Beruflich Dokumente
Kultur Dokumente
Closed-Book Practice-Ch 09 (2017!08!07)
Hochgeladen von
JuanOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Closed-Book Practice-Ch 09 (2017!08!07)
Hochgeladen von
JuanCopyright:
Verfügbare Formate
CHAPTER 9 PHASE DIAGRAMS Page 1/9
CLOSED-BOOK PRACTICE
CHAPTER 9: PHASE DIAGRAMS
CONCEPT CHECK
1. What is the difference between the states of phase equilibrium and metastability?
Ans:
For the condition of phase equilibrium the free energy is a minimum; that is, the system is completely
stable, meaning that over time the phase characteristics are constant. For metastability, the system is not at
equilibrium and there are very slight (and often imperceptible) changes of the phase characteristics with
time.
2. The phase diagram for the cobalt-nickel system is an isomorphous one. On the basis of melting temperatures
for these two metals, describe and/or draw a schematic sketch of the phase diagram for the Co-Ni system.
Ans:
From data inside the front cover, the melting temperatures for cobalt and nickel are, respectively, 1495C
and 1455C. Because this phase diagram is an isomorphous one, phase boundaries and phase regions are
similar to those for the Cu-Ni phase diagram, Figure 9.3athat is, there exists a blade-shaped L region
having intersections of liquidus and solidus lines at 1495C for 100 wt% Co and at 1455C for 100 wt% Ni.
Thus, a schematic phase diagram appears as follows:
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 9 PHASE DIAGRAMS Page 2/9
3. A copper-nickel alloy of composition 70 wt% Ni-30 wt% Cu is slowly heated from a temperature of
1300C.
(a) At what temperature does the first liquid phase form?
(b) What is the composition of this liquid phase?
(c) At what temperature does complete melting of the alloy occur?
(d) What is the composition of the last solid remaining prior to complete melting?
Ans:
Upon heating a copper-nickel alloy of composition 70 wt% Ni-30 wt% Cu from 1300C and utilizing
Fig. 9.3a:
(a) The first liquid forms at the temperature at which a vertical line at this composition intersects the
(L) phase boundaryi.e., about 1350C;
(b) The composition of this liquid phase corresponds to the intersection with the (L)L phase boundary,
of a tie line constructed across the L phase region at 1350Ci.e., 59 wt% Ni;
(c) Complete melting of the alloy occurs at the intersection of this same vertical line at 70 wt% Ni with the
(L)L phase boundaryi.e., about 1380C;
(d) The composition of the last solid remaining prior to complete melting corresponds to the intersection
with (L) phase boundary, of the tie line constructed across the L phase region at 1380Ci.e.,
about 78 wt% Ni.
4. Is it possible to have a copper-nickel alloy that, at equilibrium, consists of an phase of composition
37 wt% Ni-63 wt% Cu, and also a liquid phase of composition 20 wt% Ni-80 wt% Cu? If so, what will be
the approximate temperature of the alloy? If this is not possible, explain why.
Ans:
It is not possible to have a Cu-Ni alloy, which, at equilibrium, consists of a liquid phase of composition
20 wt% Ni-80 wt% Cu and an phase of composition 37 wt% Ni-63 wt% Cu. From Figure 9.3a, a single tie
line does not exist within the L region that intersects the phase boundaries at the given compositions. At
20 wt% Ni, the (L)L phase boundary is at about 1200C, whereas at 37 wt% Ni the (L) phase
boundary is at about 1225C.
5. At 700C (1290F), what is the maximum solubility (a) of Cu in Ag? and (b) of Ag in Cu?
Ans:
(a) From the copper-silver phase diagram, Fig. 9.7, the maximum solubility of Cu in Ag at 700C
corresponds to the position of the () phase boundary at this temperature, or about 6 wt% Cu.
(b) From this same figure, the maximum solubility of Ag in Cu corresponds to the position of the ()
phase boundary at this temperature, or about 5 wt% Ag.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 9 PHASE DIAGRAMS Page 3/9
6. The figure at right is a portion of the H2O-
NaCl phase diagram:
(a) Using this diagram, briefly explain how
spreading salt on ice that is at a
temperature below 0C (32F) can cause
the ice to melt.
(b) At what temperature is salt no longer
useful in causing ice to melt?
Ans:
(a) As shown in the figure, the eutectic
point, which is marked as E, occurs at
(76.7wt% H 2O & 23.3wt% NaCl @
21.2C). Thus, spreading salt on ice
will lower the melting temperature,
since the liquidus line decreases from
0C (at 100% H2O) to the eutectic
temperature at about 21.2C. That is,
ice at a temperature below 0C (and
above 21C) can be made to form a
liquid phase by the addition of salt.
(b) Below 21C salt is no longer useful in causing ice to melt because this is the lowest temperature at
which a liquid phase forms (i.e., it is the eutectic temperature for this system).
7. The following figure is the hafnium-vanadium phase diagram, for which only single-phase regions are
labeled. Specify temperature-composition points at which all eutectics, eutectoids, peritectics, and congruent
phase transformations occur. Also, for each, write the reaction upon cooling.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 9 PHASE DIAGRAMS Page 4/9
Ans:
There are two eutectics ( Ec ) on this phase diagram. One exists at 18 wt% V-82 wt% Hf and 1455C. The
reaction upon cooling is: L Hf HfV2 . The other eutectic exists at 39 wt% V-61 wt% Hf and 1520C.
This reaction upon cooling is: L HfV2 V solid solution .
There is one eutectoid ( Ed ) at 6 wt% V-94 wt% Hf and 1190C. Its reaction upon cooling is:
Hf Hf HfV2
There is one congruent melting point ( C ) at 36 wt% V-64 wt% Hf and 1550C. The reaction upon cooling
is: L HfV2 .
No peritectics are present.
8. For a ternary system, three components are present; temperature is also a variable. What is the maximum
number of phases that may be present for a ternary system, assuming that pressure is held constant?
Ans:
For a ternary system (C = 3) at constant pressure (N = 1), Gibbs phase rule, Eq (9.16), becomes:
P F C N 3 1 4 or P 4 F . Thus, when F = 0, P will have its maximum value of 4, which
means that the maximum number of phases present for this situation is 4.
9. Explain why a proeutectoid phase (ferrite or cementite) forms along austenite grain boundaries. Hint:
Consult Sec. 4.6.
Ans:
Associated with grain boundaries is an interfacial energy (i.e., grain boundary energySec. 4.6). A lower
net interfacial energy increase results when a proeutectoid phase forms along existing austenite grain
boundaries than when the proeutectoid phase forms within the interior of the grains.
QUESTIONS & PROBLEMS
Microstructure
Cite three variables that determine the microstructure of an alloy.
Ans:
Three variables that determine the microstructure of an alloy are:
(1) the alloying elements present,
(2) the concentrations of these alloying elements, and
(3) the heat treatment of the alloy.
Phase Equilibria
What thermodynamic condition must be met for a state of equilibrium to exist?
Ans:
In order for a system to exist in a state of equilibrium the free energy must be a minimum for some specified
combination of temperature, pressure, and composition.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 9 PHASE DIAGRAMS Page 5/9
Binary Isomorphous Systems
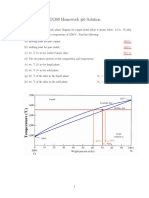
Shown below is the copper-nickel (Cu-Ni) binary isomorphous phase diagram, where L and represent
liquid and solid Cu-Ni alloy phases, respectively. Consider 100g of a copper-nickel alloy of composition
35 wt% Ni is cooled from a liquid phase to 1250C?
(a) How many grams of the alloy are in the liquid and phases, respectively?
(b) How many grams of nickel are contained in these liquid and phases, respectively?
32
1250
43
35
Ans:
(a) Given that the overall nickel composition is: C0 35 wt% . As shown in the tie line above, we have:
CL 32 wt% and C 43 wt% . From the lever rule, we obtain:
C C0 43 35
WL 72.27 wt% 72.27 grams of the Cu-Ni alloy are in the liquid phase
C CL 43 32
C CL 35 32
W 0 27.27 wt% 27.27 grams of the Cu-Ni alloy are in the phase
C CL 43 32
WNi in L CL WL 32 wt% 72.73 wt% 23.27 wt%
(b) By definition,
WNi in C W 43 wt% 27.27 wt% 11.73 wt%
The liquid phase contains 23.27 grams of nickel and the balance of 49.46 (= 72.73 23.27) grams is
copper. Similarly, the phase contains 11.73 grams of nickel and balance of 15.54 (= 27.27 11.73)
grams is copper.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 9 PHASE DIAGRAMS Page 6/9
Phase Diagrams
The lead-tin (Pb-Sn) alloy is frequently used
for soldering. Based on the Pb-Sn phase
diagram shown at right, propose a Pb-Sn
composition for soldering material that will be
the most economical. Also at what
temperature should soldering be operated for
the proposed composition? Explain why?
Also, what is the common name of this
specific composition-temperature in the Pb-Sn
phase diagram?
Ans:
As shown in the figure, the specific
composition-temperature of 61.9 wt% Sn @
183C, which is marked as E, will be the most
economical for soldering operation since the
melting temperature is the lowest among all
lead-tin (Pb-Sn) alloys. For the Pb-Sn phase diagram, E is called a eutectic point, which has the following
phase transformation: L ; that is, a liquid phase transforms into a mixture of two solid phases.
The figure at right shows a copper-zinc
(Cu-Zn) binary phase diagram. Here L stands
for the liquid phase while all Greek letters
represent various solid phases. Mark all the
invariant reaction points (i.e., eutectic,
eutectoid, peritectic, peritectoid, monotectic or
monotectoid) on the figure. Justify your
reasons.
Ans:
As shown in the figure, there are two
peritectic points, marked as P since
L
. There is also a eutectoid point,
L
marked as E since .
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 9 PHASE DIAGRAMS Page 7/9
The figure on the right shows an iron-
carbon (Fe-C) binary phase diagram.
Here L stands for the liquid phase, Fe3C
means the solid cementite phase while
all Greek letters represent other various
solid phases. Mark all the invariant
reaction points (i.e., eutectic, eutectoid,
peritectic, peritectoid, monotectic or
monotectoid) on the figure. Justify your
reasons.
Ans:
As shown in the figure, there are one
peritectic point, marked as P since
L Austenite ; one eutectic
point, marked as F since
L Austenite Fe3C Cementite ;
and one eutectoid point, marked as E since Austenite Ferrite Fe3C Cementite .
Development of Microstructure in Isomorphous Alloys
(a) Describe the phenomenon of coring and why it occurs.
(b) Cite one undesirable consequence of coring.
Ans:
(a) Coring is the phenomenon whereby concentration gradients exist across grains in polycrystalline alloys,
with higher concentrations of the component having the lower melting temperature at the grain
boundaries. It occurs, during solidification, as a consequence of cooling rates that are too rapid to allow
for the maintenance of the equilibrium composition of the solid phase.
(b) One undesirable consequence of a cored structure is that, upon heating, the grain boundary regions will
melt first and at a temperature below the equilibrium phase boundary from the phase diagram; this
melting results in a loss in mechanical integrity of the alloy.
Development of Microstructure in Eutectic Alloys
Explain why, upon solidification, an alloy of eutectic composition forms a microstructure consisting of
alternating layers of the two solid phases.
Ans:
Upon solidification, an alloy of eutectic composition forms a microstructure consisting of alternating layers
of the two solid phases because during the solidification atomic diffusion must occur, and with this layered
configuration the diffusion path length for the atoms is a minimum.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 9 PHASE DIAGRAMS Page 8/9
What is the difference between a phase and a microconstituent?
Ans:
A phase is a homogeneous portion of the system having uniform physical and chemical characteristics,
whereas a microconstituent is an identifiable element of the microstructure (that may consist of more than
one phase).
Congruent Phase Transformations/Eutectoid and Peritectic Reactions
What is the principal difference between congruent and incongruent phase transformations?.
Ans:
The principal difference between congruent and incongruent phase transformations is that for congruent no
compositional changes occur with any of the phases that are involved in the transformation. For incongruent
there will be compositional alterations of the phases.
The IronIron Carbide (FeFe3C) Phase Diagram/Development of Microstructure in IronCarbon Alloys
(a) What is the distinction between hypoeutectoid and hypereutectoid steels?
(b) In a hypoeutectoid steel, both eutectoid and proeutectoid ferrite exist. Explain the difference between
them. What will be the carbon concentration in each?
Ans:
(a) A hypoeutectoid steel has a carbon concentration less than the eutectoid; on the other hand, a
hypereutectoid steel has a carbon content greater than the eutectoid.
(b) For a hypoeutectoid steel, the proeutectoid ferrite is a microconstituent that formed above the eutectoid
temperature. The eutectoid ferrite is one of the constituents of pearlite that formed at a temperature
below the eutectoid. The carbon concentration for both ferrites is 0.022 wt% C.
FUNDAMENTALS OF ENGINEERING
Once a system is at a state of equilibrium, a shift from equilibrium may result by alteration of which of the
following?
(A) Pressure
(B) Temperature
(C) Composition
(D) All of the above
Ans: D. A shift from equilibrium may result from alteration of temperature, pressure, and/or composition.
When the concentration of solute in a solid solution exceeds the solubility limit, which of the following
situations results?
(A) The formation of a new solid solution or phase that has a composition similar to that of the original solid
solution
(B) The formation of a new solid solution or phase that has a distinctly different composition than the
original solid solution
Ans: B. When the concentration of solute in a solid solution exceeds the solubility limit, a new solid
solution or phase forms that has a distinctly different composition than the original solid solution.
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
CHAPTER 9 PHASE DIAGRAMS Page 9/9
A binary compositiontemperature phase diagram for an isomorphous system will be composed of regions
that contain which of the following phases and/or combinations of phases?
(A) Liquid
(B) (Liquid)
(C)
(D) , liquid and (liquid)
Ans: D. For an isomorphous system the two components are completely miscible in both the liquid and
solid phases; thus, the phase regions are as follows: , liquid and (liquid).
A solidus line separates which of the following phase fields?
(A) Liquid and (liquid)
(B) and (liquid)
(C) and ( )
(D) (Liquid) and (liquid)
Ans: B. A solidus line separates and (liquid) phase fields.
A eutectoid reaction involves which of the following phases?
(A) One liquid and one solid
(B) One liquid and two solid
(C) Two liquids and one solid
(D) Three solid
Ans: D. A eutectoid reaction: involves three solid phases: , and .
From the leadtin phase diagram (Figure 9.8), which of the following phases/phase combinations is present
for an alloy of composition 46 wt% Sn54 wt% Pb that is at equilibrium at 44C?
(A)
(B)
(C) liquid
(D) liquid
Ans: B. Locate this temperature-composition point on the phase diagram. Inasmuch as it is within the
region, both alpha beta phases will be present.
For a leadtin alloy of composition 25 wt% Sn-75 wt% Pb, select from the following list the phase(s)
present and their composition(s) at 200C. (The Pb-Sn phase diagram appears in Figure 9.8.)
(A) = 17 wt% Sn-83 wt% Pb; L = 55.7 wt% Sn-44.3 wt% Pb
(B) = 25 wt% Sn-75 wt% Pb; L = 25 wt% Sn-75 wt% Pb
(C) = 17 wt% Sn-83 wt% Pb; L = 55.7 wt% Sn-44.3 wt% Pb
(D) = 18.3 wt% Sn-81.7 wt% Pb; L = 97.8 wt% Sn-2.2 wt% Pb
Ans: A. Locate this temperature-composition point on the phase diagram. Inasmuch as it is within the L
region, at 200C both and liquid phases are present. A tie line drawn across the L phase field intersects
the (L) phase boundary at about 17 wt% Sn-83 wt% Pb (which is the composition of the phase) and
the L(L) phase boundary at about 55.7 wt% Sn-44.3 wt% Pb (which is the composition of the liquid
phase).
ME 46100: ENGINEERING MATERIALS CLOSED-BOOK PRACTICE
Das könnte Ihnen auch gefallen
- HW 9 2011 SolutionsDokument15 SeitenHW 9 2011 SolutionsParamesh Anugu100% (1)
- Assignment 7 SolutionsDokument11 SeitenAssignment 7 SolutionsIsaiah Qe LiewNoch keine Bewertungen
- Question & Answer Set-7Dokument12 SeitenQuestion & Answer Set-7eeng.ali651550% (2)
- Phase Diagrams: Solubility LimitDokument163 SeitenPhase Diagrams: Solubility LimitseaNoch keine Bewertungen
- Closed-Book Practice-Ch 11 (2015!03!16)Dokument17 SeitenClosed-Book Practice-Ch 11 (2015!03!16)Juan100% (1)
- Problems Phase FeDokument9 SeitenProblems Phase FeAshutoshKumarNoch keine Bewertungen
- Homework and Solutions - ch5 Ch6.IMSDokument18 SeitenHomework and Solutions - ch5 Ch6.IMSHery RobiyantoroNoch keine Bewertungen
- 17 Askeland ChapDokument4 Seiten17 Askeland ChapIngrid Zetina Tejero100% (1)
- Assigned Problems - Week of February 13Dokument5 SeitenAssigned Problems - Week of February 13Anam PirachaNoch keine Bewertungen
- Material Sheet Solution CH 4Dokument3 SeitenMaterial Sheet Solution CH 4David Sahry Mark100% (1)
- CH 13Dokument30 SeitenCH 13Laurertan TavaresNoch keine Bewertungen
- Chapter 5 Practice Problems SolutionsDokument8 SeitenChapter 5 Practice Problems SolutionsShafiq HafizullahNoch keine Bewertungen
- Question & Answer Set-5Dokument8 SeitenQuestion & Answer Set-5rahul kumar100% (1)
- Resolução Exercs. Cienc. Dos MateriaisDokument19 SeitenResolução Exercs. Cienc. Dos MateriaisRafael AraújoNoch keine Bewertungen
- ch11 AllProblem KeyDokument56 Seitench11 AllProblem Keyladyinred90Noch keine Bewertungen
- CH 21Dokument32 SeitenCH 21Ayyappa SwamyNoch keine Bewertungen
- Material Science Cht04 and Cht08Dokument43 SeitenMaterial Science Cht04 and Cht08Arnaldo Bester67% (3)
- CH 21Dokument32 SeitenCH 21Indro ParmaNoch keine Bewertungen
- Heat Transfer Final Exam Problem & SolutionsDokument8 SeitenHeat Transfer Final Exam Problem & SolutionsRonard Pa–a0% (1)
- ch12 PDFDokument66 Seitench12 PDFSurekha Pravin AlshiNoch keine Bewertungen
- CH 14Dokument41 SeitenCH 14IlhamBintangNoch keine Bewertungen
- 21 Askeland ChapDokument8 Seiten21 Askeland Chapel_dualcNoch keine Bewertungen
- Closed-Book Practice-Ch 12 (2015!03!18)Dokument7 SeitenClosed-Book Practice-Ch 12 (2015!03!18)JuanNoch keine Bewertungen
- HW8 ch07Dokument7 SeitenHW8 ch07Jeremy HernandezNoch keine Bewertungen
- 19 Askeland ChapDokument6 Seiten19 Askeland Chapel_dualcNoch keine Bewertungen
- Engineering Materials 2 - Chapter 18Dokument4 SeitenEngineering Materials 2 - Chapter 18Andres PerezNoch keine Bewertungen
- Material Science and Engineering Sol. Chap. 20Dokument45 SeitenMaterial Science and Engineering Sol. Chap. 20Patrick Gibson100% (1)
- CH 07Dokument60 SeitenCH 07IlhamBintang100% (1)
- Steam TturbineDokument2 SeitenSteam Tturbinemanish_chaturvedi_6Noch keine Bewertungen
- Material Science and Engineering Ch. 10 SolDokument72 SeitenMaterial Science and Engineering Ch. 10 SolPatrick GibsonNoch keine Bewertungen
- Exercícios CallisterDokument66 SeitenExercícios CallistermicrovilosidadesNoch keine Bewertungen
- Unit 1 Lecture 3 QFETDokument17 SeitenUnit 1 Lecture 3 QFETSaketNoch keine Bewertungen
- 20 Askeland ChapDokument10 Seiten20 Askeland Chapel_dualcNoch keine Bewertungen
- Text Cross Within TheDokument475 SeitenText Cross Within Theversion3Noch keine Bewertungen
- Closed-Book Practice-Ch 14 (2015!03!28)Dokument9 SeitenClosed-Book Practice-Ch 14 (2015!03!28)JuanNoch keine Bewertungen
- Mech 285 True or FalseDokument18 SeitenMech 285 True or FalseKayleigh RobotnikNoch keine Bewertungen
- ChE 413N Problem Set 1 - Atomic Structure-Ionic Arrangements 4-20-16vstudDokument1 SeiteChE 413N Problem Set 1 - Atomic Structure-Ionic Arrangements 4-20-16vstudLouie G NavaltaNoch keine Bewertungen
- Callister Solutions of Ch08Dokument42 SeitenCallister Solutions of Ch08Malika Navaratna100% (1)
- ch31 PDFDokument26 Seitench31 PDFRodrigo S QuirinoNoch keine Bewertungen
- Combustion: Luminous Gas Flame Flame Front Luminous Non-LuminousDokument18 SeitenCombustion: Luminous Gas Flame Flame Front Luminous Non-LuminousMd. Ahsanur RahmanNoch keine Bewertungen
- HW#5 SolutionDokument5 SeitenHW#5 Solutionmtydaics100% (2)
- Chapter 15 - Oscillations - Solutions PDFDokument9 SeitenChapter 15 - Oscillations - Solutions PDFrobin bhalroyNoch keine Bewertungen
- EN380 Homework #6 Solution: 1084 C 1455 C 70% N i L + α C = 61% C = 73% W = = = 25% W = = = 75%Dokument4 SeitenEN380 Homework #6 Solution: 1084 C 1455 C 70% N i L + α C = 61% C = 73% W = = = 25% W = = = 75%roseNoch keine Bewertungen
- MSE20OTDokument212 SeitenMSE20OTゆかり100% (1)
- Chemical EquilibriumDokument18 SeitenChemical EquilibriumCarbuncle JonesNoch keine Bewertungen
- CH 09Dokument8 SeitenCH 09Antoinette ChuaNoch keine Bewertungen
- CH 10Dokument9 SeitenCH 1000gp1200r100% (1)
- 4Dokument63 Seiten4ARJUN PESARU 19BME0161Noch keine Bewertungen
- Phase Diagram ExDokument23 SeitenPhase Diagram ExTey KaijingNoch keine Bewertungen
- Phase Diagram - : Dr. Aneela WakeelDokument21 SeitenPhase Diagram - : Dr. Aneela WakeelHassan KhanNoch keine Bewertungen
- Week 11Dokument12 SeitenWeek 11lduran_63Noch keine Bewertungen
- Assignment 8 SolutionDokument6 SeitenAssignment 8 SolutionBrishen Hawkins100% (1)
- Materials Science and Engineering Concept Check Part2 PDFDokument27 SeitenMaterials Science and Engineering Concept Check Part2 PDF李宛芸Noch keine Bewertungen
- Phase DiagramDokument47 SeitenPhase DiagramMuhammed MansNoch keine Bewertungen
- Copper NickelDokument5 SeitenCopper NickelFahri Ali ImranNoch keine Bewertungen
- 104 PhaseDiags QS2AnsDokument6 Seiten104 PhaseDiags QS2Ansnilanga123Noch keine Bewertungen
- Anup Sir PPT All PDFDokument222 SeitenAnup Sir PPT All PDFFAIQNoch keine Bewertungen
- Concept Check Qa Ch07-21Dokument106 SeitenConcept Check Qa Ch07-21ellie<3Noch keine Bewertungen
- Assignment PhaseDiaDokument5 SeitenAssignment PhaseDiaAnshu Kumar GuptaNoch keine Bewertungen
- A Modern Course in Statistical PhysicsVon EverandA Modern Course in Statistical PhysicsBewertung: 3.5 von 5 Sternen3.5/5 (2)
- Closed-Book Practice-Ch 15 (2015!03!28)Dokument21 SeitenClosed-Book Practice-Ch 15 (2015!03!28)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 15 (2015!03!28)Dokument21 SeitenClosed-Book Practice-Ch 15 (2015!03!28)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 03 (2016!12!10)Dokument8 SeitenClosed-Book Practice-Ch 03 (2016!12!10)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 07 (2016!12!24)Dokument8 SeitenClosed-Book Practice-Ch 07 (2016!12!24)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 13 (2017!04!18)Dokument9 SeitenClosed-Book Practice-Ch 13 (2017!04!18)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 10 (2017!08!08)Dokument12 SeitenClosed-Book Practice-Ch 10 (2017!08!08)Juan0% (1)
- Closed-Book Practice-Ch 14 (2015!03!28)Dokument9 SeitenClosed-Book Practice-Ch 14 (2015!03!28)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 07 (2016!12!24)Dokument8 SeitenClosed-Book Practice-Ch 07 (2016!12!24)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 12 (2015!03!18)Dokument7 SeitenClosed-Book Practice-Ch 12 (2015!03!18)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 02 (2017!06!19)Dokument9 SeitenClosed-Book Practice-Ch 02 (2017!06!19)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 06 (2016!07!31)Dokument8 SeitenClosed-Book Practice-Ch 06 (2016!07!31)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 03 (2016!12!10)Dokument8 SeitenClosed-Book Practice-Ch 03 (2016!12!10)JuanNoch keine Bewertungen
- Closed-Book Practice-Ch 16 (2017!06!01)Dokument11 SeitenClosed-Book Practice-Ch 16 (2017!06!01)Juan100% (6)
- Modul Perefect Score Matematik Tambahan Melaka 2014Dokument65 SeitenModul Perefect Score Matematik Tambahan Melaka 2014Muhammad Hazwan RahmanNoch keine Bewertungen
- Metric System Hand OutDokument3 SeitenMetric System Hand OutKenny TothNoch keine Bewertungen
- Robots in Radioactive EnvironmentsDokument21 SeitenRobots in Radioactive EnvironmentsMohimina Jawariyah67% (3)
- Leader DLP Review Test 1 QPDokument56 SeitenLeader DLP Review Test 1 QPharshit pandeyNoch keine Bewertungen
- Timeline History of ScienceDokument6 SeitenTimeline History of ScienceMatara Ligaya GarciaNoch keine Bewertungen
- Astm D3282 93 - 260421Dokument6 SeitenAstm D3282 93 - 260421Mark D VillanuevaNoch keine Bewertungen
- Sullair WSDokument70 SeitenSullair WSToso Eko Purwanto100% (1)
- DLP in Science 3Dokument12 SeitenDLP in Science 3Jefferson SisonNoch keine Bewertungen
- Quizlet Com 8175072 Atpl Ins Formulas Flash CardsDokument6 SeitenQuizlet Com 8175072 Atpl Ins Formulas Flash Cardsnathan zizekNoch keine Bewertungen
- Viscosimetro OfiteDokument20 SeitenViscosimetro OfitejomarlucaNoch keine Bewertungen
- LT Steel STDokument9 SeitenLT Steel STSyed Umair RizviNoch keine Bewertungen
- Power Electronics Unit1Dokument92 SeitenPower Electronics Unit1ashish_rewaNoch keine Bewertungen
- RF316Dokument2 SeitenRF316Marcelo Carneiro de PaivaNoch keine Bewertungen
- Spectacle Lens OptionsDokument11 SeitenSpectacle Lens OptionsJitander Dudee100% (1)
- Van Eekelen Et Al 2011 BS8006 FinalDokument15 SeitenVan Eekelen Et Al 2011 BS8006 FinalRaden Budi HermawanNoch keine Bewertungen
- Seismic Load AnalysisDokument99 SeitenSeismic Load AnalysisMANDARAW100% (1)
- IPSF Code of Points 2017-18 Final EnglishDokument135 SeitenIPSF Code of Points 2017-18 Final EnglishIsabela BiazottoNoch keine Bewertungen
- S-18 20Dokument2 SeitenS-18 20Raghavendra MurthyNoch keine Bewertungen
- Compressive Test of Cylindrical Concrete SpecimenDokument5 SeitenCompressive Test of Cylindrical Concrete SpecimenJenevive TumacderNoch keine Bewertungen
- Screw Thread MeasurementDokument55 SeitenScrew Thread MeasurementMadhuNoch keine Bewertungen
- Journal FYP 6Dokument7 SeitenJournal FYP 6Mira FazziraNoch keine Bewertungen
- Fiberbond Fiberglass Piping Systems Series 110FW: DescriptionDokument2 SeitenFiberbond Fiberglass Piping Systems Series 110FW: DescriptionIbhar Santos MumentheyNoch keine Bewertungen
- Conductivity TesterDokument4 SeitenConductivity TesterFelix PintoNoch keine Bewertungen
- Normal in Vol TimeDokument6 SeitenNormal in Vol TimeIgorNoch keine Bewertungen
- Combustion Process SimulationDokument6 SeitenCombustion Process SimulationKingsley Etornam AnkuNoch keine Bewertungen
- Fracture MechanicsDokument11 SeitenFracture MechanicsDEEPAKNoch keine Bewertungen
- P22 23 CRD PDFDokument2 SeitenP22 23 CRD PDFMikka IdanoNoch keine Bewertungen
- Astm c1439Dokument4 SeitenAstm c1439Nik TarazNoch keine Bewertungen
- PW and Wfi Storage and Distribution Systems Design and OperationDokument33 SeitenPW and Wfi Storage and Distribution Systems Design and Operationmanojdhamne5802Noch keine Bewertungen
- Computer Controlled Process Control System With SCADA and PID ControlDokument12 SeitenComputer Controlled Process Control System With SCADA and PID Controldwi meyNoch keine Bewertungen