Beruflich Dokumente
Kultur Dokumente
Lo:an1jj: II II Lilt II
Hochgeladen von
vainateyagoldarOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Lo:an1jj: II II Lilt II
Hochgeladen von
vainateyagoldarCopyright:
Verfügbare Formate
ijjJ
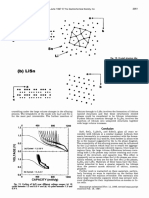
2048 J. Electrochem. Soc., Vol. 144, No.6, June 1997 The Electrochemical Society, Inc.
SCATTERING ANGLE (deg.) SCATTERING ANGLE (deg.)
20 30 40 50 20 30 40 50
100 (a)Scanl2 ---Sn
O.66V
10 ______
4 I I I I
100 (b)Scan 8
c) O.94V
10 _______
C)
0
4 I I I I I
>- I I
j.... 100 (c) Scan 4 BeO
I-
Cl)
zu-I
ii II lilt II
lo:an1jj Sf0 I- 4 I
z
>
0)
1 _
(
a0- 20 40 >0
0
SCAN NUMBER SCAN NUMBER
Fig. 4. In situ XRD results for SnO; (a) scan 12 —0.66 V, (b) scan Fig. 5. In situ XRD results for SnO; (a) scan 17 —0.41 V, (b) scan
8 —0.94 V, (c) scan 4 —1.05 V, (ci) scan 1 —1.04 V, and (e) voltaae 15 —0.50 V, (c) scan 14 —0.55 V, (ci) scan 12 —0.66 V. and (e) volt-
profile (scan number vs. voltage) for constant current cycling of cell. age profile (scan number vs. voltage) for constant current cycling of
Logarithmic intensities are Dlolted for the scans in reference to the cell. Logarithmic intensities are plotted for the scans in reference to
left-hand vertical axis and )inear intensities are plotted for the cal- the left-hand vertical axis and linear intensities are plotted for the
culated patterns (normalized to 100) in reference to the right-hand calculated patterns (normalized to 100) in reference to the right.
vertical axis. hand vertical axis. Note the change of intensity scale from Fig. 4.
From Fig. 4d (scan 1, 1.04 V) to Fig. 4b (scan 8, 0.94 V) ken lines in Fig. 8d. It is important to note that crystalline
we can observe the decline of the SnO peaks and an SnO does not reform. Figure 8a (scan 1, 1.04 V) shows the
increase in the background. At Fig. 4a (scan 12, 0.66 V) we first scan on the same scale as Fig. 8b (scan 55, 2.50 V). An
observe broad tin peaks, which confirms that the SnO increase in x-ray background has occurred during the
loses its oxygen leaving regions of tin metal.
Figure 5c (scan 14, 0.55 V) shows the disappearance of
Sn followed by the emergence of Li2Sn5, whose calculated
peak positions and intensities are shown by the broken SCATTERING ANGLE (deg.)
lines in Fig. Sc. The Li2Sn5 peaks are gone by Fig. Sa (scan 20 30 40 50
17, 0.41 V) and we see the emergence on the next Li-Sn
phase, LiSn, as shown by the broken lines in Fig. 5a. (a) Scan 27
Figure 6d shows the calculated intensities for Li7Sn3,
which has strong features at approximately 22° and 38°.
1
One could imagine that we have Li7Sn3 as at Fig. 6c (scan 4 I I
21, 0.31 V). However, it turns out that all of the remaining (b) Scan 22
Li-Sn phases, that is Li5Sn2, Li13Sn5, Li7Sn2, and Li22Sn5
are structurally similar to each other and to Li7Sn3 as
III
ihIhu
shown by the calculated peaks in Fig. 7d, again with 4
strong features at approximately 22° and 38°. Throughout
Fig. 6b, a, 7c, and b (scan 22, 0.23 V, scan 27, 0.10 V, scan
zu-j 1
(c) Scan 21
31, 0.23 V charge, and scan 43, 0.43 V charge, respectively) I.
6
we observe these broad features at 22° and 38°. These Li- 4 1
rich alloy phases Li5Sn2, Li13Sn5, Li7Sn2, and Li22Sn5 are all (d)
based on the BCC unit cell of lithium14 and differ primar- - - - U7Sn3
ily in the arrangement of Li and Sn on the lattice sites. The ,. .
phases also exhibit structural distortions and appropriate
superstructures. It is our opinion that the broad peaks in
the patterns of Fig. 6a, b, c, 7b, and c arise from these
phases or mixtures of these phases with poorly developed
long-range order. Apparently the Li and Sn atoms cannot
move effectively in these phases at room temperature to
form long-range ordered structures. This is suggested by
—
>
2
0
...I. 20
SCAN NUMBER
40
the Li-Sn phase diagram which shows that the Li-rich
phases have the highest melting point, and hence lowest Fig. 6. In situ XRD results for SnO; (a) scan 27 —0.10 V, (b) scan
atom mobility at room temperature. 22 —023 V, (c) scan 21 —0.31 V, (d) valculated peak positions for
Further charging the cell to 0.78 V by Fig. 7a (scan 45, Li7Sn3, and (e) voltage profile (scan number vs. voltage) for constant
0.78 V charge) shows the re-emergence of LiSn. From current cycling of cell. Logarithmic intensities are Dlotted for the
Fig. 8d-c and b (scan 47, 1.00 V, Scan 50, 1.44 V; and scan scans in reference to the left-hand vertical axis and linear intensi-
55, 2.50 V respectively) we observe the reformation of ties are plotted for the calculated patterns (normalized to 100) in
metallic tin whose calculated peaks are shown by the bro- reference to the right-hand vertical axis.
Das könnte Ihnen auch gefallen
- Scattering Angle (Deg.) Scattering Angle (Deg.)Dokument1 SeiteScattering Angle (Deg.) Scattering Angle (Deg.)vainateyagoldarNoch keine Bewertungen
- Grand Test-2 Key & HintsDokument9 SeitenGrand Test-2 Key & HintsM JEEVARATHNAM NAIDUNoch keine Bewertungen
- Section-A Four Three.: L-Lrr-Lfeee Date 06/12/2014Dokument22 SeitenSection-A Four Three.: L-Lrr-Lfeee Date 06/12/2014Trisha DasNoch keine Bewertungen
- 'Ffi W Ffi Ffi: Sectors Trigonometric Functions Trigonometric WithDokument25 Seiten'Ffi W Ffi Ffi: Sectors Trigonometric Functions Trigonometric WithMin KangNoch keine Bewertungen
- A1-114 Field CalcultionDokument10 SeitenA1-114 Field Calcultionnasrun nasrunNoch keine Bewertungen
- IAS Mains Electrical Engineering 2007Dokument12 SeitenIAS Mains Electrical Engineering 2007kaarthik12Noch keine Bewertungen
- DL 128Dokument2 SeitenDL 128Gagan SLNoch keine Bewertungen
- رياضيات نموذج اجابة اختبار مبارك الكبير2016Dokument8 Seitenرياضيات نموذج اجابة اختبار مبارك الكبير2016sherif elmansouryNoch keine Bewertungen
- DL 1002Dokument2 SeitenDL 1002Gagan SLNoch keine Bewertungen
- CAPE Physics 2016 U1 P2 Mark SchemeDokument12 SeitenCAPE Physics 2016 U1 P2 Mark SchemeLeah CassieNoch keine Bewertungen
- DL 1008Dokument2 SeitenDL 1008Gagan SLNoch keine Bewertungen
- Ancova: A Hybrid of Regression and Analysis of VarianceDokument21 SeitenAncova: A Hybrid of Regression and Analysis of Variancedanishrehman1985Noch keine Bewertungen
- CNT3 ExamsDokument4 SeitenCNT3 ExamsJoshua Ng'ang'aNoch keine Bewertungen
- DL 115Dokument2 SeitenDL 115Gagan SLNoch keine Bewertungen
- Ebar?At: AprilDokument2 SeitenEbar?At: AprilAthisayaraj RajNoch keine Bewertungen
- Aits-1920-Ot-Jeem - Sol PDFDokument12 SeitenAits-1920-Ot-Jeem - Sol PDFsoumadip senNoch keine Bewertungen
- Adobe Scan Mar 13, 2023Dokument7 SeitenAdobe Scan Mar 13, 2023Abhishek VermaNoch keine Bewertungen
- L XK Ileprtmmt: Udver Fo DapplhdDokument4 SeitenL XK Ileprtmmt: Udver Fo DapplhdGnanasegaran RishiNoch keine Bewertungen
- Ned University of Engineering & Technology: (Fig Q - 1 A)Dokument3 SeitenNed University of Engineering & Technology: (Fig Q - 1 A)Muhammadtayyabaziz KhanNoch keine Bewertungen
- E - Valuat, - Can Every T T: SectionDokument9 SeitenE - Valuat, - Can Every T T: SectionAlok ChoubeyNoch keine Bewertungen
- Robust Micron-Sized Silicon Secondary Particles Anchored by Polyimide As High-Capacity High-Stability Li-Ion Battery AnodeDokument10 SeitenRobust Micron-Sized Silicon Secondary Particles Anchored by Polyimide As High-Capacity High-Stability Li-Ion Battery AnodegdjNoch keine Bewertungen
- Lab 2: Nodal Analysis and Mesh Analysis Techniques: V V I V Ia V V I V Ib V I V V IDokument5 SeitenLab 2: Nodal Analysis and Mesh Analysis Techniques: V V I V Ia V V I V Ib V I V V Igurdian skyNoch keine Bewertungen
- No SeatDokument5 SeitenNo Seatnikitabhavar27052004Noch keine Bewertungen
- DL 1514Dokument2 SeitenDL 1514Gagan SLNoch keine Bewertungen
- Fiitjee All India Test Series: JEE (Advanced) - 2020Dokument14 SeitenFiitjee All India Test Series: JEE (Advanced) - 2020yashjhan2017Noch keine Bewertungen
- Chem1000 2018 & 2019 Pastpapersnm4Dokument19 SeitenChem1000 2018 & 2019 Pastpapersnm4mulengamordecai92Noch keine Bewertungen
- Aits-1920-Ot-Jeem - SolDokument12 SeitenAits-1920-Ot-Jeem - SolZomatoswiggy100% (1)
- D15be7 Com CbgsDokument8 SeitenD15be7 Com CbgsrachanaypatilNoch keine Bewertungen
- Phy3622 11 Nor 2018Dokument5 SeitenPhy3622 11 Nor 2018Jay KayyNoch keine Bewertungen
- 06ESL38 - Logic Design Lab1Dokument76 Seiten06ESL38 - Logic Design Lab1Sachin S ShettyNoch keine Bewertungen
- Ume805 2Dokument2 SeitenUme805 2Raunaq SinghNoch keine Bewertungen
- 2024-JEE Main-6 - SolutionsDokument16 Seiten2024-JEE Main-6 - Solutionssinglaanush18Noch keine Bewertungen
- Cusat PapersDokument2 SeitenCusat PapersAbheek KashyapNoch keine Bewertungen
- Bee Practical ExperimentsDokument13 SeitenBee Practical Experimentspravviinn08Noch keine Bewertungen
- 100 Question 100 Min. Series ElectrostaticsDokument13 Seiten100 Question 100 Min. Series Electrostaticsumved singh yadavNoch keine Bewertungen
- National Exam May, 2017 16-Elec-A 1 CircuitsDokument6 SeitenNational Exam May, 2017 16-Elec-A 1 CircuitsK DNoch keine Bewertungen
- Degree Electronic Engineeringe First Semester EXAMINATIONS 2020/2021Dokument3 SeitenDegree Electronic Engineeringe First Semester EXAMINATIONS 2020/2021Peter JumreNoch keine Bewertungen
- 3000 Problemas de CircuitosDokument768 Seiten3000 Problemas de Circuitosbetodias30Noch keine Bewertungen
- 10AE752-Gomputational Fluid DynamicsDokument6 Seiten10AE752-Gomputational Fluid DynamicsashwaNoch keine Bewertungen
- DL 1612Dokument2 SeitenDL 1612Gagan SLNoch keine Bewertungen
- IFS Mathematics 2002Dokument9 SeitenIFS Mathematics 2002Salman NaimNoch keine Bewertungen
- DL 541Dokument3 SeitenDL 541Gagan SLNoch keine Bewertungen
- Lecture 22 18102022Dokument14 SeitenLecture 22 18102022Nitish PutrevuNoch keine Bewertungen
- Final SKEE1013 20172018 - 2Dokument5 SeitenFinal SKEE1013 20172018 - 2Dua IslamNoch keine Bewertungen
- Network: Hrs. HuoryDokument4 SeitenNetwork: Hrs. Huorylohith sNoch keine Bewertungen
- 7.1 To 7.4 ReviewDokument2 Seiten7.1 To 7.4 ReviewAurora KongNoch keine Bewertungen
- Physics Quarterly Revision WS - AKDokument3 SeitenPhysics Quarterly Revision WS - AKKazzenseiNoch keine Bewertungen
- EE380A Experiment 1Dokument4 SeitenEE380A Experiment 1rohanNoch keine Bewertungen
- Created With Scanner Pro: Uestion.!Dokument4 SeitenCreated With Scanner Pro: Uestion.!Syed Imtiaz Ali ShahNoch keine Bewertungen
- PP5 - Solutions - 2D Moment of Force and CouplesDokument6 SeitenPP5 - Solutions - 2D Moment of Force and CouplesDamirAljosevic100% (1)
- Tutorial 5Dokument8 SeitenTutorial 5Muhammad FauziNoch keine Bewertungen
- Aits 2122 FT V Jeem SolDokument19 SeitenAits 2122 FT V Jeem Solchinmaya bakiNoch keine Bewertungen
- Drift LimitationDokument12 SeitenDrift LimitationHammad AhmedNoch keine Bewertungen
- PSPICE Asssignments For Students Excused From Lab.: VT FT F R JX JDokument4 SeitenPSPICE Asssignments For Students Excused From Lab.: VT FT F R JX JJames Adrian Abalde SaboNoch keine Bewertungen
- Praveendeepak 2302PC09 Assignment4Dokument22 SeitenPraveendeepak 2302PC09 Assignment4Faiz AhmadNoch keine Bewertungen
- Basic Electrical1.2Dokument42 SeitenBasic Electrical1.2mivel82872Noch keine Bewertungen
- Rec1I...... : Used The and Ihau Appropriate andDokument1 SeiteRec1I...... : Used The and Ihau Appropriate andAnonymous vwbLPTpjNoch keine Bewertungen
- Concussion EvaluationDokument4 SeitenConcussion Evaluationapi-385504349Noch keine Bewertungen
- Algorithm 4 Procedure Next - Gen - GA Input The Current Population Output The Next Population While SNDokument1 SeiteAlgorithm 4 Procedure Next - Gen - GA Input The Current Population Output The Next Population While SNvainateyagoldarNoch keine Bewertungen
- 1, If 0, Otherwise : Iii. The Proposed FrameworkDokument1 Seite1, If 0, Otherwise : Iii. The Proposed FrameworkvainateyagoldarNoch keine Bewertungen
- Min - Util, Ce Is Not An HUI. The TU of T T TWU (Ce) TU (T: Tid T T T T T T T T T TDokument1 SeiteMin - Util, Ce Is Not An HUI. The TU of T T TWU (Ce) TU (T: Tid T T T T T T T T T TvainateyagoldarNoch keine Bewertungen
- C. Illustrated ExampleDokument5 SeitenC. Illustrated ExamplevainateyagoldarNoch keine Bewertungen
- K-Itemset. Let D (T: Ux uXTDokument1 SeiteK-Itemset. Let D (T: Ux uXTvainateyagoldarNoch keine Bewertungen
- Standard Steady State Genetic Algorithms Can Hillclimb Faster Than Mutation-Only Evolutionary AlgorithmsDokument14 SeitenStandard Steady State Genetic Algorithms Can Hillclimb Faster Than Mutation-Only Evolutionary AlgorithmsvainateyagoldarNoch keine Bewertungen
- Proc. POWERCON 7, 1980, Pp. E3-1-E3-15.: OnclusionDokument1 SeiteProc. POWERCON 7, 1980, Pp. E3-1-E3-15.: OnclusionvainateyagoldarNoch keine Bewertungen
- S - 5 S - SS - : (A) Li2Sn5Dokument1 SeiteS - 5 S - SS - : (A) Li2Sn5vainateyagoldarNoch keine Bewertungen
- i1T5u20L.3: Scattering Angle (Deg.)Dokument1 Seitei1T5u20L.3: Scattering Angle (Deg.)vainateyagoldarNoch keine Bewertungen
- Matrix Converter Modulation Strategies: A New General Approach Based On Space-Vector Representation of The Switch StateDokument1 SeiteMatrix Converter Modulation Strategies: A New General Approach Based On Space-Vector Representation of The Switch StatevainateyagoldarNoch keine Bewertungen
- 6.4li + Sno - 8.4li + Sno, - : A Reasonable Correlation Between The Calculated andDokument1 Seite6.4li + Sno - 8.4li + Sno, - : A Reasonable Correlation Between The Calculated andvainateyagoldarNoch keine Bewertungen
- Fig. 5. Double-Sided Switching Pattern in A Cycle Period: Casadei Et Al.: Matrix Converter Modulation Strategies 375Dokument1 SeiteFig. 5. Double-Sided Switching Pattern in A Cycle Period: Casadei Et Al.: Matrix Converter Modulation Strategies 375vainateyagoldarNoch keine Bewertungen
- Fig. 2. Double-Sided Switching Pattern in A Cycle PeriodDokument1 SeiteFig. 2. Double-Sided Switching Pattern in A Cycle PeriodvainateyagoldarNoch keine Bewertungen
- Binder2 5Dokument1 SeiteBinder2 5vainateyagoldarNoch keine Bewertungen
- Matrix Converter Modulation Strategies: A New General Approach Based On Space-Vector Representation of The Switch StateDokument1 SeiteMatrix Converter Modulation Strategies: A New General Approach Based On Space-Vector Representation of The Switch StatevainateyagoldarNoch keine Bewertungen
- Fig. 1. Basic Scheme of Matrix Converters.: Casadei Et Al.: Matrix Converter Modulation Strategies 371Dokument1 SeiteFig. 1. Basic Scheme of Matrix Converters.: Casadei Et Al.: Matrix Converter Modulation Strategies 371vainateyagoldarNoch keine Bewertungen
- Casadei Et Al.: Matrix Converter Modulation Strategies 373: A. SVM TechniqueDokument1 SeiteCasadei Et Al.: Matrix Converter Modulation Strategies 373: A. SVM TechniquevainateyagoldarNoch keine Bewertungen
- Binder1 40Dokument1 SeiteBinder1 40vainateyagoldarNoch keine Bewertungen
- Ionic vs. Covalent Bonding Lab InvestigationDokument3 SeitenIonic vs. Covalent Bonding Lab InvestigationLeslieNoch keine Bewertungen
- Separating Solids From LiquidsDokument4 SeitenSeparating Solids From LiquidsRizal RusydiNoch keine Bewertungen
- Copper Nickel Concentric Reducer Din86089Dokument2 SeitenCopper Nickel Concentric Reducer Din86089Araby GamalNoch keine Bewertungen
- GulfSea DE Compressor Oil Series 100Dokument2 SeitenGulfSea DE Compressor Oil Series 100Md.Obydur RahmanNoch keine Bewertungen
- CSMB - VERNACULAR TERMSDokument2 SeitenCSMB - VERNACULAR TERMSChari Madlang-awa BunagNoch keine Bewertungen
- Display Boards For Hazardous Waste Generating UnitsDokument2 SeitenDisplay Boards For Hazardous Waste Generating UnitsBAla100% (5)
- Socotherm BrochureDokument9 SeitenSocotherm Brochuremaruc08Noch keine Bewertungen
- Horizontal Flame ArresterDokument5 SeitenHorizontal Flame ArresterMustafa PardawalaNoch keine Bewertungen
- Al-Obaidi - 2021 - IOP - Conf. - Ser. - Earth - Environ. - Sci. - 779 - 012010Dokument12 SeitenAl-Obaidi - 2021 - IOP - Conf. - Ser. - Earth - Environ. - Sci. - 779 - 012010Cladi DanaNoch keine Bewertungen
- Rack B-2Dokument535 SeitenRack B-2waqas001Noch keine Bewertungen
- AluminiumDokument19 SeitenAluminiumAnkur Jain100% (1)
- How To Inexpensively Repair A Crumbling Retaining Wall at Your Home - The Washington PostDokument7 SeitenHow To Inexpensively Repair A Crumbling Retaining Wall at Your Home - The Washington PostAdam BrownNoch keine Bewertungen
- Painting Procedure: Total Solutions Integrated ServicesDokument9 SeitenPainting Procedure: Total Solutions Integrated ServicesLi LiuNoch keine Bewertungen
- Metal Organic Frameworks Vs Buffers Case Study of Ui o 66 StabilityDokument19 SeitenMetal Organic Frameworks Vs Buffers Case Study of Ui o 66 StabilitymahahajNoch keine Bewertungen
- Tolerance As Per AWS D1.1Dokument3 SeitenTolerance As Per AWS D1.1milind bedarkarNoch keine Bewertungen
- Taloja Industrial Directory-By Taloja Industries AssociationDokument434 SeitenTaloja Industrial Directory-By Taloja Industries Associationdeva_4184% (74)
- 1 Memoriu Tehnic CS enDokument219 Seiten1 Memoriu Tehnic CS enIuly AnaNoch keine Bewertungen
- GCSE Exam QuestionsDokument155 SeitenGCSE Exam Questionsdanielphilip68Noch keine Bewertungen
- Corrosion Protection of Flanges and Valves: E. Lyublinski, G. Begunova, E. Kopilova, M. Schultz and R. SinghDokument7 SeitenCorrosion Protection of Flanges and Valves: E. Lyublinski, G. Begunova, E. Kopilova, M. Schultz and R. SinghBoulHich BoulHichNoch keine Bewertungen
- LPP BiomoleculesDokument6 SeitenLPP BiomoleculesAashiNoch keine Bewertungen
- UOP-Mercury-Removal-From-Natural-Gas-and-Liquid-Streams-Tech-Paper 2 PDFDokument9 SeitenUOP-Mercury-Removal-From-Natural-Gas-and-Liquid-Streams-Tech-Paper 2 PDFPedraza Velandia JhonNoch keine Bewertungen
- Study On Impacts of Spinning Process On Fiber CharacteristicsDokument6 SeitenStudy On Impacts of Spinning Process On Fiber CharacteristicsNeelakandan DNoch keine Bewertungen
- T02 11 Simonovic - Medic - Medic - Selimotic - GNP2020Dokument8 SeitenT02 11 Simonovic - Medic - Medic - Selimotic - GNP2020Kenan KajosevicNoch keine Bewertungen
- Gas Laws Homework IIDokument4 SeitenGas Laws Homework IIchpwalker100% (2)
- Thermodynamic Database of Multi-Component MG Alloys and Its Application To Solidification and Heat TreatmentDokument16 SeitenThermodynamic Database of Multi-Component MG Alloys and Its Application To Solidification and Heat Treatmentmridzuan69Noch keine Bewertungen
- Hot Spotet, Lista Me 31 Pikat Më Të Rrezikshme Mjedisore Në VendDokument5 SeitenHot Spotet, Lista Me 31 Pikat Më Të Rrezikshme Mjedisore Në VendTelevizioni A1 ReportNoch keine Bewertungen
- Effect of Rice Husk Ash On Properties of ConcreteDokument4 SeitenEffect of Rice Husk Ash On Properties of ConcreteEduardo Fernando Alarcon PrincipeNoch keine Bewertungen
- Community Based Project ProposalDokument5 SeitenCommunity Based Project ProposalJOMICA SANTOSNoch keine Bewertungen
- Copper Mountain Mine TR 09012020Dokument233 SeitenCopper Mountain Mine TR 09012020Thibaut LeconteNoch keine Bewertungen
- Hardtop Clear: Technical Data SheetDokument5 SeitenHardtop Clear: Technical Data SheetHussain AlsadahNoch keine Bewertungen