Beruflich Dokumente
Kultur Dokumente
Synthesis and Reactivity of Zinc and Aluminum Alkyl Derivatives Stabilized by Oxygen
Hochgeladen von
Jennifer Carolina Rosales NoriegaCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Synthesis and Reactivity of Zinc and Aluminum Alkyl Derivatives Stabilized by Oxygen
Hochgeladen von
Jennifer Carolina Rosales NoriegaCopyright:
Verfügbare Formate
Inorg. Chem.
1992, 31, 989-992
989
Contribution from the Department of Chemistry,
Columbia University, New York, New York 10027
Synthesis and Reactivity of Zinc and Aluminum Alkyl Derivatives Stabilized by Oxygen
Tripod Ligands
Adrian Looney, Mark Cornebise, Dena Miller, and Gerard Parkin*
Received May 22, 1991
Zinc and aluminum alkyl derivatives LoEtZnR(R = CH3, CH2CH3,CH(CH3)2)and LoE,AI(CH3)2[Al(CH3)3]
of the oxygen
tripod ligand system LoEt [CpCO(P(O)(OEt),J3]-(Cp = q5-C5H5)have been synthesized by the reactions of the alkyls ZnR2
and A1(CH3)3with TlLoE,. The zinc alkyls LoE,ZnRare precursors for a series of other half-sandwich complexes LoE,ZnX (X
= C1, I, CH3C02,NO3).
Introduction
We have recently described the use of the nitrogen-donor
poly(pyrazoly1)hydroborato ligands' to provide well-defined coordination environments for a series of monomeric alkyl and
hydride derivatives of the s- and pblock metals? Here we report
the use of the related oxygen-donor tripod ligand system LOEt p
[CpCo(P(O)(OEt),},]- (Cp = qS-C5Hs),recently introduced by
KlBui,3 for the synthesis of alkyl derivatives of zinc and aluminum.
Results
The thallium derivative TlLOE,, prepared by metathesis of
TIN03 with NaLoEt, is a useful reagent for the synthesis of
half-sandwich zinc alkyl derivatives b u Z n R . Thus, the complexes
LoE,ZnR (R = CH,, CH2CH3,CH(CH,),) are readily prepared
by the metathesis of ZnR, with TlLoEt (eq 1). The alkyl deZnR,
TlLoet
LoEtZnR
(1)
rivatives LoEtZnR are yellow crystalline solids that are soluble
in hydrocarbons. IH and 13CN M R spectra are in accord with
the proposed formulation, and the 'H N M R spectrum of
LoEtZnCH3,illustrated in Figure 1, is provided as a repesentative
example. Selected NMR data (in which only the cyclopentadienyl
resonances of the LoEt ligand are reported) for all new complexes
are presented in Table I, while a complete table of spectroscopic
data is available as supplementary material.
A1(CH3), reacts with TlLoEt (or NaLoEt) to give a binuclear
complex of composition LoE,A1(CH3),[A1(CH3),], in which the
tripod ligand is proposed to be coordinated to two different aluminum alkyl centers (eq 2). Evidence for the formulation of
TlLm
Al(CH3)3
LOEtAl(CH3)2[A1(CH3)31
(2)
LoE,A1(CH3)2[Al(CH3)3]is provided by the IH N M R spectrum
(Figure 2), which shows three singlets due to aluminum methyl
groups in the ratio 1:1:3. The two singlets in the ratio 1:l are
assigned to inequivalent methyl groups of the [Al(CH,),] group,
with the remaining singlet due to coordinated [Al(CH,),].
Furthermore, the observation of three sets of triplet resonances
assigned to the methyl groups of the LoEt ligand indicates that
the C, symmetry of the tripod ligand has been reduced to C, upon
coordination. A tentative structure for LoEt~(CH3)2[Al(CH3)3]
that is consistent with the N M R data is shown in Figure 2, in
which a mirror plane passes through Co and the two A1 atoms.
The reactions of the zinc alkyl complexes LoE,ZnR (R = CH,,
CH,CH,) with HC1, CH3C02H,and HNO, result in elimination
(a) Trofimenko, S.Prog. Inorg. Chem. 1986,34, 115-210. (b) Trofimenko, S.; Calabrese, J. C.; Thompson, J. S. Inorg. Chem. 1987, 26,
1507-1514. (c) Trofimenko, S.; Calabrese, J. C.; Domaille, P. J.;
Thompson, J.'S: Inorg. Chem. 1989, 28, 1091-1101.
(a) Han, R.; Looney, A.; Parkin, G . J . Am. Chem. SOC.1989, 1 1 1 ,
7276-7278. (b) Gorrell, I. E.;Looney, A.; Parkin, G . J . Chem. SOC.,
Chem. Commun. 1990, 220-222. ( c ) Han, R.; Gorrell, I. B.; Looney,
A.; Parkin G.J . Chem. SOC.,Chem. Commun. 1991, 717-719. (d)
Looney, A.; Parkin, G . Polyhedron 1990, 9, 265-276. ( e ) Han, R.;
Parkin, G. Organometallics 1991,10, 1010-1020. (0 Han, R.; Parkin,
G . J . Am. Chem. SOC.1992, 114, 748-757.
Kliiui, W. Angew. Chem., Int. Ed. Engl. 1990, 29, 627-637.
of alkane and formation of the corresponding zinc derivative
bE,ZnX (X = c1, 02CCH3,NO,). The iodide derivative LoEtZnI
is obtained from the reaction of LoEtZnR with 1,. The LoEtZnX
complexes (X = C1, I, CH3C02, NO,) may also be obtained
independently by the reaction of ZnX, with 1 equiv of MLoEt (M
= Na, T1) as shown in eq 3.
ZnX,
+ MLoE,
LoE,ZnX
+ MX
(3)
The molecular structures of the chloride and nitrato derivatives,
LoEtZnC1 and LoE,Zn(q2-O2NO),have been determined by X-ray
diffraction. Unfortunately, the tripod ligands in LoEtZnCI and
LoEtZn(q2-02NO)are disordered in a manner similar to that for
previously reported structure^,^ so that detailed analyses of the
structures are not warranted. However, the constitutions of the
molecules are clearly supported by the structure determinations,
as illustrated by Figures 3 and 4 in which only one of the disordered configurations is shown in each case.
Discussion
The zinc alkyl derivatives LoE,ZnR (R = CH,, CH,CH,,
CH(CH3)2)are conveniently obtained by the metathesis of ZnR,
with TlLoEt. The decomposition of [TlR], depositing metallic
thallium, provides an effective driving force for the reaction, in
a manner similar to that observed in the reactions of ZnR, and
MgRz with (poly(pyrazoly1)hydroborato)thallium complexes.2
Although the mononuclear 4-coordinate zinc alkyl complexes are
analogous to those obtained for the tris(pyrazoly1)hydroborato
ligand system,2b,cthe product obtained from the reaction of Al(CH,), with TlLoEt (or NaLoEt) is proposed to be a binuclear
complex of composition LoEtAl(CH3)2[Al(CH3)3],
in which the
tripod ligand is coordinated to two different aluminum alkyl
centers. Precedence for coordination of the LOEt ligand to more
than one metal center is provided by the structures of the sodium
and lithium complexes Na3(LoEt)3(H20)25
and [LiLoEt]2.6
The reactivity of the zinc alkyl complexes LoEtZnR (R = CH3,
CH,CH,) is illustrated in Scheme I. Reactions with HC1,
CH3C02H,and H N 0 3 result in elimination of alkane accompanied by formation of the corresponding zinc derivative LoEtZnX
(X = C1, CH,C02, NO,). The iodide derivative LoE;ZnI may
be obtained from the reaction of LoEtZnR with I,. Thus, a series
of half-sandwich derivatives LoEtZnX (X = C1, I, CH3CO2,NO,),
which complement the very stable sandwich complex (LoEt),Zn,'
may be prepared directly from the zinc alkyl complexes LoE,ZnR.
Alternatively, the half-sandwich complexes LoEtZnX (X = C1,
I, CH3C02,NO,) may be obtained independently by the reaction
of ZnX, with 1 equiv of MLoEt (M = Na, Tl).
The molecular structures of the chloride and nitrato derivatives,
LoE,ZnCl and LoEtZn(q2-02NO),have been determined by X-ray
diffraction, as shown in Figures 1 and 2. Although the tripod
(4) Dubler, E.; Linowsky, L.; Klaui, W. Transition Met. Chem. 1979, 4 ,
191-198.
(5) Klaui, W.; Muller, A.; Eberspach, W.; Boese, R.; Goldberg, I. J. Am.
Chem. SOC.1987, 109, 164-169.
(6) Goldberg, I.; Shinar, H.; Navon, G.; Klaui, W. J . Inclusion Phenom.
1987, 5 , 181-185.
(7) Klaui. W.; Dehnicke, K. Chem. Ber. 1978, 1 1 1 , 451-468.
0020-166919211331-0989%03.00/0 0 1992 American Chemical Society
990 Inorganic Chemistry, Vol. 31, No. 6, 1992
Looney et al.
Table I. Selected Spectroscopic Datam
compd
'H NMR
6, ppm
assgnt
q5-CsH5
Zn-CH,
q5-C5H5
Zn-CH2CH3
Zn-CH2CH,
LOEtZnCH3
LoE,ZnCH2CH3
coupling, Hz
4.96
0.01
4.96
0.92
1.91
s
s
s
q, 3 J ~=- 8~
t, ,JH-H
9
assgnt
B~-C~HS
Zn-CH3
V~-CSH~
Zn-CH2CH3
"C NMR
6, ppm
89.6
-16.9
89.6
-2.4
coupling, Hz
d, 'Jc-H= 181
q, 'Jc-H= 120
d, 'JC-H 181
t, IJC-H = 119
q. 2Jc-H= 4
Zn-CH2CH,
(LOEt)2Zn
LoE,Ahie2[AIMe3]
q5-C5Hs
q5-C5Hs
Al(CH3)2Al(CH,),
5.16
4.95
-0.37
-0.48
-0.20
AI(CH,),AI(CH,),
s
s
s
"In C,D6. Abbreviations: s = singlet, d = doublet, t = triplet, q = quartet, and spt = septet.
LOB, ligand. 'NMR assignments are consistent with observed integrations.
cyclopentadienyl resonances are given for the
CI
I
3
I
1
Figure 3. Ball and stick drawing of LoEtZnCI showing only one of the
disordered configurations (see text).
6 lwm)
Figure 1. 'H NMR spectrum of LoE,ZnCH3(R' = OEt).
042
Figure 4. Ball and stick drawing of LoE1Zn(q2-02NO)showing only one
of the disordered configurations (see text).
I
5
I
4
I
2
1
1
6 lpomi
Figure 2. 'HNMR spectrum of LoE,AI(CH,)2[A1(CH3),](R = OEt;
a-c subscripts indicate chemically equivalent groups).
ligand in these molecules is disordered, the coordination environment about the zinc center in the chloride derivative LoEtZnCI
can be seen to be closely analogous to that in the related 4-coordinate complex (v3-HB(3-Butpz),JZnC1 (3-Bu'pz =
C3N2ButHZ).* Thus, the Zn-C1 bond distance (2.163 (2) A),
the Zn-0 distances (average = 1.97 A), and 0-Zn-Cl bond angles
(average = 119') in LoEtZnClmay be compared with the Zn-Cl
bond distance (2.183 (2) A), the Zn-N distances (average = 2.05
A), and N-Zn-Cl bond angles (average = 122O) in (v3-HB(3Bu'pz),}ZnCl. However, the oxygen tripod ligand LoEt is significantly less sterically demanding than the tris(3-terf-butylpyrazoly1)hydroborato ligand, as indicated by the ready formation
of the 5-coordinated complexes LoE1Zn(v2-O2NO)and LoEtZn(8) Yoon, K.; Parkin, G . J . Am. Chem. SOC.1991, 113, 8414-8418.
Inorganic Chemistry, Vol. 31, No. 6, I992 991
Zinc and Aluminum Alkyl Derivatives
Scheme 1. Reactivity of LoBtZnR (R' = OEt)
bdn-1
H 2 0or O2
(bEt12Zn
under reduced pressure. The residue was extracted into CH2CI2(2 X 100
($-02CCH3). Although 5-coordination is not common for zinc,
mL) and filtered. The solvent was removed under reduced pressure,
several other complexes that possess 5-coordinate zinc centers have
giving TlLoEt as a yellow solid (11.7 g, 88%). Anal. Calcd for
been reported? Indeed, the zinc N-benzoylglycinate pentahydrate
C17H3509P3C~T1:
C, 27.6; H, 4.8. Found: C, 27.6; H, 4.6.
complex, Zn(C6H5CONHCH2C02)2*5H20P
is similar to
Synthesis of bEtZnCH3. Z ~ I ( C H , )(1.2
~ mL of a 1.1 M solution in
LoEtZn($-02NO) in that the zinc coordination sphere of both
pentane, 1.32 mmol) was added to a stirred solution of TlLoEt (400 mg,
compounds is comprised of only oxygen ligation.
0.54 mmol) in C6H6 (15 mL), resulting in the immediate formation of
A further illustration of the relatively small steric requirements
a precipitate of TI metal. The mixture was stirred overnight at room
of the bEl
ligand is the observation that the 6-cmrdinate sandwich
temperature and filtered, giving a yellow solution. The solvent was
complex (LoEl)2Zn7may be obtained as the product of the reremoved under reduced pressure, giving LoEtZnCH, as a yellow solid
(200 mg, 60%). LoE,ZnCH3was recrystallized by addition of pentane
actions of LoEtZnRwith a variety of substrates, including O2and
to a concentrated benzene solution. Anal. Calcd for ClsH,sO,P,CoZn:
HzO. In this regard the oxygen tripod ligand LOEt more closely
C, 35.1; H, 6.2. Found: C, 35.3; H, 5.9. MS: m / e 615 (M+ + 1).
resembles the unsubstituted tris(pyrazoly1)hydroborato ligand than
Synthesis of &,E,Z~CH~CH~.
Z I I ( C H ~ C H ~(0.73
) ~ mL of a 1.1 M
the sterically demanding tris( 3-tert-butylpyrazolyl)hydroborato
solution in hexane, 0.80 mmol) was added to a stirred solution of TILoEt
ligand system.
(300 mg, 0.4 mmol) in C6H6 (25 mL), resulting in the immediate forIn summary, alkyl derivatives of zinc and aluminum, LoEtZnR
mation of a precipitate of TI metal. The mixture was stirred for 1 h at
(R = CH3, CH2CH3,CH(CH3I2)and L o ~ ~ A ~ ( C H ~ ) ~ [ A ~ ( C Hroom
~ ) ~temperature
~,
and filtered, giving a yellow solution. The solvent was
that are stabilized by the oxygen tripod ligand system LoEt may
removed under reduced pressure, giving L ~ E , Z ~ C H ~asCa H
yellow
~ solid
be readily synthesized. The zinc complexes are analogous to those
(218 mg, 86%), which was recrystallized from pentane at -78 OC. Anal.
Calcd for Cl9HaO9PSCoZn: C, 36.2; H, 6.4. Found: C, 36.0; H, 6.2.
of the tris(pyrazoly1)hydroborato ligand system, whereas the
synthesis of &,E,ZnCH(CH3)2. Zn[CH(CH3)2]2(1.0 mL of a 0.2 M
is unique.
binuclear aluminum complex LoElAl(CH3)2[Al(CH3)3]
solution in pentane, 0.20 mmol) was added to a stirred solution of TIL,,
The reactions of the zinc alkyls LoE,ZnR result in the formation
(150 mg, 0.20 mmol) in C6H6 (20 mL), resulting in the immediate
of a variety of other half-sandwich derivatives LoE,ZnX (X = C1,
formation of a precipitate of TI metal. The mixture was stirred for 2 h
I, CH3C02,NO3).
at room temperature and filtered, giving a yellow solution. The solvent
Experimental Details
General Considerations. All manipulations were performed using a
combinationof glovebox, high-vacuum, or Schlenk techniques.I0 Solvents
were purified and degassed by standard procedures. 'H NMR spectra
were measured on a Varian VXR 200 spectrometer. Mass spectra were
obtained on a Nermag R10-10 mass spectrometer using chemical ionization (NH,) techniques. Elemental analyses were measured using a
Perkin-Elmer 2400 C H N elemental analyzer. Na[CpCo(P(O)(OEt),),].xH,O was prepared by the literature method." NMR data
for all new complexes are given in Table I.
Synthesis of TGEt. A solution of TlN03 (10 g, 37.6 mmol) in H 2 0
(125 mL) was added to a solution of NaLoEt (10 g, 17.9 mmol) in H 2 0
(90 mL), resulting in the formation of a yellow precipitate. The mixture
was stirred for 6 h at room temperature, and the water was removed
(a) Grewe, H.; Udupa, M. R.; Krebs, B. fnorg. Chim. Acta 1982,63,
119-124. (b) Bencini, A.; Bianchi, A.; Garcia-Espana, E.; Mangani,
S.; Micheloni, M.; Orioli, P.; Paoletti, P. Inorg. Chem. 1988, 27,
1104-1 107. (c) Kimura, E.; Koike, T.; Toriumi, K. fnorg. Chem. 1988,
27, 3687-3688. (d) Kato, M.; Ito, T. Inorg. Chem. 1985, 24, 504-508.
(e) Takahashi, K.; Nishida, Y.; Kida, S. Bull. Chem. SOC.Jpn. 1984,
57, 2628-2633. (f) Kai, Y.; Morita, M.; Yasuoka, N.; Kasai, N. Bull.
Chem. Soc. Jpn. 1985,58,1631-1635. (g) Harrison, P. G.; Begley, M.
J.; Kikabhai, T.; Killer, F. J . Chem. Soc.,Dalton Tram. 1986,929-938.
(h) Sola, M.; Lledos, A.; Duran, M.; Bertran, J. Inorg. Chem. 1991,30,
2523-2527 and references therein.
(a) McNally, J. P.; Leong, V. S.; Cooper, N. J. ACS Symp. Ser. 1987,
No. 357,6-23. (b) Burger, B. J.; Bercaw, J. E. ACS Symp. Ser. 1987,
NO. 357, 79-97.
KIPui, W . Z . Naturforsch.. B Anorg. Chem., Org. Chem. 1979,348,
1403-1 407.
was removed under reduced pressure, giving LoE,ZIICH(CH3)2 as a
yellow solid (40 mg, 31%), which was recrystallized from pentane at -78
OC. Anal. Calcd for C20H4209P3CoZn:C, 37.3; H, 6.6. Found: C,
38.1; H, 6.1. MS: m / e 644 (M').
Synthesis of &,E,Znl. A solution of NaLoEt (250 mg, 0.45 mmol) in
CH2CI2(20 mL) was added to a suspension of ZnI, (143 mg, 0.45 mmol)
in CH2C12(10 mL) and stirred overnight at room temperature. The
mixture was filtered, giving a yellow solution, and the solvent was removed under reduced pressure, giving LEtZnI as a yellow solid (160 mg,
49%). LoEIZnI was recrystallized from a mixture of benzene/pentane.
Anal. Calcd for C,7H3S09P3CoZnI:C, 28.1; H, 4.9. Found: C, 28.3;
H, 4.8. MS: m / e 726 (M+ - 1).
Reaction of bE,ZnR (R = CH2CH3,CH(CH3)2)with 12. A solutioil
of LoE,ZnR (R = CH2CH3,CH(CH3)2)(20 mg) in C6D6(0.7 mL) was
treated with I2 (ca. 10 mg). LoE,ZnI and RI were formed quantitatively
after 10 min at room temperature, as observed by IH NMR spectroscopy.
Synthesis of bE,ZnCI. A solution of NaLoEt(200 mg, 0.36 mmol)
in CH2C12(20 mL) was added to a suspension of ZnC1, (49 nig, 0.36
mmol) in CH2CI2(10 mL) and stirred overnight at room temperarure.
The mixture was filtered, giving a yellow solution, and the solvent was
removed under reduced pressure, giving LoEtZnC1as a yellow solid (1 20
mg, 52%). LoEtZnC1was recrystallized from a mixture of benzene/
pentane. Single crystals for X-ray diffraction were obtained from acetone. Anal. Calcd for C17H3,09CICoZn:C, 32.1; H, 5.6. Found: C,
32.2; H, 5.3. MS: m / e 634 (M').
Reaction of LoE,Z~CH$H~ with HCl(aq). A solution of
L ~ E , Z ~ C H ~(350
C Hmg,
~ 0.55 mmol) in C6H6 (15 mL) was treated with
HCl(aq) (60 pL of a 12 M solution, 0.72 mmol). The mixture was
stirred for 2 h at room temperature and filtered, giving a yellow solution.
The solvent was removed under reduced pressure, giving LoEtZnC1 as a
yellow solid (125 mg, 35%).
992 Inorganic Chemistry, Vol. 31, No. 6, 1992
Table 11. Summarv of Crvstal and Intensitv Collection Data
LoEtZn(?2-N0)
LoE,ZnC1
formula
CI7H1$N0,,P1CoZn
CI7Hl5O9P1CICoZn
._ _ _
fw
662.;636.2lattice
monoclinic
monoclinic
cell consts
a, A
11.923 (2)
11.882 (2)
b, A
20.352 (4)
20.017 (4)
c, A
12.170 (2)
12.204 (2)
8, deg
103.31 (1)
106.49 (1)
v,A
2873.8 (8)
2783.2 (7)
4
Z
4
P2/n (No. 14)
P2,/n (No. 14)
space group
Mo K a (0.71073)
radiation (A, A) Mo K a (0.71073)
1.53
1.52
p(calcd), g cm-
1.841
GOF
1.848
0.0627
0.0524
R
0.0775
0.0723
RW
~~
~~
R = EIFo- Fcl/131Fol;
Rw = IIE(Fo- ~ c ) 2 1 / [ E ~ ( ~ o ) 2 1GOF
1~2;
+ gP1-1.
= l [ P ( F o- FE)21/[Nda,a- Nparams112;
w=
Synthesis of hEtZn(q2-O2NO).A solution of T l L o ~(500
t
mg, 0.68
mmol) in THF (20 mL) was added to a suspension of Zn(NO3),.6H2O
(200 mg, 0.68 mmol) in T H F (6 mL), resulting in the formation of a
white precipitate. The mixture was stirred overnight at room temperature
and was filtered, giving a yellow solution, and the solvent was removed
under reduced pressure, giving LoEtZn(q2-02NO)as a yellow solid (345
mg, 77%). LoE1Zn(q2-O2NO)was recrystallized from a mixture of
benzene/pentane. Single crystals of X-ray diffraction were obtained from
C, 30.8; H, 5.3; N, 2.1.
THF. Anal. Calcd for C17H35012NP3C~Zn:
Found: C, 30.8; H, 5.1; N, 2.1. MS: m / e 660 (M - I).
Reaction of L o E , Z ~ C H ~ C H
with
~ HN03(aq). A solution of
LoE,Z~CH~CHJ
(130 mg, 0.21 mmol) in C6H6(10 mL) was treated with
HN03(aq) (15 pL of a 15.6 M solution, 0.23 mmol). The mixture was
stirred for 2 h at room temperature and filtered, giving a yellow solution.
The solvent was removed under reduced pressure, giving LoEtZn(q20 2 N O ) as a yellow solid (100 mg, 74%).
Synthesis of hEtZn(s2-O2CCH3).A solution of TlLoEt (250 mg, 0.34
mmol) in CH2C1, (20 mL) was added to a suspension of Zn(02CCH3)2.2H20(74 mg, 0.34 mmol) in CH2Clz (10 mL), resulting in the
formation of a white precipitate. The mixture was stirred overnight at
room temperature and was filtered, giving a yellow solution, and the
solvent was removed under reduced pressure, giving LomZn(q2-02CCH3)
as a yellow solid (100 mg, 45%). LoE,Zn(q2-O2CCH3)was recrystallized
from a mixture of toluene/pentane. Anal. Calcd for C19H380,1P3CoZn:
C, 34.6; H, 5.8. Found: C, 34.6; H, 5.8.
Reaction of LoEtZnCH2CH3with CH3C02H. A solution of
LoEtZnCH2CH, (770 mg, 1.22 mmol) in C6H6(30 mL) was treated with
CH3C02H(110 pL, 1.93 mmol). The mixture was stirred overnight at
room temperature and filtered, giving a yellow solution. The solvent was
removed under reduced pressure, giving LoE,Zn(~2-02CCH3)
as a yellow
solid (700 mg, 87%).
Synthesis Of LOE,AI(CH~)JAI(CH,),].AI(CH3) (600 pL Of a 2 M
solution in hexane, 1.20 mmol) was added to a stirred solution of TlLoEt
(200 mg, 0.27 mmol) in pentane (10 mL), resulting in the immediate
formation of a precipitate of TI metal. The mixture was stirred for 1 h
at room temperature and filtered, giving a yellow solution. The solvent
was removed under reduced pressure, giving LoE,AI(CH)2[A1(CH9)]
as a yellow solid (100 mg, 56%). Anal. Calcd for C22HS,,A12C009P3:
C, 39.8; H, 7.6. Found: C, 39.8; H, 7.5.
Reaction of hE,ZnCH2CH3with 02.A solution of L o E , Z ~ C H ~ C H ~
(100 mg, 0.16 mmol) in benzene (10 mL) was stirred for 5 min under
Looney et al.
0,(1 atm). The mixture was filtered and the solvent removed under
reduced pressure to give (LoEJ2Zn as a yellow solid (50 mg).
RePCtion Of Lr~EtznCH2cH3With H20. A solution Of L o E , Z ~ C H ~ C H ~
(20, mg) in C6D6 (0.7 mL) was treated with H 2 0 (14 pL). Over the
period of 1 day L O E , Z ~ C H ~ C
was
H ~converted to the complex (LoEl)2Zn
as shown by H NMR spectroscopy.
Synthesis of (hEJ2Zn. The following synthesis is adapted from the
original report.6 A solution of NaLoEt (200 mg, 0.36 mmol) in CH2CI2
( 15 mL) was added to a stirred suspension of ZnCI2 (24 mg, 0.18 mmol)
in CH2CI2(IO mL). The mixture was stirred overnight at room temperature. The mixture was filtered and the solvent removed under reduced pressure, giving (LoEl)2Zn(40 mg, 20%).
X-ray Structure Determination of h E t Z ~ Cand
i hEtZn(q2-O2No).
Crystal data, data collection, and refinement parameters are summarized
in Table 11. Single crystals of LoE,ZnCI and LoEtZn(q2-02NO)were
mounted in glass capillaries and placed on a Nicolet R3m diffractometer.
The unit cells were determined by the automatic indexing of 25 centered
reflections and confirmed by examination of the axial photographs. Intensity data were collected using graphite-monochromated Mo Ka Xradiation (A = 0.71073 A). Check reflections were measured every 100
reflections, and the data were scaled accordingly and corrected for
Lorentz, polarization, and absorption effects. The structures was solved
using Patterson and standard difference map techniques on a Data
General NOVA 4 computer using SHELXTL.~ Systematic absences were
consistent with the space group P2,/n (No. 14) for both crystals. A
2-fold disorder of the [P(O)(OEt,),] groups of the LoEt ligand about the
Co-Zn vector was evident for both structures, analogous to that previously reported? The 2-fold disorder is such that the OEt groups of both
orientations become coincident (or nearly so), so that in effect only two
orientations of the [P(O)] group were observed. The disorder was
therefore modeled by refining the occupancies of the P and 0 atoms of
the two sets of [P(O)] groups. The site occupancy factors refined to 0.56
and 0.44 for LoEtZn(q2-N03),and 0.69 and 0.31 for LoEtZnCI. Tables
of atomic coordinates and thermal parameters are available as supplementary material.
Acknowledgment is made to the National Science Foundation
(Grant CHE 90-07512) and to the donors of the Petroleum
Research Fund, administered by the American Chemical Society,
for support of this research. G.P. is the recipient of an A. P. Sloan
Research Fellowship (1991-1993) and a Camille and Henry
Dreyfus Teacher-Scholar Award (1991-1996). We thank Professor John E. Bercaw and Dr. John Power for a generous gift
of [CpCo(P(O)(OEt),),]Na and two of the reviewers for helpful
comments.
Registry NO. TILoE~, 137942-96-2; LoE,ZnCH,, 137942-97-3;
LOE,Z~CH~CH,
137942-98-4; LoE,ZnCH(CH,)2, 137942-99-5; LOBtZnI, 137943-00-1; LoEtZnCI, 137943-01-2; Lo~&l(?)~-02NO),
13794302-3; L o ~ ~ z n ( ~ ~ - o $ C H137943-03-4;
p),
LoEtZn, 58438-24-7; LoEtA1(CH3)2[AI( CH,),] , 137943-04-5.
Supplementary Material Available: Table SI, listing spectroscopic data
for all the new complexes, Tables SII-SV, listing crystal and intensity
collection data and atomic coordinates for LoE,ZnC1 and LoEtZn(q202NO), and ORTEP drawings (1 1 pages); listings of calculated and observed structure factors for LoEtZnC1and LoElZn(q2-02NO)(27 pages).
Ordering information is given on any current masthead page.
(12) Sheldrick,G. M. SHELXTL,
An Integrated System for Solving, Refining,
and Displaying Crystal Structures from Diffraction Data; University
of Gottingen, Gottingen, Federal Republic of Germany, 1981,
Das könnte Ihnen auch gefallen
- 634 Notes Combined PDFDokument184 Seiten634 Notes Combined PDFJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Flame Spectrometry in Environmental Chemical Analysis PDFDokument122 SeitenFlame Spectrometry in Environmental Chemical Analysis PDFJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- FluorescenciaDokument651 SeitenFluorescenciaJennifer Carolina Rosales Noriega100% (1)
- 3.2 Organic Synthesis (Reaction Pathways) : Pupil Notes Learning Outcomes Questions & AnswersDokument58 Seiten3.2 Organic Synthesis (Reaction Pathways) : Pupil Notes Learning Outcomes Questions & AnswersJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- 6cd7 PDFDokument5 Seiten6cd7 PDFJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Introduction To Computational Chemistry For Experimental Chemists... (Part 1/2)Dokument46 SeitenIntroduction To Computational Chemistry For Experimental Chemists... (Part 1/2)Jennifer Carolina Rosales NoriegaNoch keine Bewertungen
- (William Jones, C. N. R. Rao) Supramolecular OrganDokument456 Seiten(William Jones, C. N. R. Rao) Supramolecular OrganJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Comprehensive Coordination Chemistry Volumen 2 PDFDokument833 SeitenComprehensive Coordination Chemistry Volumen 2 PDFJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Reactions of Organic FamiliesDokument5 SeitenReactions of Organic FamiliesJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Organic Synthesis. Functional Group InterconversionDokument57 SeitenOrganic Synthesis. Functional Group InterconversionJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Bok:978 3 642 22860 5Dokument191 SeitenBok:978 3 642 22860 5Jennifer Carolina Rosales NoriegaNoch keine Bewertungen
- The Michael ReactionDokument4 SeitenThe Michael ReactionJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- 01 1352193505 80382 PDFDokument86 Seiten01 1352193505 80382 PDFJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Pericyclic Reactions (MCM Chapt 30) : - Polar React. (Nucleophiles and Electrophiles)Dokument30 SeitenPericyclic Reactions (MCM Chapt 30) : - Polar React. (Nucleophiles and Electrophiles)Jennifer Carolina Rosales NoriegaNoch keine Bewertungen
- A Detailed Study On Pericyclic Reactions: Raj Kumar Pandey, Dr. Neelima JainDokument3 SeitenA Detailed Study On Pericyclic Reactions: Raj Kumar Pandey, Dr. Neelima JainJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Lecture11 Moebius TopologyDokument11 SeitenLecture11 Moebius TopologyJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Thermal Pericyclic Reaction Pericyclic Reactions: "Protection of Diene"Dokument12 SeitenThermal Pericyclic Reaction Pericyclic Reactions: "Protection of Diene"Jennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Hooker 1936Dokument3 SeitenHooker 1936Jennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Frontiers in Molecular Main Group Chemistry: Web Themed IssueDokument4 SeitenFrontiers in Molecular Main Group Chemistry: Web Themed IssueJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Reactive Intermediate 4Dokument15 SeitenReactive Intermediate 4Jennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Circuitos Electricos - Joseph A. EdministerDokument294 SeitenCircuitos Electricos - Joseph A. Edministermelisa11100% (2)
- Suggested Solutions For Chapter 38: Purpose of The ProblemDokument14 SeitenSuggested Solutions For Chapter 38: Purpose of The ProblemJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Enantioselective Addition of Phenyl and Alkyl Acetylenes To Imines Catalyzed by Chiral Cu (I) ComplexesDokument4 SeitenEnantioselective Addition of Phenyl and Alkyl Acetylenes To Imines Catalyzed by Chiral Cu (I) ComplexesJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Leak Detection ReportDokument29 SeitenLeak Detection ReportAnnMarie KathleenNoch keine Bewertungen
- Fusion Implementing Offerings Using Functional Setup Manager PDFDokument51 SeitenFusion Implementing Offerings Using Functional Setup Manager PDFSrinivasa Rao Asuru0% (1)
- GPS Spoofing (2002-2003)Dokument8 SeitenGPS Spoofing (2002-2003)Roger JohnstonNoch keine Bewertungen
- Gemini Dollar WhitepaperDokument7 SeitenGemini Dollar WhitepaperdazeeeNoch keine Bewertungen
- PyhookDokument23 SeitenPyhooktuan tuanNoch keine Bewertungen
- E0 UoE Unit 7Dokument16 SeitenE0 UoE Unit 7Patrick GutierrezNoch keine Bewertungen
- Thermally Curable Polystyrene Via Click ChemistryDokument4 SeitenThermally Curable Polystyrene Via Click ChemistryDanesh AzNoch keine Bewertungen
- Hey Friends B TBDokument152 SeitenHey Friends B TBTizianoCiro CarrizoNoch keine Bewertungen
- Snapdragon 435 Processor Product Brief PDFDokument2 SeitenSnapdragon 435 Processor Product Brief PDFrichardtao89Noch keine Bewertungen
- Mcdonald 2016Dokument10 SeitenMcdonald 2016Andrika SaputraNoch keine Bewertungen
- ReadmeDokument3 SeitenReadmedhgdhdjhsNoch keine Bewertungen
- From Philo To Plotinus AftermanDokument21 SeitenFrom Philo To Plotinus AftermanRaphael888Noch keine Bewertungen
- Hamstring - WikipediaDokument21 SeitenHamstring - WikipediaOmar MarwanNoch keine Bewertungen
- Oracle - Prep4sure.1z0 068.v2016!07!12.by - Lana.60qDokument49 SeitenOracle - Prep4sure.1z0 068.v2016!07!12.by - Lana.60qLuis AlfredoNoch keine Bewertungen
- RSA - Brand - Guidelines - 2019 2Dokument79 SeitenRSA - Brand - Guidelines - 2019 2Gigi's DelightNoch keine Bewertungen
- IMS Objectives Targets 2Dokument2 SeitenIMS Objectives Targets 2FaridUddin Ahmed100% (3)
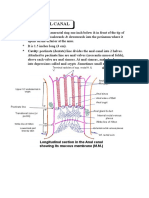
- Anatomy Anal CanalDokument14 SeitenAnatomy Anal CanalBela Ronaldoe100% (1)
- DQ Vibro SifterDokument13 SeitenDQ Vibro SifterDhaval Chapla67% (3)
- Dialogue Au Restaurant, Clients Et ServeurDokument9 SeitenDialogue Au Restaurant, Clients Et ServeurbanuNoch keine Bewertungen
- IKEA SHANGHAI Case StudyDokument5 SeitenIKEA SHANGHAI Case StudyXimo NetteNoch keine Bewertungen
- Ozone Therapy - A Clinical Review A. M. Elvis and J. S. EktaDokument5 SeitenOzone Therapy - A Clinical Review A. M. Elvis and J. S. Ektatahuti696Noch keine Bewertungen
- Kahneman & Tversky Origin of Behavioural EconomicsDokument25 SeitenKahneman & Tversky Origin of Behavioural EconomicsIan Hughes100% (1)
- Bana LingaDokument9 SeitenBana LingaNimai Pandita Raja DasaNoch keine Bewertungen
- 8.ZXSDR B8200 (L200) Principle and Hardware Structure Training Manual-45Dokument45 Seiten8.ZXSDR B8200 (L200) Principle and Hardware Structure Training Manual-45mehdi_mehdiNoch keine Bewertungen
- The Magic DrumDokument185 SeitenThe Magic Drumtanishgiri2012Noch keine Bewertungen
- What Is TranslationDokument3 SeitenWhat Is TranslationSanskriti MehtaNoch keine Bewertungen
- Presentation 11Dokument14 SeitenPresentation 11stellabrown535Noch keine Bewertungen
- Technology Management 1Dokument38 SeitenTechnology Management 1Anu NileshNoch keine Bewertungen
- UC 20 - Produce Cement Concrete CastingDokument69 SeitenUC 20 - Produce Cement Concrete Castingtariku kiros100% (2)
- Obesity - The Health Time Bomb: ©LTPHN 2008Dokument36 SeitenObesity - The Health Time Bomb: ©LTPHN 2008EVA PUTRANTO100% (2)