Beruflich Dokumente
Kultur Dokumente
613208
Hochgeladen von
Tra ThanhCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
613208
Hochgeladen von
Tra ThanhCopyright:
Verfügbare Formate
APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Sept. 1995, p.
32083215
0099-2240/95/$04.0010
Copyright q 1995, American Society for Microbiology
Vol. 61, No. 9
Stereoselective Production of (1)-trans-Chrysanthemic Acid by
a Microbial Esterase: Cloning, Nucleotide Sequence,
and Overexpression of the Esterase Gene of
Arthrobacter globiformis in Escherichia coli
MASAKO NISHIZAWA,* MASATOSHI SHIMIZU, HIDEO OHKAWA,
AND MASAHARU KANAOKA
Biotechnology Laboratory, Takarazuka Research Center, Sumitomo
Chemical Co., Ltd., 4-2-1 Takatsukasa, Takarazuka,
Hyogo 665, Japan
Received 27 June 1994/Accepted 5 June 1995
The gene coding for a novel esterase which stereoselectively hydrolyzes the (1)-trans (1R,3R) stereoisomer
of ethyl chrysanthemate was cloned from Arthrobacter globiformis SC-6-98-28 and overexpressed in Escherichia
coli. The cellular content of the active enzyme reached 33% of the total soluble protein in the recombinant E.
coli JM105 cells and 5.6 g/liter of culture by high-density cell cultivation. The hydrolytic activity of the
recombinant E. coli cells for ethyl chrysanthemate reached 605 mmol of chrysanthemic acid per min per g of
dry cells, which is approximately 2,500-fold higher than that of A. globiformis cells. The stereoselective hydrolysis by the recombinant E. coli cells was efficient at substrate concentrations of up to 40% by removing the
produced chrysanthemic acid by ultrafiltration. The (1)-trans-chrysanthemic acid produced had 100% optical
purity. The amino acid sequence of the esterase was found to be similar to that of several class C b-lactamases,
D,D-carboxypeptidase, D-aminopeptidase, 6-aminohexanoate-dimer hydrolase, and Pseudomonas esterase. The
sequence comparison also suggested that the Ser-X-X-Lys motif in the esterase was at the active site of the
enzyme.
system, stereochemically pure (1)-trans-chrysanthemic acid
was produced at a substrate concentration of up to 40%.
Chrysanthemic acid [2,2-dimethyl-3-(2-methyl-1-propenyl)
cyclopropanecarboxylic acid] has been widely used as the
acidic part of synthetic pyrethroid insecticides (48) since it was
identified as the acidic moiety of pyrethrin, the naturally occurring insecticidal compound in Chrysanthemum cineariaefolium (42). Although the (1)-trans-configuration of chrysanthemic acid found in the pyrethrin is the most effective of the
four stereoisomers (48), the acidic part of pyrethroid insecticides are generally mixtures of all stereoisomers. Therefore, a
highly stereoselective enzymatic production of the (1)-trans
isomer would have a great advantage over the conventional
chemical preparation processes (2, 6, 26) not only in simplifying the processes but also in affording a pyrethroid with higher
specific activity.
As reported in a previous paper (32), we have purified and
characterized an esterase from Arthrobacter globiformis SC-698-28 which stereoselectively hydrolyzes (1)-trans-ethyl chrysanthemate. Despite the excellent stereospecificity, the extremely low content of this esterase in A. globiformis cells made
it difficult to use the enzyme in large-scale production of (1)trans-chrysanthemic acid for pyrethroid insecticides.
To overcome this problem, we cloned the esterase gene from
A. globiformis and overexpressed it in Escherichia coli cells. By
using the recombinant E. coli cells obtained by high-density cell
cultivation in combination with a hollow-fiber ultrafiltration
MATERIALS AND METHODS
Enzymes and reagents. Achromobacter protease I (25) (EC 3.4.21.50) was
purchased from Wako Pure Chemical Co., Osaka, Japan. Restriction endonucleases, modifying enzymes, and a DNA sequencing kit were from Takara Shuzo
Co., Ltd., Kyoto, Japan. [g-32P]ATP and [a-32P]dCTP were from Amersham
Japan, Tokyo, Japan. Isopropyl-b-D-thiogalactoside (IPTG) was from Nacalai
Tesque Inc., Kyoto, Japan. Phenolphthalein dibutyrate was obtained from Research Organics Inc., Cleveland, Ohio.
Bacterial strains and plasmids. A. globiformis SC-6-98-28 (32) was used as the
source of chromosomal DNA. E. coli HB101, JM101, JM103, JM105, and JM109
were used as the host cells for cloning and expression of the esterase gene. A
cloning vector, pUC19, and an expression vector, pKK223-3, were from Takara
Shuzo Co., Ltd., and Pharmacia LKB Biotechnology, respectively. pKK223-4 was
prepared from pKK223-3 by removing one of the two BamHI sites (33).
Amino acid sequencing. The esterase which stereoselectively hydrolyzes (1)trans-ethyl chrysanthemate was purified from A. globiformis cells (32). The purified sample was digested overnight with Achromobacter protease I (molar ratio
of 20:1) at 378C in 80 mM Tris-HCl (pH 8.5) containing 8.0 M urea and 100 mM
dithiothreitol. The Achromobacter protease I-digested peptide fragments were
applied to a m-Bondapack C4 column (4.6 by 150 mm; Nihon Waters Co.)
equilibrated with 0.1% trifluoroacetic acid and separated with an acetonitrile
gradient. Amino acid sequencing of the purified esterase and the digested peptide fragments was carried out with an Applied Biosystems model 470A protein
sequencer.
Synthesis of DNA probes and linkers. Oligodeoxyribonucleotides were synthesized on an Applied Biosystems model 380A DNA synthesizer. Degenerated
oligonucleotide probes I and II, both derived from N-terminal amino acid sequencing of the esterase, correspond to Met-1 through Ala-7 and Glu-23 through
Ser-28, respectively (see Fig. 2). Probe III corresponds to the sequence from 615
to 633 in Fig. 2. Nucleotide sequences of the synthesized probes are as follows:
* Corresponding author. Phone: 81-797-74-2140. Fax: 81-797-742133.
Present address: Department of Chemical and Biochemical Engineering, University College London, London WC1E 7JE, England.
Present address: Department of Plant Protection, Kobe University, Rokkodai, Nada, Kobe 657, Japan.
59
Probe I
3208
39
ATG GAT GCT CAA ACT ATT GC
C
C
G
C
C
A
A
A
G
G
VOL. 61, 1995
STEREOSELECTIVE ETHYL CHRYSANTHEMATE ESTERASE GENE
59
39
Probe II
GAA GAT CGT GAA TAT TC
G
C
C
G
C AG
A
G
AGA
G
Probe III
GATGCGGCCGCTGTGGAAG
The 59 ends of the synthetic oligonucleotide probes were labeled with [g-32P]
ATP and polynucleotide kinase.
Cloning and sequencing of the esterase gene. Chromosomal DNA of A. globiformis SC-6-98-28 was prepared by the method of Saito and Miura (39), except
that 0.3 U of penicillin G (Wako Pure Chemical Co.) per ml was added to the
culture when the turbidity at 660 nm reached 0.3 during A. globiformis
SC-6-98-28 cell cultivation (32). The chromosomal DNA was digested with
several restriction enzymes, and DNA fragments containing the portion of the
esterase gene were identified by Southern hybridization. The nucleotide sequence was determined by the dideoxynucleotide chain termination method
using [a-32P]dCTP.
Computer analysis. Amino acid sequence comparison was done with the
FASTA program (23) and the PRF (Protein Research Foundation, Osaka, Japan) and PIR (National Biomedical Research Foundation, Washington, D.C.)
databases. Clustal V (14) was used for multiple-sequence alignment.
Construction of the expression plasmid. Briefly, a synthetic BamHI-Nsp
(7524)V oligonucleotide fragment shown below and a 1.9-kbp Nsp(7524)VHindIII fragment from pAGE-1 were inserted into the multiple-cloning site of
pKK223-4 to give the expression plasmid pAGE-203:
BamHI
Nsp(7524)V
GATCCTTTTTTAATAAAATCAGGAGGAAAAAATATGGACGCACAGACCATCGCACCGGGCTT
GAAAAAATTATTTTAGTCCTCCTTTTTTATACCTGCGTGTCTGGTAGCGTGGCCCGAAGC
High-density cultivation of the recombinant E. coli cells. A fed-batch process
(47) was used for high-density cultivation of recombinant E. coli cells. E. coli
JM105 cells harboring pAGE-203 were precultivated at 308C in 100 ml of Luria
broth (24) containing sodium ampicillin. An overnight preculture of 80 ml was
inoculated into 800 ml of medium I, composed of 0.9 g of Casamino Acids per
liter, 0.6 g of yeast extract per liter, 30 g of glycerol per liter, 4.0 g of K2HPO4 per
liter, 4.0 g of KH2PO4 per liter, 7.0 g of Na2HPO4 z 12H2O per liter, 1.2 g of
(NH4)2SO4 per liter, 0.2 g of NH4Cl per liter, metal mixture (MgSO4 z 7H2O, 2.4
g/liter; CaCl2 z 2H2O, 0.04 g/liter; FeSO4 z 7H2O, 0.04 g/liter; ZnSO4 z 7H2O,
2.0 mg/liter; CuCl2 z 2H2O, 1.0 mg/liter; CoCl2 z 6H2O, 4.0 mg/liter; Na2MoO4 z
2H2O, 2.0 mg/liter; MnSO4 z nH2O, 0.01 g/liter; H3BO4, 0.5 mg/liter), 0.01 g of
thiamine-HCl per liter, and 0.05 g of sodium ampicillin per liter. The composition of the medium was a modification of the one reported by Mori et al. (30).
Cultivation was continued overnight under agitation at 900 rpm and aeration at
1.5 liter/liter of culture broth per min in a 2.6-liter jar fermentor (working
volume, 1.0 liter; type MD-250; Marubishi Bioengineering Co., Ltd.). IPTG was
added when the turbidity at 660 nm reached 60. During the cultivation, glycerol
and acetic acid concentrations were monitored by high-pressure liquid chromatography (HPLC) (Aminex HPX-87H column [7.8 by 300 mm; Bio-Rad]; mobile
phase, 6 mM H2SO4; temperature 608C), and medium II (Casamino Acids, 6.0
g/40 ml; yeast extract, 4.0 g/40 ml; and glycerol, 16 g/40 ml) was added so that the
concentration of acetic acid was kept to a minimum (47).
Ethyl chrysanthemate hydrolysis by recombinant E. coli cells. (i) Batch reaction. The reaction mixture for the batch reaction, composed of ethyl chrysanthemate [(1)-cis/(2)-cis/(1)-trans/(2)-trans ratio 5 2.5:2.5:47.5:47.5] or (1)trans-ethyl chrysanthemate dispersed in either 50.0 ml (see Table 1) or 25.0 ml
(for kinetic analysis) of glycine-NaOH buffer (200 mM), was continuously stirred
at 1,000 rpm. The reaction was started by adding cultured cells or the purified
esterase. During the reaction, the initial pH value was maintained with 1.0 N
NaOH. Unless otherwise mentioned, the reaction was done at pH 9.5 and 458C.
(ii) Scale-up reaction. The reaction mixture (400 ml), composed of 160 g of
ethyl chrysanthemate dispersed in 200 mM glycine-NaOH (pH 9.5 at 508C), was
continuously agitated at 1,000 rpm with a magnetic stirrer in a 1-liter threenecked round flask fitted with a pH electrode coupled to an automatic controller
(FC-10; Tokyo Rikakikai Co., Ltd.). The reaction was started by adding 3.2 g of
the cultured dry cells, and the pH was kept at 9.5 with 2.0 N NaOH throughout
the reaction. During the reaction, the amount of chrysanthemic acid produced
was monitored by sampling aliquots of the reaction mixture. The reaction mixture was filtered through a polyacrylnitrile hollow-fiber ultrafiltration membrane
(AIP-0013; 20 by 130 mm; Asahi Chemical Industry Co., Ltd.) with a peristaltic
pump (flow rate, 20 ml/min) and the acid that had been produced was removed.
After filtration, the residue was fed back into the flask and replenished with
glycine buffer, and the reaction was continued. Filtration was repeated four times
within 48 h. The filtered solution was acidified, and the chrysanthemic acid that
had been produced was extracted from the solution with methylisobutyl ketone.
After the reaction, unreacted ethyl chrysanthemate was extracted from the reaction mixture with methylisobutyl ketone.
3209
Purification of the esterase from recombinant E. coli JM105. The cultured
recombinant E. coli cells were disrupted three times by sonication (80 W for 5
min each) at 48C, the cell extract was centrifuged at 100,000 3 g for 60 min, and
the resulting supernatant was subjected to ammonium sulfate precipitation. The
precipitate obtained at a 30% saturation was dissolved in 20 mM Tris-HCl (pH
7.5) and subjected to DEAE-5PW ion-exchange chromatography as described
previously (32).
Analysis. Hydrolytic activity of the esterase toward (6)-cis,trans-ethyl chrysanthemate and phenolphthalein dibutyrate was measured as described previously (32). One unit of activity is defined as the amount of enzyme that produces
1 mmol of chrysanthemic acid per min at 458C. Stereoisomeric composition of the
chrysanthemic acid was analyzed by HPLC (32). The percent conversion is
defined as the molar ratio of the liberated (1)-trans-acid to (1)-trans-ester
contained in the substrate. Protein was measured with the bicinchoninic acid
protein assay kit (Pierce, Rockford, Ill.). Sodium dodecyl sulfate-polyacrylamide
gel electrophoresis (SDS-PAGE) was carried out as described by Laemmli (21),
and proteins were stained with Coomassie brilliant blue R-250. The isoelectric
point was measured with an isoelectric focusing PAGE gel (pH 3.0 to 7.0;
TEFCO Co., Ltd., Nagano, Japan) and with an isoelectric focusing calibration kit
(pH 2.5 to 6.5; Pharmacia LKB, Uppsala, Sweden) as a standard.
Nucleotide sequence accession number. The sequence data reported in this
paper have been submitted to GenBank and assigned the accession number
L22165.
RESULTS
Cloning and nucleotide sequence of the A. globiformis esterase gene. A 0.7-kbp KpnI-digested fragment (Fig. 1, pK12) was
identified by Southern hybridization with both probes I and II
as containing an N-terminal portion of the esterase coding
sequence. The rest of the esterase gene was identified on a
3.3-kbp EcoRI fragment (pE74) with probe III, which corresponded to a sequence between the EcoRI and KpnI sites (Fig.
1, solid box) in the 0.7-kbp KpnI fragment. A 1.7-kbp EcoRIHincII fragment (pEH16) was subcloned from the 3.3-kbp
fragment. A plasmid containing the full length of the esterase
coding region (pAGE-1) was constructed from pK12 and
pEH16.
The nucleotide sequence of the 2.2-kbp KpnI-HincII segment of pAGE-1 is shown in Fig. 2. One open reading frame
with GTG as the initiation codon, coding for a protein of 375
amino acids, was found at positions 211 to 1335. The molecular
weight deduced, 39,836, was approximately in accordance with
that of the esterase purified from A. globiformis cells (32). In
addition, amino acid sequences of the Achromobacter protease
I-digested fragments as well as the N terminus of the esterase
were found within this open reading frame (Fig. 2, underlined
regions). Either of the putative ribosome-binding sites located
upstream of the translation initiation codon (at position 192 or
200; doubly underlined in Fig. 2) complements only three or
four of six bases at the 39 end of the 16S rRNA of E. coli (12).
Unlike other Arthrobacter genes (1, 4, 35, 38), consensus 210
FIG. 1. Restriction map of the A. globiformis genomic DNA fragment containing the esterase gene. The hatched box indicates the open reading frame of
the esterase gene. The positions of the DNA probes used in cloning the esterase
coding region are indicated by open (probes I and II) and solid (probe III) boxes.
Subcloned fragments are shown with the corresponding plasmid names. B,
BamHI; E, EcoRI; H, HincII; K, KpnI; N, Nsp(7524)V; P, PstI.
3210
NISHIZAWA ET AL.
APPL. ENVIRON. MICROBIOL.
FIG. 2. Nucleotide sequence and the deduced amino acid sequence of the A. globiformis esterase gene. The 2.2-kbp region cloned in pAGE-1 is shown. The putative
ribosome-binding sites are doubly underlined. The termination codon is indicated by an asterisk. The underlined amino acid sequences are those determined by protein
sequencing of the purified esterase. The positions of the oligonucleotide probes used for cloning the gene are marked with dashed lines.
and 235 regions (28) were not obvious. No inverted-repeat
sequence for a possible transcription terminator with a sufficient DG value (kilocalories per mole) was found downstream
of the termination codon (TAG). The G1C content in the
esterase coding region was 65.3%, and a strong bias was noted
in the third position of the codon (G1C, 77.9%). Such a high
G1C content (56 to 66% for the A. globiformis group [1, 4, 35]
and 70 to 74% for the A. simplex group [38]) and a strong
preference for G or C in the wobble position of the codon are
the common characteristics of the genes previously cloned
from Arthrobacter species.
Amino acid sequence analysis of the esterase. Comparison
of the sequence of the esterase with proteins in databases
revealed significant similarities to the following enzymes:
b-lactamase of Enterobacter cloacae (11) (20.9% identity within
a 302-amino-acid [aa] region), b-lactamase of Citrobacter
freundii OS60 (22) (19.3% within 305 aa), ampC cephalosporinase of E. coli K-12 (16) (20.2% within 302 aa), and D,D-
carboxypeptidase of Streptomyces strain R61 (10) (26.7%
within 146 aa). In addition to these amide hydrolases, one
esterase showed sequence similarity, i.e., the esterase of
Pseudomonas sp. strain LS107d2 (29) (29.2% identity within
356 aa). A lesser degree of homology was also noted with
D-aminopeptidase of Ochrobactrum anthropi (3) (25.7% identity within 109 aa) and 6-aminohexanoate-dimer hydrolase of
Flavobacterium species (31) (31.0% within 61 aa). Multiplesequence alignment among the esterases, D,D-carboxypeptidase, and b-lactamases is shown in Fig. 3.
The consensus sequence of Ser-X-X-Lys, perfectly conserved among all the penicillin-recognizing enzymes found so
far (5, 9, 17), surrounding the active-site serine residue (Ser
residue in this motif) was found at positions 59 to 62 in the
esterase (Fig. 3, reversed font). Weak homology was observed
between another conserved motif of Lys-Thr/Ser-Gly in the
C-terminal portion of penicillin-recognizing enzymes and the
corresponding region of the esterase (Fig. 3, Lys-Ser-His at 310
VOL. 61, 1995
STEREOSELECTIVE ETHYL CHRYSANTHEMATE ESTERASE GENE
to 312, reversed font). However, neither overall similarity to
other carboxylesterases, lipases, or proteases known to date
nor the consensus sequence of Gly-X-Ser-X-Gly around the
active-site serine residue of these enzymes (5) was found in the
A. globiformis esterase.
Expression of the esterase gene in E. coli. An expression
plasmid was constructed to overexpress the esterase, as described in Materials and Methods, since no esterase activity
was observed in E. coli JM109 cells carrying pAGE-1. E. coli
JM109 cells were transformed with the expression plasmid
pAGE-203, which contained a linker DNA corresponding to
the upstream region and initial 10 aa. The recombinant E. coli
cells were cultivated in Terrific Broth (24) at 378C, and esterase production was induced by 1 mM IPTG. A potent ethyl
chrysanthemate-hydrolyzing activity was observed with the recombinant E. coli cells, whereas no such activity was observed
with the E. coli cells transformed with the vector plasmid,
pKK223-4. Cellular esterase activity of 375 U/g of dry cells was
found at pH 9.5 and 458C, while Arthrobacter strain SC-6-98-28
cells had an activity of 0.238 U/g of dry cells under the same
conditions. Thus, the cellular activity of JM109/pAGE-203
reached approximately 1,600-fold that of A. globiformis. The
(1)-trans stereoisomer was produced exclusively by the recombinant E. coli cell reaction. Recombinant E. coli JM101,
JM103, JM105, and JM109, all of which contained pAGE-203,
were compared for the expression of the esterase. JM105 and
JM109 exhibited the highest expression level (data not shown).
High-density fed-batch cell cultivation. Temperature, pH,
concentration of IPTG, and the feeding rate of medium II
were optimized to produce the esterase in E. coli JM105/
pAGE-203 by high-density fed-batch cultivation.
The effect of temperature on the esterase production is
shown in Fig. 4. A protein corresponding to the esterase was
localized almost exclusively in the soluble fraction when the
cells were cultivated at 338C or lower temperatures, whereas at
higher temperatures, some of the proteins were found in the
precipitate fraction (Fig. 4, lanes P). The esterase activity recovered correlated well with the amount of protein in the
soluble fraction (lanes S; 77.5 U/g of dry cells at 258C, 605 U/g
at 308C, 533 U/g at 338C, 405 U/g at 378C, and no activity at
428C). No esterase activity was observed in the precipitate
fraction.
The optimized conditions were as follows: pH 7.5, 308C, 1.0
mM IPTG, feeding of medium II started when the turbidity at
660 nm reached 60 at a feeding rate of 14 ml/h. Other details
of the cultivation conditions are described in Materials and
Methods. As a result, a final cell density of 104 g of dry cells per
liter of broth with an activity of 605 U/g of dry cells was
achieved, which was 2,500-fold that of Arthrobacter cells. Almost all of the active enzyme that accumulated in the cytoplasmic fraction remained undigested during overnight cultivation.
The stability of the plasmid in E. coli cells was more than 95%.
The esterase activity obtained was 62.9 U/ml by high-density
cell cultivation but 2.1 U/ml by batch cultivation (final cell
density, 5.6 g of dry cells per liter; activity, 375 U/g of dry cells).
Thus, a 30-fold increase in the activity was achieved by the
high-density cell cultivation.
Large-scale production of (1)-trans-chrysanthemic acid by
recombinant E. coli cells. Conditions of stereoselective hydrolysis of ethyl chrysanthemate by cultivated E. coli cells were
examined. Since earlier investigations showed that the purified
enzyme was most active around pH 10.0 and 458C (32), similar
conditions were examined. As shown in Table 1, the optimum
pH and temperature were 9.5 at 508C and more than 98%
conversion occurred within 6 h at substrate concentration of
10%. The optical purity of the produced chrysanthemic acid
3211
TABLE 1. Effects of reaction conditions on enzymatic hydrolysis of
(6)-cis,trans-ethyl chrysanthemate by the recombinant E. coli cellsa
Substrate
concn (%)
pH
Temp
(8C)
Yield of
acid (g)
Conversion
(%)
Stereoisomeric
ratio (%)b
10
10
10
10
10
10
15c
20c
30c
40c
9.0
9.5
10.0
9.5
9.5
9.5
9.5
9.5
9.5
9.5
45
45
45
50
55
60
50
50
50
50
1.61
1.79
1.18
2.00
1.87
1.61
1.95
1.87
1.89
1.71
79
88
58
98
92
79
64
46
31
21
NT
NT
100:0:0:0
100:0:0:0
99.3:0.7:0:0
99.2:0.8:0:0
NT
NT
NT
NT
a
Reactions were done on a 50-ml scale. Cultured cells obtained by highdensity cell cultivation were used in each experiment. Reactions were started by
the addition of 62.5 U (1.0-ml culture broth equivalent) of the cells and continued for 6 h.
b
(1)-trans/(1)-cis/(2)-trans/(2)-cis-chrysanthemic acid ratio. NT, not tested.
c
Cells (125 U) were added at 6 h, and the reaction was continued for another
24 h.
decreased at higher temperatures. At substrate concentrations
of 15% or higher, hydrolysis of the (1)-trans stereoisomer was
arrested when up to 2.0 g of (1)-trans-chrysanthemic acid (230
mM) had accumulated in the reaction mixture and was not
completed even if sufficient numbers of cells were fed to the
reaction mixture (Table 1). From Lineweaver-Burk analysis
with the purified esterase, (1)-trans-chrysanthemic acid was
found to inhibit the enzymatic activity to (1)-trans-ethyl chrysanthemate competitively with an apparent Ki of 7.1 mM. Furthermore, the solubility of ethyl chrysanthemate in 200 mM
glycine-NaOH (pH 9.5) at 508C is approximately 20 mM, while
the chrysanthemic acid that is produced is freely soluble.
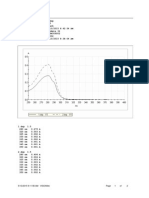
Therefore, the acid is present in increasing excess as the reaction proceeds. A scale-up reaction was done at 40% substrate
concentration with a hollow-fiber ultrafiltration membrane to
remove the produced chrysanthemic acid (Fig. 5). Substrate
ester was trapped with E. coli cells in the reaction mixture
because of its hydrophobicity. Filtration was repeated four
times during the 48-h reaction. As a result, the reaction proceeded almost uninterrupted even at ester concentrations as
high as 40%, and the conversion rate reached 70.0% in 48 h
(Fig. 5). The conversion rate remained 13.0%, however, if the
reaction mixture was not filtered (Fig. 5). The optical purity of
the (1)-trans-chrysanthemic acid produced was over 99.9%.
Residual ethyl chrysanthemate recovered from the reaction
mixture was 96.2 g, and thus the total recovery of acid and ester
was 90.1%. Unreacted substrate, thus recovered from the reaction mixture, can be chemically racemized (45) and recycled
in the hydrolytic reaction.
Purification and characterization of the recombinant esterase. The esterase was purified from E. coli JM105 harboring
pAGE-203, as summarized in Table 2. The DEAE-5PW fraction, which had a specific activity of 52.4 U/mg, was confirmed
to be pure by SDS-PAGE analysis (Fig. 6). On the basis of the
catalytic activity of the purified esterase, the expression level of
the enzyme in the recombinant E. coli JM105/pAGE-203 was
estimated to be 33.0% of the total soluble proteins. That is, the
production level of the active esterase reached 5.6 g/liter of
broth.
The recombinant esterase had enzymatic and physicochemical properties identical to those of the enzyme from A. globiformis (32): the Vmax and Km for (1)-trans-ethyl chrysanthemate were 8.7 mmol/min/mg and 3.3 mM, respectively;
3212
NISHIZAWA ET AL.
APPL. ENVIRON. MICROBIOL.
FIG. 3. Multiple amino acid sequence alignment of A. globiformis esterase, class C b-lactamases, Pseudomonas esterase, and D,D-carboxypeptidase. K12, E. coli K-12
cephalosporinase ampC (16); P99, Enterobacter cloacae b-lactamase (11); CF, Citrobacter freundii OS60 b-lactamase (22); EST, A. globiformis esterase (this study); PSE,
Pseudomonas sp. strain LS107d2 esterase (29); R61, Streptomyces R61 D,D-carboxypeptidase (10). The mature portions of the proteins are aligned. Conserved regions
are hatched. The residues forming the active site of b-lactamases and D,D-carboxypeptidase suggested from crystallographic structures are in reversed font. Dashed lines
indicate gaps introduced for better alignment. Numbers above the lines denote amino acid positions of the A. globiformis esterase. Asterisks denote amino acids
perfectly conserved in all six proteins, and dots denote well-conserved amino acids.
VOL. 61, 1995
STEREOSELECTIVE ETHYL CHRYSANTHEMATE ESTERASE GENE
3213
TABLE 2. Purification of the recombinant esterase
from E. coli JM105 cells
Purification stepa
Activity
(U)b
Yield
(%)
Amt of
protein (mg)
Sp. act.
(U/mg)
Cell extract
(NH4)2SO4 (0 to 30%)
DEAE-5PW
2,922
1,596
1,566
100.0
54.6
53.6
168.8
46.9
29.9
17.3
34.0
52.4
a
Steps carried out with 10.0 ml of culture broth obtained from a high-density
cell cultivation experiment.
b
Enzyme activity was measured with phenolphthalein dibutyrate as a substrate.
FIG. 4. SDS-PAGE analysis of the proteins produced in the recombinant E.
coli cells by high-density cultivation. Localization of the proteins produced in the
recombinant E. coli JM105/pAGE-203 cells cultivated at high density and various
temperatures is shown. Cells were disrupted by sonication and fractionated by
centrifugation at 10,000 3 g for 10 min. Lanes: S1P, total cellular fraction
(sonicated); S, supernatant; P, precipitate. In each lane, 24 mg of the cultured dry
cells or the fractions derived therefrom was applied. Other cultivation conditions: pH 7.5; induction by 1.0 mM IPTG; feeding rate of medium II, 14 ml/h.
The arrowhead indicates the position of the esterase.
isoelectric point, 5.2. No reduction of specific activity was observed when the esterase was highly expressed in E. coli cells.
DISCUSSION
We have cloned the gene encoding the esterase from A.
globiformis, which has ethyl chrysanthemate-hydrolyzing activity with excellent stereospecificity for the (1)-trans stereoisomer. Probably because of the sequence features of the esterase
in the 59-flanking region, as mentioned in Results, E. coli cells
FIG. 5. Production of (1)-trans-chrysanthemic acid by recombinant E. coli
JM105/pAGE-203 cells with the ultrafiltration system. The time course of the
production of (1)-trans-chrysanthemic acid is shown. The reaction mixture (400
ml) contained 160 g of the substrate (6)-cis,trans ester dispersed in 200 mM
glycine-NaOH (pH 9.5, 508C) (40% [wt/vol] concentration) and 3.2 g of the dry
cells (activity, 605 U/g of dry cells) obtained by high-density cell cultivation. At
8.0, 23.5, 28.5, and 48 h (indicated by arrows), the reaction mixture was filtered.
F, with filtration; E, without filtration.
carrying pAGE-1 did not show esterase activity. Therefore, to
improve the expression level of the esterase, a synthetic linker
corresponding to the upstream and translational initiation region was constructed as follows. (i) The initiation codon GTG
was replaced with ATG, since GTG is a rare initiation codon
in E. coli and an improved expression level was reported when
the initiation codon GTG or TTG was replaced by ATG (27,
46). (ii) The nucleotides between the Shine-Dalgarno sequence and the initiation codon were changed according to
those of highly expressed genes of human growth factor and
human single-chain urokinase-like plasminogen activator (8,
44), in which A-rich sequences were introduced. (iii) An ATrich sequence was introduced upstream of the Shine-Dalgarno
sequence, since this had been found to improve the heterologous protein expression in E. coli cells (40). (iv) Codon usage
preferred in E. coli was used (15). As a consequence, E. coli
cells harboring the expression plasmid pAGE-203, constructed
in this way, exhibited potent inducible ethyl chrysanthematehydrolyzing activity. As a result of the optimization of the
esterase production by high-density cell cultivation, the production level of the esterase in E. coli JM105/pAGE-203
reached 33.0% of that of the total soluble protein. Since the
content of the esterase in A. globiformis was about 0.016% of
the total soluble fraction (32), an approximately 2,000-fold
FIG. 6. SDS-PAGE analysis of the fractions from the purification step. The
esterase was purified from E. coli JM105/pAGE-203 cells cultivated at high
density under optimized conditions. Lanes: 1 to 3, each purification step in Table
2; M, molecular weight standards (phosphorylase b, 92,500; bovine serum albumin, 66,700; ovalbumin, 45,000; carbonic anhydrase, 31,000; soybean trypsin
inhibitor, 21,500; lysozyme, 14,400). In each lane, 2.0 mg of proteins was applied.
3214
NISHIZAWA ET AL.
increase in production of active enzyme in the bacterial cells
was achieved by recombinant DNA technology and high-density cultivation of the transformed E. coli cells.
Since the esterase reported here is the only enzyme found so
far which perfectly distinguishes stereoisomers of (6)-cis,transethyl chrysanthemate (36, 41), the recombinant E. coli cells
producing the esterase or the esterase obtained from the cell
extract should be suitable for a manufacturing process for
optically active pyrethroid insecticides. Thermostability (32)
and accumulation of the enzyme as a major component in the
cytoplasmic fraction of E. coli cells should be a great advantage
for use in the industrial process. On account of its stability,
immobilized esterase might be optimal for continuous (1)trans-chrysanthemic acid production in an enzyme reactor.
As a result of our earlier investigation (32), we classified A.
globiformis esterase as a carboxylesterase with broad substrate
specificity. In this study, however, the esterase was found to
have high sequence homology to several members of penicillinrecognizing enzymes (Fig. 3), all of which are amide bond
hydrolases, in addition to the esterase of Pseudomonas sp. (29).
For the following reasons, we presume the A. globiformis esterase to be a unique enzyme which has the characteristics of
the penicillin-recognizing enzymes as well as its ester-hydrolyzing activity. First, the motif Ser-X-X-Lys (Fig. 3, reversed
font) in the esterase is suggested to participate in the active site
because (i) this consensus motif has been shown to form a part
of the active-site cavity of class A and C b-lactamases and
D,D-carboxypeptidase by crystallographic studies (19, 20, 34,
43); (ii) the studies on the sensitivity of the esterase to some
inhibitors such as diisopropylfluorophosphate and phenylmethylsulfonyl fluoride (32) suggest that the esterase has a
serine residue in its active site, and comparison of the esterase
sequence with these homologous enzymes shows that Ser-59
could be the catalytic residue (Fig. 3); (iii) the esterase activity
was found to be inhibited by the substrates of b-lactamases and
D,D-carboxypeptidase in a competitive manner, although no
hydrolytic activity toward these substrates was observed (data
not shown); and (iv) b-lactamases and D,D-carboxypeptidase
were reported to have some ester-hydrolyzing activity (18),
although ethyl chrysanthemate was not hydrolyzed by b-lactamases of Bacillus cereus or Enterobacter cloacae (data not
shown). In addition, the secondary-structure profile (7) predicted for the esterase was similar to that of class C (34) and
class A (13, 43) b-lactamases and D,D-carboxypeptidase (19,
20). Although little overall sequence homology was found
among these penicillin-recognizing enzymes, striking similarities were observed in the three-dimensional structures and the
mechanism of the lactam hydrolysis (18). From these arguments, we assume that the spatial arrangement around the
active site of the A. globiformis esterase is similar to that in
these penicillin-recognizing enzymes and so is the catalytic
mechanism. Further studies are now being done to identify the
essential residues for ester hydrolysis. Site-directed mutagenesis should provide other interesting information enabling us
to understand the molecular mechanism of the hydrolysis. It is
also of great interest to elucidate how this esterase recognizes
one stereoisomer so strictly.
Asano et al. screened for microorganisms with D-aminopeptidase activity and isolated Arthrobacter sp. and Ochrobactrum
anthropi. The gene responsible for the activity was cloned from
O. anthropi and found to have an amino acid sequence homology to D,D-carboxypeptidase and b-lactamases (3). The esterase we cloned also has a sequence homology to this D-aminopeptidase, although to a lesser degree than to the penicillinrecognizing enzymes. Recently, another Arthrobacter species
with D-alaninamide hydrolase activity was found, and the en-
APPL. ENVIRON. MICROBIOL.
zyme was purified (37). The esterase we cloned also showed
hydrolytic activity to D-alaninamide (unpublished data). These
findings suggest the possibility that Arthrobacter species and
other related species have enzymes that evolved from a common ancestor and are responsible for cell wall peptidoglycan
synthesis or other analogous functions.
ACKNOWLEDGMENTS
We are grateful to Y. Yabusaki and Henrik Jrgensen, Sumitomo
Chemical Co. Ltd., for stimulating discussions.
REFERENCES
1. Anderton, T. L., P. C. Shaw, M. K. Shin, and B. S. Hartley. 1991. D-Xylose
isomerase from Arthrobacter strain N.R.R.L. B3728. Biochem. J. 277:263
271.
2. Aratani, T., Y. Yoneyoshi, and T. Nagase. 1975. Asymmetric synthesis of
chrysanthemic acid. An application of copper carbenoid reaction. Tetrahedron Lett. 21:17071710.
3. Asano, Y., Y. Kato, A. Yamada, and K. Kondo. 1992. Structural similarity of
D-aminopeptidase to carboxypeptidase DD and b-lactamases. Biochemistry
31:23162328.
4. Brandsch, R., A. E. Hinkkanen, L. Mauch, H. Nagursky, and K. Decker.
1987. 6-Hydroxy-D-nicotine oxidase of Arthrobacter oxidans. Eur. J. Biochem.
167:315320.
5. Brenner, S. 1988. The molecular evolution of genes and proteins: a tale of
two serines. Nature (London) 334:528530.
6. Campbell, I. G. M., and S. H. Harper. 1952. The chrysanthemumcarboxylic
acids. IV. Optical resolution of the chrysanthemic acids. J. Sci. Food Agric.
3:189192.
7. Chou, P. Y., and G. D. Fasman. 1978. Prediction of the secondary structure
of proteins from their amino acid sequence. Adv. Enzymol. 47:45148.
8. Dalbge, H., S. Carlsen, E. B. Jensen, T. Christensen, and H. H. M. Dahl.
1988. Expression of recombinant growth hormone in Escherichia coli: effect
of the region between the Shine-Dalgarno sequence and the ATG initiation
codon. DNA 7:399405.
9. Dale, J. W., D. Godwin, D. Mossakowska, P. Stephenson, and S. Wall. 1985.
Sequence of the OXA2 b-lactamase: comparison with other penicillin-reactive enzymes. FEBS Lett. 191:3944.
10. Duez, C., C. P. Fraipont, B. Joris, J. Dusart, M. S. Urdea, J. A. Martial, J. M.
Frere, and J. M. Ghuysen. 1987. Primary structure of the Streptomyces R61
extracellular DD-peptidase. Eur. J. Biochem. 162:509518.
11. Galleni, M., F. Lindberg, S. Normark, S. Cole, N. Honore, B. Joris, and J. M.
Frere. 1988. Sequence and comparative analysis of three Enterobacter cloacae ampC b-lactamase genes and their products. Biochem. J. 250:753760.
12. Gold, L., and G. D. Stormo. 1987. Translation initiation, p. 13021307. In
F. C. Neidhardt, J. L. Ingraham, K. B. Low, B. Magasanik, M. Schaechter,
and H. E. Umbarger (ed.), Escherichia coli and Salmonella typhimurium:
cellular and molecular biology. American Society for Microbiology, Washington, D.C.
13. Herzberg, O., and J. Moult. 1987. Bacterial resistance to b-lactam antibiotics: crystal structure of b-lactamase from Staphylococcus aureus PC1 at 2.5
resolution. Science 236:694701.
14. Higgins, D. G., A. J. Bleasby, and R. Fuchs. 1992. Clustal V: improved
software for multiple sequence alignment. Comput. Appl. Biosci. 8:189191.
15. Ikemura, T. 1981. Correlation between the abundance of Escherichia coli
transfer RNAs and the occurrence of the respective codons in its protein
genes: a proposal for a synonymous codon choice that is optimal for the E.
coli translational system. J. Mol. Biol. 151:389409.
16. Jaurin, B., and T. Grundstro
m. 1981. ampC cephalosporinase of Escherichia
coli K-12 has a different evolutionary origin from that of b-lactamases of the
penicillinase type. Proc. Natl. Acad. Sci. USA 78:48974901.
17. Joris, B., J. M. Ghuysen, G. Dive, A. Renard, O. Dideberg, P. Charlier, J. M.
Frere, J. A. Kelly, J. C. Boyington, P. C. Moews, and J. R. Knox. 1988. The
active-site-serine penicillin-recognizing enzymes as members of the Streptomyces R61 DD-peptidase family. Biochem. J. 250:313324.
18. Kelly, J. A., O. Dideberg, P. Charlier, J. P. Wery, M. Libert, P. C. Moews,
J. R. Knox, C. Duez, C. Fraipont, B. Joris, J. Dusart, J. M. Frere, and J. M.
Ghuysen. 1986. On the origin of bacterial resistance to penicillin: comparison of a b-lactamase and a penicillin target. Science 231:14291431.
19. Kelly, J. A., J. R. Knox, P. C. Moews, G. J. Hite, J. B. Bartolone, and H.
Zhao. 1985. 2.8- structure of penicillin-sensitive D-alanyl carboxypeptidasetranspeptidase from Streptomyces R61 and complexes with b-lactams. J. Biol.
Chem. 260:64496458.
20. Kelly, J. A., J. R. Knox, and H. Zhao. 1989. Crystallographic mapping of
b-lactams bound to a D-alanyl-D-alanine peptidase target enzyme. J. Mol.
Biol. 209:281295.
21. Laemmli, U. K. 1970. Cleavage of structural proteins during the assembly of
the head of bacteriophage T4. Nature (London) 227:680685.
22. Lindberg, F., and S. Normark. 1986. Sequence of the Citrobacter freundii
VOL. 61, 1995
23.
24.
25.
26.
27.
28.
29.
30.
31.
32.
33.
34.
35.
STEREOSELECTIVE ETHYL CHRYSANTHEMATE ESTERASE GENE
OS60 chromosomal ampC b-lactamase gene. Eur. J. Biochem. 156:441
445.
Lipman, D. J., and W. R. Pearson. 1985. Rapid and sensitive protein similarity searches. Science 227:14351441.
Maniatis, T., E. F. Fritsch, and J. Sambrook. 1982. Molecular cloning: a
laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor,
N.Y.
Masaki, T., T. Fujihashi, K. Nakamura, and M. Soejima. 1981. Studies on a
new proteolytic enzyme from Achromobacter lyticus M497-1. Biochim. Biophys. Acta 660:5155.
Matsuo, T., K. Mori, and M. Matsui. 1976. Synthesis of optically active
trans-chrysanthemic acid from optically active pantolactone. Tetrahedron
Lett. 23:19791982.
Mauch, L., V. Bichler, and R. Brandsch. 1990. Functional analysis of the 59
regulatory region and the UUG translation initiation codon of the Arthrobacter oxidans 6-hydroxy-D-nicotine oxidase gene. Mol. Gen. Genet. 221:
427434.
McClure, W. R. 1985. Mechanism and control of transcription initiation in
prokaryotes. Ann. Rev. Biochem. 54:171204.
McKay, D. B., M. P. Jennings, E. A. Godfrey, I. C. MacRae, P. J. Rogers, and
I. R. Beacham. 1992. Molecular analysis of an esterase-encoding gene from
a lipolytic psychrotrophic pseudomonad. J. Gen. Microbiol. 138:701708.
Mori, H., T. Yano, T. Kobayashi, and S. Shimizu. 1979. High density cultivation of biomass in fed-batch system with DO-stat. J. Chem. Eng. 12:313
319.
Negoro, S., T. Taniguchi, M. Kanaoka, H. Kimura, and H. Okada. 1983.
Plasmid-determined enzymatic degradation of nylon oligomers. J. Bacteriol.
155:2231.
Nishizawa, M., H. Gomi, and F. Kishimoto. 1993. Purification and some
properties of carboxylesterase from Arthrobacter globiformis: stereoselective
hydrolysis of ethyl chrysanthemate. Biosci. Biotechnol. Biochem. 57:594
598.
Oeda, K., K. Oshie, M. Shimizu, K. Nakamura, H. Yamamoto, I. Nakayama,
and H. Ohkawa. 1987. Nucleotide sequence of the insecticidal protein gene
of Bacillus thuringiensis strain aizawai IPL7 and its high-level expression in
Escherichia coli. Gene 53:113119.
Oefner, C., A. DArcy, J. J. Daly, K. Gubernator, R. L. Charnas, I. Heinze,
C. Hubschwerlen, and F. K. Winkler. 1990. Refined crystal structure of
b-lactamase from Citrobacter freundii indicates a mechanism for b-lactam
hydrolysis. Nature (London) 343:284288.
Okushima, M., D. Sugino, Y. Kouno, S. Nakano, J. Miyahara, H. Toda, S.
Kubo, and A. Matsushiro. 1991. Molecular cloning and nucleotide sequenc-
36.
37.
38.
39.
40.
41.
42.
43.
44.
45.
46.
47.
48.
3215
ing of the Arthrobacter dextranase gene and its expression in Escherichia coli
and Streptococcus sanguis. Jpn. J. Genet. 66:173187.
Oritani, T., and K. Yamashita. 1975. Microbial resolution of some racemic
monocyclic alcohols. Agric. Biol. Chem. 39:8996.
Ozaki, A., H. Kawasaki, M. Yagasaki, and Y. Hashimoto. 1992. Enzymatic
production of D-alanine from DL-alaninamide by novel D-alaninamide specific amide hydrolase. Biosci. Biotechnol. Biochem. 56:19801984.
Roberts, A. N., G. S. Hudson, and S. Brenner. 1985. An erythromycinresistance gene from an erythromycin-producing strain of Arthrobacter sp.
Gene 35:259270.
Saito, H., and K. Miura. 1963. Preparation of transforming deoxyribonucleic
acid by phenol treatment. Biochim. Biophys. Acta 72:619629.
Scherer, G. F. E., M. D. Walkinshaw, S. Arnott, and D. J. Morre. 1980. The
ribosome binding sites recognized by E. coli ribosomes have regions with
signal character in both the leader and protein coding segments. Nucleic
Acids Res. 8:38953905.
Schneider, M., N. Engel, and H. Boensmann. 1984. Enzymatic synthesis of
chiral building blocks from racemates: preparation of (1R,3R)-chrysanthemic, -permethrinic and -caronic acids from racemic, diastereomeric mixtures. Angew. Chem. Int. Ed. Engl. 23:6466.
Staudinger, H., and L. Ruzicka. 1924. Insektento
tende Stoffe I: u
ber Isolierung und Konstitution des wirksamen Teiles des dalmatinischen Insektenpulvers. Helv. Chim. Acta 7:177259.
Strynadka, N. C. J., H. Adachi, S. E. Jensen, K. Johns, A. Sielecki, C. Betzel,
K. Sutoh, and M. N. G. James. 1992. Molecular structure of the acyl-enzyme
intermediate in b-lactam hydrolysis at 1.7 resolution. Nature (London)
359:700705.
Surek, B., M. Wilhelm, and W. Hillen. 1991. Optimizing the promoter and
ribosome binding sequence for expression of human single chain urokinaselike plasminogen activator in Escherichia coli and stabilization of the product
by avoiding heat shock response. Appl. Microbiol. Biotechnol. 34:488494.
Suzukamo, G., M. Fukao, K. Hagiya, and Y. Sakito. 1989. Interconversion of
optical isomers of chrysanthemic acid and related reactions (II). Sumitomo
Chem. II:3542. (In Japanese.)
Watanabe, K., K. Murata, and A. Kimura. 1987. Effect of a change of
unusual initiation codon TTG to ATG by site-directed mutagenesis on g-glutamylcysteine synthetase gene expression. Agric. Biol. Chem. 51:11111114.
Yee, L., and H. W. Blanch. 1992. Recombinant protein expression in high cell
density fed-batch cultures of Escherichia coli. Bio/Technology 10:15501557.
Yoshioka, H., and J. Miyamoto. 1976. Synthetic Pyrethroids (1). Chem. Biol.
14:427434. (In Japanese.)
Das könnte Ihnen auch gefallen
- Tankguard Holding Primer: Technical DataDokument4 SeitenTankguard Holding Primer: Technical DataTra ThanhNoch keine Bewertungen
- Tankguard Holding Primer: Technical DataDokument4 SeitenTankguard Holding Primer: Technical DataTra ThanhNoch keine Bewertungen
- WelcomeDokument5 SeitenWelcomeCatNoch keine Bewertungen
- Material and Energy Balance in Industrial PlantDokument26 SeitenMaterial and Energy Balance in Industrial Plantconan45Noch keine Bewertungen
- Tankguard Holding Primer: Technical DataDokument4 SeitenTankguard Holding Primer: Technical DataTra ThanhNoch keine Bewertungen
- Material and Energy Balance in Industrial PlantDokument26 SeitenMaterial and Energy Balance in Industrial Plantconan45Noch keine Bewertungen
- 2 1 5/13/2015 9:11:56 Am Visionlite ofDokument2 Seiten2 1 5/13/2015 9:11:56 Am Visionlite ofTra ThanhNoch keine Bewertungen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5784)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (399)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (890)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (587)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (265)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (72)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2219)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (119)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Book of Abstracts of Symposium Post-Transcriptional Gene Regulation in PlantsDokument82 SeitenBook of Abstracts of Symposium Post-Transcriptional Gene Regulation in PlantsvTsygankova_96587182Noch keine Bewertungen
- The Road To ModularityDokument11 SeitenThe Road To ModularityGiovanni DummondNoch keine Bewertungen
- TB240VMDokument2 SeitenTB240VMwajeeornoNoch keine Bewertungen
- Assignment-2 XII Molecular Basis of InheritanceDokument2 SeitenAssignment-2 XII Molecular Basis of InheritanceJai Bharat0% (1)
- Textbook of Personalized Medicine, 2nd EditionDokument763 SeitenTextbook of Personalized Medicine, 2nd EditionNamık Korkmaz100% (6)
- Gene Expression and Regulation: Building To The Performance ExpectationsDokument14 SeitenGene Expression and Regulation: Building To The Performance ExpectationsCaleb Correa GarciaNoch keine Bewertungen
- BioChemistry Syllabus and MQPs 08012018Dokument33 SeitenBioChemistry Syllabus and MQPs 08012018rupaNoch keine Bewertungen
- Pinoresinol-Lariciresinol Reductases, Key To The Lignan Synthesis in PlantsDokument20 SeitenPinoresinol-Lariciresinol Reductases, Key To The Lignan Synthesis in PlantsGastón Bravo ArrepolNoch keine Bewertungen
- Introduction To Bacterial GeneticsDokument24 SeitenIntroduction To Bacterial GeneticseyoelNoch keine Bewertungen
- Molecular Biology of The Cell, Sixth Edition Chapter 7: Control of Gene ExpressionDokument36 SeitenMolecular Biology of The Cell, Sixth Edition Chapter 7: Control of Gene ExpressionIsmael Torres-Pizarro100% (2)
- GRE Biology Subject Test (For Test Takers)Dokument9 SeitenGRE Biology Subject Test (For Test Takers)uttam.micro404Noch keine Bewertungen
- Advances in Agriculture SciencesDokument23 SeitenAdvances in Agriculture SciencesLeajen CampoNoch keine Bewertungen
- #16 Ch21 SplicingDokument31 Seiten#16 Ch21 SplicingKyle BroflovskiNoch keine Bewertungen
- Novagen pET System ManualDokument68 SeitenNovagen pET System ManualJagathpriyaNoch keine Bewertungen
- Jurnal Trichomonas VaginalisDokument7 SeitenJurnal Trichomonas VaginalisHendric HariansyahNoch keine Bewertungen
- Original Pdfbiology Exploring The Diversity of Life 4th 1 3 by Peter Russell PDFDokument41 SeitenOriginal Pdfbiology Exploring The Diversity of Life 4th 1 3 by Peter Russell PDFbonnie.eaton513100% (34)
- BRCA1 Associated Epigenetic Regulation of p73 Mediates Effector Pathway For Chemosensitivity in Ovarian CarcinomaDokument10 SeitenBRCA1 Associated Epigenetic Regulation of p73 Mediates Effector Pathway For Chemosensitivity in Ovarian CarcinomaDr.Yogender SinghNoch keine Bewertungen
- Development of Recombinant Subunit Vaccine and Monoclonal Antibody Based Diagnostic Test For Infectious Bursal Disease in Chickens.Dokument217 SeitenDevelopment of Recombinant Subunit Vaccine and Monoclonal Antibody Based Diagnostic Test For Infectious Bursal Disease in Chickens.Dr-Mohammed AbdallaNoch keine Bewertungen
- GENE CONCEPTDokument12 SeitenGENE CONCEPTDavid trumpNoch keine Bewertungen
- Q Topik 6Dokument32 SeitenQ Topik 6Carin TanNoch keine Bewertungen
- HB-0436-006 - HB - RNeasy Midi Maxi - 0421 - WWDokument104 SeitenHB-0436-006 - HB - RNeasy Midi Maxi - 0421 - WWYi-Ling ChiuNoch keine Bewertungen
- Strategies of Neutrophil Diversification: Nature ImmunologyDokument10 SeitenStrategies of Neutrophil Diversification: Nature ImmunologylfjuradozNoch keine Bewertungen
- Protein Synthesis: Genes, Transcription, Translation and MutationsDokument23 SeitenProtein Synthesis: Genes, Transcription, Translation and MutationsBonnyNoch keine Bewertungen
- CG000527_ChromiumFixedRNAProfiling_MultiplexedSamples_UserGuide_Rev_E (1)Dokument95 SeitenCG000527_ChromiumFixedRNAProfiling_MultiplexedSamples_UserGuide_Rev_E (1)Henry David Mogollon GarciaNoch keine Bewertungen
- Albert Et Al., 2014 PDFDokument19 SeitenAlbert Et Al., 2014 PDFIndrani BhattacharyaNoch keine Bewertungen
- 2023 All Biology Is Cognitive Information ProcessingDokument15 Seiten2023 All Biology Is Cognitive Information ProcessingettenmollNoch keine Bewertungen
- Artigo Com ImagensDokument28 SeitenArtigo Com ImagensMarcio FonteneleNoch keine Bewertungen
- Biology: Content Knowledge (5235) Study Plan Test Content CategoriesDokument21 SeitenBiology: Content Knowledge (5235) Study Plan Test Content CategoriesEvone BarkerNoch keine Bewertungen
- Regulation of Gene Expression From Lehninger - 4e PDFDokument21 SeitenRegulation of Gene Expression From Lehninger - 4e PDFAlthea Karmylle M. BonitaNoch keine Bewertungen
- Saffron Benefits and Uses by King KesariyaDokument13 SeitenSaffron Benefits and Uses by King KesariyaKing KesariyaNoch keine Bewertungen