Beruflich Dokumente
Kultur Dokumente
Acrilic 1
Hochgeladen von
Isaac Gabriel LarreaOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Acrilic 1
Hochgeladen von
Isaac Gabriel LarreaCopyright:
Verfügbare Formate
Journalof DentistryVol. 23, No. 5, pp.
313-318, 1995
Copyright Q 1995Elsevier Science Ltd
Printed in Great Britain. All rights reserved
0300-5712/95 $10.00+ 0.00
0300-5712(94)00002-6
The effect of preparation conditions of acrylic
denture base materials on the level of residual
monomer, mechanical properties and water
absorption
A. Dotjan*,
5. Bek*, N. N. Cevikf- and A. Usanmazt
*Department of Prosthodontics,
University, Ankara, Turkey
Gazi University,
Faculty of Dentistry and tDepartment
of Chemistry,
Middle East Technical
ABSTRACT
Objectives and methods: The curing of several commercial powder/liquid
mixtures of acrylic denture
base materials was carried out at different temperatures and curing times. The levels of residual
mononer, tensile strength, percentage elongation before break and water absorption were measured.
Results and Conclusion: It was found that the level of residual monomer determined by gas-liquid
chromatography decreased with curing time and temperature increase. At the same time, the tensile
strength was improved and water absorption decreased.
KEY WORDS: PMMA, Residual monomer, Mechanical
J. Dent. 1995; 23: 313-318
(Received
properties,
10 May 1994; reviewed
Water absorption
22 July 1994; accepted 20 October 1994)
INTRODUCTION
The autoploymerizing acrylic resins are now commonly
used in processing dentures. For both autopolymerizing
and heat-cured acrylic resins, the level of the residual
monomer in the resin- and the mechanical properties1-12 are closely related to the curing conditions. It
is a general belief that tissue sensitivity to acrylic resin
denture-base materials depends on the level of residual
monomerg13,14,and increases with an increase in the
percentage of residual monomer in the resin. McCabe
and Baskerg have reported that methyl methacrylate
monomer is an irritant and sensitizer to tissues. Therefore, the level of the residual monomer must be controlled carefully when curing these types of materialslm7.
It has also been reported that a high level of residual
monomer is detrimental to mechanical properties of
the cured resin&. However, there is insufficient reported literature on the relationship between conditions of material preparations and the material properties of the cured resin.
Several physical and chemical methods have been
used to measure the level of residual monomer in
polfimethylmethacrylate) (PMMA) resin. The most accurate method is gas-liquid chromatography5. McCabe
and Basker showed by this method that the level of
the residual monomer is inversely related to the curing
time, i.e. the longer the curing time, the lower the
Correspondence
should be addressed to: Prof. Dr Arife Dogan, 10
Sokak No. 2, MutlukGy, Ankara, Turkey.
residual monomer level. Fletcher et aZ.7 reported high
levels of the residual monomer in thick polymer samples compared to thin ones. Austin and Basker3 compared the residual monomer level obtained by gas
chromatography (GC) and IR-spectral analysis (IR) for
12 specimens that had been cured for different curing
times. The results range from 0.24 to 6.33% for GC
and 0.33 to 5.50% for IR measurements. The agreement for each of the specimens was considered to be
acceptable. They also investigated the effect of sample
thickness, material preparation, sampling methods, etc.
All these factors have certain effects on the level of
residual monomer. For example, they found the residual monomer percentage to be seven times greater for
short curing cycle specimens compared to that in long
curing cycles. The effect of the curing cycle is also
studied by Harrison and Huggett15 for heat-cured
acrylic resins. They have determined the residual
monomer level after leaching most of it in water.
However, this will make the conclusion between residual monomer level and mechanical properties less
meaningful, because the residual monomer, in addition
to acting as a plasticizer in the polymer matrix, will also
cause the formation of more porosity that will also
affect the mechanical properties. Therefore, in our
study, we determine the residual monomer level without keeping the samples in water.
In this study, several commercial samples were prepared at different temperatures and curing times. The
autopolymerizing resins were prepared in a constanttemperature water-bath to keep the temperature con-
314
J. Dent. 1995; 23: No. 5
sister-nand to dissipate the exothermic reaction to heat
by water, which has a higher thermal conductivity than
air. In most of the reported work, polymerization takes
place in air, and heat formed from the exothermic
reaction might cause a high level of porosity in the
material. The effect of curing temperature for autopolymerizing resin has also not been reported before. The
level of the residual monomer, tensile strength, percentage elongation before break and water absorption
were measured. Optimum conditions for imporved mechanical properties are proposed when considering
these results.
The effect of thickness of sample plate on residual
monomer content has been studied by Sadamori et ~1.~
and other workers3J7,and therefore will not be repeated in this work.
MATERIALS
AND METHODS
The powder/liquid denture materials chosen for this
study are given in Table I. The organic solvents used
were of spectroscopic grade and they were used without further purification. Water was triple distilled and
deionized. The purity of water was tested by conductivity measurements.
The powder/liquid mixtures were prepared according to the respective manufacturers instruction. They
were mixed in proportions recommended by the manufacturer. Heat curing samples were cured at boiling
water temperature for 30,40 and 50 min. The autopolymerizing samples were cured in constant temperature
(20, 25 and 30 C) water-baths, which have been
equipped with an electronically controlled heater, for
30 min. The curing times and temperatures were adjusted accurately (+ 0.1 s and f O.OPC) to the desired
values. The mixed resins were packed into a plaster
mould designed to produce 6 x 3 x 0.5 (+ 0.1) cm sample size plates which were used for all measurements.
The Fourier Transform Infrared (FTIR) spectra of
reference monomer and liquid samples in methanol
were taken in a NaCl cell, and those of powders and
cured polymer samples from KBr pellets using FTIR
Spectrophotometry (Nicolet, Model FFP-510, Madison,
WI, USA).
The specimens for residual monomer determination
were prepared by using a 2.5 mm engineering twist drill
with 250 r.p.m. drilling velocity. Drilling velocities of
100-500 r.p.m. were tested, to determine if either the
drilling velocity or heat produced from drilling resulted
in any depolymerization or break in polymer chains.
However, no effect was observed from experimental
results for one test specimen. Therefore, all drillings
were done at 250 r.p.m. velocity. The drill cuttings were
collected and weighed, then placed in a round-bottom
250 ml extraction flask equipped with a condenser. The
sample weight was approximately 1 g (+ 0.1 mg) and
the extraction solvent was 20 (+O.l) ml methanol. The
extraction was carried out at the boiling temperature of
methanol for 6 h using a heating mantle. After the
extraction was completed, the polymer residues were
filtered off, and the methanol solution saved for the
residual monomer analysis. Further extraction of residual polymer showed no more residual monomer extract. The extraction was repeated for three experimental samples of each material in Table I, with five specimens per sample and ten specimens of material B, per
sample to test for statistical differences between samples.
The residual monomer analyses were done by
gas-liquid chromatography (GLC) (10% Carbowax 20
M packed column HP 5890 model Hewlett Packard
GLC, Neyrinz, Geneva, Switzerland). The oven temperature was set at 70C for 1 min, then raised to 80C at a
heating rate of SC/min. and kept at that temperature
for 5 min. The injection and flame ionization detector
temperatures were at 150C. Nitrogen gas was used as
the carrier gas at a flow rate of 345 kPa. The detector
output was linked to a chart recorder, with a chart
speed set at 5 mm/min. The results were also evaluated by a connected computer that gives percentage
area corresponding to each observable peak. A 1 ml
extract was injected each time and a standard methanol
solution of monomer was used as reference (Aldrich
Chemical Co. Ltd, Milwaukee, WI, USA). The reference curves were obtained for monomer. The internal
standards (known amount of monomer) were used in
the sample solution to obtain very accurate results free
of all types of systematic errors.
Table 1. Denture materials used
Notation sample
Manufacturer
A QC-20 Heat Cured
Conventional processing
B Paladon 65 Heat Cured
Conventional processing
C SR-lvocap Heat Cured
Injection technic
D Meliodent Autoploymerizing
At room temperature
E Palapress Autopolymerizing
At room temperature
De Trey Division Dentsply Ltd,
Weybridge, Surrey, UK
Kulzer & Co. GMBH Bereich Dental,
Hamburg, Germany
lvoclar AGFL Schan,
Liechtenstein
Bayer UK Limited, Newbury,
Berkshire, UK
Kulzer & Co. GMBH Dental Division,
Hamburg, Germany
A. Dogan eta/.: Acrylic denture base materials on level of residual powder/liquid
mixtures
315
Tensile strength and percentage elongation before
break for several samples cured under different conditions were measured on an Instron mechanical tester
(Tensile Testing Machine, Table Model-1102. Instron
Corp., Canton, MA, USA). The measurements were
carried out with 10 specimens of each sample (e.g. B,,
C,, etc.) prepared under identical conditions, and mean
values and standard deviations were reported.
For the measurement of water absorption, specimens
(6 x 3 x 0.5 (t-0.1) cm) were kept in triple distilled
water at 25 ( f 0.5)C for about 2 months. They were
weighed (with +O.l mg accuracy) at intervals of 15 min
for the first 2 h, every 30 min for 6 h, and then every 24
h. The excesswater on specimens was removed before
weighing by blotting with a filter paper. Measurements
were performed on three specimens and mean values
were calculated.
RESULTS
Infrared (IR)-spectral
1
loo
investigation
I
boo
I
3ooo
2500
zoo0
15co
xno
500
Wavenumber (cm)
Fig. 1. FTIR spectrum of liquid (a), powder (b) and cured polymer
(cl.
The FTIR spectra of powder, liquid and cured form of
the materials in Table I were investigated. The spectra
of different commercial samples were identical. Therefore, one example of a liquid, powder and cured mixture is shown in Fig. 1. The spectra of the liquids were
identical to those of methyl methacrylate monomer and
those of powders were identical to those of PMMA
which have been polymerized by radiation via vinyl
groups addition. The IR-spectral peaks at 1643 cm-i
corresponding to vinyl groups is observed in the spectrum of the monomer (8&. la), but not in the spectrum
of the polymer (F&J. Ib,c). The FTIR spectrum of the
cured samples (Fig. Ic) shows peak broadening as an
indication of high-molecular-weight, crosslinked polymer formation. The carbonyl (C = 0) peak at 1750
cm- is observed in all spectra. The concentration of
the residual monomer was too small to be observed
from this spectra.
The FTIR spectra of the heat-cured and the autopolymerizing powder/liquid samples were the same. The
FTIR spectra of heat-cured and autopolymerizing
powder and those of liquid samples did not give any
peak when differentiated. Therefore, the accelerators
that may exist in autopolymerizing samples are IR
inactive or have very small concentrations that are
under the detection limit of IR spectra.
showed no significant differences. Statistical analysis
revealed that the differences in mean values for each
material are significant. For example, when the t-test
was applied to two mean values closest to each other
(e.g. E, and E, in TubZeII), the results obtained were
t = 3.00, d.f. = 8, P > 0.02.
The level of the residual monomer decreased from
2.44% to 0.96% for QC-20 heat cured (sample A, to
A3), when curing time was increased from 30 to 50
min. Similar decreases in the level of the residual
monomer with increasing curing time were also
observed for other samples. The amount of reduction
for an additional 20 min of curing time is considerable.
The decreases in residual monomer levels with an
increase in curing time for heat curing samples are
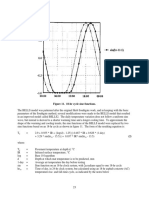
plotted in Fig. 2. In all cases,straight lines (Y = 0.99 for
A and B samples; Y= 0.94 for C samples) were observed
within the range studied. The residual monomer levels
at 50 min curing time are quite similar for different
samples.
The reduction in residual monomer level for autopolymerizing samples with an increase of temperature
from 20 to 30C is relatively small.
The levels of residual monomer
Mechanical properties
The levels of the residual monomer are tabulated in
Table 11. The values given are the means ( f SD) of five
specimens (10 for BJ for each sample of A,, A,, etc.
prepared under identical conditions. The statistical calculations for sample B,(t = 1.83, d.f. = 9, P > 0.05)
The tensile strength and percentage elongation for
some samples are shown in TabZeIII. All the measurements have been repeated for 10 specimens of each
sample (e.g. 10 specimens of A,, etc.) for statistical
evaluation, and mean values are reported. The stan-
316
J. Dent. 1995; 23: No. 5
Table II. Curing conditions and average residual monomer levels (*SD)
each sample and 10 for B3)
Sample
Ai
A2
A3
B,
B2
B3
Cl
c2
C3
Di
D2
D3
El
E2
E3
for samples in Table / (five specimens for
Curing temperature
PC)
Curing time
(mid
% Residual
monomer
Heat cured 100
Heat cured 100
Heat cured 100
Heat cured 100
Heat cured 100
Heat cured 100
Heat cured 100
Heat cured 100
Heat cured 100
Autoploymerizing 20
Autopolymerizing 25
Autopolymerizing 30
Autopolymerizing 20
Autopolymerizing 25
Autopolymerizing 30
30
40
50
30
40
50
30
40
50
30
30
30
30
30
30
2.44 i 0.02
1.51 + 0.01
0.96 + 0.03
3.28 f 0.02
2.45 + 0.02
0.92 + 0.01
2.03 + 0.02
1 .16 + 0.01
0.98 f 0.03
2.33 i 0.02
2.06 + 0.04
1.89 + 0.02
1.52 + 0.01
1.49 + 0.02
0.94 + 0.02
dard deviations of the measurements as shown in Table
111 are relatively great, but typical for these types of
measurements. In general the mechanical properties of
different commercial samples are not much different
from each other. For example, the percentage elongation before break is almost the same for all samples.
The differences between the mean values of percentage elongation for most-sample groups are not statistically significant (e.g. for each pair of B,, B,; C,, C,;
and E,, E,, the values are almost the same). However,
the differences for the means of tensile strength are
significant. For example, if the tensile strength mean
values of D, : D, which have the closest value are
compared, the t-test results are t = 15.13, d.f. = 18,
P < 0.001.
Water absorption
The results in terms of percentage decrease in the first
30 min and maximum percentage increased attained
for samples stored in water at 25 & 0.5C, are tabulated
in Table W. In the first 30 min of storage in water, a
slight decreasein weight for most sampleswas observed.
The weights then started increasing gradually, reaching
a constant value at 20-25 days, then remaining at that
constant value for the rest of the time. The changes in
weight were plotted against time, and constant weights
reached were used for calculation of percentage. The
three specimens of each sample studied gave almost
exactly the same values.
DISCUSSION
The residual monomer level decreased for longer curing times of heat-cured samples and curing at higher
temperatures for autopolymerizing samples. Although
it is very hard to predict a tolerable level of residual
monomer for tissue sensitivity for each individual, the
level of residual monomer should be very low (e.g.
t
40
1
30
Time (min)
Fig. 2. Percentage
residual monomer level against curing time. Materials:
0, A; l , B; A, C.
I
50
A. Dogan et a/,: Acrylic denture base materials on level of residual powder/liquid
mixtures
317
Table /il. Tensile strength (T) (+ SD) and percentage elongation (E) (+ SD) for some samples
in Tab/e // ( the values are means of 10 specimens for each sample that is prepared under
identical conditions)
Sample
4
5,
63
Cl
C*
C3
5,
52
53
El
E2
E3
T (MPa)
%E
26.9 + 1.1
35.8 + 0.8
56.6 + 2.1
44.7 f 1.4
49.5 i 1.o
57.1 + 2.3
25.1 + 0.9
31.9 + 1.l
50.0 i 2.1
38.1 f 0.9
48.9 + 0.8
54.6 i 1.7
4.9 + 0.4
5.0 j 0.5
6.1 jz 0.4
5.0 10.4
5.0 * 0.5
7.5 j 0.6
4.9 10.4
5.5 * 0.5
6.1 i 0.6
5.0 * 0.3
5.0 * 0.4
5.4 * 0.5
0.45% as given by Austin and Basker3). In order to
achieve such a low residual monomer level, the curing
time should be more than 50 min.
At boiling water temperature, benzoyl peroxide (initiator in powder) decomposes (above 6oC), giving free
radicals which start and propagate further polymerization. However, the boiling water temperature is also
close to the boiling temperature of methyl methacrylate (100.8C at 101 kPa), which will change into gas
form and produce bubbles in a polymer matrix when
trapped in it. This type of gas formation will also be
enhanced by exothermic heat production during polymerization. The gas bubbles in the polymer matrix
form voids, increase porosity17 and develop high internal stress. The details of porosity formation have been
reported by Wolfaardt et al.. Entrapped residual
monomer in the polymer matrix will also leave voids
when removed from it. Therefore, mechanical properties which are closely related to internal stresses and
voids in the polymer matrix will also be related to the
level of residual monomer. As an example, the residual
monomer percentage in sample B, (3.28%) is larger
than that in sample C, (2.03%), and their corresponding tensile strengths are 26.9 and 44.7 MPa, respectively. Both samples are heat cured for 30 min, and
FTIR spectra showed no structural differences even
though they are made by different companies. Thus, for
a given sample, the tensile strength increases when the
residual monomer level decreases. This behvaiour
should be expected because of the plastisizer effect of
unpolymerized monomer in the polymer matrix. The
results obtained in this work are in agreement with the
suggestion of Brauer6. The data in Table 111show that
there is an improvement in tensile strength with an
increase in curing time, which also causes a decrease in
residual monomer level.
Poly(methy1 methacrylate) is insoluble in water
therefore the first weight decrease cannot be attributed
to the dissociation of resin in water. The residual
monomer near the surface of the resin probably diffuses into water, causing a decrease in the weight of
the samples. The reason for no weight decrease for
some samples containing a higher percentage of residual monomer may be explained by the nature of residual monomer in the polymer matrix. If the residual
monomer is not close to the surface of the specimen,
entrapped in the inner layers of resin plate, it cannot
diffuse out into water in a short time. Even if it diffuses
out later, the amount of water absorption into the
polymer will be high enough to compensate for the
weight lost caused by the residual monomer diffusion
from the resin to the water. As a result, a weight
increase instead of weight decrease was observed.
Tab/e IV. Water absorption of some samples in Table I/ (the values are means of three
specimens of each group)
Sample
Ai
A2
B,
B3
C,
D3
E3
% Weight decrease in
first 30 min
Trace
0.61
0.71
0.46
Trace
0.41
0.08
% Water absorption
at 60 days
2.70 + 0.005
2.15 + 0.004
2.75 + 0.007
2.19 * 0.01
2.61 + 0.008
1.95 * 0.009
2.43 i 0.008
318
J. Dent. 1995; 23: No. 5
There is a parallel relation between the level of the
residual monomer and the percentage water absorption.
For example, in this study, both values are
highest for the B, sample. Similarly, if we compare the
values for A, and A,, the percentages of residual
monomer were 2.44 and 1.51; the corresponding percentage water absorption was 2.70 and 2.15.
The percentage water absorption is higher for A,
than for A,, and B, was higher than B,. This can be
related to the curing time. As the curing time increases, the crosslinking increases, giving a harder polymer with less porosity. This could make the diffusion
of water into polymer matrix more difficult.
The absorption and desorption of water by acrylic
resins has been investigated in detaillsm21.This is a
diffusion process with a diffusion coefficient reported
by Braden of 5.4 X 10M9 cm2 s-i at 22.5 C. Water
will diffuse into voids in the polymer mass. Therefore,
when the level of residual monomer is high, the number of voids in the resin after leaching off the residual
monomer will be high and water intake will also be
high11,9-21. This is supported by data given in Tables II
and n/. For the same material (e.g. A, : A, or B, : Ba),
when the percentage of residual monomer is high (TabZe II>, the percentage of water absorbed is also high
(Table IV).
CONCLUSIONS
1. FTIR spectra determinations showed that there
were no material or structural differences between the
denture base materials used in this study. The activator
used in autopolymerizing resin in all samples studied
could not be verified by FTIR.
2. For heat-cured resins, the longer curing times
improved the tensile strength and decreased the level
of residual monomer. However, the percentage elongation does not change much.
3. For autopolymerizing resins, the level of residual
monomer decreased with an increase in temperature
when the curing time is kept constant. The constanttemperature water-bath used in this study is very important for obtaining relatively pore free resin that has
lower residual monomer content and low water absorption.
4. The percentage of water absorption is higher for
samples containing higher levels of residual monomer.
References
1. Underwade H, Sibhaye AB. Curing acrylic resin in a
domestic pressure cooker: a study of residual monomer
content. QuintessenceZnt 1989; 20: 123-129.
2. Sadamori S, Kotani H, Hamada T. The usage period of
dentures and their residual monomer contents. .I Prosthet
Dent 1992; 68: 374-376.
3. Austin AT, BaskerRM. the level of residual monomer in
acrylic denture base materials. Br Dent J 1980; 149:
281-286.
4.
Austin AT, Basker RM. Residual monomer levels in
denture bases. The effect of varying short curing cycles.
Br Dent J 1982; 153: 424-426.
5. Bliimler VG. Vergleichende untersuchungen iiber art
und menge des restmonomer gehaltes bei dem prothesenwerkstoff piacryl. IM Dtsch Stomat 1965; 15: 651-658.
6. Brauer D. Chemishe analyse von metacrylat kunstoffen.
Dtsch Zahnaerzt Z 1975; 30: 672-679.
7. Fletcher AM, Purnaveja S, Amin WM, Ritchie GM,
Moradians S, Dodd AW. The level of residual monomer
in self curing denture base materials. J Dent Res 1983; 62:
118-120.
8. Marx H, Fukui M, Stender E. Zur frage der restmonomer
untersuchung von prothesenkunstsoffen. Dtsch Zahnaerzt
2 1983; 38: 550-553.
9. McCabe JF, Basker RM. Tissue sensitivity to acrylic
resin. Br Dent J 1976; 140: 347-350.
10. Honorez R, Catalan A, Angness A, Grimonster J. The
effect of three processing cycles on some physical and
chemical properties of a heat cured acrylic resin. J Prosthet Dent 1989; 61: 510-517.
11. Arab J, Newton JP, Lloyd CH. The effect of an elevated
level of residual monomer on the whitening of a denture
base and its physical properties. J Dent 1989; 17: 1899194.
12. Donovan TE, Hurst RG, Campagi WV. Physical properties of acrylic resin polymerized by four different techniques. J Prosthet Dent 1985; 54: 522-524.
13. Kallus T. Evaluation of the toxicity of denture base
polymers after subcutaneous implantation in guinea pigs.
J Prosthet Dent 1984; 52: 126-134.
14. Devlin H. Acrylic monomer-friend or foe. Quintessence
Dent Technol 1984; 8: 511-512.
15. Harrison A, Huggett R. Effect of the curing cycle on
residual monomer levels of acrylic resin denture base
polymers. J Dent 1992; 20: 370-374.
16. Sadamori S, Ganefiyanity T, Hamada T, Arima T. Influence of thickness and location on the residual monomer
content of denture base cured by three processing methods. J Prosthet Dent 1994; 72: 19-22.
17. Bafile M, Grasser GN, Myers ML, Li EKH. Porosity of
denture resin cured by microwave energy. J Prosthet Dent
1991; 66: 269-274.
18. Wolfaardt JF, Dent M, Cleaton-Jones P, Fatti P. The
occurrence of porosity in heat cured poly(methy1
methacrylate) denture base resin. J Prosthet Dent 1986;
55: 393-400.
19. Braden M. The absorption of water by acrylic resins and
other materials. J Prosthet Dent 1964; 14: 307-316.
20. Braden M, Wright PS. Water absorption and water solubility of soft lining materials for acrylic dentures. J Dent
Res 1983; 62: 764-768.
21. Kalachandra S, Turner DT. Water sorption of polymethacrylate networks: Bis-GMA/TEGDMA
copolymers. J Biomed Mater Res 1987; 21: 329-338.
Das könnte Ihnen auch gefallen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (399)
- 2017xu Avances en Materiales Polimericos Dental AplicacionDokument17 Seiten2017xu Avances en Materiales Polimericos Dental AplicacionIsaac Gabriel LarreaNoch keine Bewertungen
- 2016freitas Da Sivla Toxicidad de Las Protesis OcularesDokument6 Seiten2016freitas Da Sivla Toxicidad de Las Protesis OcularesIsaac Gabriel LarreaNoch keine Bewertungen
- 2013xu Refuerzo Con Fibras VegetalesDokument7 Seiten2013xu Refuerzo Con Fibras VegetalesIsaac Gabriel LarreaNoch keine Bewertungen
- Investigacoin MicroDokument1 SeiteInvestigacoin MicroIsaac Gabriel LarreaNoch keine Bewertungen
- Beyli 1981Dokument4 SeitenBeyli 1981Isaac Gabriel LarreaNoch keine Bewertungen
- Mandibular Condyle Movement During Mastication of FoodsDokument11 SeitenMandibular Condyle Movement During Mastication of FoodsIsaac Gabriel LarreaNoch keine Bewertungen
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (894)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (587)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (265)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (73)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2219)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (119)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Design of Road PavementsDokument56 SeitenDesign of Road Pavementsirbasukirahardjoces100% (1)
- Supplement 5 - Multiple RegressionDokument19 SeitenSupplement 5 - Multiple Regressionnm2007kNoch keine Bewertungen
- Bayesian Structural Time Series ModelsDokument100 SeitenBayesian Structural Time Series Modelsquantanglement100% (1)
- Measures of Central Tendency LessonDokument8 SeitenMeasures of Central Tendency LessonDonagine Ibay100% (8)
- A Critical Review of The Models Used To Estimate Solar RadiationDokument16 SeitenA Critical Review of The Models Used To Estimate Solar RadiationScribedSkypeNoch keine Bewertungen
- Concepts and Techniques: - Chapter 10Dokument97 SeitenConcepts and Techniques: - Chapter 10sebpkyNoch keine Bewertungen
- BS en 00459-2-2010Dokument68 SeitenBS en 00459-2-2010KlevisSheteliNoch keine Bewertungen
- Monthly Streamflow Simulation: User's ManualDokument104 SeitenMonthly Streamflow Simulation: User's Manualrony_curasmaNoch keine Bewertungen
- Advances in Information and Communication Technologies For Adapting Agriculture To Climate Change Proceedings of The International Conference of ICT For Adapting Agriculture To Climate ChangeDokument278 SeitenAdvances in Information and Communication Technologies For Adapting Agriculture To Climate Change Proceedings of The International Conference of ICT For Adapting Agriculture To Climate ChangeEmilio HernandezNoch keine Bewertungen
- 2000 (LTPP) FHWA-RD-98-085 Temperature Predictions and Adjustment Factors For Asphalt Pavemen PDFDokument49 Seiten2000 (LTPP) FHWA-RD-98-085 Temperature Predictions and Adjustment Factors For Asphalt Pavemen PDFTim LinNoch keine Bewertungen
- Caste and Census 1891Dokument340 SeitenCaste and Census 1891रिजु भारतीयन0% (1)
- Concentrations and Emissions of Ammonia in Livestock Buildings in Northern EuropeDokument17 SeitenConcentrations and Emissions of Ammonia in Livestock Buildings in Northern Europelg2fNoch keine Bewertungen
- Fluid Handling Engineers: U S Agency - Taco DivDokument5 SeitenFluid Handling Engineers: U S Agency - Taco DivsitehabNoch keine Bewertungen
- Anova One Way PDFDokument32 SeitenAnova One Way PDFcasperNoch keine Bewertungen
- Adaptation of The Thornthwaite Humidity Index For The Characterization of Annual Water SituationsDokument8 SeitenAdaptation of The Thornthwaite Humidity Index For The Characterization of Annual Water SituationsInternational Journal of Innovative Science and Research TechnologyNoch keine Bewertungen
- BSCM 2 PDFDokument56 SeitenBSCM 2 PDFduytay94Noch keine Bewertungen
- Stochastic Hydrology CourseDokument40 SeitenStochastic Hydrology CoursekardraNoch keine Bewertungen
- Liquid Thermal Sizing SheetDokument1 SeiteLiquid Thermal Sizing SheetapgoslingNoch keine Bewertungen
- Climate impacts in Lake TaalDokument8 SeitenClimate impacts in Lake TaalKat Dino100% (1)
- Types of Charts and GraphsDokument25 SeitenTypes of Charts and GraphslordniklausNoch keine Bewertungen
- A Leisurely Look at The Bootstrap, The Jackknife, and Cross-Validation (1983 13s) - BRADLEY EFRONDokument13 SeitenA Leisurely Look at The Bootstrap, The Jackknife, and Cross-Validation (1983 13s) - BRADLEY EFRONValentin RodriguezNoch keine Bewertungen
- Genetic Variability and Associations in Lentil GenotypesDokument24 SeitenGenetic Variability and Associations in Lentil GenotypesĐoh RģNoch keine Bewertungen
- Machine learning models forecast Kerala rainfallDokument8 SeitenMachine learning models forecast Kerala rainfallMamo SisayNoch keine Bewertungen
- Buccal Corridor SpaceDokument6 SeitenBuccal Corridor Spacepopat78Noch keine Bewertungen
- 0610 w17 Ms 62Dokument8 Seiten0610 w17 Ms 62Hacker1Noch keine Bewertungen
- IRM Chapter 6e 05Dokument42 SeitenIRM Chapter 6e 05enufeffizyNoch keine Bewertungen
- Bivariate Analysis: Measures of AssociationDokument38 SeitenBivariate Analysis: Measures of AssociationShaloo Sidhu100% (1)
- ET ISO 22935-2-2012 - Milk and Milk Product - Sensory AnalysisDokument27 SeitenET ISO 22935-2-2012 - Milk and Milk Product - Sensory AnalysisRadi TyoNoch keine Bewertungen
- Correlation AnalysisDokument20 SeitenCorrelation AnalysisVeerendra NathNoch keine Bewertungen
- Guidelines For Implementing NCHRP 1-37A M-E Design Procedures in Ohio: Volume 1 - Summary of Findings, Implementation Plan, and Next StepsDokument112 SeitenGuidelines For Implementing NCHRP 1-37A M-E Design Procedures in Ohio: Volume 1 - Summary of Findings, Implementation Plan, and Next StepsLa FoliakNoch keine Bewertungen