Beruflich Dokumente
Kultur Dokumente
Closed-Book Practice-Ch 10 (2017!08!08)
Hochgeladen von
JuanOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Closed-Book Practice-Ch 10 (2017!08!08)
Hochgeladen von
JuanCopyright:
Verfügbare Formate
CHAPTER 10 PHASE TRANSFORMATIONS Page 1/12
CLOSED-BOOK PRACTICE
CHAPTER 10: Phase Transformations
CONCEPT CHECK
1. Which is more stable, the pearlitic or the spheroiditic microstructure? Why?
Ans:
Spheroiditic microstructures are more stable than pearlitic ones. Since pearlite transforms to spheroidite, the
latter is more stable.
2. Cite two major differences between martensitic and pearlitic transformations.
Ans:
Two major differences are:
1) atomic diffusion is necessary for the pearlitic transformation, whereas the martensitic transformation is
diffusionless; and
2) relative to transformation rate, the martensitic transformation is virtually instantaneous, while the
pearlitic transformation is time-dependent.
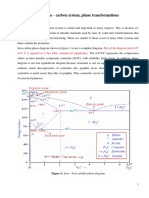
3. Make a copy of the isothermal transformation diagram for an iron-carbon alloy of eutectoid composition
(Fig. 10.22) and then sketch and label on this diagram a time-temperature path that will produce 100% fine
pearlite.
Ans:
The figure below shows an isothermal transformation diagram for a eutectoid iron-carbon alloy on which a
time-temperature path that will produce 100% fine pearlite is included.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 2/12
4. The figure below shows the continuous-cooling-
transformation (CCT) diagram for a 4340 alloy
steel, superimposed by several cooling curves.
Here, the letters M, B, F, P stand for martensite,
bainite, ferrite and pearlite, respectively.
Describe a simple continuous cooling heat
treatment procedure, including temperature
heating/cooling rates and isothermal holding
time, if necessary, that would be used to convert
a 4340 steel from (MB) into (FP).
Ans:
In order to convert from (martensite bainite)
into (ferrite pearlite) it is necessary to
(1) heat above about 720C at any heating rate
and hold isothermally for enough time to
allow complete austenitization, then
(2) cool to room temperature at a rate equal to or
less than 0.006C/s, as shown in the figure.
5. Rank the following iron-carbon alloys and associated microstructures from the highest to the lowest tensile
strength. Justify this ranking.
0.25 wt%C with spheroidite 0.25 wt%C with coarse pearlite
0.6 wt%C with fine pearlite 0.6 wt%C with coarse pearlite
Ans:
This ranking called for is as follows:
(1) 0.6 wt%C with fine pearlite (2) 0.6 wt%C with coarse pearlite
(3) 0.25 wt%C with coarse pearlite (4) 0.25 wt%C with spheroidite
The 0.25 wt% C coarse pearlite is stronger than the 0.25 wt% C spheroidite because coarse pearlite is
stronger than spheroidite (since the composition of the alloys is the same). The 0.6 wt% C coarse pearlite is
stronger than the 0.25 wt% C coarse pearlite because increasing the carbon content increases the strength
(while maintained the same coarse pearlite microstructure). Finally the 0.6 wt% C fine pearlite is stronger
than the 0.6 wt% C, coarse pearlite inasmuch as the strength of fine pearlite is greater than coarse pearlite
because of the many more ferrite-cementite phase boundaries in fine pearlite.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 3/12
6. For a eutectoid steel, describe an isothermal heat treatment that would be required to produce a specimen
having a hardness of 93 HRB.
Ans:
From Fig. 10.30(a), in order for a 0.76wt%C alloy to have a Rockwell hardness of 93HRB, the
microstructure must be coarse pearlite. Thus, utilizing the isothermal transformation diagram for this alloy,
Fig. 10.22, after austenitizing at about 760C, rapidly cool to a temperature at which coarse pearlite forms
(i.e., to about 675C), and allow the specimen to isothermally and completely transform to coarse pearlite.
At this temperature an isothermal heat treatment for at least 200s is required. Then cool to room temperature
(cooling rate is not important).
7. A steel alloy is quenched from a temperature within the austenite phase region into water at room
temperature so as to form martensite; the alloy is subsequently tempered at an elevated temperature which is
held constant.
(a) Make a schematic plot showing how room-temperature ductility varies with the logarithm of tempering
time at the elevated temperature. (Be sure to label your axes.)
(b) Superimpose and label on this same plot the room-temperature behavior resulting from tempering at a
higher tempering temperature and briefly explain the difference in behavior between these two
temperatures.
Ans:
(a) Shown below is the plot that was requested.
(b) The line for the higher temperature (labeled TH) will lie above the one at the lower temperature (labeled
TL) because the Fe3C particles in tempered martensite will grow faster at the higher temperature; thus, at
some given tempering time they will be larger at the higher temperature. The alloy tempered at the
higher temperature will be more ductile because there will be fewer -Fe3C phase boundaries (due to the
larger Fe3C particles) that may impede dislocation motion.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 4/12
QUESTIONS & PROBLEMS
The Kinetics of Phase Transformations
Name the two stages involved in the formation of particles of a new phase. Briefly describe each.
Ans:
The two stages involved in the formation of particles of a new phase are nucleation and growth. The
nucleation process involves the formation of normally very small particles of the new phase(s), which are
stable and capable of continued growth. The growth stage is simply the increase in size of the new phase
particles.
Isothermal Transformation Diagrams
The figure at right shows the phase transformation 1
diagram of an eutectoid steel, where the letters: A, P,
B and M represent austenite, pearlite, bainite and
martensite phases of the eutectoid steel, respectively.
Consider two heat treatments denoted as 1-2-3 and
1-2-4-5 in the figure, respectively?
(a) Identify the microstructures at the locations 1 to 5,
respectively.
(b) Discuss the advantage(s) and disadvantage(s) of
these two processes. Justify the reason(s).
(c) Determine if either of the two processes can be
2 4
considered as an isothermal phase transformation.
Explain why.
(d) What are the common heat-treatment names for 31.0
the processes 1-2-3 and 1-2-4-5, respectively?
(e) Estimate the cooling rate of the process 1-2-3.
Ans:
(a) Pt 1 (0s, 800C): 100% austenite 3 5
Pt 2 (0.3s, 300C): 100% austenite
Pt 3 (0.7s, 0C): 100% martensite
Pt 4 (30s, 300C): 100% austenite
Pt 5 (30s, 0C): 100% martensite
(b) Processes 1-2-3:
Advantages: fast cooling rate, no need for extra equipment and effort for isothermal processing
Disadvantages: high thermal residual stresses; thus inducing cracking
Processes 1-2-4-5:
Advantages: low thermal residual stresses; thus avoiding cracking
Disadvantages: requires extra equipment and effort for isothermal processing
(c) Process 1-2-4-5 is an isothermal heat treatment due to the constant-temperature duration of 3-4 whereas
Process 1-2-3 is a continuous-cooling heat treatment; hence, not an isothermal thermal processing.
(d) The common heat-treatment names for the processes 1-2-3 and 1-2-4-5 are direct and indirect
quenching, respectively.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 5/12
T 800C
(e) The cooling rate of the process 1-2-3 is estimated as: Q 1600 C
t 0.5 s s
Note: The time scale is logarithmic with a base of 10.
The figure at right shows the phase transformation 1
diagram of an eutectoid steel, where the letters: A, P,
B and M represent austenite, pearlite, bainite and
martensite phases of the eutectoid steel, respectively.
Consider two heat treatments denoted as 1-2-6-7 and 2 6
1-3-4-5 in the figure, respectively.
(a) Identify the microstructures at the locations 1 to
7, respectively.
(b) Which process will produce stronger end product?
Justify the reason(s). 3
4
(c) Determine if either of the two processes can be
considered as an isothermal phase transformation.
Explain why.
(d) What is the common heat-treatment name for the 31.0
process 1-3-4-5?
Ans:
(a) Pt 1 (0s, 800C): 100% austenite
5 7
Pt 2 (0.2s, 600C): 100% austenite
Pt 3 (0.3s, 400C): 100% austenite
3
Pt 4 (10 s, 400C): 100% bainite
Pt 5 (103s, 0C): 100% bainite
Pt 6 (104s, 600C): 100% pearlite
Pt 7 (104s, 0C): 100% pearlite
(b) Processes 1-3-4-5 produces stronger end product since bainite has higher tensile and yield strengths due
to its smaller grain size compared to that of pearlite.
(c) Both Processes 1-2-6-7 and 1-3-4-5 are isothermal heat treatments due to the constant-temperature
durations: 2-6 and 3-4, respectively.
(d) The common heat-treatment name for the Process 1-3-4-5 is austempering.
Note: The time scale is logarithmic with a base of 10.
Cite the differences between pearlite, bainite, and spheroidite relative to microstructure and mechanical
properties.
Ans:
The microstructures of pearlite, bainite, and spheroidite all consist of -ferrite and cementite phases. For
pearlite, the two phases exist as layers which alternate with one another. Bainite consists of very fine and
parallel needle-shaped particles of cementite that are surrounded an -ferrite matrix. For spheroidite, the
matrix is ferrite, and the cementite phase is in the shape of sphere-shaped particles.
Bainite is harder and stronger than pearlite, which, in turn, is harder and stronger than spheroidite.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 6/12
What is the driving force for the formation of spheroidite?
Ans:
The driving force for the formation of spheroidite is the net reduction in ferrite-cementite phase boundary
area.
Continuous-Cooling Transformation Diagrams
Cite two important differences between continuous-cooling transformation diagrams for plain carbon and
alloy steels.
Ans:
Two important differences between continuous cooling transformation diagrams for plain carbon and alloy
steels are:
(1) For an alloy steel, a bainite nose will be present, which nose will be absent for plain carbon alloys; and
(2) The pearlite-proeutectoid noses for plain carbon steel alloys are positioned at shorter times than for the
alloy steels.
The figure at right (Fig. 10.25, p. 381) shows the
superimposition of isothermal and continuous-cooling
transformation diagrams for a eutectoid iron-carbon
alloy. Explain why there is no bainite transformation
region on the continuous-cooling transformation
diagram for an ironcarbon alloy of eutectoid
composition.
Ans:
There is no bainite transformation region on the
continuous cooling transformation diagram for an
iron-carbon alloy of eutectoid composition because
by the time a cooling curve has passed into the bainite
region, the entirety of the alloy specimen will have
transformed to pearlite.
On the basis of diffusion considerations, explain why fine pearlite forms for the moderate cooling of
austenite through the eutectoid temperature, whereas coarse pearlite is the product for relatively slow
cooling rates.
Ans:
For moderately rapid cooling, the time allowed for carbon diffusion is not as great as for slower cooling
rates. Therefore, the diffusion distance is shorter and thinner layers of ferrite and cementite form (i.e., fine
pearlite forms).
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 7/12
Mechanical Behavior of IronCarbon Alloys
Explain why fine pearlite is harder and stronger than coarse pearlite, which in turn is harder and stronger
than spheroidite.
Ans:
The hardness and strength of iron-carbon alloys that have microstructures consisting of -ferrite and
cementite phases depend on the boundary area between the two phases. The greater this area, the harder and
stronger the alloy inasmuch as:
(1) these boundaries impede the motion of dislocations, and
(2) the cementite phase restricts the deformation of the ferrite phase in regions adjacent to the phase
boundaries.
Fine pearlite is harder and stronger than coarse pearlite because the alternating ferrite-cementite layers are
thinner for fine, and therefore, there is more phase boundary area. The phase boundary area between the
sphere-like cementite particles and the ferrite matrix is less in spheroidite than for the alternating layered
microstructure found in coarse pearlite.
Cite two reasons why martensite is so hard and brittle.
Ans:
Two reasons why martensite is so hard and brittle are:
(1) there are relatively few operable slip systems for the body-centered tetragonal crystal structure, and
(2) virtually all of the carbon is in solid solution, which produces a solid-solution hardening effect.
Explain why the hardness of tempered martensite diminishes with tempering time (at constant temperature)
and with increasing temperature (at constant tempering time).
Ans:
This question asks for an explanation as to why the hardness of tempered martensite diminishes with
tempering time (at constant temperature) and with increasing temperature (at constant tempering time). The
hardness of tempered martensite depends on the ferrite-cementite phase boundary area; since these phase
boundaries are barriers to dislocation motion, the greater the area the harder the alloy. The microstructure
of tempered martensite consists of small sphere-like particles of cementite embedded within a ferrite matrix.
As the size of the cementite particles increases, the phase boundary area diminishes, and the alloy becomes
softer. Therefore, with increasing tempering time, the cementite particles grow, the phase boundary area
decreases, and the hardness diminishes. As the tempering temperature is increased, the rate of cementite
particle growth also increases, and the alloy softens, again, because of the decrease in phase boundary area.
(a) Describe the microstructural difference between spheroidite and tempered martensite.
(b) Explain why tempered martensite is much harder and stronger.
Ans:
(a) Both tempered martensite and spheroidite have sphere-like cementite particles within a ferrite matrix;
however, these particles are much larger for spheroidite.
(b) Tempered martensite is harder and stronger inasmuch as there is much more ferrite-cementite phase
boundary area for the smaller particles; thus, there is greater reinforcement of the ferrite phase, and more
phase boundary barriers to dislocation motion.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 8/12
The figure at right shows the ductility (%RA) as a function of
carbon composition for plain carbon steels of three different
microstructures, which are designated as (a), (b) and (c).
Determine which one is coarse pearlite, which one is fine
pearlite and which one is spheroidite. Justify the reason.
Ans:
The microstructures are: (a) spheroidite, (b) coarse pearlite
and (c) fine pearlite. Since at the same carbon composition,
the coarse pearlite microstructure has a larger grain size than the
fine pearlite microstructure, implying coarse pearlite is more
ductile than coarse pearlite. The cementites in a spheroidite form
spherical-shaped particles while in a pearlite they become
needle-shaped. Hence, the spheroidite is the most ductile among
the three.
The photomicrographs below show the microstructures of coarse and fine pearlites, respectively. Rank the
following three Fe-C alloys from the highest to the lowest tensile strength. Justify the ranking.
0.3 wt%C coarse pearlite 0.6 wt%C coarse pearlite 0.6 wt%C fine pearlite
Ans:
The ranking from the highest to the lowest Rockwell hardness
is:
(1) 0.6 wt% C fine pearlite
(2) 0.6 wt% C coarse pearlite
(3) 0.3 wt% C coarse pearlite
While the composition of the alloys is the same, the 0.6wt%C
fine pearlite is stronger than the 0.6wt%C coarse pearlite
because of the smaller particle size, implying many more
ferrite-cementite phase boundaries in fine pearlite, which will
hinder dislocation motions. The 0.6wt%C coarse pearlite is
stronger than the 0.3wt%C coarse pearlite since increasing the coarse pearlite fine pearlite
carbon content increases the strength (while maintained the same coarse pearlite microstructure).
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 9/12
The photomicrographs below show the microstructures of spheroidite and coarse pearlite, respectively.
Rank the following three Fe-C alloys from the highest to the lowest ductility. Justify the ranking.
0.4 wt%C coarse pearlite 0.2 wt%C coarse pearlite 0.2 wt%C spheroidite
Ans:
The ranking from the highest to the lowest ductility is:
(1) 0.2wt%C spheroidite
(2) 0.2wt%C coarse pearlite
(3) 0.4wt%C coarse pearlite
While the composition of the alloys is the same, the
0.2wt%C spheroidite is more ductile than the 0.2wt%C
coarse pearlite because the spherical-shaped cementite
particles in spheroidites make them more ductile than the
coarse pearlites, which contain the needle-shaped
cementites. The 0.2wt%C coarse pearlite is more ductile
than the 0.4wt%C coarse pearlite since decreasing the coarse pearlite spheroidite
carbon content increases the ductility while maintained the same coarse pearlite microstructure.
The photomicrographs below show the microstructures of martensite and tempered martensite, respectively.
Rank the following three Fe-C alloys from the highest to the lowest Rockwell hardness. Justify the ranking.
0.6 wt%C martensite 0.4 wt%C martensite 0.4 wt%C tempered martensite
Ans:
The ranking from the highest to the lowest Rockwell
hardness is:
(1) 0.6 wt% C martensite
(2) 0.4 wt% C martensite
(3) 0.4 wt% C tempered martensite
The 0.6 wt% C martensite is stronger than the 0.4 wt% C
martensite since increasing the carbon content increases the
strength (while maintained the same martensite
microstructure) and the strength is proportional to Rockwell
hardness. While the composition of the alloys is the same,
0.4wt%C martensite is stronger than the 0.4wt% C tempered martensite tempered martensite
martensite because of the BCT structure of martensite, as
indicated by the needle-shaped grains, are very hard and brittle whereas the tempered martensite consists of
extremely small and uniformly dispersed smooth cementite particles embedded within a continuous, softer
ferrite matrix, making it more ductile and tough, yet lower in Rockwell hardness.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 10/12
Metastable vs. Equilibrium States
In terms of heat treatment and the development of microstructure, what are two major limitations of the
ironiron carbide phase diagram?
Ans:
Two limitations of the iron-iron carbide phase diagram are:
(1) The nonequilibrium martensite does not appear on the diagram; and
(2) The diagram provides no indication as to the time-temperature relationships for the formation of
pearlite, bainite, and spheroidite, all of which are composed of the equilibrium ferrite and cementite
phases.
(a) Describe the phenomena of superheating and supercooling.
(b) Why do these phenomena occur?
Ans:
(a) Superheating and supercooling correspond, respectively, to heating or cooling above or below a phase
transition temperature without the occurrence of the transformation.
(b) These phenomena occur because right at the phase transition temperature, the driving force is not
sufficient to cause the transformation to occur. The driving force is enhanced during superheating or
supercooling.
FUNDAMENTALS OF ENGINEERING
Which of the following describes recrystallization?
(A) Diffusion dependent with a change in phase composition
(B) Diffusionless
(C) Diffusion dependent with no change in phase composition
(D) All of the above
Ans: C. Recrystallization is diffusion-dependent with no change in phase composition.
For most technologically important metal alloys, the usual state or microstructure is:
(A) an equilibrium one
(B) a metastable one
Ans: B. For most technologically important alloys, the usual state or microstructure is a metastable one,
which is between initial and equilibrium states.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 11/12
On the basis of accompanying isothermal transformation diagram for a 0.45 wt% C iron-carbon alloy, which
heat treatment could be used to isothermally convert a microstructure that consists of proeutectoid ferrite
and fine pearlite into one that is composed of proeutectoid ferrite and martensite?
(A) Austenitize the specimen at approximately 700C, rapidly cool to about 675C, hold at this temperature
for 1 to 2 s, and then rapidly quench to room temperature
(B) Rapidly heat the specimen to about 675C, hold at this temperature for 1 to 2 s, then rapidly quench to
room temperature
(C) Austenitize the specimen at approximately 775C, rapidly cool to about 500C, hold at this temperature
for 1 to 2 s, and then rapidly quench to room temperature
(D) Austenitize the specimen at approximately 775C, rapidly cool to about 675C, hold at this temperature
for 1 to 2 s, and then rapidly quench to room temperature
Ans: D. Re-austenitize the specimen at approximately 775C, and then rapidly cool to about 675C, hold for 1
to 2 s to form proeutectoid ferrite, then rapidly quench to room temperature to convert the remaining austenite
to martensite.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 10 PHASE TRANSFORMATIONS Page 12/12
Schematic room-temperature microstructures for four ironcarbon alloys are as follows. Rank these
microstructures (by letter) from the hardest to the softest.
(A) (B) (C) (D)
(A) A>B>C>D
(B) C>D>B>A
(C) A>B>D>C
(D) None of the above
Ans: B: C>D>B>A. Specimen C will be the hardest because it has the highest carbon content inasmuch as
proeutectoid Fe3C is present; also the pearlite is fine pearlite. Specimens B and D have approximately the
same carbon content since the proportions of proeutectoid ferrite in both are about the same. However,
carbon contents for B and D are lower than for specimen C because of the presence of proeutectoid ferrite.
Specimen D is harder than B because its pearlite is fine, whereas B's is coarse. Finally, specimen A is the
softest since it is composed entirely of -ferrite and, thus, has the lowest carbon content.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
Das könnte Ihnen auch gefallen
- Question & Answer Set-13Dokument9 SeitenQuestion & Answer Set-13Himanshu SharmaNoch keine Bewertungen
- Engineering Materials PDFDokument28 SeitenEngineering Materials PDFSasikumar100% (1)
- MM435 - CDP-4 - Nernst Equation+Ref ElectrodesDokument23 SeitenMM435 - CDP-4 - Nernst Equation+Ref ElectrodesAbdul Ahad ShamsNoch keine Bewertungen
- Amem201 Group1 Multiple Choice Q&A Amem201Dokument4 SeitenAmem201 Group1 Multiple Choice Q&A Amem201Aero NaveenNoch keine Bewertungen
- Model Question Paper WeldingDokument4 SeitenModel Question Paper Weldingjasminnee100% (1)
- Metallurgists-Quiz No.6 (: Mutiple Choice QuestionsDokument6 SeitenMetallurgists-Quiz No.6 (: Mutiple Choice QuestionsRathnakrajaNoch keine Bewertungen
- Week 11Dokument12 SeitenWeek 11lduran_63Noch keine Bewertungen
- AnswersDokument34 SeitenAnswersMahmoud Elsayed MohamedNoch keine Bewertungen
- WIS5 Paper 2 Rev 3Dokument4 SeitenWIS5 Paper 2 Rev 3Ahmed ShakirNoch keine Bewertungen
- Hypoeutectiod Steel Alloys - SteelDokument52 SeitenHypoeutectiod Steel Alloys - SteelnotsofarNoch keine Bewertungen
- Welding Sample QuestionsDokument4 SeitenWelding Sample Questionsenels770% (2)
- Unit 1 Physics of Welding Arc WeldingDokument106 SeitenUnit 1 Physics of Welding Arc WeldingyashNoch keine Bewertungen
- Multiple Choice 6 Welding ConsumablesDokument8 SeitenMultiple Choice 6 Welding ConsumablesChandra Mohan0% (1)
- Test No 4ccDokument4 SeitenTest No 4ccSalman GhaffarNoch keine Bewertungen
- Engineering Materials Questions and AnswersDokument4 SeitenEngineering Materials Questions and AnswersEmıły WınıfredNoch keine Bewertungen
- Slides - 4Dokument55 SeitenSlides - 4Rahul PandeyNoch keine Bewertungen
- Metallurgy MCQ On Chp1&2Dokument3 SeitenMetallurgy MCQ On Chp1&2Ashutosh Avasekar100% (1)
- Engineering Alloys (Non Ferrous)Dokument52 SeitenEngineering Alloys (Non Ferrous)Sukhwinder Singh GillNoch keine Bewertungen
- WIS5 Paper 3 Rev 3Dokument4 SeitenWIS5 Paper 3 Rev 3Ahmed ShakirNoch keine Bewertungen
- Practice 2 - Questions - CSWIPDokument10 SeitenPractice 2 - Questions - CSWIPravichandran0506Noch keine Bewertungen
- Lecture 1Dokument37 SeitenLecture 1Sarah Syamimi RozaliNoch keine Bewertungen
- Cyaniding: M.Venkatesh Prabhu SPG 16 1431Dokument11 SeitenCyaniding: M.Venkatesh Prabhu SPG 16 1431Sathu satishNoch keine Bewertungen
- CHAPTER-16 Welding Consumable....Dokument9 SeitenCHAPTER-16 Welding Consumable....shahid khanNoch keine Bewertungen
- TEM Lecture CrystallineDokument30 SeitenTEM Lecture CrystallineSyed Abdul AhadNoch keine Bewertungen
- 1614326996093-Objective Questions On Welding & Non-Destructive Testing MRT-10Dokument8 Seiten1614326996093-Objective Questions On Welding & Non-Destructive Testing MRT-10AshishNoch keine Bewertungen
- PMMA Stress-Strain AnalysisDokument5 SeitenPMMA Stress-Strain AnalysisAnam PirachaNoch keine Bewertungen
- As Spec Ri 002 AnswerDokument6 SeitenAs Spec Ri 002 AnswerKarthikeyan GanesanNoch keine Bewertungen
- Closed-Book Practice-Ch 11 (2015!03!16)Dokument17 SeitenClosed-Book Practice-Ch 11 (2015!03!16)Juan100% (1)
- Chapter 8 Phase TransformationsDokument102 SeitenChapter 8 Phase TransformationsAnup DalalNoch keine Bewertungen
- Iron Carbon Phase DiagramDokument4 SeitenIron Carbon Phase DiagramMizanur RahmanNoch keine Bewertungen
- Answers To Materials and Heat Treatment Board Exam Review QuestionsDokument3 SeitenAnswers To Materials and Heat Treatment Board Exam Review Questionsrex tanongNoch keine Bewertungen
- Presentation On Heat TreatmentsDokument34 SeitenPresentation On Heat TreatmentsVinothkumarNoch keine Bewertungen
- Week13 Iron Carbon Phase DiagramDokument49 SeitenWeek13 Iron Carbon Phase DiagramvishalNoch keine Bewertungen
- SolidificationDokument10 SeitenSolidificationAnonymous RY3dAWN9o100% (1)
- 195 Top Engineering Materials - Mechanical Engineering Multiple Choice Questions and AnswersDokument34 Seiten195 Top Engineering Materials - Mechanical Engineering Multiple Choice Questions and AnswersAnand AshkNoch keine Bewertungen
- Afternoon Quiz Set 4Dokument66 SeitenAfternoon Quiz Set 4pchakkrapani100% (1)
- Foundry NoteDokument42 SeitenFoundry NotetaridanNoch keine Bewertungen
- Iron Carbon Phase DiagramDokument4 SeitenIron Carbon Phase DiagramnaveedNoch keine Bewertungen
- Heat TreatmentDokument21 SeitenHeat TreatmentVenkiteshNoch keine Bewertungen
- Heat Treatment ProcessDokument31 SeitenHeat Treatment ProcessAbhishek IndalkarNoch keine Bewertungen
- The Iron-Carbon Phase Diagram: Prof. H. K. Khaira Professor in MSME Deptt. MANIT, BhopalDokument38 SeitenThe Iron-Carbon Phase Diagram: Prof. H. K. Khaira Professor in MSME Deptt. MANIT, BhopalYogesh KumbharNoch keine Bewertungen
- wk7 (3) - Fe-C SystemDokument12 Seitenwk7 (3) - Fe-C Systemsaeed khaledNoch keine Bewertungen
- Soluition Charpter 4 - 1Dokument3 SeitenSoluition Charpter 4 - 1Bryan de BarrosNoch keine Bewertungen
- Bulk Deformation ProcessesDokument41 SeitenBulk Deformation ProcessesAbdullahNoch keine Bewertungen
- Engineering Materials and Metallurgy Questions For Campus Interviews - SanfoundryDokument9 SeitenEngineering Materials and Metallurgy Questions For Campus Interviews - SanfoundryGopinathan MNoch keine Bewertungen
- Api 571 CorrosionDokument5 SeitenApi 571 CorrosionArun GuptaNoch keine Bewertungen
- Welding: Solidification and Microstructure: S.A. David, S.S. Babu, and J.M. VitekDokument11 SeitenWelding: Solidification and Microstructure: S.A. David, S.S. Babu, and J.M. VitekGoriNoch keine Bewertungen
- Crystal Defects: Types, Origins and Impact on Material PropertiesDokument21 SeitenCrystal Defects: Types, Origins and Impact on Material PropertiesAbhijeet Singh KatiyarNoch keine Bewertungen
- 5 Iron-Cementite Phase DiagramDokument46 Seiten5 Iron-Cementite Phase DiagramsmrutiNoch keine Bewertungen
- Iron Carbon Phase DiagramDokument7 SeitenIron Carbon Phase Diagrampratap biswasNoch keine Bewertungen
- Visual Inspection Guide for WeldsDokument11 SeitenVisual Inspection Guide for WeldsAldy Bagus PratamaNoch keine Bewertungen
- Stainless Steels and Alloys - Why They Resist Corrosion and How They FailDokument15 SeitenStainless Steels and Alloys - Why They Resist Corrosion and How They FailMarcos LeiteNoch keine Bewertungen
- Chapter 9d FractureDokument70 SeitenChapter 9d FracturenaveenaNoch keine Bewertungen
- Test No 5Dokument3 SeitenTest No 5Muhammad AsimNoch keine Bewertungen
- Amorphous and Nano Alloys Electroless Depositions: Technology, Composition, Structure and TheoryVon EverandAmorphous and Nano Alloys Electroless Depositions: Technology, Composition, Structure and TheoryNoch keine Bewertungen
- Time Temperature Transformation (TTT) Diagrams PDFDokument108 SeitenTime Temperature Transformation (TTT) Diagrams PDFSerkan Apay100% (1)
- CH 11Dokument7 SeitenCH 11Mario Misael Machado LòpezNoch keine Bewertungen
- Ch-8 Compatibility ModeDokument58 SeitenCh-8 Compatibility Modedreamgurl9011Noch keine Bewertungen
- Effect of Austempering Time On Microstructure and Properties of A Low-Carbon Bainite SteelDokument7 SeitenEffect of Austempering Time On Microstructure and Properties of A Low-Carbon Bainite Steelمسعود بوزويرNoch keine Bewertungen
- Time Temperature Transformation With ReferncesDokument12 SeitenTime Temperature Transformation With ReferncesEllie BrooklynNoch keine Bewertungen
- Closed-Book Practice-Ch 11 (2015!03!16)Dokument17 SeitenClosed-Book Practice-Ch 11 (2015!03!16)Juan100% (1)
- Closed-Book Practice-Ch 15 (2015!03!28)Dokument21 SeitenClosed-Book Practice-Ch 15 (2015!03!28)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 03 (2016!12!10)Dokument8 SeitenClosed-Book Practice-Ch 03 (2016!12!10)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 15 (2015!03!28)Dokument21 SeitenClosed-Book Practice-Ch 15 (2015!03!28)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 13 (2017!04!18)Dokument9 SeitenClosed-Book Practice-Ch 13 (2017!04!18)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 13 (2017!04!18)Dokument9 SeitenClosed-Book Practice-Ch 13 (2017!04!18)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 09 (2017!08!07)Dokument9 SeitenClosed-Book Practice-Ch 09 (2017!08!07)Juan100% (1)
- Closed-Book Practice-Ch 08 (2017!02!26)Dokument13 SeitenClosed-Book Practice-Ch 08 (2017!02!26)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 07 (2016!12!24)Dokument8 SeitenClosed-Book Practice-Ch 07 (2016!12!24)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 11 (2015!03!16)Dokument17 SeitenClosed-Book Practice-Ch 11 (2015!03!16)Juan100% (1)
- Closed-Book Practice-Ch 14 (2015!03!28)Dokument9 SeitenClosed-Book Practice-Ch 14 (2015!03!28)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 09 (2017!08!07)Dokument9 SeitenClosed-Book Practice-Ch 09 (2017!08!07)Juan100% (1)
- Closed-Book Practice-Ch 06 (2016!07!31)Dokument8 SeitenClosed-Book Practice-Ch 06 (2016!07!31)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 12 (2015!03!18)Dokument7 SeitenClosed-Book Practice-Ch 12 (2015!03!18)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 07 (2016!12!24)Dokument8 SeitenClosed-Book Practice-Ch 07 (2016!12!24)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 02 (2017!06!19)Dokument9 SeitenClosed-Book Practice-Ch 02 (2017!06!19)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 03 (2016!12!10)Dokument8 SeitenClosed-Book Practice-Ch 03 (2016!12!10)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 16 (2017!06!01)Dokument11 SeitenClosed-Book Practice-Ch 16 (2017!06!01)Juan100% (6)
- Closed-Book Practice-Ch 04 (2017!09!13)Dokument7 SeitenClosed-Book Practice-Ch 04 (2017!09!13)JuanNoch keine Bewertungen
- Lycra Heat SettingDokument8 SeitenLycra Heat SettingKushagradhi Debnath100% (1)
- Astm b280 1997Dokument10 SeitenAstm b280 1997George GeorgeNoch keine Bewertungen
- PT Montis Energy Welding Procedure SpecificationDokument4 SeitenPT Montis Energy Welding Procedure SpecificationMuhammad Fitransyah Syamsuar PutraNoch keine Bewertungen
- Training trends in Indian retail and FMCG sectorsDokument2 SeitenTraining trends in Indian retail and FMCG sectorssana nazNoch keine Bewertungen
- Disc Coupling Selection GuideDokument28 SeitenDisc Coupling Selection GuideamericolatinoNoch keine Bewertungen
- Cost of Goods Manufactured Formula Excel TemplateDokument5 SeitenCost of Goods Manufactured Formula Excel TemplatePrecious UmingaNoch keine Bewertungen
- 32 Samss 002 PDFDokument35 Seiten32 Samss 002 PDFSahilNoch keine Bewertungen
- Young Touchstone FHF-SSF CatalogueDokument8 SeitenYoung Touchstone FHF-SSF Catalogueangie2702Noch keine Bewertungen
- Chemical Engineering Process Design: Mody and Marchildon: Formation and Processing of SolidsDokument5 SeitenChemical Engineering Process Design: Mody and Marchildon: Formation and Processing of SolidsHarshaNoch keine Bewertungen
- Reclaimed Building Products GuideDokument80 SeitenReclaimed Building Products GuidemymalvernNoch keine Bewertungen
- US Steel Corporation Case Analysis in Strategic ManagementDokument7 SeitenUS Steel Corporation Case Analysis in Strategic ManagementraymundsagumNoch keine Bewertungen
- 03 Intro ERP Using GBI Story (Letter) en v2.11Dokument11 Seiten03 Intro ERP Using GBI Story (Letter) en v2.11Gugun MaulanaNoch keine Bewertungen
- Brochure Ess Apron Feeders Refurbishments 3315 enDokument16 SeitenBrochure Ess Apron Feeders Refurbishments 3315 ensheikmoinNoch keine Bewertungen
- 5 Flooring PDFDokument6 Seiten5 Flooring PDFMasoudRmdnNoch keine Bewertungen
- Solid and Hazardous Waste - Review PPT NotesDokument4 SeitenSolid and Hazardous Waste - Review PPT Notesapi-235658421Noch keine Bewertungen
- ISO 286 - Limits & FitsDokument46 SeitenISO 286 - Limits & FitsindeceNoch keine Bewertungen
- The Global Environment and Operations StrategyDokument73 SeitenThe Global Environment and Operations StrategyJoy LuNoch keine Bewertungen
- tt-750 Tool-Less Closure Installation Operation Manual - Dec 2012 PDFDokument6 Seitentt-750 Tool-Less Closure Installation Operation Manual - Dec 2012 PDFJuan P RuizNoch keine Bewertungen
- List of Indonesian Canned Fish Exporters: Indonesian Trade Promotion Center (Itpc - Osaka)Dokument2 SeitenList of Indonesian Canned Fish Exporters: Indonesian Trade Promotion Center (Itpc - Osaka)SudarismanNoch keine Bewertungen
- 05 Samss 002Dokument8 Seiten05 Samss 002Moustafa BayoumiNoch keine Bewertungen
- Manual de Cuchilla ClarksonDokument4 SeitenManual de Cuchilla ClarksonWalter IvanNoch keine Bewertungen
- M&E Fittings and AccessoriesDokument9 SeitenM&E Fittings and AccessoriesRachel IngramNoch keine Bewertungen
- The Diesel Catalytic ConverterDokument4 SeitenThe Diesel Catalytic ConvertersayeemNoch keine Bewertungen
- A325MDokument8 SeitenA325MmsbarretosNoch keine Bewertungen
- Potable Water Storage Tank SpecificationDokument7 SeitenPotable Water Storage Tank SpecificationTeguh Setiono100% (2)
- Guyana TFO PKG W'SHP Session 3 - Paper and BoardDokument34 SeitenGuyana TFO PKG W'SHP Session 3 - Paper and BoardfrestloviNoch keine Bewertungen
- Aluminum Case - Offshore Box AL 640Dokument6 SeitenAluminum Case - Offshore Box AL 640Alu-Logic Ltd.Noch keine Bewertungen
- 12-Session Vendor Selection and DevelopmentDokument13 Seiten12-Session Vendor Selection and DevelopmentRajnish YadavNoch keine Bewertungen
- Treatment Process For Making Material Softer But Does Not Produce The Uniform Material Properties of AnnealingDokument3 SeitenTreatment Process For Making Material Softer But Does Not Produce The Uniform Material Properties of AnnealingSyahirzabidi100% (1)