Beruflich Dokumente
Kultur Dokumente
Desulfurization of Liquid Steel Containing Aluminum or Silicon With Lime PDF
Hochgeladen von
Rasul BzOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Desulfurization of Liquid Steel Containing Aluminum or Silicon With Lime PDF
Hochgeladen von
Rasul BzCopyright:
Verfügbare Formate
Desulfurization of Liquid Steel Containing
Aluminum or Silicon with Lime
R. J . FRUEHAN
The r e a c t i o n m e c h a n i s m s for the d e s u l f u r i z a t i o n of i r o n c o n t a i n i n g f r o m 0.04 to 1.5 pct
a l u m i n u m or 1.1 to 3.7 pct s i l i c o n by CaO at 1600~ have b e e n e x a m i n e d . The d e s u l f u r i z a -
tion of Fe-A1 by Ca(9 is c o n s i d e r a b l y f a s t e r than that of F e - S i . T h e b a s i c d i f f e r e n c e b e -
t w e e n the two p r o c e s s e s is that Fe-A1 a l l o y s can be d e s u l f u r i z e d by the f o r m a t i o n of
A1203, w h e r e a s for F e - S i m e l t s it is n e c e s s a r y to f o r m Ca2SiO4. T h e r a t e of d e s u l f u r i z a -
tion of F e - S i a l l o y s by Ca(3 is c o n t r o l l e d by the f o r m a t i o n of the g a s e o u s i n t e r m e d i a t e s ,
SiS and S, and is the s a m e as that for d e s u l f u r i z a t i o n in v a c u u m . T h e r a t e of d e s u l f u r i z a -
tion of F e - A 1 m e l t s is fast, and is a p p a r e n t l y c o n t r o l l e d by the diffusion of s u l f u r to the
liquid m e t a l - C a O i n t e r f a c e . E x p e r i m e n t s w e r e a l s o conducted to d e m o n s t r a t e that s u l f u r
could be t r a n s f e r r e d to CaO by the g a s e o u s i n t e r m e d i a t e s SiS and S.
THE o v e r a l l d e s u l f u r i z a t i o n r e a c t i o n s of F e - C , Si-S alloys at 1400~ About 8 g of the a l l o y was con-
F e - S i , and Fe-A1 a l l o y s with CaO can be r e p r e s e n t e d t a i n e d in a ZrO2 c r u c i b l e (1.7 c m ID) and 1 g of g r a n u -
by l a r CaO p a r t i c l e s w e r e s u s p e n d e d above the m e l t in a
S + CaO + C = CaS + CO [1]
p l a t i n u m b a s k e t at a p r e d e t e r m i n e d d i s t a n c e (x) of 0.3,
0.8, or 2.5 c m above the m e l t . The s a m p l e s w e r e
S + 2CaO + ~Si = CaS + ~CaeSiO4 [2] heated in a v e r t i c a l tube f u r n a c e (4.0 c m ID) with a top
flow of A r at 1.5 1//min. T h e l i m e p a r t i c l e s , p r e v i o u s l y
3S + 4CaO + 2A1 = 3CaS + CaA1204. [3]
c a l c i n e d at 1500~ w e r e within the s i z e r a n g e f r o m
As d i s c u s s e d in p r e v i o u s p u b l i c a t i o n s , ~-3 the r e a c t i o n s 0.5 to 1 m m and had an i n t e r n a l p o r e a r e a of 0.3 m2//g.
as w r i t t e n r e p r e s e n t the o v e r a l l r e a c t i o n ; to u n d e r - T h e i n i t i a l s u l f u r c o n t e n t of the m e l t was 0.1 pct and
s t a n d the d e s u l f u r i z a t i o n m e c h a n i s m the i n d i v i d u a l the Si c o n t e n t v a r i e d up to 3.8 pct. S e p a r a t e d u p l i -
s t e p w i s e r e a c t i o n s should be c o n s i d e r e d . cate e x p e r i m e n t s w e r e a l s o conducted in which t h e r e
F o r e x a m p l e , Eq. [2] has a m i n i m u m of t h r e e p h a s e s was no CaO p r e s e n t , with the d i s t a n c e by which the
p r e s e n t , a s s u m i n g that the CaO is i m p r e g n a t e d with c r u c i b l e wall extended above the m e l t b e i n g the s a m e
liquid s l a g . F o r this case t h e r e is only a line of as the d i s t a n c e above the m e l t at which the CaO was
contact b e t w e e n the t h r e e p h a s e s which is not f a v o r - suspended.
able for r a p i d d e s u l f u r i z a t i o n . It has b e e n p r o p o s e d In a s e c o n d s e r i e s of e x p e r i m e n t s 5000 g of an F e - C -
that an i m p o r t a n t step in the d e s u l f u r i z a t i o n of F e - S i S, Fe-A1-S, or F e - S i - S alloy c o n t a i n e d i n a 9.6 c m ID
m e l t s is the f o r m a t i o n of the v a p o r s p e c i e s SiS and z i r c o n i a c r u c i b l e was heated in a n induction f u r n a c e
that the CaO m e r e l y acts as a getter for the s u l f u r - in a p r o t e c t i v e a t m o s p h e r e of flowing A r . A f t e r the
b e a r i n g g a s e s . It is one of the p u r p o s e s of the p r e s e n t t e m p e r a t u r e r e a c h e d 1600~ a s a m p l e of the m e l t was
work to p r o v e this h y p o t h e s i s . The d e s u l f u r i z a t i o n of taken by s u c t i o n into a s i l i c a tube, a f t e r which pow-
F e - A 1 m e l t s m u s t p r o c e e d by m e a n s of a n o t h e r type d e r e d l i m e (<0.1 mm) was added. U s u a l l y 125 g of
of m e c h a n i s m s i n c e t h e r e is no s i g n i f i c a n t a l u m i n u m - CaO was used; however, in s e v e r a l c a s e s d u p l i c a t e
s u l f u r v a p o r s p e c i e s . The m e c h a n i s m of d e s u l f u r i z a - e x p e r i m e n t s w e r e conducted in which 80 or 160 g of
tion of Fe-A1 alloys with CaO will a l s o be e x a m i n e d . CaO w e r e u s e d . In a l l c a s e s the l i m e was s u f f i c i e n t
It has b e e n r e c e n t l y p r o p o s e d by T u r k d o g a n 4 that to cover the e n t i r e s u r f a c e of the m e l t , and t h e r e was
liquid s t e e l c a n be d e s u l f u r i z e d e f f i c i e n t l y by i n j e c t - no d i f f e r e n c e in the r e s u l t s when the a m o u n t of CaO
i n g CaO with A1 or Si. By doing so a l a r g e CaO7 used was v a r i e d within this r a n g e . S a m p l e s w e r e t a k e n
l i q u i d - m e t a l s u r f a c e a r e a is p r o v i d e d and a l o c a l l y at r e g u l a r i n t e r v a l s and a n a l y z e d for s u l f u r and
high A1 or Si content at the i n t e r f a c e will e x i s t , both a l u m i n u m , s i l i c o n , or c a r b o n . E x p e r i m e n t s w e r e a l s o
of which a r e f a v o r a b l e for r a p i d d e s u l f u r i z a t i o n . The made with F e - A 1 - S a l l o y s u n d e r v a c u u m (10 Pa) with
p r e s e n t work was u n d e r t a k e n to d e t e r m i n e whether no CaO addition; the p r o c e d u r e used has b e e n d e s c r i b e d
the d e s u l f u r i z a t i o n r e a c t i o n s a r e fast enough to make previously.~
this p r o p o s e d p r o c e s s f e a s i b l e .
RESULTS AND DISCUSSION
EXPERIMENTAL
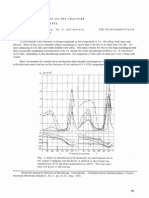
The r e s u l t s o b t a i n e d with the CaO s u s p e n d e d 0.8 c m
T o d e m o n s t r a t e the e f f e c t i v e n e s s of s u l f u r t r a n s f e r
above the s u r f a c e of the 8 g m e l t a r e shown in F i g . 1.
f r o m l i q u i d - i r o n a l l o y s to CaO via the gas phase, a A s i m i l a r r e l a t i o n s h i p was o b t a i n e d with the Ca(3 for
s e r i e s of e x p e r i m e n t s was made with F e - 3 . 0 pct C- each of the other d i s t a n c e s above the m e l t . T h e r a t e
R. J. FRUEHAN is with U.S. Steel Research Laboratory, Monroe- of d e s u l f u r i z a t i o n i n c r e a s e d with i n c r e a s i n g Si c o n t e n t
ville, PA 15146 of the m e l t . It is also i m p o r t a n t to note that with 3.8
Manuscript submitted October 5, 1977. pct Si in the m e t a l the r a t e of d e s u l f u r i z a t i o n was
ISSN 0360-2141/78/0612-0287500.75/0
METALLURGICALTRANSACTIONS B 9 1978AMERICANSOCIETYFOR METALSAND VOLUME 9B, JUNE 1978-287
THE METALLURGICALSOCIETYOF AIME
In F i g . 2 the slopes of the line for In (pet S / p c t S o ) v s
Lime a r e pioLted vs l / x ; the expected b e h a v i o r is ob-
served.
It is p o s s i b l e to e s t i m a t e the r a t e of d e s u l f u r i z a t i o n
f r o m the t h e r m o d y n a m i c s of F e - C - S i - S alloys, the
f r e e e n e r g y of f o r m a t i o n of SiS, and a n e s t i m a t e d value
of rn. At 1400~ the e q u i l i b r i u m c o n s t a n t K for Eq.
[9] is 0.496
s + s i : sis(g) [9]
-0.8 Psis
K=
a s i f s pct S
% F o r an alloy c o n t a i n i n g 3.8 pct Si and 3 pct C the
3.8O% Si
a c t i v i t y coefficient of s u l f u r (fs) is 5 and the a c t i v i t y
-,,2 [ I l
200 400 600 of s i l i c o n (asi) is 0.001.1 T h e r e f o r e the e q u i l i b r i u m
TIME, rain p a r t i a l p r e s s u r e os SiS is given by
Fig. 1--Desulfurization of Fe-3.0 pct C-X alloys at 1400~ by
vapor transport to porous lime granules suspended 0.8 em PSiS = 2.5 • 10 "a (wt pct S), a i m
above the melt. [10]
P s i s = 2.5 x 102 (wt pct S) Pa.
about the s a m e with or without the CaO at a d i s t a n c e
of 0.8 cm above the m e l t . T h e s e r e s u l t s s u g g e s t that It is p o s s i b l e to e s t i m a t e m by u s i n g Eq. [8]. The
the r a t e is c o n t r o l l e d p r i m a r i l y by diffusion of SiS value of D is e s t i m a t e d to be 3 c m 2 / s f r o m the data
v a p o r through the s t a g n a n t gas l a y e r over the m e l t . and equations given by H i r s c h f e l d e r , e t a l . 6 At a dis"
The r e a c t i o n of SiS with CaO on the pore w a i l s of Lance of x = 0.8 c m the c a l c u l a t e d v a l u e of k ' m is 7
the l i m e p a r t i c l e s may be r e p r e s e n t e d , for e x a m p l e , • 10 -5 s "I c o m p a r e d with the o b s e r v e d value of 4 • 10 -~
by s -z. The a g r e e m e n t is good c o n s i d e r i n g the u n c e r t a i n -
t i e s in the t h e r m o d y n a m i c s and the e s t i m a t i o n of m .
SiS(g) + 2CaO = CaS + ~Ca2SiO4 + ~ S i ( / ) . [4] I n p r e v i o u s work done in v a c u u m 1 it was found that
the r a t e of d e s u l f u r i z a t i o n of F e - S i - S alloys was p r i -
T h e e q u i l i b r i u m p a r t i a l p r e s s u r e of SiS for this r e a c - m a r i l y c o n t r o l l e d by a slow c h e m i c a l r e a c t i o n for the
t i o n at 1400~ is 2 • 10 "~ Pa, z'5 which is s e v e r a l o r d e r s f o r m a t i o n of SiS, the r a t e of which was 1/10 the r a t e
of m a g n i t u d e lower than that at the s u r f a c e of the a l - for f r e e v a p o r i z a t i o n of SiS. At 1400~ the r a t e con-
loys i n v e s t i g a t e d . T h e r e f o r e , the p a r t i a l p r e s s u r e of s t a n t for the f o r m a t i o n of SiS is 0.02 s -z. A p p r o x i m a t -
SiS at the CaO s u r f a c e can be a s s u m e d to be z e r o and ing m by D / x gives x = 25 /zm as the c r i t i c a l d i s t a n c e
the r a t e e q u a t i o n for d e s u l f u r i z a t i o n c a n be r e p r e s e n t e d b e t w e e n the lime p a r t i c l e s and the s u r f a c e of the m e l t
by
for a p p r o a c h to the m a x i m u m r a t e of d e s u l f u r i z a t i o n ;
d pct S 3200 A that is, the r a t e i n c r e a s e s as the d i s t a n c e between the
dt - RTW m PSiS [5] CaO p a r t i c l e and the m e l t d e c r e a s e s u n t i l that d i s t a n c e
is 25 tzm. T h e r e f o r e , in w e l l - s t i r r e d l i q u i d - F e alloys
where
c o n t a i n i n g s i l i c o n , the r a t e of d e s u l f u r i z a t i o n by l i m e in
A = s u r f a c e a r e a of the m e l t (2.27 cm2), d i r e c t contact with the m e l t will be c l o s e to that
R = gas c o n s t a n t , a c h i e v e d in v a c u u m . T h i s c o n c l u s i o n w i l l be f u r t h e r
W = weight of the m e l t (8 g), s u b s t a n t i a t e d by the r e s u l t s to follow.
T = a b s o l u t e t e m p e r a t u r e (K),
Psis = the e q u i l i b r i u m vapor p r e s s u r e of SiS which is
,=
p r o p o r t i o n a l to the s u l f u r content.
The i n t e g r a t e d f o r m of this equation is
ln Pct S = - k ' m t [6]
pct So 8 I
w h e r e k ' is a c o n s t a n t dependent on the p r e s s u r e of
SiS,
As shown in Appendix A the mass-transfer coeffi- K
cient, m, is expected to v a r y with the d i s t a n c e f r o m
the m e l t to the CaO and is given by 4
1 1 x
m m o
+
D
[7]
w h e r e m o is the m a s s - t r a n s f e r coefficient at the top
I I I
of the c r u c i b l e o r at the CaO p a r t i c l e s and D is the 1 2 3
i n t e r d i f f u s i v i t y . In the p r e s e n t work m o is r e l a t i v e l y 1
~. cm t
l a r g e and Eq. 7 r e d u c e s to
Fig. 2--Rate constant for the desulfurization of Fe-3.8 pet
D Si-3 pct C melts at 1400~ as a function of the reciprocal of
.~ : - . [8]
X the diffusion distance to lime particles above the melt.
288 V O L U M E 9B, J U N E 1 9 7 8 METALLURGICAL TRANSACTIONS B
T y p i c a l r e s u l t s obtained with 5000 g m e l t s at 1600~ 0,OOlB I |
with CaO in d i r e c t contact with F e - A 1 a l l o y s are g i v e n
in F i g . 3 for an initial sulfur content of 0.1 pct and in 3S + 2AI + 3CaO = AI20 3 + 3CaS
Fig. 4 for 0.031 pct i n i t i a l s u l f u r . The A1 content ~ EQUILIBRIUM
changed during the c o u r s e of the e x p e r i m e n t , partly b e -
0.0OB O 20 MINVTES
c a u s e of the r e a c t i o n i t s e l f but p r i m a r i l y b e c a u s e of
~ 9 120 MINUTES
oxygen pickup f r o m the a t m o s p h e r e . In the e x p e r i m e n t s
in w h i c h no CaO was added at a t m o s p h e r i c p r e s s u r e
or in v a c u u m , there was no a p p r e c i a b l e d e s u l f u r i z a -
Lion, indicating that there is no s i g n i f i c a n t v o l a t i l e a l u - u~
m i n u m - s u l f u r s p e c i e s as in the c a s e of s i l i c o n . 0.004
The i n i t i a l rate of d e s u l f u r i z a t i o n of F e - A 1 m e l t s
i s v e r y rapid. A s shown in F i g . 5, within l e s s than 20
rain the sulfur content was reduced below that for
e q u i l i b r i u m with A1203.
0.00"2
3S + 3CaO + 2A1 = 3CaS + A12Oa. [11]
FIowever, in g e n e r a l e v e n at long t i m e s , 120 min,
the s u l f u r content w a s s t i l l higher than that in e q u i l i - 4CaO + 2AI + 3S =
3CaS + CaAI204
b r i u m with CaA1204. 5 In s e v e r a l c a s e s the sulfur con- EQUILIBRIUM
tent of the m e t a l i n c r e a s e d at long t i m e s b e c a u s e the I I I I i
0.2 0.4 0.6
A1 content was d e c r e a s i n g . It appears that the r e a c -
AI, wt %
tion s e q u e n c e
F i g . 5 - - S u l f u r c o n t e n t s a s a f u n c t i o n of a l u m i n u m contents at
S + CaO = O + CaS [12] 20 and 120 rain.
2A1 + 3 0 = A1203 [13 ]
0.1
is v e r y rapid and goes to c o m p l e t i o n e a s i l y . The f o r -
0,06
0,1q
l I I I I
0.04
Al(initi~) /Ill fir~l
O 0.06 0.03
0,05 E] 0,60 0.04
9 0,14 0.0O 0.02
9 0.42 0.36
9 1,51 1.50
0,02
0.01
Si
'~ 0.01 O
u~ 0.006
0.004
0.005
0
3.70% Si
0.602
0.602 ~ 9I
0.601 I ~ "I ~ I
40 80 1120
o.601 I I I I I I
20 40 60 60 160 120 140 TIME, rain
TIME, rain F i g . 6 - - D e s u l f u r i z a t i o n of F e - S i a n d F e - C m e l t s by CaO a t
F i g . 3 - - D e s u l f u r i z a t i o n of F e - A 1 m e l t s w i t h C a O a t 1600~ 1600~ at atmospheric pressure.
0.04~ l l I l I I m a t i o n of the a l u m i n a t e is apparently m u c h s l o w e r
s i n c e this r e q u i r e s a s o l i d - s o l i d r e a c t i o n
AI(5 minuZe~l All~al~
002
D 0,09 0.04 A1203 + CaO = CaA1204 [14]
O 0,32 0,20
or the d i r e c t r e a c t i o n indicated by Eq. [3].
001
T h e i n i t i a l rate of d e s u l f u r i z a t i o n of F e - A 1 a l l o y s
by CaO is v e r y fast and is p o s s i b l y c o n t r o l l e d by the
0.606
diffusion of s u l f u r f r o m the bulk to the liquid m e t a l -
CaO s u r f a c e . The integrated f o r m of the rate equation
for m a s s - t r a n s f e r c o n t r o l i s given by
In pct S t - pct S e = - k t [15]
pct S o - pct S e
w h e r e h is the m u s s - t r a n s f e r c o e f f i c i e n t for an in-
d u c t i v e l y s t i r r e d m e l t . A s s u m i n g that the A1 c o n c e n -
o.ooi I I I I I I
20 40 60 80 100 120 140 tration at the s u r f a c e is a p p r o x i m a t e l y equal to that
TIME, min in the bulk and the o x y g e n potential is d e t e r m i n e d by
F i g . 4 - - D e s u l f u r i z a t i o n of F e - A I m e l t s w i t h C a O at 1600~ the A1 - AI~O~ e q u i l i b r i u m , it i s p o s s i b l e to c a l c u l a t e
METALLURGICAL TRANSACTIONS B VOLUME 9B, JUNE 1978-289
0 ' I I r r a t e c o n t r o l l i n g r e a c t i o n is the f o r m a t i o n of SiS
%.
AI (0,32- 0,04)
St + S -- SiS(g). [18]
SO (0,10-0,031)
1 --
The r e m a i n i n g r e a c t i o n s for d e s u l f u r i z a t i o n a r e fast
,:Z: 2P
and it is u n c l e a r what the exact r e a c t i o n s might be.
F o r e x a m p l e the r e a c t i o n might p r o c e e d by Eq. [4]
or a s follows:
=
%. SiS(g) + CaO : CaS + SiO(g) [19]
3-- ,% 8
%. SiO(g) + 2CaO + O = Ca2SiO4. [20]
X As d i s c u s s e d e a r l i e r , if the r a t e of d e s u l f u r i z a t i o n
4 I I of F e - S i a l l o y s is c o n t r o l l e d by the f o r m a t i o n of SiS,
o 4 8 the r a t e should be the s a m e as that o b s e r v e d in
TIME, rain v a c u u m . The r a t e e q u a t i o n d e r i v e d p r e v i o u s l y 1,2 is
Fig. 7--Desulfurization of Fe-A1 melts by CaO at 1600~ in
an inductively stirred melt. In pct S _ 100A (klaSi + k2)fst [21]
pcs% w
the e q u i l i b r i u m s u l f u r content (pet Se). U s i n g the where
a v e r a g e A1 c o n t e n t s the r e s u l t s a r e plotted in F i g . 7 /el = the r a t e c o n s t a n t for the f o r m a t i o n of SiS,
in a c c o r d a n c e with Eq. [15]. It should be noted that in /e2 = the r a t e p a r a m e t e r for f r e e v a p o r i z a t i o n of S,
g e n e r a l t h e r e is a dual diffusion p r o c e s s , with A1 a l s o A = the s u r f a c e a r e a of the melt,
diffusing to the s u r f a c e , but for this r o u g h a p p r o x i m a - W= the weight of the melt,
tion this was n e g l e c t e d . The r e s u l t s fit the e q u a t i o n asi = the a c t i v i t y of s i l i c o n .
o v e r a f a i r l y wide r a n g e of A1 contents; and the slope
of the l i n e , which is the m a s s - t r a n s f e r coefficient, F r o m the t h e r m o d y n a m i c p r o p e r t i e s for the alloys
a g r e e s within a f a c t o r of two with that c a l c u l a t e d f r o m given p r e v i o u s l y 2 the r a t e c o n s t a n t s d e r i v e d f r o m the
the e q u a t i o n s developed by M a c h l i n 7 for m a s s t r a n s f e r s l o p e s of the r a t e c u r v e s in F i g . 5 a r e plotted in Fig.
in an i n d u c t i v e l y s t i r r e d m e l t . T h i s a g r e e m e n t is 8 in a c c o r d a n c e with Eq. [21] along with the r e s u l t s of
r e a s o n a b l e c o n s i d e r i n g the a p p r o x i m a t i o n s made in the p r e v i o u s work o b t a i n e d in v a c u u m . 2 8-~ The p r e s e n t
calculations. r e s u l t s obtained at a t m o s p h e r i c p r e s s u r e with CaO
Although the r e s u l t s a r e i n s u f f i c i e n t to prove con- a r e in e x c e l l e n t a g r e e m e n t with those obtained under
c l u s i v e l y that the i n i t i a l r a t e of d e s u l f u r i z a t i o n of v a c u u m with no CaO. T h e r e f o r e the r a t e of d e s u l f u r i z a -
F e - A 1 a l l o y s by CaO in the p r e s e n t work is c o n t r o l l e d tion of F e - S i - S alloys by CaO is p r i m a r i l y c o n t r o l l e d
by the diffusion of s u l f u r to the s u r f a c e , this a s s u m p - by the r a t e of f o r m a t i o n of SiS and S, and the rate is
tion a p p e a r s r e a s o n a b l e and the r e s u l t s a r e c o n s i s t e n t the s a m e as that achieved in v a c u u m .
with this i n t e r p r e t a t i o n . In any case the r a t e is v e r y
f a s t and t h e r e is no e v i d e n c e of a slow c h e m i c a l r e a c - SUMMARY AND CONCLUSIONS
tion.
As shown in F i g . 6, the r a t e of d e s u l f u r i z a t i o n of T h e m e c h a n i s m s of d e s u l f u r i z a t i o n of s t e e l contain-
F e - S i a l l o y s is c o n s i d e r a b l y s l o w e r than that of Fe-A1 ing A1 o r Si by CaO have b e e n e x a m i n e d . F o r F e - S i
a l l o y s . F o r F e - S i a l l o y s the d e s u l f u r i z a t i o n r e a c t i o n a l l o y s the i m p o r t a n t step in the p r o c e s s is the f o r m a -
in which SiO2 is f o r m e d is not t h e r m o d y n a m i c a l l y tion of SiS(g). The r a t e of d e s u l f u r i z a t i o n is c o n t r o l l e d
f a v o r a b l e u n d e r the c o n d i t i o n s of the p r e s e n t work
2S + 2CaO + Si = SiOz + 2CaS. [16]
J
J
T h e e q u i l i b r i u m s u l f u r c o n t e n t for Eq. [16] at 1 pct Si t 9
J
is over 0.1 pct. 5 D e s u l f u r i z a t i o n by the f o r m a t i o n of J
f
CaSiO3 is a l s o not f a v o r a b l e , with e q u i l i b r i u m s u l f u r f
f
content b e i n g 0.07 pct at 1 pct Si. It is t h e r e f o r e f
p
n e c e s s a r y for Ca2SiO4 to f o r m a s i n d i c a t e d by Eq. [2],
for which the e q u i l i b r i u m s u l f u r c o n t e n t is about 0.001 J
J
pct at 1 pct Si. V
x /
It is difficult to conceive that the d e s u l f u r i z a t i o n VA //
m e c h a n i s m is by the d i r e c t r e a c t i o n given by Eq. [2] V o/f OHNO a ISHIDA
/ VACUO SEFIGAL
b e c a u s e it r e q u i r e s Si and S a t o m s in the m e t a l to col- v /
SEHGAL & MITCHELL
o /
lide s i m u l t a n e o u s l y with a CaO p a r t i c l e . It a l s o does -- V jl Mt FRUEHAN & TURKDOGAN
not s e e m likely that the m e c h a n i s m c o n s i s t s of a f o r - CoO O PRESENT WORK
(ATMOSPHERIC PRESSURE)
m a t i o n of SiO2 f i r s t , followed by the s o l i d - s o l i d r e a c - ~d
J
0
tion (
J I I I I J I I
SiO2 + 2CaO = Ca2SiO4. [17] 2 4 s 8 lo
~x lo 4
The following m e c h a n i s m i n v o l v i n g the v a p o r s p e c i e s Fig. 8--Desulfurization r a t e c o n s t a n t a s a f u n c t i o n of s i l i c o n
SiS is p r o p o s e d for the r e a c t i o n m e c h a n i s m . The slow activity at 1600~
2 9 0 - V O L U M E 9B, J U N E 1978 METALLURGICAL TRANSACTIONS B
by the r a t e of f o r m a t i o n of SiS and is the s a m e as that 6 I I T I I I
a c h i e v e d in v a c u u m when no CaO is p r e s e n t . T h e r a t e
of d e s u l f u r i z a t i o n of F e - A 1 with CaO is c o n s i d e r a b l y
5
f a s t e r , and the i n i t i a l r a t e in the p r e s e n t w o r k is
p r o b a b l y c o n t r o l l e d by the m a s s t r a n s f e r of s u l f u r to
the r e a c t i o n i n t e r f a c e . T h e b a s i c d i f f e r e n c e b e t w e e n
the d e s u l f u r i z a t i o n of F e - A 1 and F e - S i a l l o y s with CaO
is that f o r F e - A 1 a l l o y s s i g n i f i c a n t d e s u l f u r i z a t i o n can
o c c u r when the r e a c t i o n p r o d u c t is the s i m p l e oxide
A1203, for which the d i r e c t r e a c t i o n s a r e f a s t . F o r
F e - S i a l l o y s it is n e c e s s a r y to f o r m Ca2SiO4 to a c h i e v e
a p p r e c i a b l e d e s u l f u r i z a t i o n . T h e f o r m a t i o n of t h i s X = 0.8 cm
m o r e c o m p l e x oxide is slow and r e q u i r e d the u se of
the g a s e o u s i n t e r m e d i a t e s SiS and S f o r a f a s t r e a c - .~
X = 1,9cm
O
tion. Th e t r a n s f e r of s u l f u r to CaO by SiS and S was
d e m o n s t r a t e d in a s e p a r a t e s e t os e x p e r i m e n t s . T h e J I I I t i
t e c h n i q u e p r o p o s e d by T u r k d o g a n f o r d e s u l f u r i z i n g 2 4 6 8 10 12
s t e e l by the i n j e c t i o n of A1 and CaO with an i n e r t gas u, cm/s
(Ar fo r e x a m p l e , t h r o u g h a Q - B O P t u y e r e ) a p p e a r s Fig. A-2--Mass-transfer coefficient for Pb in flowing Ar at
ll00~
f e a s i b l e in that the k i n e t i c s of the r e a c t i o n a r e f a s t .
,sf , , , ,
APPENDIX A
u = 1.6 eml~
Gas-Phase Mass-Transfer Coefficient for
Crucible Geometry
Many experimental investigations of the rate of crn/s
1.0
chemical reactions involving liquid metals use an
experimental arrangement similar to the one employed
E
in part of the present study; that is, liquid metal in a
crucible supported in a vertical tube furnace with the
reacting or neutral gas flowing in the reaction tube.
Since in many cases gas-phase mass transfer may be
affecting the rate of the reaction, the gas-phase mass
transfer for this experimental geometry was investi-
gated. In a previous measurement of the rate of oxi-
0 u
:: .6 cm/s
I
dation of carbon by CO2 with a similar experimenta!
, I , I /
8 1.0 2.0
X. cm
Fig. A-3--Mass-transfer coefficientfor vaporizationof Pb in
He and Ar at II00~ as a functionof the distance X.
geometry, it was found that the mass transfer coeffi-
cient (m) was given approximately byz3
D
m = - [A-1]
x
d -- ~ X = 0.8 cm
where
D = interdiffusivity,
x = the d i s t a n c e f r o m the top of the c r u c i b l e to the
reaction surface.
In this s i m p l e a n a l y s i s no a c c o u n t was t a k e n of the
gas v e l o c i t y or the m a s s - t r a n s f e r c o e f f i c i e n t when x
=0.
In the p r e s e n t w o r k the r a t e of v a p o r i z a t i o n of Pb
in flowing A r or He was m e a s u r e d . T h e liquid Pb w a s
c o n t a i n e d in an a l u m i n a c r u c i b l e with a 2.5 c m ID and
I I I I I I
2 4 6 8 10 12 was heated in a v e r t i c a l tube f u r n a c e with a 5.5 c m
u, cm/s ID. Th e A r or He e n t e r e d f r o m the top of the f u r n a c e
Fig. A - l - - M a s s - t r a n s f e r coefficient for Pb in flowing He at and the gas v e l o c i t y at t e m p e r a t u r e v a r i e d f r o m 0.8
1100~ to 12.8 c m / s . T h e d i s t a n c e x was v a r i e d f r o m 0.1 to
METALLURGICAL TRANSACTIONS B VOLUME 9B, JUNE 1978-291
1.9 c m . T h e r a t e of v a p o r i z a t i o n w a s m e a s u r e d w i t h a = 0.5 D v "~/4 d 1/4 S c 1/3 R e 3/4 [A-4]
s e m i m i c r o r e c o r d i n g b a l a n c e and a l l m e a s u r e m e n t s
w e r e m a d e at l l 0 0 ~ w h e r e d is the n o z z l e d i a m e t e r , in the p r e s e n t c a s e
The mass-transfer c o e f f i c i e n t is g i v e n b y the d i a m e t e r of t h e f u r n a c e , and r is the r a d i u s of the
Jpb(RT) p l a t e . A l t h o u g h t h e gas s t r e a m in the p r e s e n t e a s e is
m- PPb [A-2] not a j e t and t h e R e y n o l d s n u m b e r (10 to 50) is l o w e r
t h a n t h a t r e c o m m e n d e d (>500) t h i s is the b e s t c o r r e l a -
where t i o n a v a i l a b l e . T h e c a l c u l a t e d v a l u e s f o r A r and He
f o r t h e p r e s e n t e x p e r i m e n t a l c o n d i t i o n s a r e a b o u t 11
Jpb = t h e f l u x of P b in m o l e s / c m 2 s, c m / s and 12.8 c m / s , a s c o m p a r e d w i t h the o b s e r v e d
R = the gas constant, v a l u e of m o of 5.5 and 11 e m / / s r e s p e c t i v e l y . W h e r e a s
T = absolute temperature, the a g r e e m e n t f o r He i s e x c e l l e n t , that f o r A r i s not
PPb = t h e v a p o r p r e s s u r e of P b . 14 v e r y good. H o w e v e r , the g e n e r a l a g r e e m e n t is f a i r
The measured mass-transfer coefficients for flowing c o n s i d e r i n g t h a t t h e e x p e r i m e n t a l c o n d i t i o n s do not
He and A r a r e g i v e n in F i g s . A - 1 and A - 2 a s a f u n c - s a t i s f y the l i m i t a t i o n s of E q . [ A - 4 ] .
t i o n of g a s v e l o c i t y . It is r e c o m m e n d e d t h a t if it is n e c e s s a r y to e s t i m a t e
O n t h e b a s i s of p r e v i o u s w o r k it is r e a s o n a b l e to the g a s - p h a s e m a s s - t r a n s f e r c o e f f i c i e n t f o r the e x -
a s s u m e t h a t the m a s s - t r a n s f e r coefficient will be p e r i m e n t a l g e o m e t r y c o n s i d e r e d , E q . [A-4] s h o u l d be
g i v e n a p p r o x i m a t e l y by u s e d to c a l c u l a t e m o and E q . [A-3] to c a l c u l a t e m . F o r
e x a m p l e , in t h e p r e s e n t w o r k the c a l c u l a t e d m a s s -
1 _ 1 + x [A-3] t r a n s f e r c o e f f i c i e n t f o r P b in He at a g a s - f l o w r a t e
m m o D of 12.8 e m / s a n d x = 0.8 e m is 4.9 c m / s , c o m p a r e d
w i t h 4.5 c m / s o b s e r v e d e x p e r i m e n t a l l y . F o r s i m i l a r
w h e r e m o i s the m a s s - t r a n s f e r c o e f f i c i e n t f o r the c o n d i t i o n s f o r A r the c a l c u l a t e d v a l u e is 1.6 c m / s and
l i m i t i n g c a s e w h e n x = 0. T h e e x p e r i m e n t a l r e s u l t s the o b s e r v e d is 1.9 c m / s . T h e s e v a l u e s c o m p a r e
a r e p l o t t e d in a c c o r d a n c e w i t h E q . [A-3] in F i g . A - 3 reasonably well so that the effect gas-phase mass
f o r g a s v e l o c i t i e s of 1.6 and 12.8 c m / s . T h e e x p e r i - t r a n s f e r m a y h a v e on t h e o v e r a l l k i n e t i c s of t h e r e a c -
m e n t a l r e s u l t s fit E q . [ A - 3 ] r e a s o n a b l y w e l l , and t h e t i o n o r p r o c e s s b e i n g c o n s i d e r e d c a n be e s t i m a t e d .
s l o p e s of t h e l i n e s a r e n e a r l y i n d e p e n d e n t of g a s v e l o c -
i t y . T h e g a s v e l o c i t y h a s a l a r g e i n f l u e n c e on t h e i n - REFERENCES
tercept or the mass-transfer coefficient when x
= O(mo). 1. R. J. Fruehan and E. T. Turkdogan: Met. Trans., 1971, vol. 2, pp. 895-902.
T h e s l o p e s of the l i n e s s h o u l d be p r o p o r t i o n a l to 1//9. 2. G. R. Belton, R. J. Fruehan, and E. T. Turkdogan: Met. Trans., 1972, vol. 3,
pp. 596-98.
T h e s l o p e of the l i n e s f o r He g i v e a v a l u e of 7.1 c m 2 / s 3. E. T. Turkdogan, R. J. Fruehan, and R. H. Tien: Physical Chemistry in
f o r t h e i n t e r d i f f u s i v i t y of P b and He, w h i c h is in e x - Metallurgy, Darken Conference,U.S. Steel Research Laboratory, Monroe-
c e l l e n t a g r e e m e n t w i t h the v a l u e of 7.2 c m 2 / s c a l c u - ville,Pa., 1976.
l a t e d f r o m the d a t a and e q u a t i o n s g i v e n by H i r s c h f e l d e r , 4. E. T. Turkdogan: private communication, U.S. Steel Corporation, Monroe-
e t a l . 6 In the c a s e of A r t h e s l o p e s g i v e v a l u e s of 2.0 ville,Pa.
5. E. T. Turkdogan: Physical Chemistry o f Oxygen Steelmaking Thermo-
and 2.2 f o r the i n t e r d i f f u s i v i t y of P b and A t , w h i c h is chemistry and Thermodynamics, vol. 2, pp. 1-190, Monograph Series on
s o m e w h a t l a r g e r t h a n the c a l c u l a t e d v a l u e of 1.5 cm2//s. Basic OxygenSteelmakingAIME, 1975.
H o w e v e r , the g e n e r a l b e h a v i o r is in good a g r e e m e n t 6. J. O. Hirschfelder,C. F. Curtiss and 1LM. Bird: Molecular Theory o f Gases
with Eq. [A-3]. and Liquids, Wiley,New York,1965.
7. E. S. Machlin: TMS-AIME, 1960, vol. 218, pp. 314-26.
T h e v a l u e of m o b e c o m e s i m p o r t a n t f o r low g a s
8. T. P. Floridis: TMS-AIME, 1970, vol. 215, pp. 870-71.
v e l o c i t i e s and l i g h t e r g a s e s . F o r e x a m p l e the u s e of 9. R. Ohno and T. Ishid: J. 1ton Steellnst., 1968, vol. 207, pp. 904-08.
the s i m p l e E q . [ A - l ] , w h i c h n e g l e c t s m o , c o u l d l e a d to 10. V. D. Sehgal:.L Iron Steellnst., 1969, vol. 207, pp. 95-100, 1507-11.
s i g n i f i c a n t e r r o r s in t h e c a s e of H e . It is a p p a r e n t the 11. V. P. Sehgal and A. Mitchell:.t Iron Steel Inst., 1964, vol. 202, pp. 216-20.
g a s v e l o c i t y a f f e c t s the v a l u e of m o . F o r a j e t s t r e a m 12. E. T. Turkdogan: Method of Producing Low Sulfur Steel, U.S. Patent No.
3,985,550, October 12, 1976.
i m p i n g i n g on a f l a t s u r f a c e , S c h o l t z and T r a s s ~ o b -
13. R. J. Fruehan and L. J. Martonik,MeL Trans., 1974, vol. 5, pp. 1027-32.
tained a correlation experimentally for the mass- 14. O. Kubaschewski, E. L. Evans, and C. B. Alcock,Metallurgical Thermo-
transfer coefficient. The average mass-transfer co- dynamics, Pergamon Press, New York, 1967.
e f f i c i e n t f o r t h e e n t i r e p l a t e (m) is g i v e n b y 15. M. T. Scholtz and O. Trass, AIChEJournal, 1963, vol. 9, pp. 548-54.
292-VOLUME 9B, JUNE 1978 METALLURGICAL TRANSACTIONS B
Das könnte Ihnen auch gefallen
- 15 - 2 - Washington DC - 09-71 - 0150Dokument8 Seiten15 - 2 - Washington DC - 09-71 - 0150Matias MancillaNoch keine Bewertungen
- Mechanical Properties of Modified Low Cobalt Powder Metallurgy UDIMET 700 Type AlloysDokument30 SeitenMechanical Properties of Modified Low Cobalt Powder Metallurgy UDIMET 700 Type AlloysEnary SalernoNoch keine Bewertungen
- Sulfuric Acid Pressure Leaching of Nickeliferous Limonites: Metallurgical and Materials Transactions B January 1977Dokument9 SeitenSulfuric Acid Pressure Leaching of Nickeliferous Limonites: Metallurgical and Materials Transactions B January 1977andyNoch keine Bewertungen
- Activities and Phase Boundaries in The Cr-Ni System Using A Solid Electrolyte TechniqueDokument6 SeitenActivities and Phase Boundaries in The Cr-Ni System Using A Solid Electrolyte Techniquehoushi_miroku_151610Noch keine Bewertungen
- Astroloy The Effects of Microstructure On Elevated Temperature Crack Growth in Nickel-Base AlloysDokument2 SeitenAstroloy The Effects of Microstructure On Elevated Temperature Crack Growth in Nickel-Base AlloysEnary SalernoNoch keine Bewertungen
- The Densities and Structures of Melts Cao-"Feo"-Sio 2 in The SystemDokument8 SeitenThe Densities and Structures of Melts Cao-"Feo"-Sio 2 in The Systemjagadish mahataNoch keine Bewertungen
- Fatigue Crack Initiation and Microcrack Growth in 4140 SteelDokument5 SeitenFatigue Crack Initiation and Microcrack Growth in 4140 SteelEdda Andrade RosalesNoch keine Bewertungen
- Kinetics of Phase Layer Growth During Aluminide Coating of NickelDokument10 SeitenKinetics of Phase Layer Growth During Aluminide Coating of NickelAbhishek ThakurNoch keine Bewertungen
- Wang 1974Dokument5 SeitenWang 1974G.L.R StudiosNoch keine Bewertungen
- Intermtionaz Tournazof Pcne:,Later, 3Dokument12 SeitenIntermtionaz Tournazof Pcne:,Later, 3RiswanNoch keine Bewertungen
- 23 - 4 - Miami Beach - 09-78 - 0072Dokument9 Seiten23 - 4 - Miami Beach - 09-78 - 0072adrian2009-2020Noch keine Bewertungen
- The Deformation Behavior of ADokument12 SeitenThe Deformation Behavior of ARoger LaraNoch keine Bewertungen
- UT Rotary Tube TestingDokument4 SeitenUT Rotary Tube Testingach soNoch keine Bewertungen
- Handling Industrial Fatty Acids - Joseph L. TrauthDokument17 SeitenHandling Industrial Fatty Acids - Joseph L. TrauthP K MisraNoch keine Bewertungen
- 1975 Precipitation Hardening of Aluminum AlloysDokument6 Seiten1975 Precipitation Hardening of Aluminum AlloysyhgrinmNoch keine Bewertungen
- The Use Sorbents To Remove Hydrogen Sulfide Coal T. L. LA: From Gases AtimtayDokument8 SeitenThe Use Sorbents To Remove Hydrogen Sulfide Coal T. L. LA: From Gases Atimtayngnm0Noch keine Bewertungen
- Fujikawa Et Al - 1978 - Diffusion of Silicon in AluminumDokument5 SeitenFujikawa Et Al - 1978 - Diffusion of Silicon in AluminumDanilo CurtolloNoch keine Bewertungen
- 10 - 3 - New York - 09-66 - 0058Dokument14 Seiten10 - 3 - New York - 09-66 - 0058Juan Bayron MuñozNoch keine Bewertungen
- Physical Properties of Uranium Process Solutions byDokument15 SeitenPhysical Properties of Uranium Process Solutions byShrishma PaikNoch keine Bewertungen
- Corrosion Fatigue of Type 4140 High Strength SteelDokument9 SeitenCorrosion Fatigue of Type 4140 High Strength Steelvarun_nagarajan_1Noch keine Bewertungen
- The Effect of Carbide and Nitride Additions On The Heterogeneous Nucleation Behavior of Liquid IronDokument9 SeitenThe Effect of Carbide and Nitride Additions On The Heterogeneous Nucleation Behavior of Liquid IronJoe Iurk PereiraNoch keine Bewertungen
- Influence of Martensite Composition Content On The Properties of Dual Phase Steels andDokument2 SeitenInfluence of Martensite Composition Content On The Properties of Dual Phase Steels andbertaNoch keine Bewertungen
- Stress-Corrosion Cracking BehaviorDokument7 SeitenStress-Corrosion Cracking BehavioroozdemirNoch keine Bewertungen
- The thermal effect due to stress-induced martensite formation in Β-CuAlNi single crystalsDokument4 SeitenThe thermal effect due to stress-induced martensite formation in Β-CuAlNi single crystalsMurilo Romagnolli Dantas de OliveiraNoch keine Bewertungen
- Metal Notes PlumbingDokument8 SeitenMetal Notes PlumbingPatrick MachariaNoch keine Bewertungen
- 1750 MT X AntennasDokument63 Seiten1750 MT X AntennasFabrício AngieneNoch keine Bewertungen
- Back Water CurveDokument20 SeitenBack Water CurveIfrah Ilham Maharsa100% (1)
- Determination of Carbohydrates by Anion Exchange Chromatography With Pulsed Amperometric DetectionDokument16 SeitenDetermination of Carbohydrates by Anion Exchange Chromatography With Pulsed Amperometric DetectionXYZUSPNoch keine Bewertungen
- Covellite To DigeniteDokument7 SeitenCovellite To DigeniteMiizoreNoch keine Bewertungen
- Awatani 1975Dokument6 SeitenAwatani 1975Ilmal YaqinNoch keine Bewertungen
- International Journal of Mine Water - © International Mine Water Association 2006 - WWW - IMWA.infoDokument13 SeitenInternational Journal of Mine Water - © International Mine Water Association 2006 - WWW - IMWA.infoGIANCARLONoch keine Bewertungen
- 0071Dokument182 Seiten0071boopelectraNoch keine Bewertungen
- Loss in Steam TurbineDokument12 SeitenLoss in Steam TurbineShankar Dayal100% (2)
- 29 6 Philadelphia 08-84 0066Dokument13 Seiten29 6 Philadelphia 08-84 0066Azza M. ElnenaeyNoch keine Bewertungen
- 27 - 1 - Las Vegas - 03-82 - 0288Dokument7 Seiten27 - 1 - Las Vegas - 03-82 - 0288Michael DavidNoch keine Bewertungen
- 32 - 3 - New Orleans - 08-87 - 0255Dokument5 Seiten32 - 3 - New Orleans - 08-87 - 0255Abdul AzizNoch keine Bewertungen
- Alaa 3Rd Joint: SdecialistDokument7 SeitenAlaa 3Rd Joint: SdecialistfienwkfwjebfNoch keine Bewertungen
- Phase Instabilities During High Temperature Exposure of 316 Austenitic Stainless Steel PDFDokument16 SeitenPhase Instabilities During High Temperature Exposure of 316 Austenitic Stainless Steel PDFgostokhelwiNoch keine Bewertungen
- Diffusion Modeling of The Carburization Process: That They InfluenceDokument2 SeitenDiffusion Modeling of The Carburization Process: That They InfluenceWahyuNoch keine Bewertungen
- 9 (I,, TillDokument4 Seiten9 (I,, TillSinhrooNoch keine Bewertungen
- Basic Solar Pond ModelDokument7 SeitenBasic Solar Pond ModelNeale NeelamegghamNoch keine Bewertungen
- 8.5 Treatment of The Leach Liquor Prior To SX-EW: Eco-Tec - Co MDokument1 Seite8.5 Treatment of The Leach Liquor Prior To SX-EW: Eco-Tec - Co MLeo Santiago GallegosNoch keine Bewertungen
- Gpo Price Csfti Price (S) $Dokument5 SeitenGpo Price Csfti Price (S) $maxwellstreetguyNoch keine Bewertungen
- Nicolle and RistDokument10 SeitenNicolle and RistMariaNoch keine Bewertungen
- BF02667283Dokument15 SeitenBF02667283Minaz DadiNoch keine Bewertungen
- Nanofiltration To Separate Salts From H2S Scrubber Solutions Continuous Microfiltration Process SelectedDokument1 SeiteNanofiltration To Separate Salts From H2S Scrubber Solutions Continuous Microfiltration Process SelectedAditya RahmatNoch keine Bewertungen
- Penthouse Spring 1985Dokument2 SeitenPenthouse Spring 1985John M. CavoteNoch keine Bewertungen
- Estimating Methane Content of Bituminous Coalbeds From AdsorptionDokument26 SeitenEstimating Methane Content of Bituminous Coalbeds From AdsorptionAdhitya KuswantoroNoch keine Bewertungen
- Design of Pillars in Shrinkage StopingDokument13 SeitenDesign of Pillars in Shrinkage StopingManuel QuirozNoch keine Bewertungen
- Discussion of "Tensile Properties To 650° C and Deformation Structures in A Precipitation-Strengthened Ti-Al Alloy"Dokument1 SeiteDiscussion of "Tensile Properties To 650° C and Deformation Structures in A Precipitation-Strengthened Ti-Al Alloy"YuliNoch keine Bewertungen
- Evaluation of Toughness in AISI 4340 Alloy Steel Austenitized at Low and High TemperaturesDokument2 SeitenEvaluation of Toughness in AISI 4340 Alloy Steel Austenitized at Low and High TemperaturesSyahid AzhariNoch keine Bewertungen
- 348Dokument14 Seiten348eyheghedgeNoch keine Bewertungen
- Field 1978Dokument4 SeitenField 1978OlgalycosNoch keine Bewertungen
- 1977 Calorimetric Studies of 7000 Series Aluminum Alloys - I. Matrix Precipitate Characterization of 7075Dokument7 Seiten1977 Calorimetric Studies of 7000 Series Aluminum Alloys - I. Matrix Precipitate Characterization of 7075yhgrinmNoch keine Bewertungen
- 1 s2.0 0376458380900242 MainDokument11 Seiten1 s2.0 0376458380900242 Main陳顥平Noch keine Bewertungen
- Szekely1976 PDFDokument9 SeitenSzekely1976 PDFlrodriguez_892566Noch keine Bewertungen
- Clifton (1985) Composite DesignDokument16 SeitenClifton (1985) Composite DesignlecupiNoch keine Bewertungen
- Thermodynamic Study of Ice and Clathrate Hydrates : Chem.Dokument10 SeitenThermodynamic Study of Ice and Clathrate Hydrates : Chem.Shurooq TaibNoch keine Bewertungen
- Simulation and Application of Tapping Online Refining in EAF Steelmaking ProcessDokument10 SeitenSimulation and Application of Tapping Online Refining in EAF Steelmaking ProcessRasul BzNoch keine Bewertungen
- Modelling of Electric Arc Furnace Off-Gas Removal System: N. Timoshenko, A. Semko and S. TimoshenkoDokument5 SeitenModelling of Electric Arc Furnace Off-Gas Removal System: N. Timoshenko, A. Semko and S. TimoshenkoRasul BzNoch keine Bewertungen
- DownloadDokument8 SeitenDownloadRasul BzNoch keine Bewertungen
- Theoretical Aspects of Scrap Dissolution in Oxygen Steelmaking ConvertersDokument12 SeitenTheoretical Aspects of Scrap Dissolution in Oxygen Steelmaking ConvertersRasul BzNoch keine Bewertungen
- Development of The Process of Electric Steel Production and Methods For Improving The Technicalandeconomic Indices of Electric Arc FurnacesDokument5 SeitenDevelopment of The Process of Electric Steel Production and Methods For Improving The Technicalandeconomic Indices of Electric Arc FurnacesRasul BzNoch keine Bewertungen
- Foaming Characteristics of BOF SlagsDokument8 SeitenFoaming Characteristics of BOF SlagsRasul BzNoch keine Bewertungen
- Aspect of Design and Developments in EAF: December 2014Dokument6 SeitenAspect of Design and Developments in EAF: December 2014Rasul BzNoch keine Bewertungen
- Thermodynamics On The Formation of Spinel Nonmetallic Inclusion in Liquid Steel PDFDokument4 SeitenThermodynamics On The Formation of Spinel Nonmetallic Inclusion in Liquid Steel PDFRasul BzNoch keine Bewertungen
- Hydrogen Pick-Up After Vacuum Degassing: J. B. Hurtig and D. SichenDokument6 SeitenHydrogen Pick-Up After Vacuum Degassing: J. B. Hurtig and D. SichenRasul BzNoch keine Bewertungen
- Removal of Hydrogen, Nitrogen and Sulphur From Tool Steel During Vacuum DegassingDokument7 SeitenRemoval of Hydrogen, Nitrogen and Sulphur From Tool Steel During Vacuum DegassingRasul BzNoch keine Bewertungen
- A Mathematical Model For The Reduction Stage of The CAS-OB ProcessDokument13 SeitenA Mathematical Model For The Reduction Stage of The CAS-OB ProcessRasul BzNoch keine Bewertungen
- The Effect of Fe O Content On Mgo Solubilities in Lime-Based SlagsDokument5 SeitenThe Effect of Fe O Content On Mgo Solubilities in Lime-Based SlagsRasul BzNoch keine Bewertungen
- Activities of SiO2 and Al2O3 and Activity Coefficients of FetO and MnO in CaO-SiO2-Al2O3-MgO SlagsDokument11 SeitenActivities of SiO2 and Al2O3 and Activity Coefficients of FetO and MnO in CaO-SiO2-Al2O3-MgO SlagsRasul BzNoch keine Bewertungen
- Selection of Stirring and Shrouding Gases For Steelmaking Applications PDFDokument20 SeitenSelection of Stirring and Shrouding Gases For Steelmaking Applications PDFRasul BzNoch keine Bewertungen
- The Physical and Mathematical Modelling of Gas Stirred Ladle Systems PDFDokument20 SeitenThe Physical and Mathematical Modelling of Gas Stirred Ladle Systems PDFRasul BzNoch keine Bewertungen
- Unit 1: Lesson 5 Soil FormationDokument12 SeitenUnit 1: Lesson 5 Soil FormationWindy CalimlimNoch keine Bewertungen
- Catalogo Empresas Solartys 2014Dokument44 SeitenCatalogo Empresas Solartys 2014Isaac RodriguezNoch keine Bewertungen
- Combustion: Luminous Gas Flame Flame Front Luminous Non-LuminousDokument18 SeitenCombustion: Luminous Gas Flame Flame Front Luminous Non-LuminousMd. Ahsanur RahmanNoch keine Bewertungen
- Energetic Chemistry TestDokument10 SeitenEnergetic Chemistry Testmuhammad naufalNoch keine Bewertungen
- Lecture Note On Photon Interection An Cross SectionDokument21 SeitenLecture Note On Photon Interection An Cross SectionpankajNoch keine Bewertungen
- Engineering Thermodynamics - Rwanda PolytechnicsDokument78 SeitenEngineering Thermodynamics - Rwanda PolytechnicsAlexis MUHIRWANoch keine Bewertungen
- Renewable Energy Systems (Inter Disciplinary Elective - I)Dokument2 SeitenRenewable Energy Systems (Inter Disciplinary Elective - I)vishallchhayaNoch keine Bewertungen
- Ee2303 Newlp ADokument3 SeitenEe2303 Newlp ARavi KannappanNoch keine Bewertungen
- Unit 8 TestDokument2 SeitenUnit 8 TestmamazookeeprNoch keine Bewertungen
- DLP in Final DemonstrationDokument10 SeitenDLP in Final DemonstrationMisty MamintaNoch keine Bewertungen
- Final Exam Study Guide 2014 (High School Chem)Dokument6 SeitenFinal Exam Study Guide 2014 (High School Chem)Brian KimNoch keine Bewertungen
- Concept: Subject-General ScienceDokument2 SeitenConcept: Subject-General SciencetrevorNoch keine Bewertungen
- IAS Mains Geology Papers 1997: ExamraceDokument3 SeitenIAS Mains Geology Papers 1997: ExamraceD DNoch keine Bewertungen
- Hydrogen - Natural Gas Blends PDFDokument8 SeitenHydrogen - Natural Gas Blends PDFLTE002Noch keine Bewertungen
- Wilson Cloud Chamber: How Does It WorkDokument3 SeitenWilson Cloud Chamber: How Does It WorkNaveed AhmedNoch keine Bewertungen
- Rocks & Energy Chapter 1 & 2 Environmental Management 0680 Geography GCSE IGCSE O-LevelsDokument108 SeitenRocks & Energy Chapter 1 & 2 Environmental Management 0680 Geography GCSE IGCSE O-LevelsIGCSE Physics & Chemistry100% (1)
- EnvisciDokument7 SeitenEnvisciPrecious CabigaoNoch keine Bewertungen
- CV - A Dutta - Updated (IF)Dokument14 SeitenCV - A Dutta - Updated (IF)Abhigyan DuttaNoch keine Bewertungen
- Pollution in The OceansDokument13 SeitenPollution in The OceansMory GamingNoch keine Bewertungen
- Lecture 3 - Protist: ProtistsDokument1 SeiteLecture 3 - Protist: ProtistsMariz RaymundoNoch keine Bewertungen
- Revision Notes - Unit 2 AQA Physics A-LevelDokument12 SeitenRevision Notes - Unit 2 AQA Physics A-LevelElliot ThomasNoch keine Bewertungen
- What Causes Seasons On Earth?Dokument3 SeitenWhat Causes Seasons On Earth?rossettejavierNoch keine Bewertungen
- Waste Management: Marco Tomasi Morgano, Hans Leibold, Frank Richter, Dieter Stapf, Helmut SeifertDokument9 SeitenWaste Management: Marco Tomasi Morgano, Hans Leibold, Frank Richter, Dieter Stapf, Helmut SeifertCarlos AlvarezNoch keine Bewertungen
- R134a TableDokument5 SeitenR134a TableAhmed Mahmoud AbouzaidNoch keine Bewertungen
- Chapter 11 Watershed Management in Nepal: Challenges and ConstraintsDokument11 SeitenChapter 11 Watershed Management in Nepal: Challenges and ConstraintsSajeet KarkeeNoch keine Bewertungen
- Chapter-01 What Is Human Comfort ?Dokument13 SeitenChapter-01 What Is Human Comfort ?kartik chopraNoch keine Bewertungen
- Geophysical ProspectingDokument16 SeitenGeophysical ProspectingMoulik MehtaNoch keine Bewertungen
- 43a-Intermidiate Check of CRM-chemicalDokument16 Seiten43a-Intermidiate Check of CRM-chemicalDeepak HoleNoch keine Bewertungen