Beruflich Dokumente
Kultur Dokumente
Chapter 18: Calculating Entropy Changes for Chemical Reactions
Hochgeladen von
Ezequiel OrellanaOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Chapter 18: Calculating Entropy Changes for Chemical Reactions
Hochgeladen von
Ezequiel OrellanaCopyright:
Verfügbare Formate
Chapter 18 Entropy, Free Energy, and Equilibrium
1. What is defined as a process that occurs under a specific set of conditions?
A) conditional process
D) spontaneous process
B) nonspontaneous process
E) conditionality law
C) specified process
Ans: D Bloom's Taxonomy: 1 Difficulty: easy
Page 324
Chapter 18 Entropy, Free Energy, and Equilibrium
2. Which of the following is an example of a nonspontaneous process?
A) ice melting at room temperature
B) sodium metal reacting violently with water
C) rusting of iron at room temperature
D) a ball rolling downhill
E) water freezing at room temperature
Ans: E Bloom's Taxonomy: 2 Difficulty: moderate
Page 325
Chapter 18 Entropy, Free Energy, and Equilibrium
3. A spontaneous endothermic reaction always
A) causes the surroundings to get colder. C) requires a spark to initiate it.
B) bursts into flame.
D) releases heat to the surroundings.
Ans: A Bloom's Taxonomy: 2 Difficulty: moderate
Page 326
Chapter 18 Entropy, Free Energy, and Equilibrium
4. The reaction rates of many spontaneous reactions are actually very slow. Which of
these statements is the best explanation for this observation?
A) Kp for the reaction is less than one.
B) The activation energy of the reaction is large.
C) G for the reaction is positive.
D) Such reactions are endothermic.
E) The entropy change is negative.
Ans: B Bloom's Taxonomy: 2 Difficulty: moderate
Page 327
Chapter 18 Entropy, Free Energy, and Equilibrium
5. When a sky diver freefalls through the air, the process is
A) nonspontaneous because he is accelerating due to the force applied by gravity.
B) nonspontaneous because he is losing potential energy.
C) nonspontaneous because he had planned the jump for two weeks.
D) spontaneous.
E) in equilibrium.
Ans: D Bloom's Taxonomy: 2 Difficulty: moderate
Page 328
Chapter 18 Entropy, Free Energy, and Equilibrium
6. Which, if any, of the following processes is spontaneous under the specified conditions?
A) H2O(l) H2O(s) at 25C
B) CO2(s) CO2(g) at 0C
C) 2H2O(g) 2H2(g) + O2(g)
D) C(graphite) C(diamond) at 25C and 1 atm pressure
E) None of these choices is spontaneous.
Ans: B Bloom's Taxonomy: 2 Difficulty: difficult
Page 329
Chapter 18 Entropy, Free Energy, and Equilibrium
7. What is defined as a measure of the randomness or disorder of a system?
A) Gibbs free energy
D) nonspontaneous process
B) entropy
E) enthalpy
C) spontaneous process
Ans: B Bloom's Taxonomy: 1 Difficulty: easy
Page 330
Chapter 18 Entropy, Free Energy, and Equilibrium
8. What is defined as the absolute disorder of a substance at 1 atm?
A) atmospheric disorder
D) specific pressure
B) atmospheric pressure
E) standard entropy
C) atmospheric entropy
Ans: E Bloom's Taxonomy: 1 Difficulty: easy
Page 331
Chapter 18 Entropy, Free Energy, and Equilibrium
9. Which statement is correct?
A) The standard entropy of a gas phase substance is less than the standard entropy of
the same substance in the solid phase.
B) For two different substances in the same phase, the substance with the more
complex structure has the lower entropy.
C) When there are allotropic forms of an element, the more highly ordered form has
a higher entropy.
D) Entropy of elements and compounds are always positive.
E) For monatomic species, the lighter substance has the greater entropy.
Ans: D Bloom's Taxonomy: 1 Difficulty: moderate
Page 332
Chapter 18 Entropy, Free Energy, and Equilibrium
10. Which statement is correct?
A) Heating always decreases the entropy of a system.
B) The reaction that results in an increase in the number of moles of gas always
decreases the entropy of a system.
C) The greater number of particles leads to a smaller number of microstates.
D) An increase in the temperature decreases all types of molecular motions.
E) The solution process usually leads to an increase in entropy.
Ans: E Bloom's Taxonomy: 2 Difficulty: difficult
Page 333
Chapter 18 Entropy, Free Energy, and Equilibrium
11. The Second Law of Thermodynamics is:
A) the energy of the universe is constant.
B) the entropy of the universe is constant.
C) the enthalpy of the universe is constant.
D) the energy of the universe is decreasing.
E) the entropy of the universe is increasing.
Ans: E Bloom's Taxonomy: 1 Difficulty: easy
Page 334
Chapter 18 Entropy, Free Energy, and Equilibrium
12. What term is given to the fact that the entropy of the universe increases in spontaneous
processes and is unchanged in an equilibrium process?
A) Second law of Thermodynamics
D) Second Law of Thermodynamics
B) First Law of Entropy
E) Third Law of Thermodynamics
C) First Universal Law
Ans: D Bloom's Taxonomy: 1 Difficulty: easy
Page 335
Chapter 18 Entropy, Free Energy, and Equilibrium
13. Which of these species has the highest entropy (S) at 25C?
A) CH3OH(l) B) CO(g) C) MgCO3(s) D) H2O(l) E) Ni(s)
Ans: B Bloom's Taxonomy: 2 Difficulty: easy
Page 336
Chapter 18 Entropy, Free Energy, and Equilibrium
14. Which of the following species has the largest entropy?
A) diamond B) C (graphite) C) H2O(l) D) Br2(l)
Ans: E Bloom's Taxonomy: 2 Difficulty: easy
Page 337
E) O2 (g)
Chapter 18 Entropy, Free Energy, and Equilibrium
15. Which species will have the greatest absolute entropy at 25C?
A) Ne(g) B) C2H2(g) C) H2O(l) D) C2H5OH(l) E) C4H10(g)
Ans: E Bloom's Taxonomy: 2 Difficulty: moderate
Page 338
Chapter 18 Entropy, Free Energy, and Equilibrium
16. Which species will have the lowest absolute entropy at 25C?
A) C2H5OH(l) B) C2H2(g) C) C3H8(g) D) C3H7OH(l) E) C2H6(g)
Ans: A Bloom's Taxonomy: 2 Difficulty: moderate
Page 339
Chapter 18 Entropy, Free Energy, and Equilibrium
17. Which response includes all of the following processes that are accompanied by an
increase in entropy?
1. I2(s) I2(g)
2. 2I(g) I2(g)
3. 2NH3(g) N2(g) + 3H2(g)
4. Mg2+(aq) + 2OH(aq) Mg(OH)2(s)
A) 1, 2 B) 1, 3 C) 3, 4 D) 3 E) 2, 4
Ans: B Bloom's Taxonomy: 2 Difficulty: difficult
Page 340
Chapter 18 Entropy, Free Energy, and Equilibrium
18. A certain process has Suniv > 0 at 25C. What does one know about the process?
A) It is exothermic.
B) It is endothermic.
C) It is spontaneous at 25C.
D) It will move rapidly toward equilibrium.
E) none of the above
Ans: C Bloom's Taxonomy: 1 Difficulty: easy
Page 341
Chapter 18 Entropy, Free Energy, and Equilibrium
19. Which of the following is necessary for a process to be spontaneous?
A) Hsys < 0 B) Ssys > 0 C) Ssurr < 0 D) Suniv > 0 E) Gsys = 0
Ans: D Bloom's Taxonomy: 1 Difficulty: moderate
Page 342
Chapter 18 Entropy, Free Energy, and Equilibrium
20. Which of the following results in a decrease in the entropy of the system?
A) O2(g), 300 K O2(g), 400 K
B) H2O(s), 0C H2O(l), 0C
C) N2(g), 25C N2(aq), 25C
D) NH3(l), 34.5C NH3(g), 34.5C
E) 2H2O2(g) 2H2O(g) + O2(g)
Ans: C Bloom's Taxonomy: 2 Difficulty: moderate
Page 343
Chapter 18 Entropy, Free Energy, and Equilibrium
21. Which reaction has the largest So?
A) 2N2H4(g) 2NH3(g) + H2(g)
D) N2(g) +3H2(g) 2NH3(g)
B) O2(g) + 2H2(g) 2H2O(l)
E) 2NO(g) N2O2(g)
C) O2(g) + 2H2(g) 2H2O(g)
Ans: A Bloom's Taxonomy: 2 Difficulty: moderate
Page 344
Chapter 18 Entropy, Free Energy, and Equilibrium
22. Which of the following is true for a system at equilibrium?
A) Ssys = Ssurr
B) Ssys = Ssurr
C) Ssys = Ssurr = 0
D) Suniv > 0
E) None of these is a sufficient condition.
Ans: B Bloom's Taxonomy: 1 Difficulty: moderate
Page 345
Chapter 18 Entropy, Free Energy, and Equilibrium
23. Which of the following is always true for an exothermic process?
A) qsys > 0, Ssurr < 0
D) qsys > 0, Ssurr > 0
B) qsys < 0, Ssurr > 0
E) w < 0
C) qsys < 0, Ssurr < 0
Ans: B Bloom's Taxonomy: 2 Difficulty: moderate
Page 346
Chapter 18 Entropy, Free Energy, and Equilibrium
24. The Third Law of Thermodynamics is:
A) the entropy of the universe is increasing.
B) the entropy of the universe is constant.
C) the entropy at T = 0 K is zero.
D) the energy of the universe is constant.
E) the entropy of all elements is zero.
Ans: C Bloom's Taxonomy: 1 Difficulty: easy
Page 347
Chapter 18 Entropy, Free Energy, and Equilibrium
25. What term is given to the fact that the entropy of a perfect crystalline solid is zero at
absolute zero?
A) First Law of Thermodynamics
D) crystalline lattice theory
B) Second Law of Thermodynamics
E) absolute crystallinity
C) Third Law of Thermodynamics
Ans: C Bloom's Taxonomy: 1 Difficulty: easy
Page 348
Chapter 18 Entropy, Free Energy, and Equilibrium
26. Determine S for the reaction SO3(g) + H2O(l) H2SO4(l).
S(J/Kmol)
SO3
256.2
H2 O
69.9
H2SO4
156.9
A) 169.2 J/Kmol
D) 29.4 J/Kmol
B) 1343.2 J/Kmol
E) 29.4 J/Kmol
C) 169.2 J/Kmol
Ans: C Bloom's Taxonomy: 3 Difficulty: easy
Page 349
Chapter 18 Entropy, Free Energy, and Equilibrium
27. Calculate S for the reaction SO2(s) + NO2(g) SO3(g) + NO(g).
S(J/Kmol)
SO2(g)
248.5
SO3(g)
256.2
NO(g)
210.6
NO2(g)
240.5
A) 53.6 J/Kmol
D) 474.8 J/Kmol
B) 53.6 J/Kmol
E) 474.8 J/Kmol
C) 22.2 J/Kmol
Ans: C Bloom's Taxonomy: 3 Difficulty: easy
Page 350
Chapter 18 Entropy, Free Energy, and Equilibrium
28. Calculate S at 25C for the reduction of PbO(s), 2PbO(s) + C(s) 2Pb(s) + CO2(g)
given these absolute entropies:
S(J/Kmol)
PbO(s)
69.45
C(s)
5.7
Pb(s)
64.89
CO2(g)
213.6
A) +198.8 J/Kmol
D) 203.3 J/Kmol
B) +488.0 J/Kmol
E) +203.3 J/Kmol
C) +353.6 J/Kmol
Ans: A Bloom's Taxonomy: 3 Difficulty: moderate
Page 351
Chapter 18 Entropy, Free Energy, and Equilibrium
29. Calculate S for the reaction
SiCl4(g) + 2Mg(s) 2MgCl2(s) + Si(s).
Substance:
SiCl4(g)
Mg(s) MgCl2(s)
S(J/Kmol):
330.73
32.68
89.62
A) 254.96 J/K
D) 254.96 J/K
B) 198.02 J/K
E) 471.86 J/K
C) 198.02 J/K
Ans: B Bloom's Taxonomy: 3 Difficulty: moderate
Page 352
Si(s)
18.83
Chapter 18 Entropy, Free Energy, and Equilibrium
30. Calculate S for the reaction
4Cr(s) + 3O2(g) 2Cr2O3(s).
Substance:
Cr(s)
O2(g)
Cr2O3(s)
S(J/Kmol):
23.77
205.138
81.2
A) 548.1 J/K B) 147.7 J/K C) 147.7 J/K D) 310.1 J/K E) 548.1 J/K
Ans: A Bloom's Taxonomy: 3 Difficulty: moderate
Page 353
Chapter 18 Entropy, Free Energy, and Equilibrium
31. Calculate So for the reaction
Fe2O3(s) + 3CO(g) = 3CO2(g) + 2Fe(s)
using the data below, T = 298.15 K.
Gfo (kJ/mole)
H fo (kJ/mole)
Fe2O3(s)
742.2
824.2
CO(g)
137.2
110.5
CO2(g)
394.4
393.5
A) 15.4 J/K B) 15.4 J/K C) 181.9 J/K D) 181.9 J/K E) 5.1 J/K
Ans: A Bloom's Taxonomy: 3 Difficulty: difficult
Page 354
Chapter 18 Entropy, Free Energy, and Equilibrium
32. Calculate S for the reaction
2Cl2(g) + SO2(g) SOCl2(g) + Cl2O(g).
Substance:
Cl2(g)
SO2(g)
SOCl2(g) Cl2O(g)
S(J/Kmol):
223.0
248.1
309.77
266.1
A) 118.2 J/K B) 104.8 J/K C) 104.8 J/K D) 118.2 J/K E) 1270.0 J/K
Ans: A Bloom's Taxonomy: 3 Difficulty: moderate
Page 355
Chapter 18 Entropy, Free Energy, and Equilibrium
33. Calculate S for the combustion of propane.
C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g)
Substance:
C3H8(g)
O2(g)
CO2(g)
H2O(g)
S(J/Kmol):
269.9
205.138
213.74
188.825
A) 100.9 J/K B) 72.5 J/K C) 72.5 J/K D) 100.9 J/K E) 877.5 J/K
Ans: D Bloom's Taxonomy: 3 Difficulty: moderate
Page 356
Chapter 18 Entropy, Free Energy, and Equilibrium
34. Calculate the entropy change for vaporization of 18 g of H2O. The molar enthalpy of
vaporization for water is 44 kJ/mol and the boiling point is 100oC.
A) 44 kJ/mol B) 440 J/K C) 117.9 J/K D) 41 kJ/K E) 440 kJ/K
Ans: A Bloom's Taxonomy: 3 Difficulty: difficult
Page 357
Chapter 18 Entropy, Free Energy, and Equilibrium
35. Elemental boron can be formed by reaction of boron trichloride with hydrogen.
BCl3(g) + 1.5H2(g) B(s) + 3HCl(g)
Substance:
BCl3(g)
H2(g)
B(s)
HCl(g)
S(J/Kmol):
?
130.6
5.87
186.8
If S = 80.3 J/K, what is S for BCl3(g)?
A) 18.2 J/Kmol
D) 355.4 J/Kmol
B) 18.2 J/Kmol
E) 450.6 J/Kmol
C) 290.1 J/Kmol
Ans: C Bloom's Taxonomy: 3 Difficulty: difficult
Page 358
Chapter 18 Entropy, Free Energy, and Equilibrium
36. The formula for G is
A) G = H TS.
B) G = H + TS.
C) G = E TS
Ans: A Bloom's Taxonomy: 1
D)
E)
G = TS.
G = q/T.
Difficulty: easy
Page 359
Chapter 18 Entropy, Free Energy, and Equilibrium
37. A negative sign for G indicates that, at constant T and P,
A) the reaction is exothermic.
D) the reaction is spontaneous.
B) the reaction is endothermic.
E) S must be > 0.
C) the reaction is fast.
Ans: D Bloom's Taxonomy: 2 Difficulty: moderate
Page 360
Chapter 18 Entropy, Free Energy, and Equilibrium
38. The themodynamical condition for a spontaneous process at constant T and P is
A) S > 0. B) S < 0. C) G < 0. D) G > 0. E) G = 0.
Ans: C Bloom's Taxonomy: 2 Difficulty: moderate
Page 361
Chapter 18 Entropy, Free Energy, and Equilibrium
39. Ozone (O3) in the atmosphere can reaction with nitric oxide (NO):
O3(g) + NO(g) NO2(g) + O2(g).
Calculate the G for this reaction at 25C. (H = 199 kJ/mol, S = 4.1 J/Kmol).
A) 1020 kJ/mol
D) 1.42 103 kJ/mol
3
B) 1.22 10 kJ/mol
E) 198 kJ/mol
C) 2.00 103 kJ/mol
Ans: E Bloom's Taxonomy: 3 Difficulty: easy
Page 362
Chapter 18 Entropy, Free Energy, and Equilibrium
40. Sodium carbonate can be made by heating sodium bicarbonate:
2NaHCO3(s) Na2CO3(s) + CO2(g) + H2O(g).
Given that H = 128.9 kJ/mol and G = 33.1 kJ/mol at 25C, above what minimum
temperature will the reaction become spontaneous under standard state conditions?
A) 0.4 K B) 3.9 K C) 321 K D) 401 K E) 525 K
Ans: D Bloom's Taxonomy: 3 Difficulty: moderate
Page 363
Chapter 18 Entropy, Free Energy, and Equilibrium
41. For a chemical reaction to be spontaneous only at high temperatures, which of the
following conditions must be met?
A) S > 0, H > 0
D) S < 0, H > 0
B) S > 0, H < 0
E) G > 0
C) S < 0, H < 0
Ans: A Bloom's Taxonomy: 2 Difficulty: difficult
Page 364
Chapter 18 Entropy, Free Energy, and Equilibrium
42. For a chemical reaction to be spontaneous at all temperatures, which of the following
conditions must be met?
A) S > 0, H > 0
B) S > 0, H < 0
C) S < 0, H < 0
D) S < 0, H > 0
E) It is not possible for a reaction to be spontaneous at all temperatures.
Ans: B Bloom's Taxonomy: 2 Difficulty: difficult
Page 365
Chapter 18 Entropy, Free Energy, and Equilibrium
43. Choose the right combination.
A) G < 0; Q > K
B) G < 0; Q < K
C) G <0; Q = K
Ans: B Bloom's Taxonomy: 2
D)
E)
Go < 0; Q = K
Go < 0; Q < K
Difficulty: difficult
Page 366
Chapter 18 Entropy, Free Energy, and Equilibrium
44. Calculate Go for the reaction
2NO(g) +Cl2(g) 2NOCl(g)
using the following Gfo (in kJ/mol): NOCl(g) 66.35, NO(g) 86.55.
A) 40.4 kJ/mol
D) 20.2 kJ/mol
B) 40.4 kJ/mol
E) 152.9 kJ/mol
C) 20.2 kJ/mol
Ans: A Bloom's Taxonomy: 3 Difficulty: moderate
Page 367
Chapter 18 Entropy, Free Energy, and Equilibrium
45. Use the given data at 298 K to calculate G for the reaction
2Cl2(g) + SO2(g) SOCl2(g) + Cl2O(g).
Substance:
Cl2(g)
SO2(g)
SOCl2(g)
Cl2O(g)
o (kJ/mol):
0
296.8
212.5
80.3
H f
S(J/Kmol):
223.0
248.1
309.77
266.1
A) 129.3 kJ B) 133.6 kJ C) 196.0 kJ D) 199.8 kJ E) 229.6 kJ
Ans: D Bloom's Taxonomy: 3 Difficulty: difficult
Page 368
Chapter 18 Entropy, Free Energy, and Equilibrium
46. Hydrogen sulfide decomposes according to the following reaction
2H2S(g) 2H2(g) + S2(g).
For this reaction at 298 K, S = 78.1 J/K, H = 169.4 kJ, and G = 146.1 kJ. What is
the value of G at 900 K?
A) 69,881 kJ B) 48.4 kJ C) 99.1 kJ D) 240 kJ E) 441 kJ
Ans: C Bloom's Taxonomy: 3 Difficulty: moderate
Page 369
Chapter 18 Entropy, Free Energy, and Equilibrium
47. Which of the following has G o = 0 at 25oC?
f
A) H2O(l) B) H2O(g) C) Na(s) D) O3(g) E) O(g)
Ans: C Bloom's Taxonomy: 2 Difficulty: easy
Page 370
Chapter 18 Entropy, Free Energy, and Equilibrium
48. Nitric oxide reacts with chlorine to form NOCl. The data refer to 298 K.
2NO(g) + Cl2(g) 2NOCl(g)
Substance:
NO(g)
Cl2(g)
NOCl(g)
90.29
0
51.71
H fo (kJ/mol):
86.60
0
66.07
G o (kJ/mol):
f
S(J/Kmol):
210.65
223.0
261.6
What is the value of G for this reaction at 550 K?
A) 143.76 kJ B) 78.78 kJ C) 22.24 kJ D) 10.56 kJ
Ans: D Bloom's Taxonomy: 3 Difficulty: difficult
Page 371
E) 66,600 kJ
Chapter 18 Entropy, Free Energy, and Equilibrium
49. Sulfuryl dichloride is formed when sulfur dioxide reacts with chlorine. The data refer to
298 K.
SO2(g) + Cl2(g) SO2Cl2(g)
Substance:
SO2(g)
Cl2(g)
o (kJ/mol):
296.8
0
H f
300.1
0
G o (kJ/mol):
f
S(J/Kmol):
248.2
223.0
SO2Cl2(g)
364.0
320.0
311.9
What is the value of G for this reaction at 600 K?
A) 162.8 kJ B) 40.1 kJ C) 28.4 kJ D) 28.4 kJ E) 162.8 kJ
Ans: D Bloom's Taxonomy: 3 Difficulty: difficult
Page 372
Chapter 18 Entropy, Free Energy, and Equilibrium
50. Calculate G for the reaction
SiCl4(g) + 2Mg(s) 2MgCl2(s) + Si(s).
Substance:
SiCl4(g)
Mg(s)
MgCl2(s)
Si(s)
o (kJ/mol):
16.98
0
591.79
0
Gf
A) 566.60 kJ B) 50.38 kJ C) 25.19 kJ D) 25.19 kJ E) 566.60 kJ
Ans: E Bloom's Taxonomy: 3 Difficulty: difficult
Page 373
Chapter 18 Entropy, Free Energy, and Equilibrium
51. Calculate G for the reaction 3NO2(g) + H2O(l) 2HNO3(l) + NO(g).
Gfo (kJ/mol)
H2O(l)
237.2
HNO3(l)
79.9
NO(g)
86.7
NO2(g)
51.8
A) 8.7 kJ/mol
D) 192 kJ/mol
B) 192 kJ/mol
E) 155 kJ/mol
C) 414 kJ/mol
Ans: A Bloom's Taxonomy: 3 Difficulty: moderate
Page 374
Chapter 18 Entropy, Free Energy, and Equilibrium
52. Calculate G for the combustion of propane.
C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g)
Substance:
C3H8(g)
O2(g)
CO2(g)
H2O(g)
o (kJ/mol):
24.5
0
394.4
228.6
Gf
A) 2073.1 kJ B) 1387.3 kJ C) 598.5 kJ D) 598.5 kJ E) 2073.1 kJ
Ans: A Bloom's Taxonomy: 3 Difficulty: moderate
Page 375
Chapter 18 Entropy, Free Energy, and Equilibrium
53. Elemental boron can be formed by reaction of boron trichloride with hydrogen.
BCl3(g) + 1.5H2(g) B(s) + 3HCl(g)
Calculate G for the reaction.
Substance:
BCI3(g)
H2(g)
B(s)
HCl(g)
388.7
0
0
95.3
Gfo (kJ/mol):
A) 293.4 kJ
D) 102.8 kJ
B) 293.4 kJ
E) None of these choices is correct.
C) 102.8 kJ
Ans: D Bloom's Taxonomy: 3 Difficulty: moderate
Page 376
Chapter 18 Entropy, Free Energy, and Equilibrium
54. Calculate G for the combustion of ethanol vapor, C2H5OH(g), at 750C in oxygen to
form carbon dioxide and water vapor. The following data is valid at 25C:
Gfo (kJ/mol)
H fo (kJ/mol)
C2H5OH(g)
234.8
167.9
O2(g)
0
0
H2O(g)
241.8
228.6
CO2(g)
393.5
394.4
A) 1407 kJ/mol
D) 4486 kJ/mol
B) 2151 kJ/mol
E) 1377 kJ/mol
C) 1307 kJ/mol
Ans: E Bloom's Taxonomy: 3 Difficulty: difficult
Page 377
Chapter 18 Entropy, Free Energy, and Equilibrium
55. Calculate G for the reaction of ammonia with fluorine.
A)
B)
C)
Ans:
2NH3(g) + 5F2(g) N2F4(g) + 6HF(g)
Substance:
NH3(g)
F2(g)
N2F4(g)
HF(g)
o (kJ/mol):
16.4
0
79.9
275.4
Gf
D) 1539.7 kJ
E) None of these choices is correct.
179.1 kJ
179.1 kJ
1539.7 kJ
D Bloom's Taxonomy: 3
Difficulty: moderate
Page 378
Chapter 18 Entropy, Free Energy, and Equilibrium
56. Consider the reaction: 2A(g) + B(g) = 2C(g). If Go = 50kJ/mol at T = 25oC and PA =
PB = 1 atm and PC = 2 atm, G is
A) 50 kJ/mol.
D) 53.5 kJ/mol.
B) 49.7 kJ/mol.
E) 49.7 kJ/mol.
C) 46.5 kJ/mol.
Ans: D Bloom's Taxonomy: 3 Difficulty: moderate
Page 379
Chapter 18 Entropy, Free Energy, and Equilibrium
57. The temperature at which the following process reaches equilibrium at 1.0 atm is the
normal boiling point of hydrogen peroxide. (R = 8.314 JK-1mol-1)
H2O2(l)
H2O2(g)
Use the following thermodynamic information at 298 K to determine this temperature.
Substance:
H2O2(l)
H2O2(g)
o (kJ/mol):
187.7
136.3
H f
120.4
105.6
G o (kJ/mol):
f
S(J/Kmol):
109.6
232.7
A) 120C B) 144C C) 196C D) 418C E) 585C
Ans: B Bloom's Taxonomy: 3 Difficulty: difficult
Page 380
Chapter 18 Entropy, Free Energy, and Equilibrium
58. The temperature at which the following process reaches equilibrium at 1.0 atm is the
normal melting point for phosphoric acid.
H3PO4(s)
H3PO4(l)
Use the following thermodynamic data at 298 K to determine this temperature.
Substance:
H fo (kJ/mol):
H3PO4(s)
1284.4
H3PO4(l)
1271.7
1124.3
1123.6
Gfo (kJ/mol):
S(J/Kmol):
110.5
150.8
A) 286 K B) 305 K C) 315 K D) 347 K E) 3170 K
Ans: C Bloom's Taxonomy: 3 Difficulty: difficult
Page 381
Chapter 18 Entropy, Free Energy, and Equilibrium
59. HI has a normal boiling point of 35.4C, and its Hvap is 21.16 kJ/mol. Calculate the
molar entropy of vaporization (Svap).
A) 598 J/Kmol
D) 0.068 J/Kmol
B) 68.6 J/Kmol
E) 89.0 J/Kmol
C) 75.2 J/Kmol
Ans: E Bloom's Taxonomy: 3 Difficulty: moderate
Page 382
Chapter 18 Entropy, Free Energy, and Equilibrium
60. The element oxygen was prepared by Joseph Priestley in 1774 by heating mercury (II)
oxide: HgO(s) Hg(l) + (1/2)O2(g), H = 90.84 kJ/mol. Estimate the temperature at
which this reaction will become spontaneous under standard state conditions.
S(Hg) = 76.02 J/Kmol
S(O2) = 205.0 J/Kmol
S(HgO) = 70.29 J/Kmol
A) 108 K B) 430 K C) 620 K D) 775 K E) 840 K
Ans: E Bloom's Taxonomy: 3 Difficulty: difficult
Page 383
Chapter 18 Entropy, Free Energy, and Equilibrium
61. The reaction of methane with water to form carbon dioxide and hydrogen is
nonspontaneous at 298 K. At what temperature will this system make the transition from
nonspontaneous to spontaneous? The data refer to 298 K.
CH4(g) + 2H2O(g)
Substance:
H fo (kJ/mol):
CH4(g)
74.87
CO2(g) + 4H2(g)
H2O(g)
241.8
CO2(g)
393.5
50.81
228.6
394.4
Gfo (kJ/mol):
S(J/Kmol):
186.1
188.8
213.7
A) 658 K B) 683 K C) 955 K D) 1047 K E) 1229 K
Ans: C Bloom's Taxonomy: 3 Difficulty: difficult
Page 384
H2(g)
0
0
130.7
Chapter 18 Entropy, Free Energy, and Equilibrium
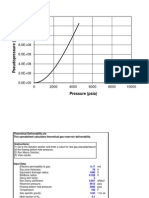
62. Consider the figure below which shows G for a chemical process plotted against
absolute temperature. Which one of the following is an incorrect conclusion, based on
the information in the diagram?
A)
B)
C)
D)
E)
Ans:
H > 0
S > 0
The reaction is spontaneous at high temperatures.
S increases with temperature while H remains constant.
There exists a certain temperature at which H = TS.
D Bloom's Taxonomy: 2 Difficulty: difficult
Page 385
Chapter 18 Entropy, Free Energy, and Equilibrium
63. Consider the figure below which shows G for a chemical process plotted against
absolute temperature. From this plot, it is reasonable to conclude that
A)
B)
C)
Ans:
H > 0, S > 0.
H > 0, S < 0.
H < 0, S > 0.
A Bloom's Taxonomy: 2
D)
E)
H < 0, S < 0.
None of these choices is correct.
Difficulty: difficult
Page 386
Chapter 18 Entropy, Free Energy, and Equilibrium
64. Consider the figure below which shows G for a chemical process plotted against
absolute temperature. From this plot, it is reasonable to conclude that
A)
B)
C)
Ans:
H > 0, S > 0.
H > 0, S < 0.
H < 0, S > 0.
B Bloom's Taxonomy: 2
D)
E)
H < 0, S < 0.
None of these choices is correct.
Difficulty: difficult
Page 387
Chapter 18 Entropy, Free Energy, and Equilibrium
65. Choose the correct equation.
A) G = RTlnK
B) G = RTlnK
C) Go = RTlnK
Ans: C Bloom's Taxonomy: 1
D)
E)
Go = RTlnQ
Go = RTlnQ
Difficulty: easy
Page 388
Chapter 18 Entropy, Free Energy, and Equilibrium
66. Which equation is correct?
A) G = Go RTlogKeq
B) G = Go + RTlnQ
C) G = RTlnQ
Ans: B Bloom's Taxonomy: 1
D)
E)
G = RTlogQ
G = RTlogKeq
Difficulty: easy
Page 389
Chapter 18 Entropy, Free Energy, and Equilibrium
67. Which equation is correct?
A) G = Go RTlogKeq
B) Go = RTlnK
C) G = RTlnQ
Ans: B Bloom's Taxonomy: 1
D)
E)
G = RTlogQ
Go = RTlogKeq
Difficulty: easy
Page 390
Chapter 18 Entropy, Free Energy, and Equilibrium
68. Which statement is correct?
A) If K < 1, lnK is negative, and Go is negative then the reaction is product favored.
B) If K > 1, lnK is negative, and Go is positive then the reaction is product favored.
C) If K > 1, lnK is positive, and Go is negative then the reaction is product favored.
D) If K > 1, lnK is negative, and Go is negative then the reaction is reactant favored.
E) If K < 1, lnK is positive, and Go is positive then the reaction is reactant favored.
Ans: C Bloom's Taxonomy: 2 Difficulty: difficult
Page 391
Chapter 18 Entropy, Free Energy, and Equilibrium
69. Which statement is correct?
A) when Q < K then G = 1
B) when Q < K then G = S
C) when Q = K then G = 0
Ans: C Bloom's Taxonomy: 1
D)
E)
when Q > K then G = 1
when Q > K then G = RT
Difficulty: easy
Page 392
Chapter 18 Entropy, Free Energy, and Equilibrium
70. What is the free energy change, G, for the equilibrium between hydrogen iodide,
hydrogen, and iodine at 453C? Kc = 0.020
2HI(g) H2(g) + I2(g)
A) 6.4 kJ B) 8.8 kJ C) 15 kJ D) 19 kJ E) 24 kJ
Ans: E Bloom's Taxonomy: 3 Difficulty: moderate
Page 393
Chapter 18 Entropy, Free Energy, and Equilibrium
71. Hydrogen peroxide (H2O2) decomposes according to the equation:
H2O2(l) H2O(l) + (1/2)O2(g).
Calculate Kp for this reaction at 25C. (H = 98.2 kJ/mol, S = 70.1 J/Kmol, R =
8.314 JK-1mol-1)
A) 1.3 1021 B) 20.9 C) 3.46 1017 D) 7.4 1020 E) 8.6 104
Ans: D Bloom's Taxonomy: 3 Difficulty: moderate
Page 394
Chapter 18 Entropy, Free Energy, and Equilibrium
72. At 1500C the equilibrium constant for the reaction CO(g) + 2H2(g)
the value Kp = 1.4 107. Calculate G for this reaction at 1500C.
A) 105 kJ/mol
D) 105 kJ/mol
B) 1.07 kJ/mol
E) 233 kJ/mol
C) 233 kJ/mol
Ans: E Bloom's Taxonomy: 3 Difficulty: moderate
Page 395
CH3OH(g) has
Chapter 18 Entropy, Free Energy, and Equilibrium
73. Calculate Kp at 298 K for the reaction SO2(g) + NO2(g) SO3(g) + NO(g). (R = 8.314
JK-1mol-1)
Gfo
SO2(g)
300.4 kJ/mol
SO3(g)
370.4 kJ/mol
NO(g)
86.7 kJ/mol
NO2(g)
51.8 kJ/mol
A) 6.99 107 B) 5.71 108 C) 14.2 D) 475 E) 1.42 106
Ans: E Bloom's Taxonomy: 3 Difficulty: difficult
Page 396
Chapter 18 Entropy, Free Energy, and Equilibrium
74. The equilibrium constant at 427C for the reaction N2(g) + 3H2(g) 2NH3(g) is Kp =
9.4 105. Calculate the value of G for the reaction under these conditions.
A) 33 kJ/mol
B) 54 kJ/mol
C) 54 kJ/mol
D) 33 kJ/mol
E) 1.3 J/mol
Ans: C Bloom's Taxonomy: 3 Difficulty: moderate
Page 397
Chapter 18 Entropy, Free Energy, and Equilibrium
75. Nitrosyl chloride (NOCl) decomposes at elevated temperatures according to the
equation 2NOCl(g) 2NO(g) + Cl2(g). Calculate Kp for this reaction at 227C. (For
this reaction H = 81.2 kJ/mol, S = 128 J/Kmol.)
A) 1.59 102 B) 2.10 107 C) 62.8 D) 4.90 106 E) 3.20 109
Ans: A Bloom's Taxonomy: 3 Difficulty: moderate
Page 398
Chapter 18 Entropy, Free Energy, and Equilibrium
76. The equilibrium constant for the reaction AgBr(s) Ag+(aq) + Br(aq) is the
solubility product constant, Ksp = 7.7 1013 at 25C. Calculate G for the reaction
when [Ag+] = 1.0 102 M and [Br] = 1.0 103 M. Is the reaction spontaneous or
nonspontaneous at these concentrations (R = 8.314 JK-1mol-1)?
A) G = 69.1 kJ/mol, nonspontaneous
B) G = 69.1 kJ/mol, spontaneous
C) G = 97.5 kJ/mol, spontaneous
D) G = 40.6 kJ/mol, nonspontaneous
E) G = 97.5 kJ/mol, nonspontaneous
Ans: D Bloom's Taxonomy: 3 Difficulty: difficult
Page 399
Chapter 18 Entropy, Free Energy, and Equilibrium
77. The solubility product constant at 25C for AgI(s) in water has the value 8.3 1017.
Calculate Grxn at 25C for the process AgI(s) Ag+(aq) + I(aq) where [Ag+] = 9.1
109 and [I] = 9.1 109.
A) +4.4 kJ/mol
D) 91.7 kJ/mol
B) +91.7 kJ/mol
E) 4.4 kJ/mol
C) 0.0 kJ/mol
Ans: C Bloom's Taxonomy: 3 Difficulty: difficult
Page 400
Chapter 18 Entropy, Free Energy, and Equilibrium
78. A sample of solid naphthalene is introduced into an evacuated flask. Use the data below
to calculate the equilibrium vapor pressure of naphthalene (C10H8) in the flask at 35C.
H fo (25C)
Gfo (25C)
C10H8(s)
78.5 kJ/mol
201.6 kJ/mol
C10H8(g)
150.6 kJ/mol
224.1 kJ/mol
A) 890. mmHg
D) 0.086 mmHg
B) 0.22 mmHg
E) 833 mmHg
C) 696 mmHg
Ans: B Bloom's Taxonomy: 3 Difficulty: difficult
Page 401
Chapter 18 Entropy, Free Energy, and Equilibrium
79. The standard free energy of formation of gaseous hydrogen iodide is 1.30 kJ/mol at
25C. Find Kp for the reaction H2(g) + I2(s) 2HI(g) at this temperature.
A) 7.0 B) 7100 C) 1.0 D) 2.4 E) 0.35
Ans: E Bloom's Taxonomy: 3 Difficulty: moderate
Page 402
Chapter 18 Entropy, Free Energy, and Equilibrium
80. Find the temperature at which Kp = 4.00 for the reaction N2O4(g) 2NO2(g). [Given:
at 25C, for NO2(g), H fo = 33.85 kJ/mol, S = 240.46 J/molK; for N2O4(g), H fo =
9.66 kJ/mol, S = 304.3 J/molK; assume that H and S are independent of
temperature.]
A) 197C B) 56C C) 36C D) 79C E) 476C
Ans: D Bloom's Taxonomy: 3 Difficulty: difficult
Page 403
Chapter 18 Entropy, Free Energy, and Equilibrium
81. Find the temperature at which Kp = 42.0 for the reaction H2(g) + I2(g) 2HI(g).
[Given: at 25C, for H2(g), H fo = 0, S = 131.0 J/molK; for I2(g), H fo = 62.26 kJ/mol,
S = 260.6 J/molK; for HI(g), H fo = 25.9 kJ/mol, S = 206.3 J/molK; assume that
H and S are independent of temperature.]
A) 1040 K B) 168 K C) 539 K D) 1400 K E) 34,200 K
Ans: A Bloom's Taxonomy: 3 Difficulty: difficult
Page 404
Chapter 18 Entropy, Free Energy, and Equilibrium
82. For the reaction HCONH2(g) NH3(g) + CO(g), Kc = 4.84 at 400 K. If H for this
reaction is 29 kJ/mol, find Kc at 500 K.
A) 5.8 B) 0.17 C) 27.5 D) 0.88 E) 10.3
Ans: C Bloom's Taxonomy: 3 Difficulty: moderate
Page 405
Chapter 18 Entropy, Free Energy, and Equilibrium
83. In the gas phase, formic acid forms a dimer, 2HCOOH(g) (HCOOH)2(g). For this
reaction, H = 60.1 kJ/mol and G = 13.9 kJ/mol at 25C. Find the equilibrium
constant (Kp) for this reaction at 75C.
A) 8960 B) 273 C) 0.120 D) 8.33 E) 1.12 104
Ans: D Bloom's Taxonomy: 3 Difficulty: moderate
Page 406
Chapter 18 Entropy, Free Energy, and Equilibrium
84. In the gas phase, methyl isocyanate (CH3NC) isomerizes to acetonitrile (CH3CN),
H3CNC(g) H3CCN(g)
with H = 89.5 kJ/mol and G = 73.8 kJ/mol at 25C. Find the equilibrium
constant for this reaction at 100C.
A) 1.68 1010
D) 4.63 1011
9
B) 5.96 10
E) 8.64 1012
C) 2.16 1010
Ans: B Bloom's Taxonomy: 3 Difficulty: moderate
Page 407
Chapter 18 Entropy, Free Energy, and Equilibrium
85. Iron (III) oxide can be reduced by carbon monoxide.
Fe2O3(s) + 3CO(g)
2Fe(s) + 3CO2(g)
Use the following thermodynamic data at 298 K to determine the equilibrium constant
at this temperature.
Substance:
Fe2O3(s)
CO(g)
Fe(s)
CO2(g)
o (kJ/mol):
824.2
110.5
0
393.5
H f
742.2
137.2
0
394.4
G o (kJ/mol):
f
S(J/Kmol):
87.4
197.7
6
3
4
A) 7.0 10
B) 1.3 10
C) 2.2 10 D) 1.4 105
Ans: D Bloom's Taxonomy: 3 Difficulty: difficult
Page 408
27.78
213.7
E) > 2.0 105
Chapter 18 Entropy, Free Energy, and Equilibrium
86. Calculate the equilibrium constant at 25C for the reaction of methane with water to
form carbon dioxide and hydrogen. The data refer to 25C.
CH4(g) + 2H2O(g)
Substance:
H fo (kJ/mol):
CH4(g)
74.87
CO2(g) + 4H2(g)
H2O(g)
241.8
CO2(g)
393.5
50.81
228.6
394.4
Gfo (kJ/mol):
S(J/Kmol):
186.1
188.8
213.7
A) 8.2 1019 B) 0.96 C) 0.58 D) 1.2 1020 E) 1.4 1046
Ans: D Bloom's Taxonomy: 3 Difficulty: difficult
Page 409
H2(g)
0
0
130.7
Chapter 18 Entropy, Free Energy, and Equilibrium
87. Use the thermodynamic data at 298 K below to determine the Ksp for barium carbonate,
BaCO3 at this temperature.
Substance:
Ba2+(aq)
CO32 (aq)
676.26
BaCO3(s)
538.36
1219
H fo (kJ/mol):
560.7
528.1
1139
Gfo (kJ/mol):
S(J/Kmol):
13
-53.1
112
A) 5.86 B) 6.30 108 C) 1.59 10-9 D) 5.47 10-21 E) 2.18 10-27
Ans: C Bloom's Taxonomy: 3 Difficulty: difficult
Page 410
Chapter 18 Entropy, Free Energy, and Equilibrium
88. The formation constant for the reaction
Ag+(aq) + 2NH3(aq)
Ag(NH3) +2 (aq)
is Kf = 1.7 107 at 25C. What is G at this temperature?
A) 1.5 kJ B) 3.5 kJ C) 18 kJ D) 23 kJ E) 41 kJ
Ans: E Bloom's Taxonomy: 3 Difficulty: moderate
Page 411
Chapter 18 Entropy, Free Energy, and Equilibrium
89. Which statement is correct?
A) Oxygen is formed during the metabolism of food.
B) The metabolism process results in a positive Go.
C) ADP is hydrolyzed to ATP and phosphoric acid in the body.
D) A protein is a polymer made of carbohydrate units.
E) ATP functions to store energy until it is needed by the body.
Ans: E Bloom's Taxonomy: 1 Difficulty: moderate
Page 412
Chapter 18 Entropy, Free Energy, and Equilibrium
90. Which statement is correct?
A) Reaction of ADP and alanine results in ATP in the body.
B) A positive Go value means that the reaction is not product favored.
C) CO2 is released when ADP reacts with an amino acid.
D) Phosphoric acid is one of the products of the reaction of ADP with alanine.
E) Hydrolysis of ATP requires NaOH.
Ans: B Bloom's Taxonomy: 1 Difficulty: moderate
Page 413
Chapter 18 Entropy, Free Energy, and Equilibrium
91. True or False: The following reaction is spontaneous under standard state conditions
at 25C:
AgCl(s) Ag+(aq) + Cl(aq) (G = 55 kJ/mol)
Ans: False Bloom's Taxonomy: 2 Difficulty: easy
Page 414
Chapter 18 Entropy, Free Energy, and Equilibrium
92. True or False: Suniv = 1 for a spontaneous reaction.
Ans: False Bloom's Taxonomy: 1 Difficulty: easy
Page 415
Chapter 18 Entropy, Free Energy, and Equilibrium
93. True or False: The higher the pressure of a gas sample, the greater is its entropy.
Ans: False Bloom's Taxonomy: 2 Difficulty: moderate
Page 416
Chapter 18 Entropy, Free Energy, and Equilibrium
94. True or False: In a spontaneous process, the entropy of the system always increases.
Ans: False Bloom's Taxonomy: 2 Difficulty: moderate
Page 417
Chapter 18 Entropy, Free Energy, and Equilibrium
95. True or False: The entropy of vaporization of a compound is always positive.
Ans: True Bloom's Taxonomy: 2 Difficulty: moderate
Page 418
Chapter 18 Entropy, Free Energy, and Equilibrium
96. True or False: The entropy change S for the reaction NH4Cl(s) NH3(g) + HCl(g)
will be negative.
Ans: False Bloom's Taxonomy: 2 Difficulty: easy
Page 419
Chapter 18 Entropy, Free Energy, and Equilibrium
97. True or False: All elements in their standard state have standard entropies of formation
equal to zero.
Ans: False Bloom's Taxonomy: 1 Difficulty: easy
Page 420
Chapter 18 Entropy, Free Energy, and Equilibrium
98. True or False: For a given reaction, a change in the temperature may result in a change
in the sign of G.
Ans: True Bloom's Taxonomy: 2 Difficulty: moderate
Page 421
Chapter 18 Entropy, Free Energy, and Equilibrium
99. True or False: For any reaction, if G > 0, then K < 1.
Ans: True Bloom's Taxonomy: 2 Difficulty: moderate
Page 422
Chapter 18 Entropy, Free Energy, and Equilibrium
100. True or False: At equilibrium Go = 0.
Ans: False Bloom's Taxonomy: 1 Difficulty: easy
Page 423
Chapter 18 Entropy, Free Energy, and Equilibrium
101. True or False: As a chemical reaction proceeds toward equilibrium, the free energy of
the system decreases at constant temperature and constant pressure.
Ans: True Bloom's Taxonomy: 2 Difficulty: moderate
Page 424
Chapter 18 Entropy, Free Energy, and Equilibrium
102. What is the general condition for a spontaneous process?
Ans: Suniverse > 0
Bloom's Taxonomy: 1 Difficulty: easy
Page 425
Chapter 18 Entropy, Free Energy, and Equilibrium
103. Which thermodynamical property is a measure of the disorder in the system?
Ans: entropy
Bloom's Taxonomy: 1 Difficulty: easy
Page 426
Chapter 18 Entropy, Free Energy, and Equilibrium
104. Predict the sign of S for the reaction O2(g) 2O(g).
Ans: positive
Bloom's Taxonomy: 2 Difficulty: moderate
Page 427
Chapter 18 Entropy, Free Energy, and Equilibrium
105. Is the reaction SiO2(s) + Pb(s) PbO2(s) + Si(s) spontaneous?
Gfo (PbO2) = 217 kJ/mol
Ans: no
Bloom's Taxonomy: 3
Gfo (SiO2) = 856 kJ/mol
Difficulty: difficult
Page 428
Chapter 18 Entropy, Free Energy, and Equilibrium
106. Predict the sign of S for the reaction 6CO2(g) + 6H2O(g) C6H12O6(g) + 6O2(g).
Ans: negative
Bloom's Taxonomy: 3 Difficulty: easy
Page 429
Chapter 18 Entropy, Free Energy, and Equilibrium
107. How does the entropy change when a solid is melted?
Ans: increases
Bloom's Taxonomy: 2 Difficulty: moderate
Page 430
Chapter 18 Entropy, Free Energy, and Equilibrium
108. How does the entropy change when a gas is liquefied?
Ans: decreases
Bloom's Taxonomy: 2 Difficulty: moderate
Page 431
Chapter 18 Entropy, Free Energy, and Equilibrium
109. What is the Third Law of Thermodynamics?
Ans: At 0 K, all perfectly crystalline compounds have S = 0.
Bloom's Taxonomy: 1 Difficulty: easy
Page 432
Chapter 18 Entropy, Free Energy, and Equilibrium
110. What is the relationship between G and Go?
Ans: G = Go RTlnQ
Bloom's Taxonomy: 1 Difficulty: easy
Page 433
Chapter 18 Entropy, Free Energy, and Equilibrium
111. For a process that is spontaneous when TS > H then S is _________.
Ans: less than zero or negative or, < 0
Bloom's Taxonomy: 2 Difficulty: difficult
Page 434
Chapter 18 Entropy, Free Energy, and Equilibrium
112. ______ is the sign of H for a spontaneous process at high temperatures but not at low
temperatures.
Ans: Positive or H > 0
Bloom's Taxonomy: 2 Difficulty: moderate
Page 435
Chapter 18 Entropy, Free Energy, and Equilibrium
113. For a process where a gas condenses to a liquid then the entropy ________.
Ans: decreases
Bloom's Taxonomy: 2 Difficulty: moderate
Page 436
Chapter 18 Entropy, Free Energy, and Equilibrium
114. _________ is the pressure required for standard entropy conditions.
Ans: 1 atm
Bloom's Taxonomy: 1 Difficulty: easy
Page 437
Chapter 18 Entropy, Free Energy, and Equilibrium
115. The _________ Law of Thermodynamics states that the entropy of the universe in a
spontaneous process increases and is constant in equilibrium processes.
Ans: Second
Bloom's Taxonomy: 1 Difficulty: easy
Page 438
Chapter 18 Entropy, Free Energy, and Equilibrium
116. a. Explain what is meant by a spontaneous process.
b. Is a spontaneous process necessarily a rapid one? Explain, and provide a real reaction
as an example to illustrate your answer.
Ans: a. A spontaneous process is one which will occur naturally, given enough time.
b. No, a spontaneous process may be immeasurably slow. An example is the
conversion of diamond to graphite at room temperature and one atmosphere.
Bloom's Taxonomy: 2 Difficulty: moderate
Page 439
Chapter 18 Entropy, Free Energy, and Equilibrium
117. State the Second and Third Laws of Thermodynamics.
Ans: All spontaneous processes are accompanied by an increase in the total entropy of
the universe. The entropy of a perfect crystal at absolute zero, is zero.
Bloom's Taxonomy: 1 Difficulty: easy
Page 440
Chapter 18 Entropy, Free Energy, and Equilibrium
118. For the reaction of xenon and fluorine gases to form solid XeF4, H = 251 kJ and G
= 121 kJ at 25C. Calculate S for the reaction.
Ans: S = 436 J/K
Bloom's Taxonomy: 3 Difficulty: easy
Page 441
Chapter 18 Entropy, Free Energy, and Equilibrium
119. Consider the reaction CO(g) + 2H2(g) CH3OH(l) at 25C.
Gfo (CO) = 137.3 kJ/mol
Gfo (CH3OH) = 166.3 kJ/mol
H fo (CO) = 110.5 kJ/mol
H fo (CH3OH) = 238.7 kJ/mol
S (CO = 197.9 J/Kmol
S (CH3OH) = 126.8 J/Kmol
Calculate value of the equilibrium constant (Kp) for this reaction at 25C.
Ans: 1.20 10 5
Bloom's Taxonomy: 3 Difficulty: difficult
Page 442
Chapter 18 Entropy, Free Energy, and Equilibrium
120. The complete combustion of liquid benzene is represented by the equation:
C6H6(l) + 7 1 2 O2(g) 6CO2(g) + 3H2O(l).
Using the data below, calculate, for this reaction
a. H
b. S
c. G at 25C
Substance:
C6H6(l)
O2(g)
o (kJ/mol):
49
0
H f
S(J/molK):
173
205
Ans: a. H = 3271 kJ
b. S = 217 J/K
c. G = 3206 kJ
Bloom's Taxonomy: 3 Difficulty: difficult
Page 443
CO2(g)
394
H2O(l)
286
214
70
Das könnte Ihnen auch gefallen
- CH 6 PracticeDokument11 SeitenCH 6 PracticeMichel zakhariaNoch keine Bewertungen
- Chemical Equlibria Test-1Dokument4 SeitenChemical Equlibria Test-1newlifelabsNoch keine Bewertungen
- Kinetics Practice Test 1Dokument25 SeitenKinetics Practice Test 1noelNoch keine Bewertungen
- Test BanksDokument21 SeitenTest Banksalex_flutistNoch keine Bewertungen
- Fall 2020 CHEM 112 Exam 3 Practice ProblemsDokument12 SeitenFall 2020 CHEM 112 Exam 3 Practice Problemskimber brownNoch keine Bewertungen
- PLTL Ch. 16 AssignmentDokument6 SeitenPLTL Ch. 16 AssignmentJules BrunoNoch keine Bewertungen
- Ch123 Exam II Practice Exam Spring2011Dokument7 SeitenCh123 Exam II Practice Exam Spring2011christopher92530% (1)
- Third Periodical Examination Chemistry I 2011-2012Dokument9 SeitenThird Periodical Examination Chemistry I 2011-2012Rogelio PontejoNoch keine Bewertungen
- ChemTeam - Assorted Gas Law Problems 26-50Dokument13 SeitenChemTeam - Assorted Gas Law Problems 26-50Koh Jiun AnNoch keine Bewertungen
- Electrochemistry and The Nernst EquationDokument53 SeitenElectrochemistry and The Nernst EquationMaha RajNoch keine Bewertungen
- 102 MSJC 13Dokument11 Seiten102 MSJC 13noelNoch keine Bewertungen
- Mole To Grams, Grams To Moles Conversions WorksheetDokument3 SeitenMole To Grams, Grams To Moles Conversions WorksheetjomonNoch keine Bewertungen
- CM011 Reviewer: Chemistry Combustion and Electrochemistry QuestionsDokument5 SeitenCM011 Reviewer: Chemistry Combustion and Electrochemistry QuestionsSofia Isabelle GarciaNoch keine Bewertungen
- Electrode Potential, Ecell, Nernst Equation and EcsDokument8 SeitenElectrode Potential, Ecell, Nernst Equation and Ecssasanka shawNoch keine Bewertungen
- Cairo University 3rd Year Chemistry ExamDokument3 SeitenCairo University 3rd Year Chemistry ExamKhaled AbeedNoch keine Bewertungen
- Examview - Chemistry Applications Yearly AssessmentDokument25 SeitenExamview - Chemistry Applications Yearly Assessmentapi-232424041Noch keine Bewertungen
- NEET UG Chemistry Solutions PDFDokument31 SeitenNEET UG Chemistry Solutions PDFAlexa Siddhi0% (1)
- Determining The Empirical Formula of Copper ChlorideDokument3 SeitenDetermining The Empirical Formula of Copper Chloridezack123321Noch keine Bewertungen
- 4LE Chem 22Dokument3 Seiten4LE Chem 22Adrian NavarraNoch keine Bewertungen
- Final Exam PracticeDokument17 SeitenFinal Exam PracticeKayla Dollente100% (1)
- Chapter 6 TestDokument5 SeitenChapter 6 TesthelloblargNoch keine Bewertungen
- Chapter 14Dokument20 SeitenChapter 14Angelica UyNoch keine Bewertungen
- Tutorial 2 StudentDokument6 SeitenTutorial 2 StudentIrsyad KamilNoch keine Bewertungen
- Chemistry 2 Answer KeyDokument8 SeitenChemistry 2 Answer KeyMarielle BuesingNoch keine Bewertungen
- Test PaperDokument4 SeitenTest PaperAnonymous doCtd0IJDNNoch keine Bewertungen
- Carboxylic Acids Vapor Pressure Chemistry ExamDokument9 SeitenCarboxylic Acids Vapor Pressure Chemistry ExamAmira AbdallahNoch keine Bewertungen
- ElectrochemDokument75 SeitenElectrochemMhyl HitsuyagamikaelsonNoch keine Bewertungen
- Fundamental Laws of Chemistry QuestionsDokument3 SeitenFundamental Laws of Chemistry QuestionsJsn Jsn100% (1)
- Rates of Reaction TestDokument10 SeitenRates of Reaction TestSaya MenangNoch keine Bewertungen
- Assignment 151Dokument5 SeitenAssignment 151Hai Xuan DoNoch keine Bewertungen
- Thermo ChemistryDokument15 SeitenThermo ChemistrySachin Kumar50% (2)
- CHEM 1406 Practice Exam # 2 (CH 6, 7, 9)Dokument6 SeitenCHEM 1406 Practice Exam # 2 (CH 6, 7, 9)ngterry1Noch keine Bewertungen
- 12 Chemistry Electrochemistry Test 01 Answer 8b9mDokument2 Seiten12 Chemistry Electrochemistry Test 01 Answer 8b9mMohit SahuNoch keine Bewertungen
- Midterms ChemDokument27 SeitenMidterms ChemAndrei Dela CruzNoch keine Bewertungen
- 1.1 Introduction To Physical Chemistry QuestionsDokument2 Seiten1.1 Introduction To Physical Chemistry QuestionsWrl RossNoch keine Bewertungen
- 2022 U.S. NATIONAL Chemistry Olympiad: Local Section Exam Olympiad Examinations Task ForceDokument9 Seiten2022 U.S. NATIONAL Chemistry Olympiad: Local Section Exam Olympiad Examinations Task ForceNajmusawwa Aulia RahmahNoch keine Bewertungen
- Organic Chemistry ACS Sample QuestionsDokument20 SeitenOrganic Chemistry ACS Sample QuestionsNajmusawwa Aulia RahmahNoch keine Bewertungen
- 3LE Chem 22Dokument5 Seiten3LE Chem 22Adrian NavarraNoch keine Bewertungen
- General Chemistry II Practice Problems EntropyDokument7 SeitenGeneral Chemistry II Practice Problems EntropyKalina EllisNoch keine Bewertungen
- Chem 181 Chemistry of GasesDokument15 SeitenChem 181 Chemistry of GasesJoey PooleNoch keine Bewertungen
- Chemical ThermodynamicsDokument65 SeitenChemical ThermodynamicsCacey Daiwey Calixto0% (1)
- Unit 5 Practice Test Multiple ChoiceDokument10 SeitenUnit 5 Practice Test Multiple ChoiceMadhavan Vijay100% (1)
- Quantum Theory and The Electronic Structure of Atoms: Multiple Choice Questions 1Dokument18 SeitenQuantum Theory and The Electronic Structure of Atoms: Multiple Choice Questions 1Khawla MustafaNoch keine Bewertungen
- Physics Republic of The PhilippinesDokument13 SeitenPhysics Republic of The PhilippinesGlenda AstodilloNoch keine Bewertungen
- JCE Online - Conceptual Questions and Challenge Problems - Chemical Concepts Inventory PDFDokument6 SeitenJCE Online - Conceptual Questions and Challenge Problems - Chemical Concepts Inventory PDFMenna GalalNoch keine Bewertungen
- Test2 Ch17a Acid-Base Practice Problems PDFDokument12 SeitenTest2 Ch17a Acid-Base Practice Problems PDFRaphael CastilloNoch keine Bewertungen
- Chemistry QuestionsDokument21 SeitenChemistry QuestionsNahwera EvalyneNoch keine Bewertungen
- R A e R A K X X R: InstructionsDokument2 SeitenR A e R A K X X R: InstructionsAdrian NavarraNoch keine Bewertungen
- Open Stax Chem 2e Answer With Explanation 1Dokument1 SeiteOpen Stax Chem 2e Answer With Explanation 1siewyonglimNoch keine Bewertungen
- Canada Chemistry OlympiadDokument1 SeiteCanada Chemistry OlympiadCorneliaNoch keine Bewertungen
- Quiz 2 - Measurements and DensityDokument4 SeitenQuiz 2 - Measurements and DensityCarolyn CampitaNoch keine Bewertungen
- Bonding Test ReviewDokument5 SeitenBonding Test ReviewRonaldo ManaoatNoch keine Bewertungen
- Ideal Gas Law ProblemsDokument7 SeitenIdeal Gas Law ProblemsJamie Nguyen0% (1)
- Multiple Choice Chemistry QuestionsDokument6 SeitenMultiple Choice Chemistry QuestionsGraceljaneNoch keine Bewertungen
- Savina Thompson - 5 e Lesson PlanDokument6 SeitenSavina Thompson - 5 e Lesson Planapi-377315743Noch keine Bewertungen
- Diagnostic Test in General Chemistry 1Dokument13 SeitenDiagnostic Test in General Chemistry 1Dearest Notes100% (1)
- Chemistry Chemical Reactions CH 18 Samplepractice Exam Questions and AnswersDokument20 SeitenChemistry Chemical Reactions CH 18 Samplepractice Exam Questions and Answersdao hoangNoch keine Bewertungen
- Energetics MCQDokument44 SeitenEnergetics MCQNg Swee Loong Steven100% (8)
- PhychemDokument7 SeitenPhychemChrystylyn VictorioNoch keine Bewertungen
- Ch12 Free Energy and ThermodynamicsDokument8 SeitenCh12 Free Energy and ThermodynamicsCitrus_EscapeNoch keine Bewertungen
- Final Project ReportDokument47 SeitenFinal Project ReportShashank Dubey100% (1)
- Pump Lecture 1Dokument7 SeitenPump Lecture 1juan villarroelNoch keine Bewertungen
- Surface Tension: SynopsisDokument3 SeitenSurface Tension: SynopsisWillSmithNoch keine Bewertungen
- Design of Reinforced Concrete D Regions Using Strut and Tie Models and Nonlinear Finite Element Modelling - Research Groups - Imperial College LondonDokument4 SeitenDesign of Reinforced Concrete D Regions Using Strut and Tie Models and Nonlinear Finite Element Modelling - Research Groups - Imperial College LondonDEYBI ALEJANDRO CHINCHAY POMANoch keine Bewertungen
- Isotropic Hardening Flow Rule for Plastic MaterialsDokument4 SeitenIsotropic Hardening Flow Rule for Plastic Materialsabbasiftikhar1657Noch keine Bewertungen
- Fatigue Analysis of Welded JointsDokument16 SeitenFatigue Analysis of Welded Jointsangelobuijtels100% (1)
- Stress Sample Calculation On Base EllDokument1 SeiteStress Sample Calculation On Base EllLionel TorradoNoch keine Bewertungen
- Apuntes TurbomaquinasDokument5 SeitenApuntes TurbomaquinasTeodoro HernandezNoch keine Bewertungen
- HVAC Terminology Guide Under 40 CharactersDokument2 SeitenHVAC Terminology Guide Under 40 CharactersNaresh SewdinNoch keine Bewertungen
- Problems From Past Board ExamsDokument4 SeitenProblems From Past Board ExamsMarielle Eden Ulanday TamboleroNoch keine Bewertungen
- Cooling Tower Fill EfficiencyDokument11 SeitenCooling Tower Fill EfficiencyMubarik Ali100% (1)
- Fluid Properties and Kinematics GuideDokument1 SeiteFluid Properties and Kinematics Guideshalom_pkl100% (1)
- Hacon TMDokument68 SeitenHacon TMTiago CoutoNoch keine Bewertungen
- Earthquake Analysis and Design of StructuralDokument15 SeitenEarthquake Analysis and Design of StructuralNarayanan Moorthy100% (1)
- Theoretical DeliverabilityDokument197 SeitenTheoretical Deliverabilitymath62210Noch keine Bewertungen
- Lesson 4 - Heat TransferDokument16 SeitenLesson 4 - Heat TransferJoanna Ruth SeproNoch keine Bewertungen
- Prestressing of Compression Springs-Part1Dokument9 SeitenPrestressing of Compression Springs-Part1Prihastya WishnutamaNoch keine Bewertungen
- Design of Cooling TowerDokument17 SeitenDesign of Cooling TowerMohan VarkeyNoch keine Bewertungen
- Gas Dynamics Report ValidationDokument8 SeitenGas Dynamics Report ValidationM.t. HabibiNoch keine Bewertungen
- Transport Phenomena Fluid Mechanics Problem Solution BSL - Radial Flow of A Newtonian Fluid Between Two Parallel DisksDokument4 SeitenTransport Phenomena Fluid Mechanics Problem Solution BSL - Radial Flow of A Newtonian Fluid Between Two Parallel Disksyudhaputra100% (1)
- Experiment 3Dokument15 SeitenExperiment 3Ashwin PurohitNoch keine Bewertungen
- Ramberg Osgood Stress-Strain - WIKIPEDIADokument3 SeitenRamberg Osgood Stress-Strain - WIKIPEDIAalbertoNoch keine Bewertungen
- Route Selection and Deepwater Pipeline DesignDokument19 SeitenRoute Selection and Deepwater Pipeline DesignSampurnanand PandeyNoch keine Bewertungen
- Propeller Design Workshop Part I PDFDokument92 SeitenPropeller Design Workshop Part I PDFYvessNoch keine Bewertungen
- The Design of Vertical Pressure Vessels Subjected To Applied ForcesDokument3 SeitenThe Design of Vertical Pressure Vessels Subjected To Applied ForcesMANOJ M100% (1)
- A10VSO32Dokument72 SeitenA10VSO32王涛Noch keine Bewertungen
- The Predictive Capability of Failure Mode Concept-Based Strength Criteria For Multi-Directional Laminates-Part BDokument30 SeitenThe Predictive Capability of Failure Mode Concept-Based Strength Criteria For Multi-Directional Laminates-Part BChris KořánNoch keine Bewertungen
- Page 1 Detailed Structural Analysis and Design For F1C1Dokument3 SeitenPage 1 Detailed Structural Analysis and Design For F1C1Jholo BuctonNoch keine Bewertungen
- Semester 2 - Unit 1 - Examples - AnnotatedDokument13 SeitenSemester 2 - Unit 1 - Examples - AnnotatedAatif shaikhNoch keine Bewertungen
- Nonlinear Analysis & Performance Based Design FundamentalsDokument86 SeitenNonlinear Analysis & Performance Based Design FundamentalsvinujohnpanickerNoch keine Bewertungen