Beruflich Dokumente
Kultur Dokumente
Mercury in Petroleum
Hochgeladen von
hortalemosOriginalbeschreibung:
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Mercury in Petroleum
Hochgeladen von
hortalemosCopyright:
Verfügbare Formate
Fuel Processing Technology 63 2000.
127
www.elsevier.comrlocaterfuproc
Review
Mercury in petroleum
S. Mark Wilhelm
a
a,)
, Nicolas Bloom
Mercury Technology Serices, 23014 Lutheran Church Rd., Tomball, TX 77375, USA
b
Frontier Geosciences, Seattle, WA, USA
Received 30 September 1999; accepted 30 September 1999
Abstract
The understanding of the chemistry of geologic mercury has evolved due to technical advances
that allow differentiation of the various chemical forms of mercury in hydrocarbon matrices.
Newly developed techniques can measure the concentration of mercury and mercury compounds
in some hydrocarbon matrices to better than 1 part in 10 10. Accurate determinations of total
mercury and speciation of mercury compounds depend on sophisticated sampling techniques and
rigorous analytical procedures. The mercury species that are present in crude oil and gas
condensates include several that are seldom accounted for in routine analytical methodology.
Although only limited amounts of data are presently available, it appears that the distribution of
mercury compounds in petroleum samples varies widely. The amounts and relative distribution of
mercury compounds in liquid hydrocarbons depend on the sample source and history and include
classes of compounds that have specific negative effects on people, equipment and catalysts.
Crude oil and unprocessed gas condensates contain significant amounts of suspended mercury
compounds, mostly mercuric sulfide. The dominant dissolved species in petroleum are elemental
mercury and ionic halides. Ionic mercury compounds have been found in significant proportions in
liquids but it is not known if they are abundant naturally or if they exist due to post-collection
conversion of other mercury species. Detection and quantification of dialkylmercury in liquid
hydrocarbons have been accomplished analytically but only limited data are available concerning
its prevalence in petroleum and processed fuels. The recent understanding of the toxicology of
dialkylmercury identifies this class of compounds as a potential health hazard to those exposed to
gas condensate and oil, but hard evidence of exact concentrations is needed to support the
hypothesis of possible risk. Mercury removal systems that employ sorbents to capture mercury in
feeds to gas and liquid hydrocarbon processes do not work effectively on all chemical forms of
mercury. Suspended colloidal. forms, such as mercuric sulfide, evade capture by sorbent beds
and organic mercury compounds are captured to varying degrees depending upon sorbent
chemistry. Understanding the reaction of chemisorbents with each of the various species that are
Corresponding author. Tel.: q1-281-255-3775; fax: q1-281-357-0721; e-mail: smw@hgtech.com
0378-3820r00r$ - see front matter q 2000 Elsevier Science B.V. All rights reserved.
PII: S 0 3 7 8 - 3 8 2 0 9 9 . 0 0 0 6 8 - 5
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
present in feeds is essential to the design of mercury removal systems. Mercury is universally
detrimental to petroleum processing systems. In gas processing, mercury damages equipment and
fouls cryogenic heat exchangers. In chemical manufacturing and refining, mercury poisons
catalysts and contaminates waste water, thus impacting regulatory compliance. Contamination of
primary gas treatment systems amine and glycol. and accumulation of toxic sludge deposits in
separators generate waste streams that are difficult to dispose of. Maintenance workers in the
petroleum industry can be at risk due to inhalation of mercury vapor and dermal absorption of
organic mercury compounds unless proper precautions are implemented. q 2000 Elsevier Science
B.V. All rights reserved.
Keywords: Mercury; Petroleum; Hydrocarbon
1. Introduction
In the recently submitted US EPA Mercury Report to Congress w1x, combusted
hydrocarbons are identified as major anthropogenic sources of mercury emissions to the
atmosphere in the US. It is estimated that approximately 11 tons 10 Mg. of mercury are
discharged to the atmosphere annually from boilers that burn oil. The 11 tons are
approximately 7% of the US domestic total 158 tons. from all sources. Water
discharges from refineries and petrochemical plants have been identified as sources of
aquatic mercury contamination.
The mercury in combusted fuels originates from petroleum and coal. The concentration of mercury in crude oil and natural gas is highly dependent on geologic location and
varies between approximately 0.01 ppb and 10 ppm wt... Eventually, the mercury in
combusted hydrocarbons contributes to the atmospheric mercury cycle where it distributes, by recently identified mechanisms and pathways, to marine and lacustrine
ecosystems w2x. The causes and consequences of mercury deposition to marine environments have been reviewed in Volume 4 of the Mercury Report to Congress.
The contribution of mercury in petroleum-based fuels to the global mercury cycle is
difficult to estimate because of the lack of data on concentrations of mercury compounds
in common fuels and the lack of mechanistic information on the fate of mercury
compounds in liquid fuel combustion. The mercury compounds in exhaust gas from
burning petroleum fuels are likely to be different than those originating from coal
combustion. In spite of the fact that the total amount of mercury compounds in
petroleum-based fuels is thought to be low, the sheer volume of liquid fuel combustion
requires that the concentrations of mercury compounds in petroleum be accurately
assessed.
In addition to the contribution of geologic mercury to atmospheric pollution, mercury
in oil and gas has a direct negative impact on petroleum processes. The consequences of
mercury in feeds on processing systems include equipment degradation, toxic waste
generation, increased risk to the health and safety of workers and poisoning of catalysts.
Mercury in plant feeds often requires process modifications to avoid the negative
consequences and to comply with product specifications. Speciation of mercury compounds in liquid hydrocarbons is necessary to understand the mechanisms of the
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
interaction of the various species with equipment, catalysts, mercury removal systems
and personnel.
Recent technical advances have improved our understanding of the chemistry of
mercury in petroleum systems. Analytical methods are now capable of measuring
mercury concentrations at very low levels and to differentiate quantitatively many of the
chemical forms of mercury. The newly acquired understanding assists in identifying
exposure risks, selection of mercury removal systems and waste processing for disposal.
2. Background
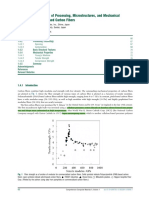
Elemental mercury and several mercury compounds occur naturally in geologic
hydrocarbons. At ambient temperature, elemental mercury Hg 0 . is soluble in liquid
aliphatic hydrocarbons to a few 13. ppm see Fig. 1., which is several times greater
than its 0.05-ppm solubility in water. Elemental mercury is highly adsorptive and
adsorbs on metallic surfaces and on solid materials sand. suspended in liquids. Hg 0
reacts with iron oxide corrosion products on pipe and equipment walls. The solubility of
Hg 0 in produced hydrocarbons depends strongly on temperature, hence, when saturated
liquids are cooled, Hg 0 can precipitate in equipment.
Fig. 1. Solubility of elemental mercury in normal alkanes as a function of temperature.
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
Table 1
Boiling points of volatile mercury compounds
Boiling point 8C.
Hg compound
0
Hg
CH 3 . 2 Hg
C 2 H 5 . 2 Hg
C 3 H 7 . 2 Hg
C 4 H 9 . 2 Hg
357
96
170
190
206
Organic mercury compounds RHgR and RHgCl, where R s CH 3 , C 2 H 5 , etc.. are
highly soluble in crude oil and gas condensate. Dialkylmercury compounds partition to
hydrocarbon liquids in separations and to distillation fractions according to their boiling
points see Table 1.. Monomethylmercury halides partition preferentially to water in
primary separations. The natural abundance of dialkylmercury compounds in produced
hydrocarbons is a matter of some debate. The existence of organic mercury compounds
is inferred when analysis for total mercury in a liquid matrix does not mass balance with
operationally speciated forms. Dialkylmercury compounds have been detected in gas
condensate and crude oil using chromatographic techniques, but typically at low
concentrations.
Mercuric halides HgCl 2 . are about 10 times more soluble than Hg 0 see Table 2. in
gas condensate and oil but partition to the water phase hydrocarbonrwater separations.
The degree to which HgCl 2 distributes between water and the liquid hydrocarbon in
primary separations depends on the salinity of the produced brine and its pH. The
natural abundance of ionic compounds is uncertain because Hg 0 has been found to
oxidize in hydrocarbon liquid samples exposed to oxygen thus casting doubt on some
published speciation data.
Mercury sulfur. complexes HgK where K is an organic sulfide, thiol, thiophene or
mercaptan. are postulated to exist in some crude oils and condensates. The existence of
such compounds in produced hydrocarbons is speculative at present. Specific complexes
of mercury have not been identified with certainty but their existence is inferred in some
chromatographic and operational speciation experiments.
Unprocessed hydrocarbon liquids and many process streams contain suspended
mercury compounds. The most common example is mercuric sulfide HgS., which is
insoluble in water and oil. Particle sizes are typically small. Elemental mercury can also
be suspended in liquids that become saturated due to cooling such as in cryogenic liquid
Table 2
Solubility of some mercury compounds in hexane
Species
0
Hg
HgCl 2
CH 3 HgCl
CH 3 . 2 Hg
Solubility ppb.
Temperature 8C.
1,200
11,500
)1,000,000
`
27.5
27.5
20.0
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
separations. Also included in this category are mercury compounds that are not
dissolved but rather adsorbed on inert particles such as formation fines.
The various chemical forms of mercury exhibit significantly different chemical and
physical behavior. Each chemical form requires analytical methodology that is specific
to its particular characteristics. Analytical procedures are formulated to take advantage
of the differences in species adsorptive character, solubility and volatility. Boiling points
of organic compounds Table 1. assist prediction of the distribution of compounds in
distillations of crude oil. The solubilities of some mercury compounds in hexane are
compiled in Table 2 and are useful to predict the distribution of compounds in phase
separations. The solubility of elemental mercury in alkanes as a function of temperature
is shown in Fig. 1 w3x.
3. Analytical methods
Analytical methods for mercury and mercury compounds in hydrocarbon matrices are
differentiated by collection sampling., species conversionrseparation digestion, extraction, filtration, vaporization. and detection methods. The methods used to determine
total mercury in liquid and gaseous hydrocarbons are reasonably well-established and
accurate. The methods to quantitatively speciate mercury compounds in liquid hydrocarbons are more recent and less verified. The speciation methodologies now practiced are
categorized as either operational or species-specific.
3.1. Sampling
Sampling of hydrocarbons containing mercury is difficult to accomplish because of
partitioning of mercury compounds among phases, species interconversion in some
sample containers, loss of non-polar compounds in plastic containers and due to
adsorption of mercury on sample container surfaces. In samples derived from hot
andror pressurized streams, the volatile mercury compounds Hg 0 and HgCH 3 . 2 .
partition between the vapor and liquid phases in sample containers. Analysis of both the
liquid and gas phases and a mass balance is therefore required if the concentration of
mercury in the produced phase is required.
The amounts of Hg 0 and RHgR that partition to water is usually a small percentage
of the total mercury concentration in coexisting phases because of their low solubilities
in water. Ionic mercury compounds, however, if present, will partition to the water
phase but the hydrocarbonrwater partition ratios depend on pH, salinity, temperature
and other factors. Acidic water can encourage formation of a particle rich layer at the
waterroil interface that can be very high in mercury concentration. Sampling and
analysis protocols should be designed to take these factors into account.
The adsorption of mercury on surfaces can result in considerable uncertainty because
the time to equilibrate plumbing is often unknown. Mercury concentrations in metal
containers used for pressurized liquid samples often exhibit lower than actual results due
to adsorption or reaction with corrosion products on container walls. The material of
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
construction for pressurized sample containers must be selected carefully to obtain
quantitative samples.
3.2. Elemental mercury in gas
Gas matrices typically contain mostly Hg 0 and some lesser amount of HgCH 3 . 2 .
The concentration of Hg 0 in a hydrocarbon gas matrix is difficult to measure directly by
spectroscopic methods because of interference by the hydrocarbon. Trapping the mercury in a known gas volume on a collector facilitates analysis by enrichment of mercury
in the matrix that is analyzed.
A common wet collection method traps mercury in gas in permanganate solution. The
permanganate oxidizes Hg 0 to Hg 2q. Hg 2q is then reduced using stannous chloride to
Hg 0 and separated by sparging into an inert gas stream for detection. This method is
accurate and reasonably sensitive if sufficient volumes of gas are used, but the apparatus
required to collect the samples is somewhat cumbersome and the required sample
volumes are large.
Dry collection methods include several that involve amalgamation of Hg 0 on Ag, Au
or AurPt sputtered on quartz.. The gas containing Hg 0 must contact the metal collector
at a very low flow rate for quantitative trapping. The amalgam is heated in an inert Ar.
gas stream to volatilize mercury for detection. Amalgam trapping is very effective for
light, dry gas. If the stream to be sampled contains heavier components, heating the traps
508C to 1508C. minimizes hydrocarbon condensation without compromise of collection
efficiency.
The efficiency of amalgamation traps for HgCH 3 . 2 depends on the composition of
the gas matrix since the heavier molecules tend to adsorb on the traps and interfere with
amalgamation of HgCH 3 . 2 . This obstacle has been overcome by using AurPt 90r10.
traps that are heated slightly 808C. w4x.
Activated carbon impregnated with iodide is also used to scavenge mercury from gas
matrices. After Hg is collected from a known volume, the carbonrKI traps are subjected
to routine digestive analysis see Section 3.3. to determine total Hg. Iodated carbon traps
are less sensitive to some contaminants than gold traps and have a higher capture
capacity, thus allowing large sample volumes. The iodated carbon traps are suitable for
moist gas if they are maintained above the condensation temperature for water. Water
condensation dissolves the iodide. The trapping efficiency for dimethylmercury is near
100%.
3.3. Total mercury in liquids
Analytical methods for total mercury in hydrocarbon liquids include combustion, wet
hot. digestion and wet extraction. Combustionrvaporization techniques vaporize the
entire liquid matrix and mercury in the exhaust gas is trapped and analyzed using
procedures that are similar to those employed for natural gas w5,6x. Wet methods oxidize
all mercury species in the hydrocarbon liquid matrix to Hg 2q that separates to an
aqueous phase. Wet digestive methods that use strong acids and heat require pressurized
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
digestion bombs. Extractive methods BrCl. are preferred for light hydrocarbon liquids
w7x.
Digestates and extracts are treated with stannous chloride or sodium borohydride to
generate Hg 0 and then sparged Ar.. The sparge gas is either sent directly to a detector
or collected on a trap amalgamation. and then thermally evolved into an inert gas
stream for detection. The amalgamation step concentrates the sample and eliminates
interference from hydrocarbons.
The most common forms of detection are cold vapor atomic absorbance w8x CVAA.
and cold vapor atomic fluorescence CVAF.. CVAF is a very sensitive method w6x with
reported absolute detection limits 100 to 700 pg. The low detection limit allows accurate
quantitation of very small samples and dramatically reduces matrix effects common to
other methods by allowing extreme dilution prior to analysis.
Other types of total mercury analysis methods include inductively coupled plasma
ICP. or microwave induced plasma MIP. followed by mass spectrometry MS. w9x or
atomic emission spectrometry AES. w10x detection. The ICPrMIP techniques avoid
digestion of the sample, hence, minimizing some of the potential errors that can occur in
multi-step wet chemical processing of liquid samples. Neutron activation analysis
NAA. methods, which also avoid wet processing of samples, also have been used to
examine crude oils w11x. NAA, ICPrMIP and MSrAES all report detection limits less
than 0.1 ngrg.
3.4. Speciation of mercury compounds
Operationally defined speciation w12,13x of liquid samples involves multiple and
sequential analyses for the various forms and a mass balance exercise.
Total Hg s Hg 0 q RHgRq HgK . q HgCl 2 q RHgCl. q suspended Hg.
1.
Total mercury concentration typically is determined by oxidative extraction. Suspended mercury is quantitatively determined by measuring total mercury of an agitated
sample followed by measuring total mercury of a filtered portion of the agitated sample.
Ionic and monoalkyl forms HgCl 2 q RHgCl. are determined by non-oxidative extraction of filtered samples using dilute acids. Hg 0 is determined by sparging and collecting
the volatile component on a trap. The sum of the concentrations of dialkylmercury and
complexed mercury often is estimated from the discrepancy in the mass balance.
Chromatographic techniques have been applied successfully to hydrocarbon liquids
and are capable to separate the various mercury species. Both gas chromatography GC.
and high-performance liquid chromatography HPLC. techniques have been used successfully.
Schickling and Broekaert w14x used HPLC, matrix destruction dichromate., borohydride reduction and CVAA to examine gas condensates. Detection limits 3 s . of
approximately 10 ng were achieved for RHgR R s phenyl., HgCl 2 and RHgCl.
Important in this work were observations of the reaction of ionic and organic mercury.
Chromatographic separation of standard mixtures of mercuric chloride and diphenylmercury produced only a peak confirmed to be monophenylmercury chloride.
RHgRq HgCl 2
2RHgCl.
2.
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
Whether reactions of ionic and other organic mercury compounds occur was not
examined and remains uncertain.
Snell et al. w15x used GC, post-column combustion, amalgamation PtrAu. and
MIPAES. MIPAES achieved a detection limits 3 s . between 0.25 and 5 ngrml for
the various species. Separation of organic, inorganic and monomethylmercury species
was accomplished using GC, following derivatization using Grignard reagent butylmagnesium chloride.:
CH 3 HgCl q C 4 H 9 MgCl
HgCl 2 q 2C 4 H 9 MgCl
CH 3 HgC 4 H 9 q MgCl 2
3.
C 4 H 9 HgC 4 H 9 q 2MgCl 2 .
4.
Zettlitzer et al. w16x used HPLC, post-column oxidation UV-PCO. and CVAA to
examine monoalkylmercury and GCMS to examine dialkylmercury in German condensates. The speciation scheme included operational steps for Hg 0 and HgCl 2 . The
detection limit for the GCMS procedure was greater than 1 ppm and thus, did not
allow quantitative determination of dialkylmercury.
Tao et al. w17x used GCICPMS to examine a variety of condensates and naphthas.
The procedures developed by Tao were rigorously verified as to recovery and sensitivity
using spikes, replicates and blanks. Tao optimized injection procedures for the GC and
verified recoveries close to 100% for all of the mercury species examined. Absolute
detection limits were orders of magnitude better than previously reported fg..
4. Mercury compounds in hydrocarbons
Because of its volatility, elemental mercury vapor is the dominant species in natural
gas. Dialkylmercury may also be present in gas, especially close to the wellhead where
the gas is hot. The organic mercury compounds in gas partition to separated hydrocarbon
liquids as the gas is cooled. The trapping process for determination of total Hg in gas
precludes any speciation of volatile mercury components.
Crude oil and gas condensate, when sampled soon after primary separation of water
and gas, typically contain significant amounts of suspended mercury compounds andror
mercury adsorbed on suspended solids. The suspended compounds are mostly HgS but
include mercury adsorbed on silicates and other suspended solid material. The amount of
suspended mercury is determined by difference of total mercury in agitatedrfiltered 0.8
m . samples and agitated samples. Mild extraction of the filtered solids yields the
adsorbedrsuspended fraction and the HgS in filtered solids is quantified by X-ray
methods. The amount of suspended mercury can be a substantial percentage of the total
concentration of mercury in liquid samples of produced hydrocarbons and they must be
separated prior to any speciation of dissolved forms.
Gas condensate samples originate at several locations in a gas processing scheme see
Fig. 2.. A generic unprocessed condensate is the hydrocarbon liquid that separates in the
primary separator, either at the wellhead or at the gas plant. Processed condensate is the
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
Fig. 2. Gas plant liquid processing.
C5 q fraction that is a product from a gas separation plant. Naphthas typically originate
from the primary distillation of oil in the range of 508C to 1508C see Fig. 3.. The
distribution of hydrocarbon compounds in both condensates and naphthas are similar
and mostly in the range C4 to C10. Processed condensate and naphthas typically do not
contain suspended mercury compounds.
Published Hg concentrations in condensate, naphthas and crude oil often do not fully
disclose sampling procedures or analytical processing steps filtration, centrifugation,
exposure to air.. For these reasons, some data are suspect in that the total mercury
concentrations reported could include a contribution from suspended forms or the
distribution of compounds could reflect species conversion. Aerobic processing of
samples is suspected to promote oxidation of Hg 0 to ionic forms and thus to alter the
distribution of species.
The total Hg concentration reported in hydrocarbons varies widely. Some condensates and crude oils are close to saturation with Hg 0 at concentrations of 14 ppm as
determined by sparging of fresh, filtered samples. Adding suspended, ionic and organic
forms, total mercury concentrations in crude oil over 5 ppm are known. Gas condensates
in SE Asia have dissolved total Hg concentrations in the 10800 ppb range. Most crude
oils in the US have relatively low mercury concentrations.
Musa et al. w11x reported total mercury in Libyan crude oils to be in the range of 0.1
to 12 ppb. Bloom w13x found total Hg in unfiltered crude oils ranging between sub-ppb
levels to over saturation see Table 3.. The mean for crude oil 1.5 ppm, N s 76. likely
is skewed high due to samples with some suspended mercury. The mean of the lower 38
samples was close to 1 ppb. Data for crude oils are compiled in Table 3 and Fig. 4.
10
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
Fig. 3. Primary distillation products.
Total mercury concentrations in gas condensates w1417x are shown in Fig. 5.
Zettlitzer analyzed condensates from the from the Rottliegend formation Germany. and
found total mercury in liquids in the range 36 mgrl and corresponding to mercury
concentrations in gas of 1000 to 4400 ugrNm3. It is not known if the liquid samples
Table 3
Total Hg in liquids w13x
Oil type
Crude oil all.
Crude oil highest half.
Crude oil lowest half.
Condensate all.
Condensate highest half.
Condensate lowest half.
Light distillates
Utility fuel oil
Asphalt
76
39
37
18
10
8
14
32
10
wHgx ngrg.
Mean
SD
1505
3009
1.20
3964
7113
20.4
1.32
0.67
0.27
3278
4140
1.49
11,665
15,240
19.7
2.81
0.96
0.32
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
11
Fig. 4. Total mercury in crude oil.
were filtered prior to analysis and thus, may have contained some suspended compounds. These concentrations are in the very high end of the range of total Hg
concentration in condensate. Taos data for total mercury in condensates are included in
Fig. 5 and range between 10 and 200 ugrl. Note: data in the figures have been
converted to grg for uniformity..
The presently published data for total Hg in naphthas Fig. 6. are similar to
condensates and range approximately between 5 and 200 ppb. High concentrations have
not been reported in the limited published data. Naphthas originating from distillations
would be expected to have lower concentrations than the raw produced liquids from
which they originate.
Only limited data are available that allow examination of the distribution of concentrations of mercury compounds in hydrocarbon liquids. Of interest are the natural
abundance of mercury compounds, the relative distribution of compounds in liquid
samples, the partitioning of compounds in separations and distillations and transformation of species during processing.
The data of Tao et al. w17x on gas condensates, naphthas and a crude oil, are compiled
in Table 4 and graphically depicted in Fig. 7. The origin process location. of samples
12
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
Fig. 5. Total mercury in condensates.
analyzed by Tao were not disclosed. Taos data indicate that ionic mercury was the
dominant species in the condensates examined. Hg 0 did not exceed 25% of the total in
any of the condensate samples. The dialkyl species was detected ) 10%. in some
condensates. The monoalkyl species was detected but at very low concentrations. Hg 0
was not seen in naphthas as would be expected assuming a normal distillation profile.
The more volatile Hg 0 would be expected to partition to the lighter gas fraction. RHgR
appeared to be the dominant species in one naphtha sample. Ionic forms of mercury
were seen in all of the samples.
Zettlitzer et al. w16x used two methods to measure concentrations of mercury species.
The method for monoalkylmercury HPLCUVPCOCVAA. provided suitable detection limits. The concentrations of monoalkylmercury in the condensate analyzed by
Zettlitzer were low and generally agree with the data of Tao. GCMS was used to
examine RHgR but the detection limit was high and the methodology suspect. Extracting
condensate with HCl was postulated to remove ionic and organic forms. The concentration of acid-extractable mercury was operationally defined as the difference between the
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
13
Fig. 6. Total mercury in naphthas.
total amount extracted using HCl and the sum of ionic and monoalkylmercury determined independently.
Zettlitzers distributions of compounds, using operationally defined values for extracted mercury, are compiled in Table 5. The unprocessed condensate sample exhibited
a 2-ppm concentration of Hg 0 which is close to the saturation value for elemental
mercury in hydrocarbon liquids. These data do not show the dominance of ionic species
seen in the data of Tao.
Snell et al. w15x analyzed two condensates and found most of the total mercury in
ionic form. The dialkyl form accounted for approximately 10% and the monoalkyl form
less than 1%. Similarly, Schickling and Broekaert w14x analyzed two condensates and
found mostly ionic compounds. Blooms operationally defined speciation Table 6. data
account for the majority of total dissolved mercury as either Hg 0 or KCl extractable
mostly ionic..
In spite of the fact that dialkylmercury has been detected in some samples, the
concentrations found for this class of compounds see Table 4. are very low - 10 ppb.
except for one naphtha Tao. in which it was found at a concentration of approximately
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
14
Table 4
Analytical results for natural gas condensates, condensate naphthas and crude oil mgrl Hg. w17x
The average values of both injection modes were adopted for DMeHg, MeEtHg and DEtHg; nd not
determined; - below detection limit J. Anal. At. Spectrom., 1998, 13, 1085..
Sample
Mode
Condensate 1
Percent
Condensate 2
Percent
Condensate 3
Percent
Condensate 4
Percent
Condensate 5
Percent
Condensate naphtha 8
Percent
Condensate naphtha 9
Percent
Condensate naphtha 10
Percent
Crude oil 11
Percent
Condensate 6
Percent
Condensate 7
Percent
a
Hg 0
HgCl 2 DMeHg MeEtHg DEtHg MeHgCl EtHgCl Suma
1.5
4.6
0.5
3.3
28.8
16.6
2.66
21.8
0.9
3.2
0.0
0.0
0.03
0.4
0.03
4.8
0.3
3.7
1.7
11.8
30.9
95.4
8.1
54.0
116
67.1
7.4
60.7
26.8
96.8
48.4
100.0
0.68
1.2
0.14
1.8
0.60
95.2
3.8
46.9
9.8
67.8
2.0
13.3
9.3
5.4
0.9
7.4
0.0
0.0
22.4
38.3
3.9
51.5
0.0
2.6
32.1
2.3
16.1
3.2
21.3
14.0
8.1
1.0
8.2
0.0
0.0
28.9
49.4
3.1
40.9
0.0
0.9
6.0
5.1
2.9
0.1
0.8
0.0
0.0
6.1
10.4
0.2
2.6
0.0
1.0
12.3
0.3
2.1
0.2
1.3
0.3
0.2
0.1
0.8
0.0
0.0
0.5
0.9
0.1
1.3
0.0
0.3
3.7
0.2
1.4
0.05
0.3
CVAAS
32.4 30.7
15.0 12.1
173
162
0.0
12.2 12.6
0.0
0.0
0.0
0.0
0.07
0.9
0.0
0.1
1.2
0.1
0.7
27.7 34.0
48.4 nd
58.5 nd
7.58 8.49
0.63 1.50
8.1 nd
14.4 nd
Sum s Hg 0 qHgClqDMeHgqMeEtHgqDEtHgqMeHgClqEtHgCl.
50 ppb. Based on the limited data, it is by no means apparent at this point in time that
dialkylmercury is prevalent in petroleum.
Stability of mercury species in synthetic gas condensate was examined by Snell et al.
w18x who demonstrated conclusively that Hg 0 and HgCl 2 react to form Hg 2 Cl 2 that is
insoluble in hydrocarbons and precipitates.
Hg 0 q HgCl 2
Hg 2 Cl 2
5.
The reaction exhibited a half-life on the order of about 10 days at ambient
temperature. The reaction of Hg 0 and ionic forms suggests that reservoirs may contain
solid Hg 2 Cl 2 and that, in the reservoir, an equilibrium is maintained that allows the
coexistence of both species. Production of liquids from reservoir conditions disrupts the
equilibrium and allows Hg 0 to decay. Most condensate samples contain both species
thus implying, given the clearly defined observations of Snell, that species conversion is
likely in gas condensate samples.
Bloom w13x likewise, examined sample stability and found standard solutions of Hg 0 ,
.
HgCHq
3 and Hg CH 3 2 stable in paraffin oil stored in glass. HgCl 2 was not stable in
0
paraffin oil and Hg and HgCl 2 were unstable in natural crude oil. Blooms data
generally support those of Snells.
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
15
Fig. 7. Distribution of mercury compounds in liquid hydrocarbons data of Tao et al...
Oxidative mechanisms may operate in hydrocarbon samples that are exposed to
oxygen, that contact metal surfaces or that are treated with impure reagents as part of the
analytical method. If this is the case, then the high concentrations of ionic forms in some
samples may be an artifact of collection procedures, sample age and analytical processing methodologies. The authors experience with crude oils and gas condensate samples
is that very fresh samples typically exhibit the dominance of the Hg 0 species. No
reductive mechanisms are known that would account for generation of Hg 0 in samples
Table 5
Concentrations of mercury compounds in natural gas condensates mgrl Hg. w16x
Sample
Hg 0
HgCl 2
Other
RHgCl
Suma
Total
HgS
Low temperature separator
Percent.
Ambient temperature separator
250
19.2
2000
39.2
200
11.8
400
30.8
400
7.8
200
11.8
644
49.5
2600
51.0
1250
73.5
6
0.5
100
2.0
50
2.9
1300
100.0
5100
100.0
1700
100.0
3500
2200
5500
400
4300
2600
Storage tank
a
Sum s Hg 0 qHgCl 2 qRHgClqother; other sacid extractedyHgCl 2 ; HgSs Totalysum.
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
16
Table 6
Operational Hg speciation in petroleum samples w13x
Sample ID
Condensate a1
Condensate a2
Crude oil a1
Crude oil a2
Crude oil a3
Crude oil a4
Crude oil a5
Crude oil a6
Crude oil a7
U
Unfiltered Hg ngrg.
0
Total
Hg
20,700
49,400
1990
4750
4610
4100
15,200
1.51
0.42
3060
U
34,500
408
1120
536
1250
2930
0.09
0.17
0.8 m filtered Hg ngrg.
Dissolved total
HgII.
CH 3 Hg
5210
36,800
821
1470
1680
1770
3110
1.01
0.41
2150
2370
291
433
377
506
489
0.39
0.02
3.74
6.24
0.25
0.26
0.27
0.62
0.45
0.15
0.11
This sample was contained particulate Hg 0 that was re-dissolved in hexane.
of geologic hydrocarbons, hence, the transformation of ionic or organic species to
elemental is not likely.
The primary separation of water in gas or oil production would be expected to
segregate the majority of ionic species naturally present to the water phase. Produced
water that has low dissolved mercury content is associated with co-produced hydrocarbon liquids contain high concentrations of ionic species analyzed days after collection..
Such high percentage concentrations of ionic species in the hydrocarbon liquid are not
expected based upon the rationalization that the ionic species should partition to the
separated water phase during primary separations.
If one compares the concentrations of Hg 0 in co-produced hydrocarbon liquid and
gas, Hg 0 typically is dominant is both. This suggests that Hg 0 is the dominant species in
the reservoir fluids and the ionic forms are derived from it. Reaction mechanisms
associated with sample stability certainly require further investigation. If the ionic
content of liquid samples is merely an artifact of sample aging, then the distribution of
mercury compounds previously cited is suspect. There is also considerable doubt that
dialkylmercury exists abundantly in crude oil and condensate.
Monoalkylmercury does not appear to be a dominant species in petroleum. If
dialkylmercury were abundant then the monoalkyl species would be expected to be
similarly abundant. Given the low concentrations of HgCHq
3 in condensate, it is
unlikely that discharges of produced water to the ocean would contain significant
amounts and thus would not have a major, direct contribution to monomethylmercury
levels in fish in proximity to platforms.
5. Mercury distribution in processes
Computational methods are used to estimate the distribution of mercury and mercury
compounds in processes. Predictive computations are useful to identify process locations
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
17
where mercury can condense or accumulate. Calculations of the distribution of mercury
in a process require accurate information on the concentrations of the various dissolved
and suspended forms that exist in liquid and gas feeds. Other data necessary for
calculations include vapor pressures and solubilities of Hg species as a function of
temperature and pressure and gasrliquid partition ratios.
Vapor pressure and solubility for elemental mercury are reasonably well-known or
easily estimated. The solubilities of dialkylmercury compounds in hydrocarbons are
assumed to be infinite over the range of temperatures encountered in most petrochemical
processes. Partitioning of mercury species between liquid and gas phases can be
estimated using chemical approximation principles and some limited empirical data
w13,19x.
In low temperature processes, transformations of one mercury species to another
typically do not occur, so a species mass balance is assumed. Thermal transformation of
Hg 0 to ionic compounds or HgS likely occurs in some high temperature refinery
processes, thus making predictive calculations more difficult. Pressure and temperature
Fig. 8. Distribution of mercury compounds in distillation cuts.
18
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
changes during processing produce the major redistribution of mercury compounds in
process separations.
Predictions of redistribution of mercury species based on assumptions of thermodynamic equilibrium may not account for some major kinetic factors. Rates of condensation and dissolution of Hg 0 are slow in liquids at low temperature. Likewise, the rates of
partition of mercury and organic mercury to separated phases are slow compared to the
rates of phase separation. Purely thermodynamic models thus require major corrections
for non-equilibrium conditions.
The distribution of total mercury in crude oil to primary distillation products w20x is
shown in Fig. 8 and generally trends toward lower concentration in the higher temperature fractions. In a gas separation plant having both gas and liquid feeds 10% of total
Hg entering the plant is in liquid., and in which mercury removal systems are not
employed, the majority of mercury remains in the sales gas see Fig. 9.. Adding a gas
Fig. 9. Distribution of mercury compounds in gas plant products.
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
19
phase mercury removal system to a gas separation process with both gas and liquid
feeds does not eliminate mercury in liquid products.
The amount of mercury in refinery atmospheric emissions depends on the species that
are present in the crude oil. Suspended HgS in crude oil stays with the bottom fractions
in the primary distillation. The HgS in resid and other bottom fractions used to fire
boilers is converted in combustion to volatile forms Hg 0 , HgO. that can be emitted to
the atmosphere.
6. Mercury removal systems
6.1. Hydrocarbons
Mercury removal sorbent beds are used to scavenge mercury from gas and liquid
hydrocarbon streams. The sorbents consists of granular or pelletized material consisting
of a substrate support zeolite, activated carbon, metal oxide or alumina. and a reactive
component Ag, KI, CuS, metal sulfide, etc.. that is bonded to the support. The
proprietary nature of commercial sorbents derives from the chemistry of the reactive
component, the adsorptive nature of the support and the method by which the reactive
component is attached to the support. Sorbents function by reacting mercury or a
mercury compound. to a chemical form HgS, HgI 2 or amalgam. that is insoluble in
hydrocarbon liquid and chemically inert to the components of process stream.
Several commercial processes are available for removing mercury and mercury
compounds from hydrocarbons. Some commercial mercury removal systems are targeted
at gas phase treatment and some are targeted at liquids. Gas phase treatment Table 7.
systems primarily consist of sulfur-impregnated carbon, metal sulfide on carbon or
alumina, and regenerative molecular sieve Ag on zeolite. w2125x. Mercury removal
processes for hydrocarbon liquids consist of iodide-impregnated carbon, metal sulfide on
carbon or alumina, a mol-sieve amalgam system and a two-step process consisting of a
hydrogenation conversion catalyst followed by metal sulfide reaction with elemental
mercury w2629x. The various liquid removal systems have both advantages and
disadvantages that depend on feed composition and stream location.
Table 7
Mercury removal systems for hydrocarbons
Reactant
Substrate
Complexed form
Application
Sulfur
Metal sulfide
Iodide
PdqH 2 ; metal sulfide
Ag
Metal oxidersulfide
Carbon, Al 2 O 3
Carbon, Al 2 O 3
Carbon
Al 2 O 3
Zeolite
Oxide
HgS
HgS
HgI 2
HgS
AgrHg amalgam
HgS
Gas
Gas, liquid
Dry liquid
Liquid
Gas, light liquid
Gas, liquid
20
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
Mercury removal systems sorbents. for hydrocarbon liquids Table 7. function by
reacting primarily with elemental mercury and with some mercury compounds to form
an insoluble chemical form. Removal systems are ineffective in reacting with suspended
mercury such as HgS. If HgS is present in a liquid feed, the feed liquid must be filtered
or otherwise treated. to remove suspended solids if the system is to function effectively.
Most mercury removal systems for liquids are chemically specific to one form of
mercury, usually elemental. If the feed contains substantial amounts of ionic, dialkyl or
complexed mercury, then the removal system may not achieve design criteria for
removal efficiency. One mercury removal system for liquids employs process steps that
are designed to hydrogenate dialkylmercury and subsequently treating to remove Hg 0 .
Dialkylmercury compounds do not amalgamate rapidly hence, the mol-sieve amalgamation sorbents do not operate at high efficiencies if organic forms of mercury are present
in significant concentrations. Similarly, the metal sulfide systems are chemically specific
to the elemental form and will not react with covalently bound mercury compounds.
Removal system chemistry is sensitive to minor feed contaminants w30x. The sulfur in
sulfur-impregnated carbons are soluble in liquid hydrocarbon and cannot be used in
process locations where liquid condensation can occur or where process upsets can
introduce liquid hydrocarbon into the removal bed. Likewise, the iodide-impregnated
carbons are sensitive to liquid phase water and process locations where water condensation can occur must be avoided. Metal sulfides are less sensitive to water and liquid
hydrocarbons but their inherent reactivity to mercury may be somewhat less than some
other systems, thus requiring longer bed residence times.
6.2. Water
In petroleum production operations, produced water containing high ) 100 ppb.
concentrations of total dissolved mercury contain mostly ionic or suspended forms
because of the solubility of Hg 0 in water is low - 50 ppb.. Suspended mercury in
produced water can be separated using filtration and centrifugal systems w31x. It is not
common to treat produced water chemically to remove mercury prior to disposal.
In refineries and gas plants, sour water streams tend to attract mercury because of
reactive mechanisms with the sulfides. Waste waters in plants that process feeds
containing mercury accumulate ionic mercury and other dissolved forms in concentrations proportional to the concentration in the feeds. Even when the mercury concentration in plant feeds is relatively low, water streams that are by-products of equipment
cleaning and water condensed from glycol regeneration can contain high concentrations
of mercury.
Mercury removal systems for petroleum waste waters are summarized in Table 8. A
common method for removal of mercury from aqueous process effluents is chemical
precipitation by oxidation and sulfide addition. The sulfide reacts with ionic forms of
mercury to form the insoluble HgS that is separated by coagulation and filtration. In
precipitative systems, it is often necessary to polish the effluent with activated carbon or
synthetic resins to further reduce the residual levels necessary to meet stringent
regulatory requirements. Special proprietary precipitating agents have been developed
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
21
Table 8
Mercury removal systems for water
Method
Waste stream
Lower limit of treatment capability mgrl.
Sulfide precipitation
Other precipitant
Ion exchange
Iron cementation
Activated carbon
Thiol resins
HgS sludge
Sludge, Hg complex
Resin, Hg 2q solution
Hg metal, iron oxide
CarbonqHg complex
ResinqHg complex
1020
110
15
0.55
0.25
- 0.1
that can achieve lower effluent mercury concentration and thus eliminate effluent
polishing. The precipitating agents form strongly insoluble complexes with mercury and
include mercaptotriazine, thiocarbonate and thiocarbamates.
Specially formulated activated carbons are used to adsorb chemisorb. elemental,
ionic and complexed mercury from water streams that do not contain significant
amounts of hydrocarbons. Activated carbons are specific and selective for mercury but
typically do not have high capacity and are used mostly for waste streams that are
relatively low in mercury content. Waste water streams can also be treated with thiol
resins that achieve very low mercury concentrations in the resin bed effluent and are
used in situations where regulatory requirements are stringent.
7. Effects of mercury on processing
Mercury has several detrimental impacts on gas processing operations.
1. Mercury deposits in cryogenic equipment, sometimes cause cracking of welded
aluminum heat exchangers w32x. Numerous cases of cold box failure are recorded in
older gas processing plants and steam cracking ethylene plants, however, the introduction of cold box designs that are resistant to mercury and mercury removal systems have
served to reduce the incidence of failure.
2. Mercury in gas plant products affects downstream processes. Gas plant products
used for chemical manufacture, especially olefins, ethylene, aromatics and MTBE, are at
risk to mercury in process feeds due to the cited equipment problems and due to catalyst
poisoning.
3. Mercury contaminates treatment processes such as molecular sieve and glycol
dehydration units, and amine acid gas removal systems. Contaminated treatment liquids
and spent mol-sieve sorbents are difficult to dispose of and to regenerate.
4. Mercury sorbent materials used for gas or liquid treatment, when spent, constitute
a generated hazardous waste that plant operators must store or process for disposal.
5. Mercury deposition in equipment poses a health and safety risk for workers
involved in maintenance or inspection activities.
6. Sludge containing mercury from water treatment systems, separators, desalters
and heat exchangers represents a toxic waste stream that is difficult to store or process
for disposal.
22
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
7. Waste water streams that contain high levels of mercury must be treated to
remove mercury prior to discharge thus adding significant costs to plant operational
expense.
7.1. Health and safety [33]
The major mercury-specific considerations for health and safety of workers in
petroleum processing involve exposure of workers to mercury vapor and dermal
absorption of dialkylmercury. Mercury and its compounds are neurotoxins. Inhalation of
mercury vapor, ingestion of ionic mercury or dermal absorption of mercury compounds
ultimately results in neurological dysfunction. The period of time between exposure and
exhibition of symptoms varies considerably depending upon the type absorbed mercury
species and magnitude of exposure. Chronic exposure to mercury vapor results in
psychological anomalies excitability, memory loss, insomnia, and depression. and
physical symptoms weakness, fatigue, anorexia, weight loss.. Tremors may develop in
more advanced cases. Compromise of renal function is seen in acute, high dose cases.
Analysis of blood and urine are the most common diagnostic tools for the discovery
and quantification of occupational exposure. Reference levels background level for
populations not occupationally exposed. for mercury total. in blood, urine and scalp
hair are compiled in Table 9. Assessment of worker exposure requires testing for
mercury in blood or urine at a frequency that is dictated by the exposure risks. The
concentrations of mercury in blood and urine decrease with time but not in the same
manner.
Hg 0 is readily absorbed into the blood stream via the lungs. Dermal absorption
efficiencies for elemental mercury in vapor are typically low less than 3% of the
absorbed dose. but nonetheless, must be strictly avoided. Closed space atmospheres in
equilibrium with elemental mercury deposits can acquire mercury vapor concentrations
Table 9
Reference values for total mercury concentrations in biological media general population. w1x
Matrix
Concentration
Reference
Whole blood
Fish consumption
No fish meals
Two mealsrweek
Two to four mealsrweek
18 mgrl
WHO 1990.
2.0 mgrl
4.8 mgrl
8.4 mgrl
Urine
45 mgrl
WHO 1990.
Scalp hair
2 mgrg
WHO 1990.
Fish consumption
Oncermonth
Oncer2 weeks
Oncerweek
Oncerday
1.4 mgrg
1.9 mgrg
2.5 mgrg
11.6 mgrg
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
23
that exceed 20 mgrm3. The most common locations for mercury accumulation are
separators and heat exchangers. Cleaning and inspection of equipment must be planned
carefully if processed hydrocarbons contain mercury total. above a few ppb. Mercury
accumulates and concentrates in vessels and will produce interior vapor concentrations
that can be much higher than the concentration of mercury in the process stream.
Accumulation mechanisms include adsorption on equipment surfaces and dissolution in
sludge.
Dialkylmercury is estimated to be many times more toxic than elemental mercury on
an equivalent weight. dose basis but exact quantification of toxicokinetics awaits
focused studies. The current OSHA standard for dialkylmercury is 0.01 mgrm3 of air
averaged over an 8-h work shift, with a ceiling level of 0.04 mgrm3. The derivation of
these limits is suspect and they should not be relied on. The concentrations of organic
mercury in unprocessed petroleum are generally assumed to be lower than for elemental
and the exposure pathways for organic mercury are more hindered in petroleum
occupations.
Estimation of the potential risk to oil-field workers due to dialkylmercury compounds
in petroleum liquids is uncertain both because of the lack of data on prevalent
concentrations and the lack of data on dermal absorption efficiency. Inhalation of large
amounts of dialkylmercury in petroleum work environments is less likely than for
elemental mercury due to its lower volatility and higher solubility in liquids. This is
offset by the higher toxicity of the dialkyl species and may not be operative in situations
involving hot work or closed space entry in warm vessels.
If dialkylmercury is discovered in substantial concentrations in crude or condensates,
dermal exposure could be a significant health risk because transdermal absorption of
organic mercury is more facile than that of elemental mercury, based on lipid solubility.
Data are lacking on the precise rates and efficiencies in both animals and humans. It is
known that petroleum workers are routinely exposed to liquid hydrocarbons that
reportedly contain significant amounts of dialkylmercury. Chronic health deficiencies
have not been reported suggesting that either the amount of dialkylmercury is over
estimated or the rate of dermal absorption is low or both.. A recent case of exposure
and death. of a laboratory worker suggests the former hypothesis is more likely w34,35x.
The toxicokinetics of RHgR has not been studied sufficiently to have definitive data
on the mechanisms of transport and elimination. It is estimated that dialkylmercury is
readily absorbed dermally and readily crosses tissue barriers. Hence, its interactions with
the body are likely more similar to CH 3 Hgq than to Hg 0 . This would mean that the
contribution of dialkylmercury to the total concentration of mercury in blood would
reflect the dose, equilibria with tissue and demethylation rate. By analogy with CH 3 Hgq,
little dialkylmercury should eliminate via urine. Monitoring total mercury in blood is the
better method for determination of occupational exposure to RHgR. Analysis of urine is
better suited to determine inhaled Hg 0 .
Ingestion of mercury is not a typical occupational hazard for workers, but ingested
mercury is often a major contributing dose factor for those whose diets contain a high
percentage of fish. The mercury compound in fish is monomethylmercury that is
bio-accumulated and bio-concentrated in piscivorous populations w36x. The predatory
marine fish species shark, tuna, swordfish, barracuda. accumulate high concentrations,
24
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
as do all predatory fresh water species pike, bass.. Mercury in fish that constitute a
major portion of dietary intake can contribute significantly to the total concentrations in
blood but not in urine.. The combination of dietary and occupational mercury can cause
total exposure to exceed the threshold for chronic detriment. High fish diets also can
obfuscate monitoring programs based on blood analysis for worker exposure to the
extent that occupational intake can be overestimated.
Because of the subtleties of mercury poisoning, toxic influences to neural function
can go unnoticed for very long periods of time, if they are detected at all. Chronic, low
level exposures may require years to diagnose unless exposure risks are apparent and
efforts are made to evaluate those potentially affected. Chronic mercury toxicity is
extremely difficult to diagnose from symptoms in their early stages. At the point of
conclusive symptomatic diagnosis, neuralgic impairment is usually at an advanced stage
and remediative therapies are mostly ineffective.
Atmospheres inside vessels and in the proximity of opened vessels and piping are
analyzed for mercury vapor to assist decisions on worker protection. Since the local
concentration of mercury vapor varies considerably depending upon temperature and
convection, rapid and numerous analyses are useful to understand the source and
concentration of mercury vapor in work areas. Atmospheres are typically characterized
using portable vapor analyzers w37,38x. Monitoring concentrations of mercury vapor in
plant equipment is not a routine operation in most processing plants unless there is a
historical recognition of mercury in processed hydrocarbons and prior discovery in
closed space atmospheres. The ability of portable analyzers to detect organic mercury is
uncertain.
7.2. Waste [39]
A wide variety of waste streams that contain mercury are generated in conjunction
with petroleum production and processing. The operational procedures to accumulate,
characterize and dispose of these streams likewise vary widely and depend on numerous
factors and variables. Liquid stream volumes are difficult to reduce by evaporation or
distillation because of the volatility of elemental mercury. Volume reduction by separation of mercury and compounds in solid matrices is likewise difficult to accomplish.
Waste materials that contain mercury, once identified and characterized, are treated to
remove the mercury prior to disposal of residue or debris. Treatment for disposal is not a
common practice for small volume waste streams in petroleum processing operations.
Storage of waste that contains mercury is more common in remote locations where
treatment facilities are logistically difficult to access. The US EPA is presently under
mandate to characterize major waste streams from refineries as to toxic constituents.
Some early reports w40x have examined desalter sludge and found concentrations
averaging approximately 30 ppm average of samples from three major US refineries..
Speciation of mercury compounds in petroleum processing waste has not been reported.
Removal of mercury from complex mixtures is accomplished chemically, thermally,
and sometimes physically. Physical methods vacuuming, filtration, particle separation.
rely on elemental mercurys high density and surface tension to allow segregation.
Separation of liquid mercury from soil or sludge by physical means is often warranted to
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
25
reduce the amount of mercury present prior to treatment by another method. Mercury
compounds typically cannot be physically separated. Chemical treatments have been
applied to soil remediation and equipment decontamination. Mercury is removed from
liquids by adding chemicals that precipitate of solid mercury compounds HgS. that can
be filtered or physically separated.
Outside the oil and gas industry, most mercury-contaminated sludge is subjected to a
thermal treatment to remove mercury, thus allowing disposal of the residuum. These
processes, referred to as roasting or retorting, vaporize mercury and mercury compounds
and condense Hg 0 . Excess heat is require to convert some compounds HgS. to the
condensable elemental form. In combustion systems, oxygen reacts with mercury
compounds to form mercuric oxide that decomposes to elemental mercury vapor. The
use of such systems to treat waste streams generated in conjunction with petroleum
processing has only recently gained acceptance in the industry.
8. Conclusions
Mercury and mercury compounds are common components of petroleum and the
products derived from it. The chemical and physical characteristics of the various
species account for the distribution of compounds in process streams. Analytical
techniques now exist to accurately determine all of the species known to be prevalent in
petroleum.
Speciation of mercury in hydrocarbon feedstocks and distilled or separated products
is necessary to determine the mechanisms that account for discharges of mercury to the
environment. As has been the case with coal-fired utilities, refineries are likely to be
closely scrutinized by regulatory bodies for mercury emissions, both atmospheric and
solid or liquid waste streams.
At present, the elemental species and ionic mercury compounds appear to be
dominant in crude oil and gas condensate. The natural abundance of organic mercury in
petroleum requires further definition. Ionic mercury in liquid hydrocarbon samples is
more prevalent than would be expected based on its concentration in coexisting phases.
This observation may be due in part to oxidation of Hg 0 in some sampling and analysis
protocols. The current data suggest monoalkylmercury compounds are not present in
produced fluids in substantial concentrations.
Removal systems that employ sorbents to react with mercury in liquid and gas
streams are available, but selection of the most effective system must be predicated on
good analytical data and on an understanding of the sorbent chemistries. Some existing
Hg removal systems for liquids do not remove the dialkyl species. Removal of mercury
from produced water or waste water streams presently consists mostly of removing
suspended hydrocarbons and solids. Chemical treatments to remove mercury from
produced water are not common in the petroleum industry.
The risks to personnel from mercury that deposits in equipment come primarily from
inhalation of vapors and from dermal absorption of dialkylmercury in condensate. The
risks due to organic mercury are uncertain and require further investigation. Health and
safety plans should be based on a quantitative analysis of risk that requires knowledge of
26
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
the amounts of the various mercury species that may be present. Monitoring for mercury
during maintenance and inspection activities is a key ingredient to risk assessment.
Waste that contains mercury and that originates in conjunction with petroleum
processing is difficult to segregate, store and process for disposal. Systems to treat such
waste are commercially available but seldom employed except for large volume waste
streams. Storage or burial of waste material containing mercury is a poor option that
carries residual liability to generators, but both are common practices in many remote
locations.
References
w1x Mercury Study Report to Congress, US EPA, EPA-452RrR-97-004 1997..
w2x D. Porcella, Mercury in the environment, biogeochemistry, in: C. Watras, J. Huckabee Eds.., Mercury
Pollution, Integration and Synthesis, Lewis Publishers, 1994.
w3x H. Clever Ed.., Mercury in Liquids, Compressed Gases, Molten Salts and Other Elements, IUPAC
Solubility Data Series, Vol. 29, Pergamon, 1987.
w4x W. Frech et al., On the determination of total mercury in natural gases using the amalgamation technique
and cold vapor atomic absorption spectrometry, J. Anal. At. Spectrom. 10 1995. 769.
w5x US EPA Method, 7473 1996..
w6x A. Shafawi et al., Determination of total mercury in hydrocarbons and natural gas condensate by atomic
fluorescence spectrometry, Analyst 124 1999. 185.
w7x L. Liang, M. Horvat, P. Danilchik, A novel analytical method for determination of picogram levels of
total mercury in gasoline and other petroleum based products, Sci. Total Environ. 187 1996. 57.
w8x R. Heyward et al., Determination of the recovery of dimethylmercury and diphenylmercury extracted
from organic solvents and a liquid condensate with bromine water using cold vapour atomic absorption
spectrometry, Anal. Commun. 10 1997. 279.
w9x S.D. Olsen et al., Analysis of metals in condensates and naphthas by ICP-MS, Analyst 122 1997. 12.
w10x W. Frech, J. Snell, R. Sturgeon, Performance comparison between furnace atomization plasma emission
spectrometry and microwave induced plasmaatomic emission spectrometry for the detection of mercury
species in gas chromatography effluents, J. Anal. At. Spectrom. 13 1998. 1347.
w11x M. Musa et al., Neutron activation analysis of major and trace elements in crude petroleum, J. Radioanal.
Nucl. Chem. 198 1. 1995. 17.
w12x W. Frech, D.C. Baxter, B. Bakke, J. Snell, Y. Thomasson, Determination and speciation of mercury in
natural gases and gas condensates, Anal. Commun. 33 1996. 7H.
w13x N.S. Bloom, Analysis and stability of mercury speciation in petroleum hydrocarbons, Fresenius J. Anal.
Chem., in press 1999..
w14x C. Schickling, J. Broekaert, Determination of mercury species in gas condensates by on-line coupled
HPLC and CVAA spectrometry, Appl. Organomet. Chem. 9 1995. 29.
w15x J.P. Snell, W. Frech, Y. Thomasson, Performance improvements in the determination of mercury species
in natural gas condensate using an on-line amalgamation trap or solid-phase micro-extraction with
GCMIPAES, Analyst 121 1996. 1055.
w16x M. Zettlitzer, R. Scholer, R. Falter, Determination of elemental, inorganic and organic mercury in North
German gas condensates and formation brines, Proceedings of Symposium: Oil and Gas Chemistry,
Houston, SPE 37260 Feb. 1997..
w17x H. Tao et al., Mercury speciation in natural gas condensate by gas chromatographyinductively coupled
plasma mass spectrometry, J. Anal. At. Spectrom. 13 1998. 1085.
w18x J. Snell, M. Johansson, W. Frech, K. Smit, Stability and reactions of mercury species in organic solution,
Analyst 5 1998. 905.
w19x B. Edmonds, R. Moorwood, R. Szczepanski, Mercury Partitioning in Natural Gases and Condensates,
GPA European Chapter Meeting, London March 1996..
S.M. Wilhelm, N. Bloomr Fuel Processing Technology 63 (2000) 127
27
w20x P. Sarrazin, C. Cameron, Y. Barthel, Processes prevent detrimental effects from As and Hg in feedstocks,
Oil Gas J., Jan. 25, 1993.
w21x H. Nishino et al., Process for Removal of Mercury Vapor and Adsorbant Therefor, U.S. Patent 4,500,327;
Feb. 1985..
w22x T.M. Matviya et al., Mercury Adsorbent Carbon Molecular Sieves and Process for Removing Mercury
Vapor from Gas Streams, U.S. Patent 4,708,853 Nov. 1987..
w23x Y. Barthel, C.J. Cameron, F. Gracco, A. Orieux, P. Sarrazin, Mercury Removal from Wet Natural Gas,
Proceedings: European Gas Processors Association, 10th Continental Meeting June 1993..
w24x A. Sugier et al., Process for Removing Mercury from a Gas or Liquid by Absorption on a Copper Sulfide
Containing Solid Mass, U.S. Patent 4,094,777 1978..
w25x J. Markovs, Purification of Fluid Streams Containing Mercury, U.S. Patent 4,874,525 1988..
w26x J. McNamara, Process for Removal of Mercury From Liquid Hydrocarbon, US Patent 5,336,835 Aug.
1994..
w27x C. Cameron et al., Method of Eliminating Mercury or Arsenic From a Fluid in the Presence of a
MercuryrArsenic Recovery Mass, U.S. Patent 5,245,106 1993..
w28x Roussell et al., Process for Removing Mercury and Possibly Arsenic in Hydrocarbons, U.S. Patent
4,911,825 1990..
w29x J. Markovs, Removal of Mercury from Process Streams, U.S. Patent 5,223,145 1993..
w30x S. Wilhelm, Conceptual design of mercury removal systems for hydrocarbon liquids, Hydrocarbon
Processing April 1999..
w31x N. Pongsiri, 1997 SPE Asia Pacific Oil and Gas Conference, SPE Paper 38087 1997..
w32x D. Nelson, Mercury Attack of Brazed Aluminum Heat Exchangers in Cryogenic Gas Service, Proceedings 73rd GPA Convention, New Orleans 1994..
w33x S. Wilhelm, Avoiding Exposure to Mercury during Inspection and Maintenance, Plant Safety Progress,
AIChE, in press 1999..
w34x M.B. Blayney, J.S. Winn, D.W. Nierenberg, Letters: chemical safety handling dimethylmercury,
Chemical and Engineering News, May 12, 1997.
w35x D.W. Nierenberg, Letters: more on working with dimethylmercury, Chemical and Engineering News,
June 16, 1997.
w36x C. Watanabe, H. Satoh, Evolution of Our Understanding of Methylmercury as a Health Threat, Workshop
on Risk Assessment Methodology for Neurobehavioral Toxicity SGOMSEC., Rochester, New York
June 1994..
w37x Arizona Instruments, Phoenix, AZ, USA.
w38x Nippon Instruments, Tokyo, Japan.
w39x S. Wilhelm, Generation and disposal of petroleum processing waste that contains mercury, Env. Progress,
AIChE June 1999..
w40x U.S. EPA, Office of Solid Waste, Study Of Selected Petroleum Refining Residuals, an Industry Study,
Part 1 August 1996..
Das könnte Ihnen auch gefallen
- Crude Oil Distilation CurvesDokument11 SeitenCrude Oil Distilation Curvesvicktorinox230388100% (1)
- Flammability of Mixtures of Co2 and HydrocarbonsDokument17 SeitenFlammability of Mixtures of Co2 and HydrocarbonsLakshmi Binoy MenonNoch keine Bewertungen
- AGR OptimizationDokument43 SeitenAGR Optimizationinara amatullahNoch keine Bewertungen
- PromaxDokument20 SeitenPromaxehsan_sa405100% (1)
- Material Studio 5.0 Quick Start TutorialDokument21 SeitenMaterial Studio 5.0 Quick Start Tutorialstarscientist100% (1)
- Gas Sweetening and Processing Field ManualVon EverandGas Sweetening and Processing Field ManualBewertung: 4 von 5 Sternen4/5 (7)
- Direct Methane to Methanol: Foundations and Prospects of the ProcessVon EverandDirect Methane to Methanol: Foundations and Prospects of the ProcessNoch keine Bewertungen
- Experiences in The OperationDokument7 SeitenExperiences in The OperationUsama Bin SabirNoch keine Bewertungen
- Chapter 3 Separation of Oil and Gas - 1987 - Developments in Petroleum ScienceDokument50 SeitenChapter 3 Separation of Oil and Gas - 1987 - Developments in Petroleum ScienceFalokid RaboNoch keine Bewertungen
- Comparison of Approaches To Determine Hydrogen Consumption During Catalytic Hydrotreating of Oil Fractions PDFDokument9 SeitenComparison of Approaches To Determine Hydrogen Consumption During Catalytic Hydrotreating of Oil Fractions PDFNoheilly Vásquez100% (1)
- Auto Ignition TemperatureDokument3 SeitenAuto Ignition TemperatureThammisetty SrinivasNoch keine Bewertungen
- 7171 Oxyfuel Combustion For Below Zero CO2 EmissionsDokument462 Seiten7171 Oxyfuel Combustion For Below Zero CO2 EmissionsJoão13@Noch keine Bewertungen
- Thermodynamic Modelling of Asphaltene Precipitation and Related Phenomena 2015 Advances in Colloid and Interface ScienceDokument12 SeitenThermodynamic Modelling of Asphaltene Precipitation and Related Phenomena 2015 Advances in Colloid and Interface ScienceEzequiel AbrilNoch keine Bewertungen
- Gpa 2174 - 93Dokument15 SeitenGpa 2174 - 93jairo7662Noch keine Bewertungen
- Natural Gas Hydrocarbon Contamination in AmineDokument8 SeitenNatural Gas Hydrocarbon Contamination in AmineRicardo BecNoch keine Bewertungen
- 03 - BRIM TechnologyDokument45 Seiten03 - BRIM TechnologyFrank Seipel6Noch keine Bewertungen
- Steam Methane Reforming Reaction Process Intensification by Using A ReactorDokument14 SeitenSteam Methane Reforming Reaction Process Intensification by Using A ReactorserchNoch keine Bewertungen
- Mercury in Extraction and Refining Process of Crude Oil and Natural GasDokument68 SeitenMercury in Extraction and Refining Process of Crude Oil and Natural GasSaif EvonyNoch keine Bewertungen
- ROSE® Process Offers Energy Savings For Solvent ExtractionDokument14 SeitenROSE® Process Offers Energy Savings For Solvent Extractiona_abbaspourNoch keine Bewertungen
- Petroleum Fluids CompositionDokument46 SeitenPetroleum Fluids CompositionAustinNoch keine Bewertungen
- Muñoz Et Al. Comparison of Correlations For Estimating Product Yields From Delayed CokingDokument12 SeitenMuñoz Et Al. Comparison of Correlations For Estimating Product Yields From Delayed CokingSergioDanielContreras100% (1)
- Treat LPGs With AminesDokument12 SeitenTreat LPGs With Amineskaaskopdawie5755Noch keine Bewertungen
- Energy Optimization of Crude Oil Distillation Using Different Designs of Pre-Flash Drums PDFDokument7 SeitenEnergy Optimization of Crude Oil Distillation Using Different Designs of Pre-Flash Drums PDFGabriela Urdaneta100% (1)
- Catalytic Dehydration of Methanol To Dimethyl Ether Dme Using The Alcufe Quasicrystalline Alloy 2157 7048.1000164Dokument8 SeitenCatalytic Dehydration of Methanol To Dimethyl Ether Dme Using The Alcufe Quasicrystalline Alloy 2157 7048.1000164Aditya BayuNoch keine Bewertungen
- Makogon Gas Hydrates - 2010Dokument11 SeitenMakogon Gas Hydrates - 2010Alfino Harun LubisNoch keine Bewertungen
- Isomax ProcessDokument8 SeitenIsomax ProcessKarthick RaghuNoch keine Bewertungen
- Ullmann S Encyclopedia of Industrial Chemistry - MethanolDokument24 SeitenUllmann S Encyclopedia of Industrial Chemistry - MethanolKellyCristinaNoch keine Bewertungen
- Hydrotreating - UOP - A Honeywell CompanyDokument59 SeitenHydrotreating - UOP - A Honeywell CompanySamNoch keine Bewertungen
- Simulation and Performance Analysis of Propane Propylene SplitterDokument4 SeitenSimulation and Performance Analysis of Propane Propylene SplitterAnonymous UY9QffNoch keine Bewertungen
- Room Temperature Synthesis of Copper Oxide Nanoparticles Morphological Evaluation and Their Catalytic Applications For Degradation of Dyes and C-N Bond Formation Reaction PDFDokument11 SeitenRoom Temperature Synthesis of Copper Oxide Nanoparticles Morphological Evaluation and Their Catalytic Applications For Degradation of Dyes and C-N Bond Formation Reaction PDFAshpavi ArunNoch keine Bewertungen
- Propylene-Propane - Howat Swift - McCabe ThieleDokument19 SeitenPropylene-Propane - Howat Swift - McCabe ThieleFred FaberNoch keine Bewertungen
- Part 1: Design, Modeling and Simulation of Post-Combustion CO Capture Systems Using Reactive SolventsDokument24 SeitenPart 1: Design, Modeling and Simulation of Post-Combustion CO Capture Systems Using Reactive SolventsBenjamin F ZavalaNoch keine Bewertungen
- Handling Mercaptans in The LaboratoryDokument7 SeitenHandling Mercaptans in The LaboratoryjoesuhreNoch keine Bewertungen
- High-Pour-Point and Asphaltic Crude Oils and CondensatesDokument5 SeitenHigh-Pour-Point and Asphaltic Crude Oils and CondensatesarispriyatmonoNoch keine Bewertungen
- Enthalpy PDFDokument119 SeitenEnthalpy PDFEstuardo Javier Gan RodríguezNoch keine Bewertungen
- Solvent ImportanceDokument6 SeitenSolvent ImportanceAbhiNoch keine Bewertungen
- CO2 Capture Over K2CO3 MgO Al2O3 Dry Sorbent in A Fluidized BedDokument8 SeitenCO2 Capture Over K2CO3 MgO Al2O3 Dry Sorbent in A Fluidized BedFarah Talib Al-sudaniNoch keine Bewertungen
- DVLS VUV Analyzer User Manual: Based On ASTM D8071Dokument22 SeitenDVLS VUV Analyzer User Manual: Based On ASTM D8071Duy DangNoch keine Bewertungen
- Cryogenic and Its ApplicationDokument11 SeitenCryogenic and Its ApplicationBhoomika MansharamaniNoch keine Bewertungen
- 1 - CL619 RPD - Properties Calculation PDFDokument166 Seiten1 - CL619 RPD - Properties Calculation PDFPankaj Kumar SainiNoch keine Bewertungen
- Application Crude Distillation InstrumentsDokument1 SeiteApplication Crude Distillation InstrumentsHARISHKOTHARU48Noch keine Bewertungen
- Characterization and Prediction of Water Droplet Size in Oil Water Flow - J Yao - MSDokument183 SeitenCharacterization and Prediction of Water Droplet Size in Oil Water Flow - J Yao - MSGianmarco Corticelli100% (1)
- Microreactors: W. Ehrfeld, V. Hessel, H. LöweDokument20 SeitenMicroreactors: W. Ehrfeld, V. Hessel, H. LöweAnonymous Ksq1dyPRhNoch keine Bewertungen
- Hydrodesulfurization Often Abbreviated To HDSDokument16 SeitenHydrodesulfurization Often Abbreviated To HDSigor0104100% (1)
- Gasification of Plastic Waste As Waste-To-Energy or Waste-To-Syngas Recovery RouteDokument10 SeitenGasification of Plastic Waste As Waste-To-Energy or Waste-To-Syngas Recovery RouteJalcamNoch keine Bewertungen
- PC 2 2008 MohaddecyDokument8 SeitenPC 2 2008 MohaddecyAnonymous 1FaavtNoch keine Bewertungen
- CrudeOil Desulfurication ThesisDokument150 SeitenCrudeOil Desulfurication ThesisJyri PaulasaariNoch keine Bewertungen
- A Correlation For Calculating HHV From Proximate Analysis of Solid FuelsDokument8 SeitenA Correlation For Calculating HHV From Proximate Analysis of Solid FuelsiadraelNoch keine Bewertungen
- Koya University Faculty of Engineering Chemical Engineering DepartmentDokument17 SeitenKoya University Faculty of Engineering Chemical Engineering Departmentwhah11Noch keine Bewertungen
- PanizzoloTerrin Federico Tesi PDFDokument112 SeitenPanizzoloTerrin Federico Tesi PDFbalajiNoch keine Bewertungen
- Prediction of Viscosities of Petroleum Fractions, Yavuz Yorulmaz, 2009Dokument13 SeitenPrediction of Viscosities of Petroleum Fractions, Yavuz Yorulmaz, 2009Anonymous Xy309m9Sm9Noch keine Bewertungen
- Liquid Fuels and Propellants For Aerospace Propulsion PDFDokument19 SeitenLiquid Fuels and Propellants For Aerospace Propulsion PDFsyaplinNoch keine Bewertungen
- Methods To Determine The Mine Gas Explosibility e An OverviewDokument11 SeitenMethods To Determine The Mine Gas Explosibility e An OverviewMohanad El-HarbawiNoch keine Bewertungen
- Application of Ionic Equiibria Process Simulation For Atmospheric Distillation Overhead SystemsDokument15 SeitenApplication of Ionic Equiibria Process Simulation For Atmospheric Distillation Overhead SystemsGraham 'Bruce' LavenderNoch keine Bewertungen
- IMM MicroreactorsDokument90 SeitenIMM MicroreactorsGoutam GottumukkalaNoch keine Bewertungen
- MScHandbook Imperial Chemical EnggDokument87 SeitenMScHandbook Imperial Chemical EnggMurugeshNoch keine Bewertungen
- Method Computes PVT Properties For Gas CondensateDokument5 SeitenMethod Computes PVT Properties For Gas CondensateSergio FloresNoch keine Bewertungen
- Liquefaction Process EvaluationDokument15 SeitenLiquefaction Process Evaluationapi-3715370100% (1)
- Dehydration Plant Twenty Years PaperDokument5 SeitenDehydration Plant Twenty Years Papersejerson07Noch keine Bewertungen
- Chemical Reactor Design - NotesDokument4 SeitenChemical Reactor Design - NotesJanaki Devi ParratNoch keine Bewertungen
- Spe 97099 MS P PDFDokument20 SeitenSpe 97099 MS P PDFCarlos Alberto Mora100% (1)
- 03-23-16 NEWS - Indonesia Rejects Inpex's Mega Floating LNG Project, Wants To Stay Onshore - RigzoneDokument4 Seiten03-23-16 NEWS - Indonesia Rejects Inpex's Mega Floating LNG Project, Wants To Stay Onshore - RigzonehortalemosNoch keine Bewertungen
- 12-30-16 2016 A Year of ActionDokument3 Seiten12-30-16 2016 A Year of ActionhortalemosNoch keine Bewertungen
- 11-26-16 2017 General State Budget Unanimously Approved in GeneralityDokument1 Seite11-26-16 2017 General State Budget Unanimously Approved in GeneralityhortalemosNoch keine Bewertungen
- 11-26-16 2017 General State Budget Unanimously Approved in GeneralityDokument1 Seite11-26-16 2017 General State Budget Unanimously Approved in GeneralityhortalemosNoch keine Bewertungen
- 11-27-16 Happy Birthday Timor! Our Gift To You Is To Steal Your Oil, and Your Future - New MatildaDokument3 Seiten11-27-16 Happy Birthday Timor! Our Gift To You Is To Steal Your Oil, and Your Future - New MatildahortalemosNoch keine Bewertungen
- 03-15-16 ASIS Chief Nick Warner Slammed Over East Timor Spy ScandalDokument2 Seiten03-15-16 ASIS Chief Nick Warner Slammed Over East Timor Spy ScandalhortalemosNoch keine Bewertungen
- 03-24-16 The Morally Bankrupt Foreign Policy That Entrenches Australia's Reputation As A Regional Bully - Tom Clarke - Opinion - The GuardianDokument2 Seiten03-24-16 The Morally Bankrupt Foreign Policy That Entrenches Australia's Reputation As A Regional Bully - Tom Clarke - Opinion - The GuardianhortalemosNoch keine Bewertungen
- 11-22-16 RIGZONE - Singapore's SGX, Japan's TOCOM To Jointly Develop Asian LNG MarketDokument1 Seite11-22-16 RIGZONE - Singapore's SGX, Japan's TOCOM To Jointly Develop Asian LNG MarkethortalemosNoch keine Bewertungen
- East Timor Playing High-Stakes Gas Game: Vol. 6 / No. 138 / 20 July 2016Dokument8 SeitenEast Timor Playing High-Stakes Gas Game: Vol. 6 / No. 138 / 20 July 2016hortalemosNoch keine Bewertungen
- 07-15-16 Government Congratulates Prime Minister Elect of AustraliaDokument1 Seite07-15-16 Government Congratulates Prime Minister Elect of AustraliahortalemosNoch keine Bewertungen
- Pressure Rising On East Timor To Make A Deal: Vol. 6 / No. 225 / 21 November 2016Dokument9 SeitenPressure Rising On East Timor To Make A Deal: Vol. 6 / No. 225 / 21 November 2016hortalemosNoch keine Bewertungen
- Government Reaffirms Commitment To UNCLOS As The Constitution of The Sea'Dokument1 SeiteGovernment Reaffirms Commitment To UNCLOS As The Constitution of The Sea'Warren WrightNoch keine Bewertungen
- 07-27-17 RIGZONE - Indonesia President Joko Widodo Names Archandra Tahar As Energy MinisterDokument1 Seite07-27-17 RIGZONE - Indonesia President Joko Widodo Names Archandra Tahar As Energy MinisterhortalemosNoch keine Bewertungen
- 06-13-16 RIGZONE - Coming Wave of Gas Puts Focus On Finding New ShoresDokument2 Seiten06-13-16 RIGZONE - Coming Wave of Gas Puts Focus On Finding New ShoreshortalemosNoch keine Bewertungen
- 02-16-16 Estonia To East Timor - The 10 Best-Value Holiday Destinations For 2016 - BTDokument7 Seiten02-16-16 Estonia To East Timor - The 10 Best-Value Holiday Destinations For 2016 - BThortalemosNoch keine Bewertungen
- Engie Shelves Cameroon As LNG Moves Offshore: Vol. 6 / No. 134 / 14 July 2016Dokument9 SeitenEngie Shelves Cameroon As LNG Moves Offshore: Vol. 6 / No. 134 / 14 July 2016hortalemosNoch keine Bewertungen
- 02-15-16 How Far Is Fair Enough - New Moves in Timor Gap's 40-Plus Years of Boundary BattlesDokument3 Seiten02-15-16 How Far Is Fair Enough - New Moves in Timor Gap's 40-Plus Years of Boundary BattleshortalemosNoch keine Bewertungen
- One-Day Public Seminar - Maritime Boundaries in The Timor Sea: "Perspectives in International Law"Dokument14 SeitenOne-Day Public Seminar - Maritime Boundaries in The Timor Sea: "Perspectives in International Law"hortalemosNoch keine Bewertungen
- 02-12-17 Japan Remains Top LNG Shipowner, Reports VesselsValueDokument1 Seite02-12-17 Japan Remains Top LNG Shipowner, Reports VesselsValuehortalemosNoch keine Bewertungen
- Media Release: Landscape Is Changing For Development and Delivery of LNG Mega Projects - Shell Vice-PresidentDokument2 SeitenMedia Release: Landscape Is Changing For Development and Delivery of LNG Mega Projects - Shell Vice-PresidenthortalemosNoch keine Bewertungen
- 02-12-17 Japan Remains Top LNG Shipowner, Reports VesselsValueDokument1 Seite02-12-17 Japan Remains Top LNG Shipowner, Reports VesselsValuehortalemosNoch keine Bewertungen
- 02-17-16 The Guardian - Timor-Leste End Australia's "Shameful Policy"Dokument2 Seiten02-17-16 The Guardian - Timor-Leste End Australia's "Shameful Policy"hortalemosNoch keine Bewertungen
- East Timorese Government Welcomes Policy of The Australian Labor Party's To Establish Maritime BoundariesDokument1 SeiteEast Timorese Government Welcomes Policy of The Australian Labor Party's To Establish Maritime BoundariesWarren WrightNoch keine Bewertungen
- 02-17-16 Joint Media Release - Timor-Leste and ConocoPhillips Australia Settle Tax DisputesDokument1 Seite02-17-16 Joint Media Release - Timor-Leste and ConocoPhillips Australia Settle Tax DisputeshortalemosNoch keine Bewertungen
- BS 88 Sec2.2 - 1988Dokument21 SeitenBS 88 Sec2.2 - 1988hortalemosNoch keine Bewertungen
- BS 648 - 1964Dokument51 SeitenBS 648 - 1964hortalemosNoch keine Bewertungen
- BS 00341-3-2002Dokument33 SeitenBS 00341-3-2002Rachid TidjaniNoch keine Bewertungen
- Keys To Soil Taxonomy Tenth Edition 2006Dokument341 SeitenKeys To Soil Taxonomy Tenth Edition 2006hegegeNoch keine Bewertungen
- British Standard: A Single Copy of This British Standard Is Licensed ToDokument17 SeitenBritish Standard: A Single Copy of This British Standard Is Licensed TohortalemosNoch keine Bewertungen
- BS 88-4 Fuse Links - 1986Dokument43 SeitenBS 88-4 Fuse Links - 1986hortalemosNoch keine Bewertungen
- F2 IS Exam1 (04-05)Dokument9 SeitenF2 IS Exam1 (04-05)羅天佑Noch keine Bewertungen
- LuckyDokument61 SeitenLuckylucky tiwariNoch keine Bewertungen
- En 10083 C50 Steel Plate High Carbon SteelDokument2 SeitenEn 10083 C50 Steel Plate High Carbon SteelMatteo BuffoNoch keine Bewertungen
- IES 1992 - II Question PaperDokument10 SeitenIES 1992 - II Question PaperK.v.Singh0% (1)
- Guidance For The Storage Handling and Transportation of Solid Mineral Fertilizers - Fertilizers EuropeDokument84 SeitenGuidance For The Storage Handling and Transportation of Solid Mineral Fertilizers - Fertilizers EuropeLarry JorgensonNoch keine Bewertungen
- CLS JEEAD-19-20 XI Che Target-4 Level-1 Chapter-11Dokument14 SeitenCLS JEEAD-19-20 XI Che Target-4 Level-1 Chapter-11Vaibhav JaimanNoch keine Bewertungen
- MSDS C2H3ClDokument9 SeitenMSDS C2H3ClJ4MAmailNoch keine Bewertungen
- JEE Main 2019 Question Paper 12th Jan Morning PDFDokument24 SeitenJEE Main 2019 Question Paper 12th Jan Morning PDFDebi prasad jenaNoch keine Bewertungen
- Hot Applied Coal Tar Coatings PDFDokument155 SeitenHot Applied Coal Tar Coatings PDFmattarchiNoch keine Bewertungen
- Chemistry of MilkDokument44 SeitenChemistry of Milkabdouam50% (2)
- TFM 1600Dokument1 SeiteTFM 1600Rendi MulyadiNoch keine Bewertungen
- Virtual Lab For Methyl OrangeDokument9 SeitenVirtual Lab For Methyl OrangeKENT BENEDICT PERALESNoch keine Bewertungen
- Carbono 14Dokument3 SeitenCarbono 14Umeko NaraNoch keine Bewertungen
- Review Worksheet 1: Use The Diagram Below To Answer Questions 6-8Dokument2 SeitenReview Worksheet 1: Use The Diagram Below To Answer Questions 6-8Natalia LisekNoch keine Bewertungen
- Elements WorksheetDokument5 SeitenElements WorksheetmangayarkarasirNoch keine Bewertungen
- 9Dokument2 Seiten9woojaeNoch keine Bewertungen
- 100 Chemistry One Liner GK Questions EnglishDokument4 Seiten100 Chemistry One Liner GK Questions EnglishVignesh100% (1)
- Experimental Determination of Organic StructuresDokument11 SeitenExperimental Determination of Organic StructuresJochebed MirandaNoch keine Bewertungen
- The 3designers: Our 3D Printed Components From Carbon and Silicon CarbideDokument16 SeitenThe 3designers: Our 3D Printed Components From Carbon and Silicon CarbideGanda PrajaNoch keine Bewertungen
- Chemistry Class 12 ProjectGrapheneDokument14 SeitenChemistry Class 12 ProjectGrapheneKaran RMW100% (1)
- Scimich WQ TestDokument12 SeitenScimich WQ TestShita AlemieNoch keine Bewertungen
- H2S Dispersion ModelDokument52 SeitenH2S Dispersion Modelrsb_4192Noch keine Bewertungen
- Ethylene ProductionDokument34 SeitenEthylene ProductionWesleyNoch keine Bewertungen
- Unit 4 Carbon Materials Fo Stealth, Health and Energy NotesDokument12 SeitenUnit 4 Carbon Materials Fo Stealth, Health and Energy NotesSanjay KumarNoch keine Bewertungen
- Historical Review of Processing, Microstructures, and Mechanical Properties of PAN-Based Carbon FibersDokument20 SeitenHistorical Review of Processing, Microstructures, and Mechanical Properties of PAN-Based Carbon FibersFlor GINoch keine Bewertungen
- 5070 s14 QP 11Dokument16 Seiten5070 s14 QP 11Sr71Noch keine Bewertungen
- Environmental Engineering MCQs PDFDokument33 SeitenEnvironmental Engineering MCQs PDFZubair MalikNoch keine Bewertungen
- lectut-MTN-513-pdf-Structure of Crystalline CeramicsDokument53 Seitenlectut-MTN-513-pdf-Structure of Crystalline CeramicsAkash AgarwalNoch keine Bewertungen
- Tabla Railsback de ElementosDokument1 SeiteTabla Railsback de ElementosgeoremixNoch keine Bewertungen