Beruflich Dokumente
Kultur Dokumente
2 MohamedKhairy1
Hochgeladen von
Azhar AdityaOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
2 MohamedKhairy1
Hochgeladen von
Azhar AdityaCopyright:
Verfügbare Formate
See
discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/261992936
Applicability of Biosensor and Oxygen Sensor
for Monitoring Spoilage and Bacterial
Contaminants of Packed Minced Beef and ....
Conference Paper · April 2014
DOI: 10.13140/2.1.2549.6001
CITATIONS READS
0 451
5 authors, including:
Prof. Ashraf Mahdy Sharoba Mohamed Morsy
Benha University Benha University
116 PUBLICATIONS 291 CITATIONS 19 PUBLICATIONS 69 CITATIONS
SEE PROFILE SEE PROFILE
Some of the authors of this publication are also working on these related projects:
food sciences View project
Production of new food blends as baby foods and school foods from millet View project
All content following this page was uploaded by Prof. Ashraf Mahdy Sharoba on 01 May 2014.
The user has requested enhancement of the downloaded file.
2nd International Conference On Biotechnology Applications In Agriculture (ICBAA), Benha University,
Moshtohor and Hurghada, 8-12,April 2014, Egypt
Applicability of Biosensor and Oxygen Sensor for Monitoring Spoilage and Bacterial
Contaminants of Packed Minced Beef and Poultry
Morsy, M.K.,1,2 Khalaf, H.H.1, Sharoba, A.M.1 and El-Tanahi, H.H.1
1
Department of Food Science, Faculty of Agriculture, Benha University, Qaluobia, Egypt
2
Department Micro-Nanotech, Technical University of Denmark, Denmark
E-mail:mohamed.abdelhafez@fagr.bu.edu.eg
Abstract
New technologies have revolutionized the food safety sector. Nano-biosensor based enzymeand oxygen
sensor have recently gained much attention in the field of food safety. Two prototypes of the sensor capable of
handling of different samples were developed and tested with beef and chicken fillets that stored at 4 °C±1 for 11
days, as wellthe physicochemical, freshness and microbiologicaltests were done. The results from this study
demonstrated that there are strong color changes were observed in biosensor array from a green color to red color
after 9and 10 days of storage beef and chicken fillets, respectively. Likewise, increased in pH, TVB-N and TBA
values during the storage periods. Moreover, the TVBC and psychrophilic bacteria were increased. Also, noticed that
the remaining oxygen content in the package headspace of different samples decreased continuously during
storage. These data present promising evidence for possible application of bio- and oxygen sensor for rapid
detection of meat spoilage for food defense and food safety application.
Keywords:nanotechnology, biosensor, oxygen sensor, beef, chicken fillets, microbiological quality.
Introduction contamination becoming an increasingly urgent
(Scindia et al., 2007).
Nanotechnology has emerged as one of the most Biosensors are compact analytical devices which
innovative technologies and has the potential to detect, record and transmit information pertaining to
improve food safety and quality. Safe food is an biological reactions (Yam et al., 2005). They consist
essential requirement of modern society, and of a bio-receptor specific to a target analyte and a
governmental authorities throughout the world are transducer to convert biological signals to a
constantly monitoring the food supply chain in an quantifiable electrical response. Bio-receptors are
attempt to ensure an adequate safety organic materials such as enzymes, antigens,
level(Neethirajan and Jayas, 2011). Likewise, this microbes, hormones and nucleic acids. Transducers
concern for food safety has recently been further may be electrochemical, optical, or calorimetric and
exacerbated by the growing threat of bioterrorism are system dependent (Zhang et al., 2014). Intelligent
(Ko et al., 2010). packaging systems incorporating biosensors have the
According to published CDC 2011 (US Centers potential for extreme specificity and reliability. A
for Disease Control and Prevention) data, foodborne market analysis of pathogen detection and safety
diseases account for approximately 76 million systems for the food packaging industry suggests that
illnesses, 325.000 hospitalizations and 5000 deaths businesses offer considerable promise for future
each year in the US alone. While meat and poultry growth (Alocilja and Radke, 2003).Biosensor
have been implicated in several foodborne outbreaks methods can be applied, as rapid screening tools, to
over the years, these products also are prone to detect such contaminants. They can be developed
spoilage (Cardenas et al., 2008). using several types of transducer (Reder-Christ and
Many methods have been employed to detect Bendas,2011) one of which is based on fluorescence
pathogens and microbial contaminant; however, either through quenching (Wang et al., 2011) or
some methods, such as traditional colony counting, labeling of the biological recognition elements, e.g.,
are excessively time consuming while others, PCR fluorescently labeled antibody(Taitt et al.,2008). The
for example, involve complex instrumentation and market for biosensors was estimated as $17.3 billion
handling (Lazcka et al., 2007). Moreover, by year 2015 as per some business reports (Sadana
determination of the TVB-N is one of the most and Sadana, 2011).
widely used methods to measure the freshness of Fluorescence-based oxygen sensors represent the
meats. The total volatile base measured is composed most promising systems to date for remote
principally of ammonia, trimethylamine (TMA) and measurement of headspace gases in packaged meat
dimethylamine (DMA) (Ocano-Higuera et al., 2009). products. A number of disposable oxygen sensing
All these methods are relatively time consuming, prototypes have been developed which may be
require experienced personal and expensive produced at low cost and provide rapid determination
apparatus.The demand of novel technologies for the of oxygen concentration (Kerry and Papkovsky,
rapiddetection of food spoilage and bacterial 2002). The active component of a fluorescence-based
Nano-Biotechnologyand Bio-systems Engineering, 7-14 7
Applicability of Biosensor and Oxygen……….
oxygen sensor usually consists of a long-delay measurement in 10 s. For O2 measurements, the
fluorescent or phosphorescent dye encapsulated in a instrument need to be brought in an optical contact
solid polymer matrix (Borchert et al., 2012). with the sensor (5–10 mm distance) to produce an O2
The aim of the present research was to develop, reading (% of O2, compensated for temperature and
from first principles, a low cost, potentially portable, pressure variation).The OxyDot-indicators were
optic biosensor capable of detecting low levels of placed in the headspace of the sample jars using
microbial contaminants and compounds of interest in transparent adhesive. A Windows-based software
food analysis. Also, attempt to use non-invasive was used for the recording of measured parameters
oxygen determination method based on fluorescence (oxygen concentration) and data storage (Molinaro et
quenching to evaluate the shelf-life of meat. al., 2013).
Materials and Methods Physicochemical analysis
Moisture content, crude protein, crude fat and
Sample collection and preparation total ash were determined according to A.O.A.C.
Minced beef and chicken fillet samples used in (2005). The pH measurements were carried out using
this study were purchased from a local market in a digital pH-meter (model MA 5736, Metrel, Iskra,
Lyngby City, Denmark (DK). The samples were Slovenia). The contents of TVB-N and TMA were
transported in the isothermal ice box to the determined by steam distillation according to the
laboratory within 10 min. The samples were cut into method described by (Haroldet al.,1987) and
portions 5 cm x5 cm; this procedure was performed expressed as mg N/100g of muscle. TBA was
in a hood cabinet with an aseptic knife. All prepared determined using a spectrophotometric method
samples were put in a sealed plastic bag, and stored according to (Vyncke, 1970)and the results were
at 4±1°C. expressed as mg malondialdehyde (MDA)/kg of
muscles.
Design of biosensor array
Biosensor array is an enzymatic system, was Microbiological Analysis
based on color change caused by a pH decrease A sample of 25 g was taken aseptically from each
which is the result of a controlled enzymatic fillets and transferred to a stomacher bag, and 225
hydrolysis of a lipid substrate. Hydrolysis of the mL of sterilized peptone water (Becton, Dickinson
substrate cause acid release and the pH drop is and Co., Le Pont de Claix, France) was added. The
translated in a color change of a pH indicator from mixture was homogenized for 2 min with a
deep green to bright yellow to orange red. Different Stomacher 3500 (Seward Medical, Worthing, U.K).
combinations of enzyme-substrate types and Samples (0.1 mL) of serial dilutions of fillet
concentrations can be used to give a variety of homogenates were spread on the surface of the
response lives, temperature dependencies and appropriate dry medium in Petri dishes for
spoilage stage. Biosensor is inactive until pressure is determination of the total viable bacterial count
applied to break a barrier between two “ampoules” (TVBC) and psychrophilic bacteria (Psy.) on plate
and the contents are mixed together. One ampoule count agar (Oxoid, CM325), and incubated at 37 °C
contains a proprietary lipase enzyme and pH for 48 hrs (TVBC) and/or 7°C for 5 days
indicating dye while the other ampoule contains an (Psy.)Yousef and Carlstrom (2003).
enzyme substrate (triglyceride). A green to yellow to
orange red color change occurs as the pH is lowered Statistical Analysis
via liberation of fatty acids from triglyceride by the The statistical analysis was carried out using
lipase enzyme. Since excess enzyme is present, the ANOVA with one factor under significance level of
reaction rate is governed primarily by temperature. 0.05 for the obtained results using SPSS and data
Concentrations of lipase enzyme and triglyceride can were treated as a complete randomization design
be manipulated during manufacture to yield targeted according to (Steel et al., 1997). The experiments
activation energies, Ea, providing design flexibility were performed in triplicate, using three samples per
(Vitsab A.B., Malmö, Sweden). treatment (Siragusa et al., 1999). Multiple
comparisons were carried out applying the LSD test.
Oxygen sensor technique
The oxygen in the jar’s headspace of meat Results and Discussion
samples was monitored, using an optical measuring
system Optech™ Platinum O2 sensor device and Physicochemical analysis and microbiological
disposable O2 sensor stickers from Mocon quality of beef and chicken fillets
(Minneapolis, USA). The optical O2 sensor works on Physicochemical properties and microbiological
the principle of a phosphorescent dye that is quality of beef and chicken fillet given in Table
incorporated in a polystyrene polymer membrane as (1)show the moisture content was 73.48 and 74.37%,
adherent agent and for environmental protection. The crude protein was 20.46 and 21.59%, ether extract
Optech device uses LED technology to take was4.76 and 2.83%, ash content was 1.1856 and
Nano-Biotechnology and Bio-systems Engineering 8
Applicability of Biosensor and Oxygen……….
1.0483%, pH value was 5.77 and 5.8, TVN was (7.47 fillets, respectively. These results confirm thatbeef
and 9.1 mg/100g), TBA was (0.263 and 0.169 mg and poultry rich in protein. These results are in
MDA/Kg), total viable bacterial count was 8.9X103 agreement with those reported by Al-Najdawi and
and7.2 X103 CFU/g, and psychrophilic bacteria were Abdullah (2002) and Salem et al. (2010).
8.8 X102 and 5.6 X102 CFU/g in beef and chicken
Table 1.Physicochemical properties and microbiological quality of beef and chicken fillet.
Meat Types
Components*
Beef Chicken Fillet
Moisture (%) 73.48±0.18 74.37±0.44
Crude protein (%) 20.46±0.8 21.59±0.08
Ether extract (%) 4.76±0.02 2.83±0.16
Ash (%) 1.1856±0.04 1.0483±0.02
pH value 5.77±0.03 5.8±0.03
TVN (mg/100g) 7.47±0.23 9.1±0.4
TBA (mg MDA/Kg) 0.263±0.014 0.169±0.011
Total viable bacterial count 8.9X103 7.2 X103
2
Psychrophilic bacteria 8.8 X10 5.6 X102
* Mean of triplicate determinations ±SE.
Biosensor array for monitoring quality of beef
and chicken fillets as smart packaging
The design of the biosensor is based on the strong
dye-analyte interactions, which is quite different
from other electronic nose technologies that
generally rely on weak, nonspecific intermolecular
interactions, primarily van der Waals and physical Fig.1. Biosensor array reactions.
adsorption interactions according to Fig. (1).
As shown in Fig. (2) after 4 days of storage beef,
no color change was observed in biosensor array its
the same as of the original one (control), it still green
color, this mean fresh beef, after that observed
gradually color changes of biosensor array to orange
color till 8 days of storage, this mean beef is still
fresh and should use soon. After 9 days of storage
there are strong color changes observed in biosensor
array to red color, this mean beef not guaranteed.
Regards of chicken fillets it noticed that after 6
days of storage, no color change in biosensor array,
was nearly the same as that of the original one
(control) it still green color this mean fresh chicken, Exposure time (days)
after that observed gradually color changes of Fig.2. Utilization of biosensor to monitor spoilage in
biosensor array to orange color till 9 days of storage, package headspace of beef during storage at 4°C.
this mean chicken is still fresh and should use soon.
After 10 days of storage there are strong color
changes observed in biosensor array to red color, this
mean chicken fillet is not guaranteed as illustrated in
Fig. (3). The color changes of biosensor array due to
volatile organic components (VOCs) emitted by the
meat changed as a result of enzymatic activities
taking place after the postmortem stage had started.
At the early time of storage, the concentration of
short-chain alcohols and hydroxyl group still low,
then increased and microbes started to multiply.
Metabolites such as amines were produced from the
decomposition of protein due to microorganisms.
Exposure time (days)
Trimethylamine, TVBN and other amines gradually Fig.3. Utilization of biosensor to monitor spoilage in
increased during the storage period. These obtained package headspace of chicken fillet during storage at
results generally are in agreement with those 4°C.
previously reported by Huang et al. (2011) and Pires
et al. (2011).
Nano-Biotechnology and Bio-systems Engineering 9
Applicability of Biosensor and Oxygen……….
Application of oxygen sensor array to Physicochemical changes of beef and chicken
monitorquality of beef and chicken fillets fillets during storage
Oxygen is an essential element for all living Data illustrated in (Fig. 5) show changes in the
organisms and also plays an important role in many moisture content of beef and chicken fillets during
chemical industrial processes, including those in storage at 4°C. Generally, moisture content of beef
which an absence of oxygen is required (Borchert et and chicken samples decreased continuously during
al., 2012). It is also one of the most commonly storage period at 4°C. The initial moisture content in
analyzed chemical species, although many of the beef and chicken was 73.48 and 74.37%,
preferred detection methods, such as gas respectively, while after ten days of storage, it was
chromatography and electrochemical techniques, observed to be 62.52 and 68.72%, respectively.
require rather expensive equipment and trained ANOVA indicated that there were significant
operators. Optical oxygen indicators are fast differences (P ≥ 0.05) between samples in time zero
responding, inexpensive and easy to use (Kozak and and ten days of storage. The loss in moisture content
Samotyja, 2013). of meat samples during storage may be due to the
In recent years there has been dedicated non- reduction of water holding capacity and increase the
invasive oxygen determination method developed drip as a result of protein denaturation. In addition,
based on fluorescence quenching of specific optical evaporate of moisture through the polyethylene bags
sensors sensitive to oxygen (Mills, 2005) and used to pack. These results are in agreement with
(Wolfbeis and Weidgans, 2006). The method was those observed by (Rhee et al., 2012).
validated and implemented for common use in 2008 The changes in pH value of beef and chicken
as ASTM F2714-08 standard. fillets during storage at 4°C are illustrated (Fig. 6).
Results illustrated in (Fig. 4) show the remaining The results indicate that the pH value of beef and
oxygen percentage in the package headspace of beef chicken samples increased during the storage periods
and chicken fillet during storage at 4°C using oxygen from (5.77 to 7.19) and (5.80 to 7.23), respectively.
sensor. It noticed that the remaining oxygen content ANOVA indicated that there were significant
in the package headspace of different samples such differences (P ≥ 0.05) between samples in time zero
as beef and chicken fillet were 21.53 and 21.52%, and ten days of storage. The change of pH value was
respectively. Generally, remaining oxygen content in due to an increase in volatile bases from the
package headspace decreased continuously during decomposition of nitrogenous compounds by
storage at 4°C, it decreased from (21.53 to 0%) in endogenous or microbial enzymes. These results are
beef sample after 9 days of storage, and (21.52 to in agreement with those observed by (Gheisari et al.,
0%) in chicken fillet sample after 10 days of storage. 2009).
The decrease in remaining oxygen content during Total volatile basic nitrogen (TVB-N) and
storage might be due to activation of aerobic bacteria thiobarbituric acid (TBA) have been used as indices
that consume the oxygen during decomposition of to assess the spoilage of refrigerated beef and
nitrogenous compounds. These obtained results chicken fillets. TVB-N was used for determination of
generally are in agreement with those previously the spoilage level of meat during the storage period.
reported by (Papkovsky et al., 2002). Total volatile nitrogen of beef and chicken fillets
during storage at 4°C is shown in (Fig. 7). It noticed
25 that TVN increased continuously during storage
Chicken
period (Sun and Holley, 2012). The initial TVN of
Beef
20 beef was 7.47 mg/100g, while after ten days of
storage, it was observed to be 28.47 mg/100g,
Oxygen %
15 whereas the initial TVN of chicken fillets were 9.10
mg/100g, while after ten days of storage, it was
10
observed to be 33.83 mg/100g. ANOVA indicated
5 that there were significant differences (P ≥ 0.05)
between samples in time zero and ten days of
0 storage. The level 25 mg/100g have been considered
the upper limit, above which meats are considered
spoiled (Gheisari et al., 2009). Generally, the
Storage days increase in TVN value during storage of meat
samples might be due to rapid breakdown of protein
Fig.4. Oxygen sensor monitor O2 percentage in through microbial decomposition.These results are in
package headspace of beef and chicken fillet agreement with those reported by (Edris et al., 2012).
during storage at 4°C.
Nano-Biotechnology and Bio-systems Engineering 10
Applicability of Biosensor and Oxygen……….
8 beef
7 chicken
6
5
4
3
2
1
0
Fig.5.Changes in moisture content on beef and chicken Fig.6.Changes in pH value on beef and chicken fillets
fillets during storage at 4 °C. during storage at 4 °C.
40 1.6
35 1.4
30 1.2
25 1
20 0.8
15 Beef Beef
0.6
chicken Chicken
10 0.4
5 0.2
0 0
Fig.7.Changes in the TVB-N on beef and chicken fillets Fig.8.Changes in the TBA on beef and chicken fillets
during storage at 4 °C. during storage at 4 °C.
Changes in thiobarbituric acid value during respectively. The TBVC of beef and chicken fillets
storage of beef and chicken fillets at 4°C were samples increased during the storage periods. It
illustrated in (Fig. 8). The TBA values of beef and increased from 8.9x103 to 5.5x107 CFU/g of beef,
chicken fillets increased up to 10and11 days, while it increased from 7.2x103 to 7.1x107 CFU/g on
respectively, depending upon storage time. The TBA chicken fillets. Also, psychrophilic bacteria increased
results as the same trend in TVN. It increased from from 8.8x102 to 9.1x104 CFU/g of beef, while it
(0.263 to 1.347mg MDA/Kg) of beef, while on increased from 5.6 x102 to8.5x104 CFU/g on chicken
chicken fillets increased from (0.169 to 1.352 mg fillets during storage periods at 4°C.
MDA/Kg). ANOVA indicated that there were As recommended by the International
significant differences (P ≥ 0.05) between samples in Commission on Microbiological Specification for
time zero and ten days of storage. These may be due Food (ICMSF, 1986), an increase of total plate count
to fat content which would speed up lipid oxidation. (TPC) up to levels exceeding the value of 6 log
These results are in agreement with those observed CFU/g is regarded as microbial spoiled meat muscle
by (Edris et al., 2012). not fit for human consumption. These results are in
agreement with those observed by (Ohk and Bhunia,
Microbial examination of beef and chicken fillets 2013).
during storage
The microbiological quality of beef and chicken
fillets is dependent on a number of factors such as
raw materials, sanitation during process and storage
period. Data in Table (2) indicate that the total viable
bacterial count (TBVC) of beef and chicken fillets
was 8.9x103 and 7.2x103 CFU/g, while psychrophilic
bacteria was 8.8x102 and 5.6x102 CFU/g,
Nano-Biotechnology and Bio-systems Engineering 11
Applicability of Biosensor and Oxygen……….
Table 2.Microbiological quality of beef and chicken fillet during storage at 4 °C.
Meat Types
Storage period Beef Chicken fillet
(Days) TVBC Psychrophilic TVBC Psychrophilic
0 8.9X103 8.8X102 7.2X103 5.6X102
4 2 3
1 1.14X10 9.4X10 7.9X10 6.5X102
4 3 4
2 5.6X10 1.12X10 1.04X10 7.9X102
4 3 4
3 6.3X10 1.24X10 1.27X10 9.2X102
4 3 4
4 7.8X10 5.1X10 4.4X10 1.03X103
4 3 4
5 9.2X10 7.1X10 6.9X10 5.3X103
5 3 4
6 1.13X10 8.7X10 9.2X10 7.0X103
5 4 5
7 4.1X10 1.03X10 1.05X10 8.9X103
5 4 5
8 5.7X10 1.15X10 1.19X10 1.02X104
6 4 5
9 5.7X10 6.8X10 7.6X10 5.4X104
10 5.5X107 9.1X104 9.6X106 6.7X104
7
11 ND ND 7.1X10 8.5X104
Mean of duplicate determinations.
ND: Not detect
Conclusion Borchert, N.; Hempel, N.; Walsh, H.; Kerry, J.P. and
Papkovsky, D.B. (2012) High throughput quality
In conclusion, new scientific disciplines and and safety assessment of packaged green produce
technologies have revolutionized the food sector. The using two optical oxygen sensor based systems.
results of this study have demonstrated that strong Food Control 28, 87-93.
color changes were observed in biosensor array from Cardenas, F.C.; Giannuzzi, L. and Zaritzkay, N.E.
a green color to red color that applied in beef and (2008) Mathematical odelling of microbial
chicken fillet during storag, this mean these sample growth in ground beef from Argentina. Effect of
not guaranteed. Also, noticed that the remaining lactic acid addition, temperature and packaging
oxygen content in the package headspace of different film. Meat Science 79, 509-520.
samples decreased continuously during storage. Bio- CDC (2011) Centers for Disease Control and
and oxygen sensors are playing an important role in Prevention, Food-related illness and death in the
the formation of food processing technique and United States, Emerg. Infect Dis. 5607-5625.
detection of spoilage and contaminants. Edris, A.M.; Hemmat, M.I.; Shaltout, F.A.; Elshater,
M.A. and Eman, F.M.I. (2012) Study on incipient
Acknowledgments spoilage of chilled chicken cuts-up. Benha
The authors would like to express their sincere Veterinary Medical Journal 23 (1): 81-86.
gratitude and appreciation for the financial support Gheisari, H.R.; Aminlari, M. and Shekarforoush, S.S.
from the Danish Agency for Universities and (2009) A comparative study of the biochemical
Internationalization. The authors want to thank and functional properties of camel and cattle meat
(Department Micro-Nanotech, Technical University during frozen storage. Veterinarski Arhiv Journal
of Denmark, University, DTU) for hosting 79 (1): 51-68.
thisresearch project. Harold, E.; Roland, S.K. and Roland, S. (1987)
Pearson's chemical analysis of foods. 8th Ed.
References Longman House, Burnt, M., Harlow, Essex CM
202 JE, England.
A.O.A.C. (2005) Official methods of analysis. Huang, X.; Xin, J. and Zhao, J. (2011) A novel
Association of Official Analytical, Chemists 18th technique for rapid evaluation of fish freshness
ed., Washington, D.C., U.S.A. using colorimetric sensor array. Journal of Food
Al-Najdawi, R. and Abdullah, B. (2002) Proximate Engineering 105, 632–637.
composition, selected minerals, cholesterol ICMSF. (1986) Microorganisms in Foods. In:
content and lipid oxidation of mechanically and Application of the Hazard Analysis Critical
hand-deboned chickens from the Jordanian Control Point (HACCP) System to Ensure
market. Meat Science 61, 243–247. Microbiological Safety and Quality. 2nd ed.
Alocilja, E.C. and Radke, S.M. (2003) Market Blackwell Scientific Publications. 42, 181-195.
analysis of biosensors for food safety. Biosensors Kerry, J.P. and Papkovsky, D.B. (2002)
and Bioelectronics 18, 841–846. Development and use of non-destructive,
ASTM F 2714-08 (2008) Standard test method for continuous assessment, chemical oxygen sensors
oxygen headspace analysis of packages using in packs containing oxygen sensitive foodstuffs.
fluorescent decay. Research Advances in Food Science 3, 121-140.
Nano-Biotechnology and Bio-systems Engineering 12
Applicability of Biosensor and Oxygen……….
Ko, S.; Gunasekaran, S. and Yu, J. (2010) Self- Sadana, A. and Sadana, N. (2011) Handbook of
indicating nanobiosensor for detection of 2,4- biosensors and biosensor kinetics. Elsevier B.V,
dinitrophenol. Food Control 21, 155–161. Amsterdam.
Kozak, W. and Samotyja, U. (2013) The use of Salem, A.M.; Amin, R.A. and Afifi, G.S. (2010)
oxygen content determination method based on Studies on antimicrobial and antioxidant
fluorescence quenching for rapeseed oil shelf-life efficiency of some essential oils in minced beef.
assessment. Food Control 33, 162-165. Journal of American Science 6, 691-700.
Lazcka, O.; Del-Campo, F.J. and Munz, F.X. (2007) Scindia, Y.; Silbert, L.; Volinsky, R.; Kolusheva, S.
Pathogen election: a perspective of traditional and Jelinek, R. (2007) Colorimetric detection and
methods and biosensors. Biosensors and fingerprinting of bacteria by glass-supported
Bioelectronics 22, 1205–1217. lipid/polydiacetylene films. Langmuir 23, 4682–
Mills, A. (2005) Oxygen indicators and intelligent 4687.
inks for packaging food. Platinum Metals Review Siragusa, G.R.; Cutter, C.N. and Willet, J.L. (1999)
34, 1003-1011. Incorporation of bacteriocin in plastic retains
Molinaro, S.; Romero, M.C.; Boaro, M.; Sensidoni, activity and inhibits surface growth of bacteria on
A.; Lagazio, C.; Morris, M. and Kerry, j. (2013) meat. Food Microbiology 16, 229–235.
Effect of nanoclay-type and PLA optical purity Steel, R.; Torrie, J. and Dickey, D. (1997) Principles
on the characteristics of PLA-based and Procedures of Statistics: A Biometrical
nanocomposite films. Journal of Food Approach, 3rd ed, McGraw-Hill, New York, NY.
Engineering 117, 113–123. Sun, X.D. and Holley, R.A. (2012) Antimicrobial
Neethirajan, S. and Jayas, D.S. (2011) and antioxidative strategies to reduce pathogens
Nanotechnology for the food and bioprocessing and extend the shelf life of fresh red meats.
industries. Food Bioprocess Technology 4, 39– Comprehensive Reviews in Food Science and
47. Food Safety 11, 340-354.
Ocano-Higuera, V.M.; Marquez-Ros, E.; Canizales- Taitt, C.R.; Shriver-Lake, L.C.; Ngundi, M.M. and
Dvila, M.; Castillo-Yez, F.J.; Pacheco-Aguilar, Ligler, F.S. (2008) Array biosensor for toxin
R. and Lugo-Snchez, M.E. (2009) Postmortem detection: Continued Advances. Sensors 8, 8361-
changes in Cazon fish muscle stored on ice. Food 8377.
Chemistry 116(4): 933–938. Vyncke, W. (1970) Direct determination of the
Ohk, S. and Bhunia, A.K. (2013) Multiplex fiber thiobarbituric acid value in trichloroacetic acid
optic biosensor for detection of Listeria extracts of fish as a measure of oxidative
monocytogenes, Escherichia coli O157:H7 and rancidity. Fette Seifen Anstrichmittel72, 1084 -
Salmonella enterica from ready-to-eat meat 1087.
samples. Food Microbiology 33, 166-171. Wang, K.; Wang, L.; Jiang, W. and Hu, J. (2011) A
Papkovsky, D.B.; Smiddy, M.A.; Papkovskaia, N.Y. sensitive enzymatic method for paraoxon
and Kerry, J.P. (2002) Nondestructive detection based on enzyme inhibition and
measurement of oxygen in modified atmosphere fluorescence quenching Talanta 84, 400–405.
packaged hams using a phase-fluorimetric sensor Wolfbeis, O.S. and Weidgans, B.M. (2006) Fiber
system. Journal of Food Science 67, 3164–3169. optic chemical sensors and biosensors: a view
Pires, A.C.; Soaresa, N.F.; Silva, L.H.; Silva, M.C.; back. In F. Baldini (Ed.), Optical chemical
Almeidac, M.V.; Hyaricc, M.L.; Andradea, N.J.; sensors (pp.17-44). Dordrecht: Springer.
Soaresa, R.F.; Magesteb, A.B. and Reis, S.G. Yam, K.L.; Takhistov, P.T. and Miltz, J. (2005)
(2011)A colorimetric biosensor for the detection Intelligent packaging: Concepts and applications.
of foodborne bacteria. Sensors and Actuators B Journal of Food Science 70, 1–10.
153, 17–23. Yousef, A.E. and Carlstrom, C. (2003) "Food
Reder-Christ, K. and Bendas, G. (2011) Biosensor Microbiology: A laboratory Manual". John Wiley
Applications in the Field of Antibiotic Research- and sons, Inc.
A Review of Recent Developments. Zhang, F.; Zhang, Q.; Zhang, D.; Lu, Y.; Liu, Q. and
Sensors 11(10): 9450-9466. Wang, P. (2014) Biosensor analysis of natural
Rhee, K.S.; Cho, S.H. and Pradahn, A.M. (2012) and artificial sweeteners in intact taste
Composition, storage stability and sensory epithelium. Biosensors and Bioelectronics 54,
properties of expanded extrudates from blends of 385-392.
corn starch and goat meat, lamb, mutton, spent
fowl meat, or beef. Meat Science 52, 135-141.
Nano-Biotechnology and Bio-systems Engineering 13
Applicability of Biosensor and Oxygen……….
ﺘطﺒﻴق اﻟﻤﺴﺘﺸﻌر اﻟﺤﻴوى و ﻤﺴﺘﺸﻌر اﻷوﻛﺴﺠﻴن ﻟﻛﺸف اﻟﻔﺴﺎد واﻟﺘﻠوث اﻟﺒﻛﺘﻴرى
ﻓﻰ اﻟﻠﺤم اﻟﺒﻘرى اﻟﻤﻔروم وﻟﺤم اﻟدواﺠن اﻟﻤﻌﺒﺄة
1
ﻤﺤﻤد ﺨﻴرى ﻋﺒد اﻟﺤﺎﻓظ ﻤرﺴﻰ ، 2،1ﺤﺴن ﺤﺴن ﺨﻠف ، 1أﺸرف ﻤﻬدى ﺸروﺒﻪ ، 1ﺤﺴن ﺤﺴن اﻟطﻨﺎﺤﻰ
1ﻗـﺴم ﻋﻠـوم اﻷﻏــذﻴﺔ -ﻛﻠﻴﺔ اﻟز ارﻋــﺔ -ﺠﺎﻤﻌــﺔ ﺒﻨﻬﺎ -ﻗﻠﻴوﺒﻴﺔ -ﻤﺼر
2ﻗﺴم ﻤﻴﻛرو -ﻨﺎﻨو ﺘﻛﻨوﻟوﺠﻰ -اﻟﺠﺎﻤﻌﺔ اﻟﺘﻛﻨﻴﻛﻴﺔ ﺒﺎﻟدﻨﻤﺎرك -اﻟدﻨﻤﺎرك
اﻟﻤﻠﺨص اﻟﻌرﺒﻰ
ﺘﻌﺘﺒر ﺘﻛﻨوﻟوﺠﻴﺎ ﻤﺘﻨﺎﻫﻴﺔ اﻟﺼﻐر ﻤن اﻟﺘﻛﻨوﻟوﺠﻴﺎت اﻟﻤﺒﺘﻛرة ﻓﻲ اﻵوﻨﺔ اﻷﺨﻴرة ﺤﻴث ﺘﻠﻌب دو ًار ﺠوﻫرﻴ ًﺎ ﻓﻰ ﺘﺤﺴﻴن ﺴﻼﻤﺔ وﺠودة اﻷﻏذﻴﺔ،
ﻛﻤﺎ ﻴﻌد إﻨﺘﺎج ﻋﺒوات ﻨﺎﻨو ذﻛﻴﺔ ﺘﺴﺘطﻴﻊ ﻛﺸف ﻓﺴﺎد اﻷﻏذﻴﺔ ﻫدﻓﺎً ﻫﺎﻤﺎً ﻷﺠﻬزة اﻟرﻗﺎﺒﺔ ﻋﻠﻰ اﻟﻐذاء وﻛذﻟك اﻟﻤﺴﺘﻬﻠك .ﻴﻌﺘﺒر اﻟﻤﺴﺘﺸﻌر اﻟﺤﻴوى و
ﻤﺴﺘﺸﻌر اﻷوﻛﺴﺠﻴن أﺤد ﺘطﺒﻴﻘﺎت اﻟﻨﺎﻨو ﺘﻛﻨوﻟوﺠﻰ اﻟﺘﻲ إﻛﺘﺴﺒﺎ ﻓﻲ اﻵوﻨﺔ اﻷﺨﻴرة اﻟﻛﺜﻴر ﻤن اﻹﻫﺘﻤﺎم ﻓﻲ ﻤﺠﺎﻝ ﺴﻼﻤﺔ اﻷﻏذﻴﺔ .ﺘﻀﻤن ﻫذا اﻟﺒﺤث
ﺘطوﻴر وﺘطﺒﻴق ﻨﻤوذﺠﻴن ﻤن وﺤدات اﻹﺴﺘﺸﻌﺎر اﻟﻘﺎدرة ﻋﻠﻰ اﻟﺘﻌﺎﻤﻝ ﻤﻊ ﻋﻴﻨﺎت اﻷﻏذﻴﺔ اﻟﻤﺨﺘﻠﻔﺔ،ﺤﻴث ﺘم اﺨﺘﺒﺎرﻫﻤﺎ ﻋﻠﻰ ﻛﻝ ﻤن ﻟﺤوم اﻟﺒﻘر و
1 ± 4م °ﻟﻤدة 11ﻴوﻤﺎ.ﻛﻤﺎ أﺠرى ﺘﻘدﻴر اﻟﺨﺼﺎﺌص اﻟطﺒﻴﻌﻴﺔ ،اﻟﻛﻴﻤﺎوﻴﺔ وﻛذﻟك أﺨﺘﺒﺎرات اﻟطزاﺠﺔ ،واﻟﺠودة ﻓﻴﻠﻴﻪ اﻟدﺠﺎج اﻟﻤﺨزﻨﺔ ﻋﻠﻰ
اﻟﻤﻴﻛروﺒﻴوﻟوﺠﻴﺔ ﻋﻠﻰ ﻋﻴﻨﺎت اﻟﻠﺤوم اﻟﻤﺨزﻨﺔ ﻋﻠﻰ ﻓﺘرات ﻤﻨﺘظﻤﺔ ﻤﺘزاﻤن ﻤﻊ وﺤدات اﻹﺴﺘﺸﻌﺎر .أظﻬرت ﻨﺘﺎﺌﺞ ﻫذﻩ اﻟدراﺴﺔ أن ﻫﻨﺎك ﺘﻐﻴر ﻟوﻨﻰ
ﻗوي ﻓﻲ اﻟﻤﺴﺘﺸﻌر اﻟﺤﻴوﯩﺤﻴث ﺘﺤوﻝ ﻤن اﻟﻠون اﻷﺨﻀر )طزاﺠﺔ اﻟﻠﺤوم( إﻟﻰ اﻟﻠون اﻷﺤﻤر اﻟﺒرﺘﻘﺎﻟﻰ )ﻓﺴﺎد اﻟﻠﺤوم( ﺒﻌد اﻟﻴوم اﻟﺘﺎﺴﻊ واﻟﻌﺎﺸرﻓﻲ ﻛﻝ
ﻤن ﻟﺤوم اﻟﺒﻘر و ﻓﻴﻠﻴﻪ اﻟدﺠﺎج ،ﻋﻠﻰ اﻟﺘواﻟﻲ .أﻴﻀﺎً أوﻀﺤت ﻨﺘﺎﺌﺞ اﻻﺨﺘﺒﺎرات أن ﻫﻨﺎﻛزﻴﺎدة ﻓﻲ ﻗﻴم ﻛﻝ ﻤﻨرﻗم اﻟﺤﻤوﻀﺔ ،اﻟﻨﻴﺘروﺠﻴن اﻟﻛﻠﻰ
اﻟﻤﺘطﺎﻴر ،ﺤﻤض اﻟﺜﻴوﺒﺎراﺒﻴوﺘرﻴك ﺨﻼﻝ ﻓﺘرات اﻟﺘﺨزﻴن .ﻛﻤﺎ ﻟوﺤظ أﻴﻀﺎ زﻴﺎدة ﻓﻲ ﻤﺤﺘوى اﻟﻌد اﻟﻛﻠﻰ ﻟﻠﺒﻛﺘﻴرﻴﺎ وﻛذﻟك اﻟﺒﻛﺘﻴرﻴﺎ اﻟﻤﺤﺒﺔ ﻟﻠﺒرودة .أﻴﻀﺎ
ﺒﻴﻨت اﻟدراﺴﺔ أن ﻤﺤﺘوى اﻻوﻛﺴﺠﻴن اﻟﻤﺘﺒﻘﻴﻔﻲ ﻋﻴﻨﺎت اﻟﻠﺤوم وﻓﻴﻠﻴﻪ اﻟدﺠﺎج اﻟﻤﺨزﻨﺔ إﻨﺨﻔض ﺒﺸﻛﻝ ﻤﺴﺘﻤر ﺨﻼﻝ ﻓﺘرات اﻟﺘﺨزﻴن .ﻫذﻩ اﻟﻨﺘﺎﺌﺞ
اﻟﻤﺘﺤﺼﻝ ﻋﻠﻴﻬﺎ ﺘﻘدم دﻟﻴﻼ واﻋداً ﻹﻤﻛﺎﻨﻴﺔ ﺘطﺒﻴق ﻛﻝ ﻤن اﻟﻤﺴﺘﺸﻌر اﻟﺤﻴوي وﻤﺴﺘﺸﻌر اﻷوﻛﺴﺠﻴن ﻟﻠﻛﺸف اﻟﺴرﻴﻊ ﻋن ﻓﺴﺎد اﻟﻠﺤوم أﺜﻨﺎء اﻟﺘداوﻝ
واﻟﺘﺨزﻴﻨﻤن أﺠﻝ ﺘﺄﻤﻴن ﺴﻼﻤﺔ اﻷﻏذﻴﺔ.
Nano-Biotechnology and Bio-systems Engineering 14
View publication stats
Das könnte Ihnen auch gefallen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (587)
- Gantt Chart Pertamina CilacapDokument1 SeiteGantt Chart Pertamina CilacapAzhar AdityaNoch keine Bewertungen
- Modeling of Extraction of Using Supercritical CO in Packed Bed ExtractorDokument9 SeitenModeling of Extraction of Using Supercritical CO in Packed Bed ExtractorAzhar AdityaNoch keine Bewertungen
- FP214 - Camera ReadyDokument5 SeitenFP214 - Camera ReadyAzhar AdityaNoch keine Bewertungen
- Milliequivalents, Millimoles, and Milliosmoles ExplainedDokument17 SeitenMilliequivalents, Millimoles, and Milliosmoles ExplainedMuhammad SafdarNoch keine Bewertungen
- Beda Rambut Orang Dewasa Vs AnakDokument13 SeitenBeda Rambut Orang Dewasa Vs AnakAzhar AdityaNoch keine Bewertungen
- We Are Intechopen, The World'S Leading Publisher of Open Access Books Built by Scientists, For ScientistsDokument31 SeitenWe Are Intechopen, The World'S Leading Publisher of Open Access Books Built by Scientists, For ScientistsAzhar AdityaNoch keine Bewertungen
- Alkalinity ConversionsDokument1 SeiteAlkalinity Conversionsahmedmagdi2009Noch keine Bewertungen
- Gantt Chart CilacapDokument1 SeiteGantt Chart CilacapAzhar AdityaNoch keine Bewertungen
- Brimac Char Product SpecificationDokument1 SeiteBrimac Char Product SpecificationAzhar AdityaNoch keine Bewertungen
- Biosensors: A Tutorial Review: IEEE Potentials April 2006Dokument13 SeitenBiosensors: A Tutorial Review: IEEE Potentials April 2006Azhar AdityaNoch keine Bewertungen
- Characteristics of Drug Substances in Oily Solutions. Drug Release Rate, Partitioning and SolubilityDokument11 SeitenCharacteristics of Drug Substances in Oily Solutions. Drug Release Rate, Partitioning and SolubilityAzhar AdityaNoch keine Bewertungen
- 1 Algal Culture Media1Dokument3 Seiten1 Algal Culture Media1ssaygun100% (1)
- Article Review Form Name: Azhar Aditya Rahman Student Number: 1506675756 Faculty & Class: Faculty of Engineering, MPK-B.Inggris 18 (S.204)Dokument2 SeitenArticle Review Form Name: Azhar Aditya Rahman Student Number: 1506675756 Faculty & Class: Faculty of Engineering, MPK-B.Inggris 18 (S.204)Azhar AdityaNoch keine Bewertungen
- Sourcesofcelluloseandtheirapplications AreviewDokument21 SeitenSourcesofcelluloseandtheirapplications AreviewAzhar AdityaNoch keine Bewertungen
- Recycle 1Dokument23 SeitenRecycle 1kiny810% (1)
- Indonesia 2017 Salary GuideDokument48 SeitenIndonesia 2017 Salary Guidediki100% (2)
- Oleochemical Process Flow DiagramDokument1 SeiteOleochemical Process Flow DiagramAzhar Aditya100% (2)
- Manual CSTR v31 FinalDokument42 SeitenManual CSTR v31 FinalAzhar AdityaNoch keine Bewertungen
- Sbe Uisc 2017 - Staff Recruitment FormDokument8 SeitenSbe Uisc 2017 - Staff Recruitment FormAzhar AdityaNoch keine Bewertungen
- Short Form ResumeDokument1 SeiteShort Form ResumeAzhar AdityaNoch keine Bewertungen
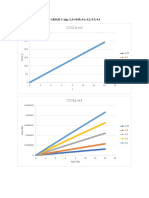
- CCO2, G Vs T: VARIASI C Alga, L, 0 0.05 0.1 0.2 0.3 0.4Dokument5 SeitenCCO2, G Vs T: VARIASI C Alga, L, 0 0.05 0.1 0.2 0.3 0.4Azhar AdityaNoch keine Bewertungen
- Sbe Uisc 2017 - Staff Recruitment FormDokument8 SeitenSbe Uisc 2017 - Staff Recruitment FormAzhar AdityaNoch keine Bewertungen
- ICAOAOPall EXDokument174 SeitenICAOAOPall EXAzhar AdityaNoch keine Bewertungen
- Routefinder: Elstree (Egtr) To London Luton (Eggw) : 2 FixesDokument2 SeitenRoutefinder: Elstree (Egtr) To London Luton (Eggw) : 2 FixesMikeNoch keine Bewertungen
- Energy & Environmental Science: Accepted ManuscriptDokument11 SeitenEnergy & Environmental Science: Accepted ManuscriptAzhar AdityaNoch keine Bewertungen
- FAA Policy Letter PL-34Dokument3 SeitenFAA Policy Letter PL-34Azhar AdityaNoch keine Bewertungen
- New Doc 2017-03-30 PDFDokument8 SeitenNew Doc 2017-03-30 PDFAzhar AdityaNoch keine Bewertungen
- A Citizens Guide To Bioremediation PDFDokument2 SeitenA Citizens Guide To Bioremediation PDFAzhar AdityaNoch keine Bewertungen
- Biopharming and BeyondDokument30 SeitenBiopharming and BeyondAzhar AdityaNoch keine Bewertungen
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (894)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (399)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (73)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2219)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (265)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (119)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Moot CourtDokument7 SeitenMoot CourtsushmaNoch keine Bewertungen
- Eugenics in The United StatesDokument14 SeitenEugenics in The United StatesSnark Jacobs100% (1)
- FAQ: Product RegistrationDokument5 SeitenFAQ: Product RegistrationCalvin WangNoch keine Bewertungen
- Coffee Master Program Flashcards - QuizletDokument23 SeitenCoffee Master Program Flashcards - QuizletmuuayliiNoch keine Bewertungen
- Calculate Size of Transformer / Fuse / Circuit Breaker: Connected Equipment To TransformerDokument16 SeitenCalculate Size of Transformer / Fuse / Circuit Breaker: Connected Equipment To TransformerHari OM MishraNoch keine Bewertungen
- Nabertherm RHTH Tube Furnace SOPDokument4 SeitenNabertherm RHTH Tube Furnace SOPIyere PatrickNoch keine Bewertungen
- Laccase Mediator Pretreatment of Wheat StrawDokument15 SeitenLaccase Mediator Pretreatment of Wheat StrawMeyerling RandfordNoch keine Bewertungen
- Chapter 1: Abnormal Behavior in Historical ContextDokument22 SeitenChapter 1: Abnormal Behavior in Historical ContextEsraRamos100% (2)
- Fermenting For Health - Pip MagazineDokument2 SeitenFermenting For Health - Pip MagazinePip MagazineNoch keine Bewertungen
- Mitsubishi v500 VFD IB NA 0600065-F FR-V500-NA Instruction Manual-DetailedDokument221 SeitenMitsubishi v500 VFD IB NA 0600065-F FR-V500-NA Instruction Manual-DetailedMROstop.comNoch keine Bewertungen
- 6th Class EM All LessonsDokument33 Seiten6th Class EM All LessonsSathish PurushothamNoch keine Bewertungen
- Fundamentals of Plant BreedingDokument190 SeitenFundamentals of Plant BreedingDave SubiyantoNoch keine Bewertungen
- 1 6Dokument58 Seiten1 6Emiliano.Armando Aguilera.Vázquez100% (1)
- Ipao Program Flyer 17novDokument1 SeiteIpao Program Flyer 17novapi-246252391Noch keine Bewertungen
- KPI and Supplier Performance Scorecard ToolDokument7 SeitenKPI and Supplier Performance Scorecard ToolJayant Kumar JhaNoch keine Bewertungen
- Un Primer Acercamiento A La Escritura Científica: OPI LabDokument66 SeitenUn Primer Acercamiento A La Escritura Científica: OPI LabLolaNoch keine Bewertungen
- c19 Standard Operating ProceduresDokument36 Seitenc19 Standard Operating ProceduresahsNoch keine Bewertungen
- Unit Test-Unit 5-Basic TestDokument2 SeitenUnit Test-Unit 5-Basic Testphamleyenchi.6a8txNoch keine Bewertungen
- Yoga Nidra MethodDokument13 SeitenYoga Nidra MethodPrahlad Basnet100% (2)
- Sodium Chloride MSDSDokument5 SeitenSodium Chloride MSDSIbaharmovic LpuNoch keine Bewertungen
- Cash Flow Analysis: Restaurant Business PlanDokument44 SeitenCash Flow Analysis: Restaurant Business Plankavirao87Noch keine Bewertungen
- Review of Literature Review of LiteratureDokument31 SeitenReview of Literature Review of LiteratureeswariNoch keine Bewertungen
- Q A With Jaquelyn BurrerDokument3 SeitenQ A With Jaquelyn Burrerapi-480718823Noch keine Bewertungen
- Surgical Item - Price ListDokument2 SeitenSurgical Item - Price ListRamnish MishraNoch keine Bewertungen
- NTFPP-Module 3 Microwave Processing of Foods - AjitKSinghDokument12 SeitenNTFPP-Module 3 Microwave Processing of Foods - AjitKSinghKeshav RajputNoch keine Bewertungen
- A Humanistic Approach To Medical PracticeDokument3 SeitenA Humanistic Approach To Medical PracticeFilipos ConstantinNoch keine Bewertungen
- NCM 117 Rle NAME: Guerzo, Danniel Dave Y. DATE/TIME: March 9, 2023 Section/Year Level: BSN 3B Group No:10 CIDokument2 SeitenNCM 117 Rle NAME: Guerzo, Danniel Dave Y. DATE/TIME: March 9, 2023 Section/Year Level: BSN 3B Group No:10 CISherlyn Miranda GarcesNoch keine Bewertungen
- Ganga Pollution CasesDokument3 SeitenGanga Pollution CasesRuchita KaundalNoch keine Bewertungen
- HRLM - Catalogue # Ex Apparatus - AC-Z Series Explosion Proof Plug and ReceptaclesDokument2 SeitenHRLM - Catalogue # Ex Apparatus - AC-Z Series Explosion Proof Plug and Receptaclesa wsNoch keine Bewertungen
- Deltair BrochureDokument4 SeitenDeltair BrochureForum PompieriiNoch keine Bewertungen