Beruflich Dokumente
Kultur Dokumente
Soluition Charpter 2 PDF
Hochgeladen von
Bryan de Barros0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
33 Ansichten3 SeitenOriginaltitel
soluition charpter 2.pdf
Copyright
© © All Rights Reserved
Verfügbare Formate
PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
33 Ansichten3 SeitenSoluition Charpter 2 PDF
Hochgeladen von
Bryan de BarrosCopyright:
© All Rights Reserved
Verfügbare Formate
Als PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 3
EMA 4324 Problem Set 3a
2.3 Assuming standard states for all reactants and products,
determine the spontaneous direction of the following reactions
by calculating the cell potential (sic):
[a] Cu + 2HCl = CuCl2 + H2 [Cu + 2H+ = Cu+2 + H2]
*****************************************************
Jones is really asking for a calculation of cell voltage!!
[1] Cu+2 + 2e-= Cu, Eo(Cu+2/Cu) = 0.342 v;
[2] 2H+ + 2e-= H2, Eo(H+/H2) = 0.000 v
Reaction [a] proceeds spontaneously in reverse to what is written...
Vcell = 0.342v, reaction [1] proceeds as written [reduction]; reaction
[2] proceeds in reverse [oxidation] the reaction [written as a
reduction reaction] with the more positive potential will proceed as
the reduction reaction in a cell with the less positive [more
negative] reduction reaction proceeding as an oxidation [reduction
in reverse].
[b] Fe + 2HCl = FeCl2 + H2; Fe + 2H+= Fe+2 + H2
Fe+2 + 2e- = Fe, Eo(Fe+2/Fe) = -0.440v [by insp., oxidation]
2H+ + 2e- = H, Eo(H+/H2) = 0.00 [by inspection, reduction]
Vcell = 0.000 - [-0.440] = 0.440 and Fe + 2H + = Fe+2 + H2
[c] 2AgNO3 + Fe = Fe(NO3)2 + 2Ag
Ag+ + 1e- = Ag, Eo(Ag+/Ag) = 0.799v
Fe+2 + 2e- = Fe, Eo(Fe+2/Fe) = -0.440
and Fe + 2Ag+ = Fe+2 + 2Ag, Ecell = 0.799 - (-0.440) = 1.239v
[d] Ag + FeCl3 = FeCl2 + AgCl
Ag+ + 1e- = Ag, Eo(Ag+/Ag) = 0.799v
Fe+3+ 1e- = Fe+2, Eo(Fe+3/Fe+2) = 0.771v
Fe+2 + Ag+ = Fe+3 + Ag, Ecell = 0.799 - 0.771 = 0.028v
[e] 2Al + 3ZnSO4 = Al2(SO4)3 + 3Zn
Zn+2 + 2e- = Zn, Eo(Zn+2/Zn) = -0.763v
Al+3 + 3e- = Al, Eo(Al+3/Al) = -1.662v
2Al + 3Zn+2 = 2Al+3 + 3Zn, Ecell = -0.763 - (-1.662) = 0.899v
2.5 Write the electrochemical half-cell reactions for oxidation and
reduction during uniform corrosion in the following:
[a] aluminum in air-free sulfuric acid
*****************************************************
Al+3 + 3e- = Al [-1.662 v, oxidation];
H+ + 1e- = 1/2H2[0.00 v, reduction]
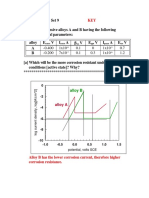
[b] iron in air-free ferric sulfate solution
*****************************************************
Fe+2 + 2e- = Fe [-0.440, oxidation]
Fe+3 + 1e- = Fe+2 [+0.771, reduction]
SO4-2 + 6e- +8H+ = S + 4H20 [+0.357, depends.....]
[c] carbon steel in aerated seawater [careful here...]
*****************************************************
Fe+2 + 2e- = Fe [-0.440, oxidation]
O2 + 2H2O + 4e- = 4OH- [0.401, reduction]
H+ + 1e- = 1/2H2 [0.00 v, depends....]
Others???
[d] zinc-tin alloy in an oxygen saturated solution of cupric chloride,
stannic chloride and hydrochloric acid
****************************************************
Zn+2 + 2e- = Zn [-0,763, oxidation]
O2 + 2H2O + 4e- = 4OH- [0.401, reduction]
Cu+2 + 2e- = Cu [+0.337, probable reduction]
Cu+2 + 1e- = Cu+1 [+0.153, depends...]
Sn+4 + 2e- = Sn+2 [+0.15, depends]
[e] copper in deaerated seawater [again, be careful...]
*****************************************************
Cu+2 + 2e- = Cu [+0.337, reduction!!!]
H+ + 1e- = 1/2H2 [0.00 v, oxidation!!!]
2.6 Copper immersed in concentrated pure hydrochloric acid is observed
to corrode rapidly with the evolution of gas bubbles. Is this contrary
to the results of problem 2.3a? Calculate a cell potential [sic] to
prove your answer, making any necessary assumptions.
******************************************************
According to the chemical [CRC] handbook, the concentration of
“ pure” HCl is 12.0 M. Assuming unit activity coefficients [poor
assumption], pH = -log10[12] = -1.08. Since E(H+/H2) = -0.0592pH,
E(H+/H2) = E(Cu+2/Cu) = 0.064 volts.
E(Cu+2/Cu) = 0.064 = 0.334 - [0.0592/2]log[1/[Cu+2]]
[Cu+2] = 7.56x10-10M!!!!!
Das könnte Ihnen auch gefallen
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- Astm - D573.25993Dokument6 SeitenAstm - D573.25993Bryan de BarrosNoch keine Bewertungen
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Astm - D4400.39339Dokument4 SeitenAstm - D4400.39339Bryan de BarrosNoch keine Bewertungen
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Vulcanized Rubber and Thermoplastic Elastomers - Tension: Standard Test Methods ForDokument14 SeitenVulcanized Rubber and Thermoplastic Elastomers - Tension: Standard Test Methods ForPaulo Venicio Alves Vieira100% (1)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- Astm - D1475.12152Dokument4 SeitenAstm - D1475.12152Bryan de BarrosNoch keine Bewertungen
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- Astm - D624.6776Dokument9 SeitenAstm - D624.6776Bryan de BarrosNoch keine Bewertungen
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- Astm G58.25944Dokument8 SeitenAstm G58.25944Bryan de BarrosNoch keine Bewertungen
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- Astm - G168.12911Dokument10 SeitenAstm - G168.12911Bryan de BarrosNoch keine Bewertungen
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Astm - E2180.29580Dokument4 SeitenAstm - E2180.29580Bryan de BarrosNoch keine Bewertungen
- Vulcanized Rubber and Thermoplastic Elastomers - Tension: Standard Test Methods ForDokument14 SeitenVulcanized Rubber and Thermoplastic Elastomers - Tension: Standard Test Methods ForPaulo Venicio Alves Vieira100% (1)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Astm D2196-10Dokument5 SeitenAstm D2196-10Bryan de BarrosNoch keine Bewertungen
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Astm - D2196.12408Dokument5 SeitenAstm - D2196.12408Bryan de BarrosNoch keine Bewertungen
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- Hydrogen Permeation Barriers - Basic Requirements, Material Selection, Deposition Methods, and Quality EvaluationDokument7 SeitenHydrogen Permeation Barriers - Basic Requirements, Material Selection, Deposition Methods, and Quality EvaluationBryan de BarrosNoch keine Bewertungen
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Astm - G8.12737Dokument9 SeitenAstm - G8.12737Bryan de BarrosNoch keine Bewertungen
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- Preparing, Cleaning, and Evaluating Corrosion Test SpecimensDokument8 SeitenPreparing, Cleaning, and Evaluating Corrosion Test SpecimensLê CôngNoch keine Bewertungen
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (344)
- Effect of Cathodic Potentials On The SCC Behavior of E690 Steel in Simulated SeawaterDokument10 SeitenEffect of Cathodic Potentials On The SCC Behavior of E690 Steel in Simulated SeawaterBryan de BarrosNoch keine Bewertungen
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- G95-87 Standard Test MethodDokument4 SeitenG95-87 Standard Test MethodandresjypNoch keine Bewertungen
- Astm - D2805.16103Dokument6 SeitenAstm - D2805.16103Bryan de Barros100% (2)
- Techniques To Measure Hydrogen Content in SS 304LDokument30 SeitenTechniques To Measure Hydrogen Content in SS 304LBryan de BarrosNoch keine Bewertungen
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- Effect of Cathodic Potentials On The Hydrogen Embrittlement Susceptibility of 10Ni5CrMo SteelDokument15 SeitenEffect of Cathodic Potentials On The Hydrogen Embrittlement Susceptibility of 10Ni5CrMo SteelBryan de BarrosNoch keine Bewertungen
- StrainGage MeasurementDokument37 SeitenStrainGage MeasurementfifafifaNoch keine Bewertungen
- Assessing Atmospheric Corrosion of Metal by A Novel Electrochemical Sensor Combining With A Thin Insulating Net Using Electrochemical Noise TechniqueDokument19 SeitenAssessing Atmospheric Corrosion of Metal by A Novel Electrochemical Sensor Combining With A Thin Insulating Net Using Electrochemical Noise TechniqueBryan de BarrosNoch keine Bewertungen
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- A Study of Strain and Deformation Measurement Using The Arduino Microcontroller and Strain Gauges DevicesDokument7 SeitenA Study of Strain and Deformation Measurement Using The Arduino Microcontroller and Strain Gauges DevicesDavid VilcaNoch keine Bewertungen
- Atmospheric Corrosion Sensor Based On Strain MeasurementDokument21 SeitenAtmospheric Corrosion Sensor Based On Strain MeasurementBryan de BarrosNoch keine Bewertungen
- NI Strain Gauge TutorialDokument12 SeitenNI Strain Gauge TutorialViki Hari Fitrianto100% (3)
- Solution Charpter 4 - 2Dokument5 SeitenSolution Charpter 4 - 2CANDYNoch keine Bewertungen
- Astm - G33.34087 - 2015Dokument3 SeitenAstm - G33.34087 - 2015Bryan de BarrosNoch keine Bewertungen
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- Soluition Charpter 3 - 3Dokument1 SeiteSoluition Charpter 3 - 3Bryan de BarrosNoch keine Bewertungen
- Soluition Charpter 5 - 1Dokument3 SeitenSoluition Charpter 5 - 1Bryan de BarrosNoch keine Bewertungen
- Soluition Charpter 6 - 1Dokument4 SeitenSoluition Charpter 6 - 1CANDYNoch keine Bewertungen
- Assimilable Organic Carbon Formation During Ultraviolet - Hydrogen Peroxide Advanced Oxidation Treatment of Surface WaterDokument207 SeitenAssimilable Organic Carbon Formation During Ultraviolet - Hydrogen Peroxide Advanced Oxidation Treatment of Surface WaterMonicaRossetimNoch keine Bewertungen
- SECTION 01010 Summary of WorkDokument14 SeitenSECTION 01010 Summary of Workabdullah amanullahNoch keine Bewertungen
- P-Block 15 To 16 GroupDokument38 SeitenP-Block 15 To 16 GroupBharti GoelNoch keine Bewertungen
- Nippon SteelDokument7 SeitenNippon SteelAnonymous 9PIxHy13Noch keine Bewertungen
- Essential and Non-Essential Variables For WPS - PQRDokument4 SeitenEssential and Non-Essential Variables For WPS - PQRNatarajan MurugesanNoch keine Bewertungen
- Carbo Graphite Grade JP 1300: Properties ofDokument1 SeiteCarbo Graphite Grade JP 1300: Properties ofMykmen SánchezNoch keine Bewertungen
- Kenya Final Report 2020Dokument93 SeitenKenya Final Report 2020macatahkNoch keine Bewertungen
- Selecting Amines For Sweetening Units PDFDokument9 SeitenSelecting Amines For Sweetening Units PDFGui Do EspNoch keine Bewertungen
- Rubber World HNBR Article - Therban AT PDFDokument9 SeitenRubber World HNBR Article - Therban AT PDFAnusha Rajagopalan100% (1)
- Thermal Conductivity of Liquids: Ka X T TDokument34 SeitenThermal Conductivity of Liquids: Ka X T TShiv KkNoch keine Bewertungen
- Steam and Cooling Water Saving in Sugar Factory by The Use of Phase Change MaterialsBDokument8 SeitenSteam and Cooling Water Saving in Sugar Factory by The Use of Phase Change MaterialsBParthosarothy K MukherjiNoch keine Bewertungen
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- Preventing Slips, Trips and Falls at Work: June 2017Dokument15 SeitenPreventing Slips, Trips and Falls at Work: June 2017Waseem RofaelNoch keine Bewertungen
- Mark Scheme (Results) January 2007: GCE Chemistry (6244/01)Dokument23 SeitenMark Scheme (Results) January 2007: GCE Chemistry (6244/01)Ajmaeen Mahtab GalibNoch keine Bewertungen
- 730al 370-Automotive LRDokument14 Seiten730al 370-Automotive LRlongNoch keine Bewertungen
- 1.8 Non Destructive Testing DisciplineDokument1 Seite1.8 Non Destructive Testing DisciplineTechie ndtNoch keine Bewertungen
- Taper AssemblyDokument36 SeitenTaper Assemblyויליאם סן מרמיגיוסNoch keine Bewertungen
- Mirror RadiusDokument14 SeitenMirror RadiusalphatoolsNoch keine Bewertungen
- Surface Plasmon Resonance Spectroscopy - SPRDokument15 SeitenSurface Plasmon Resonance Spectroscopy - SPRKayshu GroverNoch keine Bewertungen
- Oberbeck ConvectionDokument19 SeitenOberbeck ConvectionNAGARAJUNoch keine Bewertungen
- Lab Report8Dokument5 SeitenLab Report8wanjiaNoch keine Bewertungen
- Article WJPR 1405426296Dokument25 SeitenArticle WJPR 1405426296Joel Morales PerezNoch keine Bewertungen
- BioDoor BrochureDokument6 SeitenBioDoor BrochureJob ANoch keine Bewertungen
- Mekanika BatuanDokument38 SeitenMekanika BatuanKRISNA ADITYA SITANGGANGNoch keine Bewertungen
- Epa Calculation For TowersDokument11 SeitenEpa Calculation For Towersfadil3m2422Noch keine Bewertungen
- 1-s2.0-S0301479717307521-Main - PDF Eliminacin de Metales Por NanofiltracionDokument6 Seiten1-s2.0-S0301479717307521-Main - PDF Eliminacin de Metales Por NanofiltracionJaime CazaNoch keine Bewertungen
- An Overview of Silicone SoftenerDokument3 SeitenAn Overview of Silicone SoftenersiliconemanNoch keine Bewertungen
- Cleaning Supplies Tools Chemicals and EquipmentsDokument97 SeitenCleaning Supplies Tools Chemicals and EquipmentsErlyn Alcantara100% (2)
- Utkarsh ChromatographyDokument17 SeitenUtkarsh ChromatographyUtkarsh Pratap SinghNoch keine Bewertungen
- Summative DRRR q2Dokument2 SeitenSummative DRRR q2roberto limNoch keine Bewertungen
- ANSYSd BListDokument114 SeitenANSYSd BListAnonymous nfHBPXz178Noch keine Bewertungen
- AP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeVon EverandAP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeBewertung: 5 von 5 Sternen5/5 (1)
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincVon EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincBewertung: 3.5 von 5 Sternen3.5/5 (137)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactVon EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactBewertung: 5 von 5 Sternen5/5 (5)
- Guidelines for Chemical Process Quantitative Risk AnalysisVon EverandGuidelines for Chemical Process Quantitative Risk AnalysisBewertung: 5 von 5 Sternen5/5 (1)
- Sodium Bicarbonate: Nature's Unique First Aid RemedyVon EverandSodium Bicarbonate: Nature's Unique First Aid RemedyBewertung: 5 von 5 Sternen5/5 (21)
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideVon EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNoch keine Bewertungen