Beruflich Dokumente
Kultur Dokumente
Articulos de Betacaroteno
Hochgeladen von
yonkaniOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Articulos de Betacaroteno
Hochgeladen von
yonkaniCopyright:
Verfügbare Formate
CLIN. CHEM.
3215, 874-876 (1986)
Retinol,a-Tocopherol,Lycopene,and a- and f3-CaroteneSimultaneously
Determinedin Plasmaby IsocraticLiquidChromatography
David B. Mime and James Botnen
Retinol, a-tocopherol, Iycopene, and a- and /3-carotene can Materials and Methods
be simultaneously determined in human plasma by reversed-
Apparatus. The chromatographic instrumentation was a
phase liquid chromatography. Plasma-0.5 mL plus added Model 1090 LC System (Hewlett-Packard, Avondale, PA)’
internal standard, retinyl acetate-is deproteinized with 0.5 with a variable-wavelength diode-array detector, attached
mL of ethanol, then extracted with 1.0 mL of petroleum ether. to the Hewlett-Packard 3388 integration system and con-
The organic layer is removed and evaporated, the residue is trolled by a HP85B computer. For all analyses we used a 60
redissolved in 0.25 mL of ethanol, and 8-4 samples are x 4.6mm column of 3-nm Hypersil ODS (Hewlett-Packard),
injected into a 60 x 4.6 mm column of Hypersil ODS 3-.tm operated at 35#{176}C.
The mobile phase was methanol, the flow
particles at 35 #{176}C.
An isocratic methanol mobile phase, rate 0.9 mLlmin.
flow rate 0.9 mLimin, is used for the 9-mm run. Retinol and Reagents. Retinol, retinyl acetate, a-tocopherol, lycopene,
retinyl acetate are monitored at 305 nm, the tocopherols at a-carotene, and (3-carotene were all from Sigma Chemical
292 nm, and the carotenoids at 460 nm. Between-run CVs Co., St. Louis, MO. All solvents were “HPLC” grade.
were 3.1, 6.9, 6.1, and 6.5% for retinol, a-tocopherol, lyco- We dissolved the standards individually in chloroform.
pene, and (3-carotene, respectively. Small sample require- We then further diluted them in ethanol to obtain approxi-
ment, simplicity of extraction, short run time, and good mate final concentrations of 1 mg/L for retinol and the
reproducibility make this procedure ideal for clinical or re- carotenoids, 40 mg/L for a-tocopherol. The actual concentra-
search use. tions of the standards were determined from the absor-
bances and the absorptivities (in L mol’ cm’): 1780 at
AddItional Keyphrases: vitamin A vitamin E . nutrition 325 nm for retinol, 75.8 at 292 nm for a-tocopherol, 3450 at
effect of variousanticoagulants 472 nm for lycopene, 2800 at 444 nm for a-carotene, and
2396 at 465 nm for (3-carotene.
Clinical interest in evaluation of vitamin A andvitamin E Procedures. All procedures were carried out in a labora-
nutriture has increased in recent years, mainly owing to the tory equipped with yellow lights that did not includethe
possible roles of retinol (vitamin A), p-carotene, and a- ultraviolet end of the spectrum. This minimized light-
tocopherol (vitamin E) in decreasing the risk of cancer (1). induced degradation of the vitamins. We added0.5 mL of
These vitamins are also important in premature infants and ethanol that contained 0.6 g of retinyl acetate per milliliter
patients who are receiving long-term total parenteral nutri- to 0.5 mL of plasma in a 10 x 75 mm glass tube, vortex-
tion. Thus, a rapid, sensitive, relatively simple, and specific mixed the mixture for 1.0 mm, added 1.0 mL of petroleum
method for determination of these vitamins in plasma is ether (bp 36.6-56.6 #{176}C),
and vortex-mixed again for 1 mm.
desirable. Conventional colorimetric or fluorimetric meth- After centrifuging (600 x g, 5 mm) we transferred the
ods for determining retinol, carotene, or a-tocopherol (2-3) supernate to a separate tube with a Pasteur pipet and
are either nonspecific, time consuming, or insensitive. evaporated it to dryness in a 60#{176}C water bath, under a
Many of these problems are eliminated when these micro- stream of nitrogen. We then redissolved the extract in 0.25
nutrientsare determined by “high-performance” liquid mL of ethanol,2 vortex-mixed for 2.0 mm, then filtered the
chromatography (LC). Several LC procedures have already mixture through a Millipore ifiter (no. SFHOO4NS; Milli-
been described for determination of retinol anda-tocopherol, pore Corp., Bedford, MA) into injection vials. These were
either separately (4-6) or simultaneously (7-10). Additional placed in the autosampler and 84 was injected into the
procedures can separate lycopene, a-carotene, and /3-caro- chromatograph, which then was eluted as described above.
tene from other carotenoids (11-14). Nevertheless, an assay We programmed the diode array detector as follows: 0 to
that could resolve and quantify all of these compounds 1.6 mm at 305 ± 40 nm, to determine retinol and retinol
simultaneously would be highly desirable from the stand- acetate; from 1.6 mm to 2.5 mm at 290 ± 10 nm, to
point of clinical assessmentand studies of their metabolism. determine a- and $ocopherol; and from 2.5 to 9.0 mm at
Procedures recently described for determination of reti- 460 ± 25 mu, to determine the carotenoids. The reference
nol, a-tocopherol, p-carotene, a-carotene, and lycopene in a wavelength was 575 nm throughout the run. The baseline
single run (15, 16) involve either a multiple-solvent gradi- was automatically adjusted to zero with each wavelength
ent system or complex solvent mixtures and 15 to 30 mm of change. Four minutes after injection, the attenuation was
analysis times. Here we describe an isocratic LC procedure increased fourfold.
in which only methanol is used in the mobile phase. With it,
retinol, a-tocopherol, lycopene, a-carotene, and p-carotene
1 Mention of a trademark or proprietary product doesnot consti-
extracted from 0.5 mL of plasma can be resolved and
estimated in a single 9-mm run. tute a guaranteeor warranty by the U.S. Dept. of Agriculture, and
doesnot imply its approval to the exclusionof other products that
may alsobe suitable.
USDA, ARS, Grand Forks Human Nutrition Research Center, 2The sampleswere redisaolved in ethanol, becausethe fat-soluble
P.O. Box 7166,University Station, Grand Forks, ND 58202. vitamins and carotenoids are much more soluble in ethanol than in
ReceivedSeptember 30, 1985;acceptedJanuary 31, 1986. (more polar) methanol.
874 CLINICALCHEMISTRY, Vol. 32, No. 5, 1986
Peak heights were compared with those for known stan- Results and Discussion
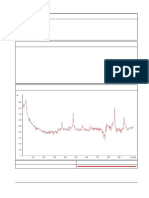
dards, run with retinyl acetate as the internal standard. The Figure 1A depicts a typical chromatogram of fat-soluble
concentrations of the analytes were determined by the peak- vitamins from plasma. It compares well with a chromato-
height ratio method (10). Samples for assessment of analyti- gram of pure standards (Figure 1B). Comparisons of reten-
cal recovery were run by adding known standards, dissolved tion times and spectra of fractions with those of pure
in 0.5 mL of ethanol, to 0.5 mL of plasma before extraction standards established the peak identities. According to a
with petroleum ether, as follows:retinol, 0.14 to 0.68 g; a- recent report (14), the two unidentified peaks after a-
tocopherol, 0.985 to 4.925 j.zg; lycopene,0.14 to 0.25 ig and tocopherolmay be precryptoxanthin and cryptoxanthin.
(3-carotene, 0.14 to 0.25 g. Analytical recoveries of different concentrations of stan-
Effect of anticoagulants. A 30-mL specimen of blood was dards added to fivedifferent plasma samples before extrac-
collected from each of six different individuals into a plastic tion averaged 99.1 (SD 1.38), 101.7(8.9), 96.5(6.6), and 98.8
syringe, for use in testing the effects of different anticoagu-
(1.9) percent forretinol, a-t.ocopherol, lycopene, and (3-
lants on results for these fat-soluble vitstmins. Each sample carotene, respectively. The corresponding correlation coeffi-
was divided into five aliquots, which were handled as
cients forexpected vs foundwere respectively 0.986, 0.995,
follows. One was allowed to clot and the serum obtained.
0.983, and 0.998 over these two- to fivefold concentration
The others were added to Vacutainer Tubes containing
ranges. The detection limits were 0.1,0.3, and 0.02 mg/L for
heparin, citrate, EDTA, and oxalate, respectively, and plas-
retinol, a-tocopherol, and thecarotenoids, respectively.
ma was collected foranalysis.
Table 1 summarizes within-run and day-to-day CVs. We
have successfully used smaller volumes of serum (down to
200 4) with proportional reductions in the other reagents
withoutaffecting recovery, precision, or final result.
Effects of different anticoagulants are summarized in
Table 2. Values obtained with heparin present in the sample
were not significantly (p >0.05) different from values for
serum. Citrate significantly depressed the values forappar-
entretinol and a-tocopherol in plasma; oxalate significantly
depressed only the a-tocopherol concentrations. EDTA and
oxalate produced insignificantly (p >0.05) lower values for
retinol and a-carotene. Insignificantly lower values for a-
tocopherol were also seen when EDTA was used as anticoag-
ulant. None of the anticoagulants affected values obtained
for either lycopeneor (3-carotene. These results are consist-
It ent with those found by McClean et al. (18), who reported
lower results forretinol when eitherEDTA or oxalate was
01
I
234567890
I I I
i i4 i#{233} used as anticoagulant. Nierenberg (19) noted that under the
Retention Time Retention Time conditionof his assay EDTA, oxalate, or citrate adversely
(mm) (mirL) affected values for (3-carotene, and he suggested that these
Fig. 1. Chrumatogramsof fat-soluble vitaminsextractedfrom 0.5 mLof compounds may catalyze reactions resulting in the isomer-
(A, left)plasma and (B, right) of standards (see text) ization or oxidation of retinol and (3-carotene. Our results
Peaks are: A re5no; B, retinyl acetate;C, y4ocopherol; 0, a-tocopherol; E, indicate that thevarious anticoagulants affect a-tocopherol
lycopene; F, a.camtene; and G, a-carotene. Peaks A and B were measuredat in a similar manner. Therefore, we recommend either serum
305 nm,peaksCandDat 290 nm,and peaksEthrough Get 460 nm.Thesnows
indicate the wavelengthchanges.Responseset at 0.012A kiII.scalefor peaks A or heparinized plasma in this assay.
through 0, and 0.003 A for peaks E through G As recommended by others (19,20), we tested the useful-
Table 1. WithIn-Runand Day-to-DayVariatIona
R.tlnol ia.TocopheroI Lycopene -Csrotene
Day 1
Meanb 0.485 (0.016) 7.3 (0.12) 0.418 (0.017) 0.109 (0.003)
CV, % 3.3 1.6 4.1 2.75
Day 2
Mean 0.502 (0.03) 7.6 (0.16) 0.432 (0.01) 0.112(0.006)
CV, % 6.0 2.1 2.3 5.4
Day 3
Mean 0.466 (0.01) 6.9 (0.15) 0.408 (0.01) 0.100(0.005)
CV, 2.1 2.2 2.5 5.0
Day 4
Mean 0.485 (0.03) 8.3 (0.17) 0.476 (0.02) 0.120 (0.007)
CV. % 6.3 2.0 4.2 5.8
Day 5
Mean 0.503 (0.03) 7.8 (0.4) 0.446 (0.03) 0.112 (0.005)
CV, % 6.0 5.1 6.7 4.5
Overall (day-to-day)
Mean 0.488 (0.015) 7.58 (0.53) 0.436 (0.027) 0.111 (0.007)
CV, % 3.1 6.9 6.1 6.5
ofa singleplasmapool sample.
VarlabiIity Fourreplicates,
determined
oneachoffiveseparatedays. a-Carotenewas notdetectedin this sample.
“Mean (and SD), in mg/L
CLINICAL CHEMISTRY, Vol. 32, No. 5, 1986 875
Table 2. Effect of Antlcoagulants on Values Observed for Fat-Soluble Vltamlns
Retinol a-Tocophirol Lycopsne
Meanconcn, mg/L (and SD)
Serum 0.51 (0.14) 9.10 (1.18) 0.49 (0.12) 0.14 (0.06) 0.16(0.05)
Heparin 0.51 (0.17) 8.70 (1.67) 0.45 (0.11) 0.14 (0.13) 0.14 (0.05)
Citrate 0.40 (0.08)#{176} 7.06 (0.70)d 0.43 (0.08) 0.11(0.003) 0.15 (0.04)
EDTA 0.45 (0.14) 8.90 (1.18) 0,47 (0.14) 0.09 (0.09) 0.16 (0.06)
Oxalate 0.43 (0.12) 7.40 (0.86)e 0.45 (0.12) 0.08 (0.06) 0.14 (0.04)
Blood fromsix individuals;each samplewasdividedintofive aliquots,which weretreatedwiththe anticoagulantsIndicated.
“a-Carotenewasnotdetectableon 3/6 samplesof serum, 4/6 sampleswithheparln,3/6 sampleswith citrate,1/6sampleswithEDTA,and 4/6 samples with
oxalate.
contrasts
cSigniflcantlylowerthan serum or heparin(p <0.04) by Scheff#{233} (21).
d Significantlylowerthanserum,heparin,or EDTA (p <0.001).
Signlflcantlylowerthan serum, heparin,or EDTA (p <0.01).
Table 3. Fat-Soluble VitamIns and Carotenes In Plasma
5. NilssonB, JohanssonB, JanasonL, Ho!mburg L Determination
of plasma a-tocophero! by high-performance liquid chromatogra-
from 12 Healthy Persons phy. J Chromatogr 1978;145:169-72.
6. DeLeenheer AP, DeBevere VO, Gruyl AA, Claeys AE. Determi-
Retinol a-Tocophorol Lycopsee a-Carotene p-Carotene nation of serum a-tocopherol (vitamin E) by high-performance
liquid chromatography. Clin Chem 197824:585-90.
Concn, tng/L 7. DeLeenheer AP, DeBevere V, DeRuyter MGM, Claeys AR.
0.620 9.6 0.655 0.107 0.230 Simultaneous determination of retino! and a-tocophero! in human
0.611 12.0 0.370 0.082 0.177 serum by high-performance liquid chromatography. J Chromatogr
0.531 10.7 0.197 0.037 0.077 1979;162:408-13.
0.445 8.2 0.510 0.108 0.196 8. Bieri JG, Tolliver TJ, Catignani GL. Simultaneous determina-
0.591 11.5 0.489 0.144 0.337 tion of a-tocophero! and retino! in plasma or red cells by high-
0.637 13.5 0.843 0.113 0.296 pressure liquid chromatography.Am J Cliii Nutr 1979;32:2143-9.
0.393 10.5 0.674 0.029 0.192
0.450 11.3 0.155 nda 0.215 9. Driskel! WJ, Neese JW, Bryant CC, Bashor MM. Measurement
0.532 11.5 0.276 0.041 0.102 of vitamin A and vitamin E in human serum by high-performance
0.094 liquid chromatography. J Chromatogr 1982231:439-44.
0.850 10.5 0.356 nd
0.679 7.3 0.241 nd 0.189 10. Catignani GL, Bieri JG, Driskell WJ, et al. Proposed selected
0.696 8.8 0.344 nd 0.076 method: simultaneous determination of retinol and a-tocopherol in
Mean 0.586 9.6 0.426 0.055 0.182 serum or plasma by liquid chromatography. Clin Chem
SD 0.127 3.3 0.212 0.053 0.084 1983;29:708-12.
a nd, not detectable, <20 tg/L. 11. Driskell WJ, BashorMM, Neese JW. Bets-carotene determined
in serumby liquid chromatographywith an internal standard. Cliii
Chem 1983;29:1042-4.
ness ofBHT (butylated hydroxytoluene) forstabilizing the 12. NeelsHJCF,Do Leenheer AP. Isocratic non-aqueousreversed-
fat-soluble vitamins and carotenoids during the extraction phase liquid chromatography of carotenoids. Anal Chem
and drying steps. When 38 mg of BHT was added to the 1983;55:270-5.
ethanol per liter before the deproteinization and extraction 13. Tangney CC. Individual carotenoid determination in human
steps, we found no differences in the values for a-tocopherol plasma by high-performance liquid chromatography. J Liq Chroma-
or any of the carotenoids as compared with aliquots of the togr 1984;7:2611-30.
same samples extracted without added BHT. Under the 14. Bieri JG, Brown ED, Smith Jr JC. Determination of individual
conditions of our assay, BHT is eluted near the retinol peak carotenoids in human plasmaby high performance liquid chroma-
and thus interferes with its determination. Nevertheless, tography.J Liq Chromatogr 1985;8:473-84.
addition of an antioxidant such as BHT may be necessary in 15. Miller KW, Lorr NA, Yang CS. Simultaneous determination of
systems that contain free-radical-producing or oxidizing plasma retinol, cr-tocopherol,lycopene,a-carotene,and p-carotene
by high-performance liquid chromatography. Anal Biochem
agents such as tetrahydrofuran or HC1O4 (19). 1984;138:340-5.
Values for plasma from 12 apparently healthy adults 16. Miller KW, Yang CS. An isocratic high-performance liquid
(Table 3) are similar to those reported by others for retinol chromatography method for the simultaneousanalysis of plasma
(15, 17), a-tocopherol (8, 15), lycopene(14, 15), a-carotene retinol, a.tocopherol and various carotenoids. Anal Biochem
(14), and (3-carotene (14, 15, 19). 1985;145:21-6.
17. Vuilleumier JP, Keller HE, Gysel D, Hunziker F. Clinical
References chemical methodsfor the routine assessment of the vitamin status
1. Willett WC, Polk BF, Underwood BA, et al. Relation of serum in human populations.hit J Vit Nutr Res 1983;53:265-72.
vitamins A and E and carotenoids to the risk of cancer. N EngiJ 18. McClean SW, RuddelME, Gross EG, et al. Liquid-chromato-
Med 1984;310:430-4. graphic assay for retinol (vitamin A) and retinol analogs in thera-
2. Sauberlich HE, Dowdy RP, Skala JR. Laboratory tests for the peutic trials. Clin Chem 1982;28:693-6.
assessment of nutritional status. Boca Raton, FL: CRC Press, 19. Nierenberg DW. Serum andplasma fl-carotene levels measured
19’74;6,7,76. with an improvedmethodof high performance liquid chromatogra-
3. Arroyave G, Chichester CO, Flores H, et al. Biochemical meth- phy.J Chromatogr1985;339:273-84.
odology for the assessmentof vitamin A status. Washington, DC: 20. Chow Fl,Omaye ST.Use of antioxidanta in the analysis of
The Nutrition Foundation, 1982:60-4. vitamins A and E in mammalian plasma by high performance
4. DeRuyter MGM, DeLeenheer AP. Determination of serum reti- liquid chromatography. Lipids 1983;18:827-41.
no! (vitamin A) by high-speed liquid chromatography. Clin Chem 21. Scheffe H. The analysis of variance. New York: John Wiley,
197622:1593-5. 1959:90-137.
876 CLINICAL CHEMISTRY, Vol. 32, No. 5, 1986
Das könnte Ihnen auch gefallen
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5795)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1091)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Introduction To Fiscal MeteringDokument26 SeitenIntroduction To Fiscal MeteringmulkaNoch keine Bewertungen
- New AQA ELC Science Component 4: Chemistry in Our WorldDokument2 SeitenNew AQA ELC Science Component 4: Chemistry in Our WorldChris HarrisNoch keine Bewertungen
- MSDS TKSA 11, 21, 31, 41, 51 Lithium SDS 2021Dokument9 SeitenMSDS TKSA 11, 21, 31, 41, 51 Lithium SDS 2021ketevan.melkadzeNoch keine Bewertungen
- Plant Design For The Production of Sodium CarbonateDokument29 SeitenPlant Design For The Production of Sodium CarbonateMuhammad Adeel KhalidNoch keine Bewertungen
- Chemical Aspect of Document ExaminationDokument6 SeitenChemical Aspect of Document Examinationgenelord opalla100% (2)
- Orca Share Media1519993790396 PDFDokument10 SeitenOrca Share Media1519993790396 PDFSecret SecretNoch keine Bewertungen
- Systematics of The Spinel Structure TypeDokument23 SeitenSystematics of The Spinel Structure TypeAlin DrucNoch keine Bewertungen
- PRG - PT - GEN - 0001 - R00 - E - S - (Blending Manual) PDFDokument47 SeitenPRG - PT - GEN - 0001 - R00 - E - S - (Blending Manual) PDFsudjonoNoch keine Bewertungen
- Feasibility of Malunggay As A Cleaning AgentDokument19 SeitenFeasibility of Malunggay As A Cleaning AgentElloisa Pauline L. CuetoNoch keine Bewertungen
- Constant Falling Head Permeability Test Lab ReportDokument12 SeitenConstant Falling Head Permeability Test Lab ReportHaziq Zuhaimi100% (3)
- Physical Science ReviewerDokument3 SeitenPhysical Science ReviewerMary Angeline L. BatacNoch keine Bewertungen
- PURELAB Quest Brochure 12 Pages.Dokument12 SeitenPURELAB Quest Brochure 12 Pages.alex_antitodoNoch keine Bewertungen
- Analysis Results Report E MDokument3 SeitenAnalysis Results Report E MViorel PopNoch keine Bewertungen
- Investigation On CorrosionDokument19 SeitenInvestigation On CorrosionSudip RokayaNoch keine Bewertungen
- Tablet Coating - Fundamentals & TechniquesDokument36 SeitenTablet Coating - Fundamentals & TechniquesSatyabrata SahuNoch keine Bewertungen
- Endotoxin Removal: The Solution With Membrane Separation TechnologyDokument4 SeitenEndotoxin Removal: The Solution With Membrane Separation TechnologykapilNoch keine Bewertungen
- Molecular Genetics: (BCHEM 365)Dokument32 SeitenMolecular Genetics: (BCHEM 365)DesmondNoch keine Bewertungen
- Module 6-7Dokument2 SeitenModule 6-7Duchess Juliane Jose MirambelNoch keine Bewertungen
- DHI Eutrophication Model 1 Sediment Benthic VegDokument54 SeitenDHI Eutrophication Model 1 Sediment Benthic VegAlina DanilaNoch keine Bewertungen
- Empirical Formula: From Percentage To FormulaDokument21 SeitenEmpirical Formula: From Percentage To FormulaPrincess KimNoch keine Bewertungen
- Auger Electron Spectroscopy: Principles and ApplicationsDokument26 SeitenAuger Electron Spectroscopy: Principles and ApplicationsPrashant ParshivnikarNoch keine Bewertungen
- Currentprotocolsforresin-Bondeddentalceramics: Markus B. Blatz,, Julian Conejo,, Amirah Alammar,, Jose AyubDokument23 SeitenCurrentprotocolsforresin-Bondeddentalceramics: Markus B. Blatz,, Julian Conejo,, Amirah Alammar,, Jose AyubPablo Gutiérrez Da VeneziaNoch keine Bewertungen
- Chem Unit 5 Organic QuestionsDokument77 SeitenChem Unit 5 Organic Questionsareyouthere92Noch keine Bewertungen
- Engineering Admissions Assessment D564/11 Thursday 2 November 2017 80 Minutes Section 1Dokument44 SeitenEngineering Admissions Assessment D564/11 Thursday 2 November 2017 80 Minutes Section 1xingchenNoch keine Bewertungen
- Experiment 9 10Dokument4 SeitenExperiment 9 10sujaraghupsNoch keine Bewertungen
- Previous Year Solution of Solid Waste ManagementDokument15 SeitenPrevious Year Solution of Solid Waste Managementudishasinghus09Noch keine Bewertungen
- AS Q1 w1-w2 Physical ScienceDokument2 SeitenAS Q1 w1-w2 Physical Scienceemmah adiong50% (2)
- Advances in Functionalized Nanoparticles Based Drilling Inhibitors For Oil ProductionDokument12 SeitenAdvances in Functionalized Nanoparticles Based Drilling Inhibitors For Oil ProductionMario Valentino DioNoch keine Bewertungen
- Language of ChemoDokument20 SeitenLanguage of Chemokaransharma690Noch keine Bewertungen
- Guidelines For Rationalised Use of High Performance ConductorsDokument163 SeitenGuidelines For Rationalised Use of High Performance ConductorsRajeev KumarNoch keine Bewertungen