is replaced with an
equal molar quantity of helium gas, which will be
changed?
a.
pressure
b.
temperature
c.
density
d.
pressure & temperature
e.
temperature and density
34.
The volume was changed while temperature held
constant until the pressure was 1.6 atm. Which
is equal to the new volume?
a.
5.0 x 1.5 / 1.6 (4.7 L)
b.
5.0 x 1.6 / 1.5
c.
25 x 1.5 / 1.6
d.
0.08 x 1.6 / 1.5
e.
0.08 x 1.5 / 1.6
35.
36.
37.
38.
39.
40.
41.
42.
A flask contains three times as many moles of H2
as it does O2. If hydrogen and oxygen are the
only gases present, what is the total pressure in
the flask if the partial pressure of oxygen is P?
a.
4P
b.
3P
c.
4/3P
d.
3/4P
e.
7P
The gas in a large cylinder is at a pressure of
3040 torr. Assuming constant temperature and
ideal gas behavior, what volume of this gas could
you compress into a 100 L box at 8 atm?
a.
20 L
b.
200 L
c.
5000 L
d.
50,000 L
e.
500,000 L
Which of the following generalizations CANNOT
be made about the phase change of a pure
substance from solid to liquid?
a.
It involves a change in potential energy
b.
It involves no change in temperature
c.
It involves a change in kinetic energy
d.
It involves a change in entropy
e.
It may occur at different temperatures for
different compounds
If the pressure of a gas sample is doubled at
constant temperature, the volume will be
a.
4 x the original
b.
2 x the original
c.
of the original
d.
of the original
e.
1/8 of the original
Three canisters, A, B, and C, are all at the same
temperature, with volumes of 2.0, 4.0, and 6.0 L,
respectively. Canister A contains 0.976 g Ar at
120 torr, Canister B contains 1.37 g N2 at 120
torr, and Canister C is completely empty at the
start. Assuming ideality, what would be the
pressure in canister C if the contents of A and B
are completely transferred to C?
a.
180 torr
b.
330 torr
c.
675 torr
d.
0.25 atm
e.
none of the above
When a fixed amount of gas has its Kelvin
temperature and pressure doubled, the new
volume of the gas is
a.
Four times greater than its original volume
b.
Twice its original volume
c.
Unchanged
d.
One half its original volume
e.
One fourth its original volume
A 600 mL container holds 2 mol O2, 3 mol H2,
and 1 mol He. The total pressure within the
container is 760 torr. What is the partial pressure
of O2?
a.
127 torr
b.
253 torr
c.
380 torr
d.
507 torr
e.
760 torr
An ideal gas has a volume of 10 L at 20 deg C
and 750 mmHg. Which of the following
expressions is needed to determine the volume
of the same amount of gas at STP?
a.
10 x (750/760) x (0/20)
b.
10 x (750/760) x (293/273)
c.
d.
e.
10 x (760/750) x (0/20)
10 x (760/750) x (273/293)
10 x (750/760) x (273/293)
c.
d.
e.
23
43.
What volume does a sample of 1.50 x 10
of helium at STP represent?
a.
5.6 L
b.
11.2 L
c.
17.8 L
d.
22.4 L
e.
none of the above
44.
Which of the following will always decrease the
volume of a gas?
i. Decrease the pressure with the
temperature held constant
ii. Increase the pressure with a temperature
decrease
iii. Increase the temperature with a pressure
increase
a.
I only
b.
II only
c.
I and III
d.
II and III only
e.
I, II and III
45.
46.
47.
atoms
A gas has a volume of 10 L at 50 deg C and 200
mmHg. What conversion factor is needed to give
a volume at STP?
a.
10 x (0/50) x (200/760)
b.
10 x (0/50) x 760/200)
c.
10 x (273/323) x (200/760)
d.
10 x (273/323) x (760/200)
e.
10 x (323/273) x (760/200)
The temperature above which a liquid cannot
exist is indicated by
a.
the triple point
b.
the critical point
c.
the eutectic point
d.
the boiling point
e.
the sublimation point
A change of phase never accompanies
a.
a change in volume
b.
a change in pressure
c.
a change in temperature
d.
a change in density
e.
a change in structure
48.
The relationship P1V1 = P2V2 is
a.
Boyles law
b.
Chaless law
c.
Van der Waals law
d.
the combined gas law
e.
the ideal gas law
49.
The rate of diffusion of hydrogen gas as
compared to that of oxygen gas is
a.
as fast
b.
identical
c.
twice as fast
d.
four times as fast
e.
eight times as fast
50.
The ratio of the rate of diffusion of oxygen to
hydrogen is
a.
1:2
b.
1:4
c.
1:8
d.
1:16
e.
1:32
51.
Standard conditions using a Kelvin thermometer
are
a.
760 torr, 273 K
b.
760 torr, 273 K, 1 L
760 torr, 0 K
0 torr, 0 K
0 torr, 273 K, 1 L
52.
The relation between the pressure and the
volume of a gas at constant temperature is given
by
a.
Boyles law
b.
Charless law
c.
the combined gas law
d.
the ideal gas law
e.
none of the above
53.
The relation between the absolute temperature
and volume of a gas at constant pressure is
given by
a.
Boyles law
b.
Charless law
c.
the combined gas law
d.
the ideal gas law
e.
none of the above
54.
The relation between the pressure, volume and
absolute temperature is given by
a.
Boyles law
b.
Charless law
c.
the combined gas law
d.
the ideal gas law
e.
none of the above
55.
At a certain temperature and pressure, ice, water
and steam are found to coexist at
1.
E
equilibrium. This pressure and
2.
A
temperature corresponds to:
3.
B
a.
the critical temperature
4.
C
b.
the critical pressure
5.
D
c.
the sublimation point
6.
B
d.
the triple point
7.
A
e.
two of the above
8.
B
9.
C
How many atoms are present in
10. D
22.4 L of O2 at STP?
11. T, T, CE
a.
3 x 1023
12. T, F
b.
6 x 1023
13. F, T
23
14. T, T, CE
c.
9 x 10
23
15. T, T, CE
d.
12 x 10
23
16. T, F
e.
15 x 10
17. F, T
18. F, F
a gas at STP that contains 6.02 x
23
19. T, T, CE
10 atoms and forms diatomic
20. B
molecules will occupy
21. B
a.
11.2 L
22. A
b.
22.4 L
23. B
c.
33.6 L
24. C
d.
67.2 L
25. C
e.
1.06 quarts
26. C
27. D
Inelastic collisions occur in
28. A
a.
Real and ideal gases
29. B
b.
Ideal gases and fusion
30. D
reactions
31. B
32. E
c.
Real gases and fusion
33. C
reactions
34. A
d.
Real gases
35. A
e.
Ideal gases
36. B
37. C
The extremely high melting point
38. C
of diamond (carbon) may be
39. E
explained by the
40. C
a.
network covalent bonds
41. B
b.
ionic bonds
42. E
c.
hydrogen bonds
43. A
d.
van der Waals forces
44. B
e.
none of the above
45. C
46. B
47. C
48. A
49. D
50. B
51. A
52. A
53. B
54. C
55. D
56. D
57. A
58. C
59. A
56.
57.
58.
59.
Das könnte Ihnen auch gefallen
- Chapter 7 - Acids Bases and SaltsDokument26 SeitenChapter 7 - Acids Bases and SaltsBiologyNoch keine Bewertungen
- Chapter 12 - The Petroleum IndustryDokument18 SeitenChapter 12 - The Petroleum IndustryBiologyNoch keine Bewertungen
- Chapter 8 - Inorganic Carbon ChemistryDokument3 SeitenChapter 8 - Inorganic Carbon ChemistryBiologyNoch keine Bewertungen
- Chapter 6 - Electrolysis and Its UsesDokument39 SeitenChapter 6 - Electrolysis and Its UsesBiologyNoch keine Bewertungen
- Chapter 11 - Rates of ReactionDokument18 SeitenChapter 11 - Rates of ReactionBiologyNoch keine Bewertungen
- Chapter 9 - Metal Extraction and The Reactivity SeriesDokument30 SeitenChapter 9 - Metal Extraction and The Reactivity SeriesBiologyNoch keine Bewertungen
- Chapter 2 - Elements, Compounds and MixturesDokument8 SeitenChapter 2 - Elements, Compounds and MixturesBiologyNoch keine Bewertungen
- Chapter 1 - All About MatterDokument3 SeitenChapter 1 - All About MatterBiologyNoch keine Bewertungen
- Urdu Idoms and Proverbs PDFDokument38 SeitenUrdu Idoms and Proverbs PDFBiologyNoch keine Bewertungen
- Key Question 3 War of Independence 1857Dokument4 SeitenKey Question 3 War of Independence 1857shabiheNoch keine Bewertungen
- May June 2014 IGCSE Zone 3 ScheduleDokument2 SeitenMay June 2014 IGCSE Zone 3 ScheduleBiologyNoch keine Bewertungen
- 2015 Specimen Paper 2Dokument26 Seiten2015 Specimen Paper 2Khawaja ShoaibNoch keine Bewertungen
- Medical MnemonicsDokument256 SeitenMedical MnemonicssitalcoolkNoch keine Bewertungen
- ABC Dedvgsdv DfvsavDokument1 SeiteABC Dedvgsdv DfvsavBiologyNoch keine Bewertungen
- Sample Cover Page For Past PapersDokument2 SeitenSample Cover Page For Past PapersBiologyNoch keine Bewertungen
- Eda2phys 4113 6Dokument2 SeitenEda2phys 4113 6Afrah Mir100% (1)
- Answers To AS Physics Hodder BookDokument29 SeitenAnswers To AS Physics Hodder BookBiology60% (5)
- 2-As Biology Core Practical SummaryDokument2 Seiten2-As Biology Core Practical SummaryJoel Oh SukJuNoch keine Bewertungen
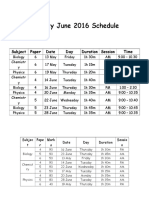
- A2 May June 2016 Schedule: Subject Paper Date Day Duration Session TimeDokument2 SeitenA2 May June 2016 Schedule: Subject Paper Date Day Duration Session TimeBiologyNoch keine Bewertungen
- Edexcel A2 Biology Practicals (Complete)Dokument45 SeitenEdexcel A2 Biology Practicals (Complete)Biology77% (22)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (399)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (73)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- 01-Kinetic Theory of Gases - (Theory)Dokument44 Seiten01-Kinetic Theory of Gases - (Theory)RAMESHNoch keine Bewertungen
- Property Tables and Charts (Si Units) : AppendixDokument31 SeitenProperty Tables and Charts (Si Units) : AppendixNero sasukeNoch keine Bewertungen
- Gujarat Technological University: InstructionsDokument3 SeitenGujarat Technological University: InstructionsRenieNoch keine Bewertungen
- GasesDokument32 SeitenGasesShruti BhatiaNoch keine Bewertungen
- Physics Ia FinalDokument11 SeitenPhysics Ia FinalscarletNoch keine Bewertungen
- UntitledDokument16 SeitenUntitledapi-233404189100% (1)
- 4th Quarter Exam FinalDokument8 Seiten4th Quarter Exam FinalMaria Fe VibarNoch keine Bewertungen
- BFF2233 THERMO Chap 3 PDFDokument48 SeitenBFF2233 THERMO Chap 3 PDFمحمد الشافعيNoch keine Bewertungen
- Student Exploration: Ideal Gas LawDokument10 SeitenStudent Exploration: Ideal Gas Lawarshia nikithaNoch keine Bewertungen
- Module 3 (Lesson 1 and 2)Dokument10 SeitenModule 3 (Lesson 1 and 2)rocky.mercado2Noch keine Bewertungen
- Fundamental of Statistical Mechanics BB-laud PDFDokument286 SeitenFundamental of Statistical Mechanics BB-laud PDFajay kumar25% (4)
- CH 4 - Gases Quiz: Name: DIRECTIONS: Read Each Question Carefully and Choose The Best AnswerDokument5 SeitenCH 4 - Gases Quiz: Name: DIRECTIONS: Read Each Question Carefully and Choose The Best Answerapi-393155483Noch keine Bewertungen
- Ideal Gas EquationDokument20 SeitenIdeal Gas EquationsamNoch keine Bewertungen
- Moodle Basic ThermodynamicsDokument14 SeitenMoodle Basic ThermodynamicsArpan BiswasNoch keine Bewertungen
- Problems On Statistical Physics FinalDokument4 SeitenProblems On Statistical Physics FinalMohamed MazouziNoch keine Bewertungen
- Non-Ideal GasesDokument8 SeitenNon-Ideal GasesMatthewNoch keine Bewertungen
- CH 1 Mixture of Ideal GasesDokument38 SeitenCH 1 Mixture of Ideal GasesFasil GetachewNoch keine Bewertungen
- 2843neet Opt PDFDokument12 Seiten2843neet Opt PDFsubhash sahuNoch keine Bewertungen
- Spe 115 G PDFDokument14 SeitenSpe 115 G PDFJuan SantosNoch keine Bewertungen
- Kinetic Molecular TheoryDokument4 SeitenKinetic Molecular TheoryJOHN ROLIE MAMELOCONoch keine Bewertungen
- Thermal Physics A2 Matter (Ideal Gas) WS FinalDokument4 SeitenThermal Physics A2 Matter (Ideal Gas) WS FinalMahbub KhanNoch keine Bewertungen
- College Physics 9th Edition Serway Test BankDokument35 SeitenCollege Physics 9th Edition Serway Test Bankshannonsellersdfrjxwiabt100% (34)
- GENERAL PHYSICS 1 Quarter 2 Activity SheetDokument26 SeitenGENERAL PHYSICS 1 Quarter 2 Activity SheetSeverus S Potter100% (1)
- A. N. Matveev-Molecular Physics-Mir Publishers (1985)Dokument450 SeitenA. N. Matveev-Molecular Physics-Mir Publishers (1985)DũngNoch keine Bewertungen
- Teorema Del VirialDokument13 SeitenTeorema Del VirialJOHAN STIWAR FIERRO FIERRONoch keine Bewertungen
- F Y B SC - ChemistryDokument15 SeitenF Y B SC - ChemistryAAANoch keine Bewertungen
- Modeling An Simulation of Steam Turbine ProcessesDokument26 SeitenModeling An Simulation of Steam Turbine Processescoronel_goNoch keine Bewertungen
- EG335 Notes Part 1 NewDokument20 SeitenEG335 Notes Part 1 NewAlan Kottommannil ThomsonNoch keine Bewertungen
- Marcet Boiler Lab ReportDokument15 SeitenMarcet Boiler Lab ReportAnna Kay100% (7)