Beruflich Dokumente
Kultur Dokumente
CH11SB061
Hochgeladen von
Rez Caitlina0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
6 Ansichten1 SeiteChemistry 11
Copyright
© © All Rights Reserved
Verfügbare Formate
PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenChemistry 11
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
6 Ansichten1 SeiteCH11SB061
Hochgeladen von
Rez CaitlinaChemistry 11
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 1
Chapter 1 Summary
Key Expectations Key Terms
Throughout this chapter you have had the actinide law of constant composition
opportunity to do the following: alchemy line spectrum
• Demonstrate an understanding of the periodic law, and alkali metal mass number (A)
describe how electron arrangements and forces within alkaline earth metal mass spectrometer
atoms can explain periodic trends such as atomic analogy metal
radius, ionic radius, ionization energy, electron affinity, atom metalloid
and electronegativity. (1.1, 1.2, 1.4, 1.5) atomic mass model
• Identify chemical substances and reactions in everyday atomic number (Z) neutron
use or of environmental significance. (1.1, 1.3) atomic radius neutron number (N)
• Define and describe the relationship among atomic carbon-14 dating noble gas
number, mass number, and atomic mass, isotope, and compound nonmetal
radioisotope. (1.3) continuous spectrum nucleus
• Identify and describe careers associated with matter. (1.3) diagnostic test orbit
• Relate the reactivity of a series of elements to their posi- electron period
tion in the periodic table. (1.5) electron affinity periodic law
electron cloud periodic trend
• Analyze data involving periodic properties such as ion-
electronegativity principal quantum number
ization energy and atomic radius in order to recognize
element proton
general trends in the periodic table. (1.5)
empirical definition quantitative
empirical knowledge quantum mechanics
energy level radioactive
Make a excited radioisotope
Summary fission representative element
flame test SATP
Summarize the concepts that you have learned ground state STP
in this chapter by creating a concept map. Begin group theoretical knowledge
with the word “element” and use as many words
half-life theory
as possible from the list of key words.
halogen transition
ion transition metal
ionization energy transuranic element
Reflectyour
on
Learning
isotope
IUPAC
trend
triad
Revisit your answers to the Reflect on Your Learning ques- lanthanide unified atomic mass (u)
tions at the beginning of this chapter. law of conservation of valence electrons
mass
• How has your thinking changed?
• What new questions do you have?
The Nature of Matter 61
Das könnte Ihnen auch gefallen
- Year 9 End of Year Assessment 2023 PLCDokument1 SeiteYear 9 End of Year Assessment 2023 PLCVaidile JonikasNoch keine Bewertungen
- Syllabus PH 5118 & 5218Dokument5 SeitenSyllabus PH 5118 & 5218Findx proNoch keine Bewertungen
- B.Sc. Physics Fifth Semester Elective IDokument11 SeitenB.Sc. Physics Fifth Semester Elective ISavitri BhandariNoch keine Bewertungen
- Phymerge PDFDokument181 SeitenPhymerge PDFvenkateshNoch keine Bewertungen
- Course: Department Specific Elective 2-: HPHDS5021T Nuclear and Particle PhysicsDokument3 SeitenCourse: Department Specific Elective 2-: HPHDS5021T Nuclear and Particle PhysicsGrim Reaper Kuro OnihimeNoch keine Bewertungen
- Physics NotesDokument131 SeitenPhysics NotesshridharNoch keine Bewertungen
- Sokolov - Quantum MehcanicsDokument566 SeitenSokolov - Quantum MehcanicsGianniNicheli100% (1)
- AL Chemistry SyllabusDokument33 SeitenAL Chemistry Syllabusapi-3734333Noch keine Bewertungen
- Bscpe Sci1 SLM 6Dokument36 SeitenBscpe Sci1 SLM 6April Joy GonzalesNoch keine Bewertungen
- UTAR Centre for Foundation Studies Inorganic Chemistry CourseDokument6 SeitenUTAR Centre for Foundation Studies Inorganic Chemistry CourseBrandon OoiNoch keine Bewertungen
- Faraday Research Article Reactive Scattering Atoms and RadicalsDokument22 SeitenFaraday Research Article Reactive Scattering Atoms and RadicalsgiampieroNoch keine Bewertungen
- H Chem Electron STructure Test Prep WKSHTDokument4 SeitenH Chem Electron STructure Test Prep WKSHTErin RiegoNoch keine Bewertungen
- Chapter 1 NMR TheoryDokument63 SeitenChapter 1 NMR TheoryabhiNoch keine Bewertungen
- Decay of Radioactivity: Robert Miyaoka, PHDDokument9 SeitenDecay of Radioactivity: Robert Miyaoka, PHDTieku NobleNoch keine Bewertungen
- Organic Spectroscopic AnalysisDokument186 SeitenOrganic Spectroscopic AnalysisNguyễn Ngọc Phước VươngNoch keine Bewertungen
- Scheme DSCE 1st Yr UG Scheme 2016Dokument61 SeitenScheme DSCE 1st Yr UG Scheme 2016Saathvik Bhat100% (1)
- DSCE ENGINEERING MATHEMATICS-I AND PHYSICS SYLLABUSDokument119 SeitenDSCE ENGINEERING MATHEMATICS-I AND PHYSICS SYLLABUSNandan MaheshNoch keine Bewertungen
- 5 Studyguide KeyDokument3 Seiten5 Studyguide KeyAnonymous PersonNoch keine Bewertungen
- Chemistry Study Guide MYP 4Dokument1 SeiteChemistry Study Guide MYP 4JamesMartinNoch keine Bewertungen
- Topic 3 - Periodicity SLDokument20 SeitenTopic 3 - Periodicity SLnikes 1Noch keine Bewertungen
- Silabus Material Teknik ElektroDokument4 SeitenSilabus Material Teknik ElektroNajmi KertasafariNoch keine Bewertungen
- Periodic Trends Think Ib SlidfesDokument10 SeitenPeriodic Trends Think Ib Slidfesamaya chopraNoch keine Bewertungen
- 07AMSCH23 - Physical Chemistry - IIDokument260 Seiten07AMSCH23 - Physical Chemistry - IIALPHY100% (1)
- Atomic Structure and Gaseous State ModuleDokument122 SeitenAtomic Structure and Gaseous State Moduleruchir angraNoch keine Bewertungen
- Unit 2+12 TARGET SHEET HL Atomic StructureDokument2 SeitenUnit 2+12 TARGET SHEET HL Atomic Structurediya joshiNoch keine Bewertungen
- CH 7 Atomic TheoryDokument73 SeitenCH 7 Atomic TheoryMurk FatimaNoch keine Bewertungen
- Seperation TechniquesDokument17 SeitenSeperation TechniquesJagadeesh YNoch keine Bewertungen
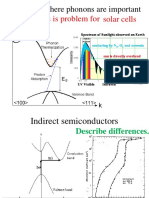
- Solar Cells Energy Loss Is Problem For: Examples Where Phonons Are ImportantDokument51 SeitenSolar Cells Energy Loss Is Problem For: Examples Where Phonons Are Importantjose mirandaNoch keine Bewertungen
- 1.1 Revision Guide Atomic Structure AqaDokument10 Seiten1.1 Revision Guide Atomic Structure AqadodoNoch keine Bewertungen
- Paper V Unit-I: Classical Mechanics Ii (25 Marks) LECTURES 25 + 5 TutorialDokument5 SeitenPaper V Unit-I: Classical Mechanics Ii (25 Marks) LECTURES 25 + 5 Tutorialzoo zooNoch keine Bewertungen
- Tenorio 2005Dokument10 SeitenTenorio 2005LuisOctavioJojoLosadaNoch keine Bewertungen
- Intro To Phonons-CastepDokument57 SeitenIntro To Phonons-CastepgadasNoch keine Bewertungen
- Nuclear Chemistry BasicsDokument10 SeitenNuclear Chemistry BasicsAlemkeng BrendaNoch keine Bewertungen
- Chem 113E Module 5 Nuclear ChemistryDokument20 SeitenChem 113E Module 5 Nuclear ChemistryKenneth John FerrarizNoch keine Bewertungen
- A Level Chemistry Two Year Linear Course Scheme of WorkDokument64 SeitenA Level Chemistry Two Year Linear Course Scheme of WorkRehan RafiqNoch keine Bewertungen
- Topic 3 Periodicity SLDokument21 SeitenTopic 3 Periodicity SLLyv SantosaNoch keine Bewertungen
- ASP2012 - Particle InteractionsDokument79 SeitenASP2012 - Particle InteractionsvictorNoch keine Bewertungen
- m1 Properties Structure of Matter ChemistryDokument8 Seitenm1 Properties Structure of Matter ChemistrysarahsyedazakiNoch keine Bewertungen
- Periodic Trends, Valence e- & LEDSDokument5 SeitenPeriodic Trends, Valence e- & LEDSJoseph ZafraNoch keine Bewertungen
- Materials 10 01148Dokument20 SeitenMaterials 10 01148Lin TianNoch keine Bewertungen
- Syll Btech. 1st Year New Syllabus FINAL 2008 BPUTDokument22 SeitenSyll Btech. 1st Year New Syllabus FINAL 2008 BPUTkamalkantmbbsNoch keine Bewertungen
- Btech. 1st Year New Syllabus FINAL 2008 BPUTDokument22 SeitenBtech. 1st Year New Syllabus FINAL 2008 BPUTRajesh KumarNoch keine Bewertungen
- Time-Dependent Density-Functional Theory: Carsten A. Ullrich Neepa T. MaitraDokument98 SeitenTime-Dependent Density-Functional Theory: Carsten A. Ullrich Neepa T. MaitraFahd ElmourabitNoch keine Bewertungen
- Fundamentals: The Quantum-Mechanical Many-Electron Problem and The Density Functional Theory ApproachDokument56 SeitenFundamentals: The Quantum-Mechanical Many-Electron Problem and The Density Functional Theory ApproachAaron ArvindNoch keine Bewertungen
- PhysicsDokument126 SeitenPhysicsKishanJhaNoch keine Bewertungen
- Atomic Structure and The Periodic TableDokument95 SeitenAtomic Structure and The Periodic TableSarah 丹鳳Noch keine Bewertungen
- Periodic Table Lab: Part 1: Exploring Periodic Trends (Data Collection)Dokument4 SeitenPeriodic Table Lab: Part 1: Exploring Periodic Trends (Data Collection)Sasha Lucero QuintanaNoch keine Bewertungen
- Aqa A Level Chemistry Cheatsheet 3Dokument24 SeitenAqa A Level Chemistry Cheatsheet 3David AdigboNoch keine Bewertungen
- Development of the Periodic TableDokument25 SeitenDevelopment of the Periodic Tablebm OUREMNoch keine Bewertungen
- PHYSICS: CONCEPTS AND APPLICATIONSDokument3 SeitenPHYSICS: CONCEPTS AND APPLICATIONSHercules ShahNoch keine Bewertungen
- Lectures 10-11: 9.0 OverviewDokument18 SeitenLectures 10-11: 9.0 OverviewHusna TajNoch keine Bewertungen
- Phy1001 Engineering-Physics LTP 1.0 1 Phy1001-Engineering PhysicsDokument3 SeitenPhy1001 Engineering-Physics LTP 1.0 1 Phy1001-Engineering PhysicsDevarsh ShahNoch keine Bewertungen
- Soft X-Ray Photoionization of Atoms and Molecules Svensson2005Dokument19 SeitenSoft X-Ray Photoionization of Atoms and Molecules Svensson2005jbmacielNoch keine Bewertungen
- Che101 Chap 8Dokument68 SeitenChe101 Chap 8Ruhi AfsaraNoch keine Bewertungen
- Engineering Physics Syllabus PDFDokument3 SeitenEngineering Physics Syllabus PDFNamanNoch keine Bewertungen
- SSP SyllabusDokument2 SeitenSSP SyllabusHaze Subin CNoch keine Bewertungen
- Fundamental Principles of Modern Theoretical Physics: International Series of Monographs in Natural PhilosophyVon EverandFundamental Principles of Modern Theoretical Physics: International Series of Monographs in Natural PhilosophyNoch keine Bewertungen
- Ionic Bonding: AnalysisDokument7 SeitenIonic Bonding: AnalysisRez CaitlinaNoch keine Bewertungen
- Toward A Modern Atomic Theory: Sections 1.2-1.3 QuestionsDokument12 SeitenToward A Modern Atomic Theory: Sections 1.2-1.3 QuestionsRez CaitlinaNoch keine Bewertungen
- Covalent Bonding: Formation of Covalent BondsDokument8 SeitenCovalent Bonding: Formation of Covalent BondsRez CaitlinaNoch keine Bewertungen
- CH11SB026Dokument11 SeitenCH11SB026Quach Pham Thuy TrangNoch keine Bewertungen
- Trends in The Periodic Table: Section 1.4 QuestionsDokument13 SeitenTrends in The Periodic Table: Section 1.4 QuestionsRez CaitlinaNoch keine Bewertungen
- CH11SB022Dokument5 SeitenCH11SB022Thien DinhNoch keine Bewertungen
- Classifying Compounds: Investigation 2.1.1Dokument3 SeitenClassifying Compounds: Investigation 2.1.1Rez CaitlinaNoch keine Bewertungen
- CH11SB064Dokument2 SeitenCH11SB064Rez CaitlinaNoch keine Bewertungen
- CH11SB062Dokument2 SeitenCH11SB062Rez CaitlinaNoch keine Bewertungen
- 44890-1537-93977-2-10-20190110 3Dokument1 Seite44890-1537-93977-2-10-20190110 3Narayan BhosekarNoch keine Bewertungen
- AiM Wheel Speed Sensor DataDokument8 SeitenAiM Wheel Speed Sensor DataDIECA STNoch keine Bewertungen
- BPMSX60 64Dokument4 SeitenBPMSX60 64Bel BarretoNoch keine Bewertungen
- Gyrosmart™: Directional Surveying in All EnvironmentsDokument2 SeitenGyrosmart™: Directional Surveying in All EnvironmentsMiguel SalcedoNoch keine Bewertungen
- Nordson VL200 PDFDokument275 SeitenNordson VL200 PDFManuel Rengifo RiveroNoch keine Bewertungen
- Questions and AnswersDokument2 SeitenQuestions and AnswersDevarakonda KondayyaNoch keine Bewertungen
- Anh 2017Dokument11 SeitenAnh 2017Võ Quốc CườngNoch keine Bewertungen
- 330D and 336D Excavator Electrical SystemDokument7 Seiten330D and 336D Excavator Electrical SystemMaquinaria Pesada ServisNoch keine Bewertungen
- LG 42lc2d LCD TV Service ManualDokument57 SeitenLG 42lc2d LCD TV Service ManualTvcrepair100% (2)
- Wireless Embedded BoardsDokument6 SeitenWireless Embedded BoardsGatroxxNoch keine Bewertungen
- Study of Electrochemical Energy ConversionDokument13 SeitenStudy of Electrochemical Energy Conversionilias1973Noch keine Bewertungen
- Dell XPS 13 Technical Specifications: Height: 0.2"-0.7"/ (6-18mm) / Width: 12.4" (316mm) / Depth 8.1" (205mm)Dokument1 SeiteDell XPS 13 Technical Specifications: Height: 0.2"-0.7"/ (6-18mm) / Width: 12.4" (316mm) / Depth 8.1" (205mm)Eric HubbardNoch keine Bewertungen
- Esp32 CamDokument4 SeitenEsp32 CamAnwarul Huda100% (2)
- Ds1307 Application NoteDokument17 SeitenDs1307 Application NoteShamsunder31Noch keine Bewertungen
- 15-03-06 Samsung Opening Brief (Fed. Cir.)Dokument497 Seiten15-03-06 Samsung Opening Brief (Fed. Cir.)Florian MuellerNoch keine Bewertungen
- Trafindo Product Catalogue Cast Resin TransformersDokument2 SeitenTrafindo Product Catalogue Cast Resin TransformersAliq FazliNoch keine Bewertungen
- T Spice ExamplesDokument42 SeitenT Spice ExamplesVENKI50% (2)
- Open Your Gate by Mobile Phone!: Installation and User's ManualDokument27 SeitenOpen Your Gate by Mobile Phone!: Installation and User's ManualsimionlNoch keine Bewertungen
- Erroor Code SamsungDokument5 SeitenErroor Code SamsungZdravko PeranNoch keine Bewertungen
- Using The ATF-10236 in Low Noise Amplifier Applications Inêthe UHF Through 1.7 GHZ Frequency RangeDokument10 SeitenUsing The ATF-10236 in Low Noise Amplifier Applications Inêthe UHF Through 1.7 GHZ Frequency RangeAbhay KulkarniNoch keine Bewertungen
- Akoto Nationbuilding 2 PDFDokument121 SeitenAkoto Nationbuilding 2 PDFrayaliblackNoch keine Bewertungen
- Telos Price List PDFDokument16 SeitenTelos Price List PDFmihaidumitruNoch keine Bewertungen
- Datasheet Tea 5757hDokument32 SeitenDatasheet Tea 5757hJose BenavidesNoch keine Bewertungen
- Darkness SensorDokument4 SeitenDarkness SensorRomela Grace BenigNoch keine Bewertungen
- Easergy Micom P544/P546: Current Differential Protection RelayDokument72 SeitenEasergy Micom P544/P546: Current Differential Protection RelayNermina MalićevićNoch keine Bewertungen
- Overline KXTVP200 Voicemail User Programming ManualDokument406 SeitenOverline KXTVP200 Voicemail User Programming ManualRashidi Abdul RafarNoch keine Bewertungen
- Ligand Subsitution Reactions of Square Planar ComplexesDokument6 SeitenLigand Subsitution Reactions of Square Planar ComplexesSharon DunneNoch keine Bewertungen
- ZMP 60211 Ho 80 W 865Dokument7 SeitenZMP 60211 Ho 80 W 865vcxbNoch keine Bewertungen
- Spacecraft Design and Mission OperationsDokument10 SeitenSpacecraft Design and Mission OperationsDavid GaleaNoch keine Bewertungen
- CIGRE2008 G1IF4 Rev0.6Dokument2 SeitenCIGRE2008 G1IF4 Rev0.6QUANGTUNGHTDNoch keine Bewertungen