Beruflich Dokumente
Kultur Dokumente
Aaa Endovasc
Hochgeladen von
Cayo NoMáCopyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Aaa Endovasc
Hochgeladen von
Cayo NoMáCopyright:
Verfügbare Formate
Eur J Vasc Endovasc Surg 29, 560–562 (2005)
doi:10.1016/j.ejvs.2005.04.001, available online at http://www.sciencedirect.com on
EDITORIAL
Registries versus Trials for the Evaluation of the
Endovascular Treatment of Abdominal Aortic Aneurysms
M.G. Wyatt*
Northern Vascular Centre, Freeman Hospital, Freeman Road, High Heaton,
Newcastle upon Tyne NE7 7DN, UK
Introduction retrospective and prospective data on 1823 pro-
cedures. Annual audit reports are produced on behalf
Since the introduction of aortic stent grafting to repair of the Vascular Society of Great Britain and Ireland and
abdominal aortic aneurysm (EVAR),1 much of the data the British Society of Interventional Radiology. The
concerning this new technology has been derived from database has been an invaluable source of data on the
registries rather than trials. The two most important performance of EVAR devices over the last 8 years, but
registries have been the UK registry for Endovascular as with all registries suffers in that it is voluntary and
Treatment of Aneurysms (RETA) based in Sheffield2 audited in an ‘open’ fashion, possibly leading to
and the European Collaborators on Stent-Graft Tech- selection bias.
niques for Abdominal Aortic Aneurysm Repair The mid-term results from the RETA registry show
(EUROSTAR) registry based in Holland.3 These that at 30 days, 90.4% of aneurysms were successfully
registries, although voluntary, have provided import- excluded and 5.8% of patients had died. The conver-
ant information on EVAR and data from the RETA sion rate to open repair was 3.3%, but this rate has
registry was used in the design of the UK EVAR trials been falling with time (9.1% in 1996 and 0.3% in 1999).
and as an audit tool to assess centres for trial entry. Post-procedural complications within 30 days
The 5-year RETA registry results are published in occurred in 27.8% of cases and 6.1% had persistent
this issue of the European Journal of Vascular and endoleaks. The data shows significantly more deaths
Endovascular Surgery.2 Nevertheless, such registry in unfit (ASA IV) patients (14.8 versus 3.3%), and a
data, especially that related to follow up, is often higher mortality rate for aorto-uni-iliac devices (12.5
incomplete and may present a biased view of the versus 3.3%) and in women (5.7 versus 1.9%).
overall performance of new technologies. It is, there- Mortality in the first year was 11% (mostly cardiac
fore, essential that data derived from these registries is disease and malignancy) and 10% per year thereafter
interpreted with caution and, more importantly, that up to 5 years. Most of these deaths were not graft
definitive clinical practice does not change until the related, the cumulative 5-year risk of rupture being
on-going randomised controlled clinical trails have only 2%.
had an opportunity to report their data. Complications related to the device and aneurysm
were reported at a rate of approximately 15% per year.
These included secondary endoleaks, graft migration,
kinking, and limb occlusions. Treatment was by
RETA Registry further interventional radiology or surgery, with only
38% of patients surviving with no secondary inter-
The RETA registry was set up in 1996 to audit EVAR vention at 5 years.
deployments within the UK. It contains both
* Corresponding author. Michael G. Wyatt, MBBS, MSc, MD, FRCS,
Consultant Vascular Surgeon, Northern Vascular Centre, Freeman EUROSTAR Registry
Hospital, Freeman Road, High Heaton, Newcastle upon Tyne NE7
7DN, UK.
E-mail address: mike.wyatt@nuth.nhs.uk The EUROSTAR registry shows similar results.3
1078–5884/000560 + 03 $35.00/0 q 2005 Published by Elsevier Ltd.
Registries versus Trials for EVAR 561
Launched in 1996, it is a prospective audit of EVAR treatment or best medical treatment alone. Three other
performance across 135 centres in 18 European randomised controlled trials have also been initiated.
countries. This registry is also voluntary, but is the These are the Dutch DREAM, the French ACE and the
largest registry of devices in Europe with a total of American OVER trials.
5466 cases of EVAR on record by July 2003. Results
from first generation devices were characterised by
EVAR 1 trial 30-day results
excellent early performance, but poor durability. The
latest data from newer devices show significant
Early results of the EVAR 1 and Dream trials were
improvements in all outcome measures, most impor-
published in 2004 and appear to support the use of
tantly durability. One hundred and eighty seven
EVAR. The EVAR 1 trial5 randomised 1082 patients
patients have been followed up for more than 5
over 60 years of age with an aneurysm of 5.5 cm or
years. Life table analysis shows a 5-year cumulative
greater to either open repair or EVAR. The 30-day
results as follows; survival 79.2% and freedom from all
mortality for EVAR was 1.7% compared to 4.7% for
endoleaks 71.9%, persistent endoleaks 91.4%, second-
open repair (pZ0.007). This mortality was lower than
ary intervention 76.3% and rupture 98.6%.
that reported by the RETA and EUROSTAR registries,
The main conclusions from analysis of the EURO-
perhaps because the patients randomised in EVAR 1
STAR database are that the incidence of rupture
were those deemed fit to have an open repair
increases progressively with time at least up to 5
compared to the registry data which includes ‘unfit’
years, but that serious adverse events occur less often
patients treated with primary stent grafts. In addition,
with newer devices, EVAR in high-risk patients is
the EVAR 1 trial 30-day results report that secondary
probably justified if they are not expected to die within
procedures were necessary in 9.8% of EVAR patients
1 year from the effects of their co-morbid conditions,
compared to 5.8% in the open repair group. Endoleaks
and the medium-term outcome is significantly better
needed treatment in 3.4% of EVAR patients and 10
in patients with small aneurysms.
patients required conversion to open repair
The above data clearly shows that open audit, in the
intraoperatively.
form of voluntary registries, is an essential tool for the
clinical evaluation of new technologies, but they do
not replace randomised controlled trials in that they DREAM trial 30-day results
are incomplete and open to data interpretation bias.
They do provide data as to the long term performance The Dutch DREAM trial6 has also published its 30-day
of EVAR, but in a ‘selected’ group of patients. Such mortality results for endovascular versus open repair.
registry data is considered useful in the evaluation of Despite only 345 patients being randomised, repre-
new technologies and organisations such as the UK senting a 12% under recruitment, operative mortality
National Institute of Clinical Excellence (NICE) now was similar to the UK EVAR trial at 1.2% in the EVAR
specifically recommend that, for EVAR, patients are group versus 4.6% in the open repair group. Never-
either entered into a registry or treated within the theless, these results did not reach significance.
confines of a randomised controlled trial. Intervention for endoleak occurred in 1.2% and the
combined rate of operative mortality and severe
complication was 4.7% for the endovascular group
Randomised Controlled Trials versus 9.8% for the open group.
To date, registries provide the only evidence we have
as to the long term durability of EVAR, and no level 1 Conclusions
evidence exists to support the use of aortic stent grafts
for the repair of abdominal aortic aneurysm. Never- It is essential that, as for registry data, the 30-day
theless, the uncertainty about how EVAR compares to randomised controlled trial data are interpreted with
conventional open repair has led to the instigation of caution, since the long term benefit of EVAR has yet to
several randomised controlled multi-centred trials. be proven. At the time of writing, the 30-day EVAR 1
The UK EVAR trials were initiated in 1999.4 EVAR 1 and DREAM randomised trial data should not be used
randomises suitable patients to either endovascular to advocate the widespread use of EVAR over open
stent graft repair or conventional open repair. Patients repair. The authors of the EVAR 1 trial conclude that
who are unfit to undergo open repair due to significant the results of 30-day mortality serve only as evidence
co-morbidity are entered into the EVAR 2 trial. This to continue investigating the use of EVAR and stress
randomises patients to either EVAR and best medical the importance of obtaining the long-term data. The
Eur J Vasc Endovasc Surg Vol 29, June 2005
562 M. G. Wyatt
DREAM study authors state that, taken together, these generation devices appear to be performing better,
results indicate that EVAR is preferable over open but it will require the positive reporting of the mid
repair in those who are suitable despite the results not term data from the randomised controlled trials before
reaching significance. This conclusion is later muted, we have sufficient evidence to allow EVAR to be fully
suggesting that further long term data are required to integrated into everyday surgical practice.
determine if the early benefit is sustained in favour of
EVAR.
The EVAR 1 and 2 trials are due to report their References
medium term results in June 2005. Professor Roger
Greenhalgh will present these results at an extraordi- 1 Parodi JC, Palmez JC, Barone HD. Transfemoral intraluminal
graft implantation for abdominal aortic aneurysms. Ann Vasc Surg
nary meeting of the Endovascular Forum to be held at 1991;5:491–499.
the Belfry, Warwickshire, England on June 16th 2005. 2 THOMAS S, BEARD JD, IRELAND M, AYERS S. Results from the
On the same day the Lancet will publish the scientific Prospective Registry of Endovascular Treatment of Abdominal
Aortic Aneurysms (RETA): mid term results to five years. Eur J
paper. By this time, one third of EVAR 1 patients will Vasc Endovasc Surgery.
have been followed for 4 years and these results will 3 Harris P. The EUROSTAR EVAR vigilance message. In:
give us the first robust evidence of the medium term Greenhalgh R, ed. Vascular and endovascular challenges. UK:
BIBA Publishing, 2004:103–112.
durability of EVAR. Funding has also recently been 4 Brown LC, Epstein D, Manca A, Beard JD, Powell JT,
secured to enable this cohort of patients to be followed Greenhalgh RM. The UK Endovascular Aneurysm Repair
up for a further 5 years. The Dutch DREAM trial (EVAR) trials: design, methodology and progress. Eur J Vasc
Endovasc Surg 2004;27(4):372–381.
patients are also to be followed for longer and the 5 Greenhalgh RM, Brown LC, Kwong GP, Powell JT,
French ACE and American OVER trials are Thompson SG, on behalf of the EVAR Trial participants.
continuing. Comparison of endovascular aneurysm repair with open repair
in patients with abdominal aortic aneurysm (EVAR1 trial), 30 day
In summary, the early and mid-term registry data operative mortality results; randomised controlled trial. Lancet
appears promising for the use of EVAR. The early 2004;364(9437):843–848.
EVAR 1 and Dream trial data supports the use of 6 Prinssen M, Verhoeven EL, Buth J, Cuypers PW, van
Sambeek MR, Balm R et al. A randomized trial comparing
EVAR in fit patients, but concerns still exist regarding conventional and endovascular repair of abdominal aortic
stent graft durability and the need for continued aneurysms. N Engl J Med 2004;351(16):1607–1618.
intervention. The registries show us that newer
Eur J Vasc Endovasc Surg Vol 29, June 2005
Das könnte Ihnen auch gefallen
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeVon EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeBewertung: 4 von 5 Sternen4/5 (5794)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreVon EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreBewertung: 4 von 5 Sternen4/5 (1090)
- Never Split the Difference: Negotiating As If Your Life Depended On ItVon EverandNever Split the Difference: Negotiating As If Your Life Depended On ItBewertung: 4.5 von 5 Sternen4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceVon EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceBewertung: 4 von 5 Sternen4/5 (895)
- Grit: The Power of Passion and PerseveranceVon EverandGrit: The Power of Passion and PerseveranceBewertung: 4 von 5 Sternen4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeVon EverandShoe Dog: A Memoir by the Creator of NikeBewertung: 4.5 von 5 Sternen4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersVon EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersBewertung: 4.5 von 5 Sternen4.5/5 (345)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureVon EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureBewertung: 4.5 von 5 Sternen4.5/5 (474)
- Her Body and Other Parties: StoriesVon EverandHer Body and Other Parties: StoriesBewertung: 4 von 5 Sternen4/5 (821)
- The Emperor of All Maladies: A Biography of CancerVon EverandThe Emperor of All Maladies: A Biography of CancerBewertung: 4.5 von 5 Sternen4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Von EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Bewertung: 4.5 von 5 Sternen4.5/5 (121)
- The Little Book of Hygge: Danish Secrets to Happy LivingVon EverandThe Little Book of Hygge: Danish Secrets to Happy LivingBewertung: 3.5 von 5 Sternen3.5/5 (400)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyVon EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyBewertung: 3.5 von 5 Sternen3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)Von EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Bewertung: 4 von 5 Sternen4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaVon EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaBewertung: 4.5 von 5 Sternen4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryVon EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryBewertung: 3.5 von 5 Sternen3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnVon EverandTeam of Rivals: The Political Genius of Abraham LincolnBewertung: 4.5 von 5 Sternen4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealVon EverandOn Fire: The (Burning) Case for a Green New DealBewertung: 4 von 5 Sternen4/5 (74)
- The Unwinding: An Inner History of the New AmericaVon EverandThe Unwinding: An Inner History of the New AmericaBewertung: 4 von 5 Sternen4/5 (45)
- Energetic Codependency: Break Free From The Toxic Energies That Hijack AuthenticityDokument26 SeitenEnergetic Codependency: Break Free From The Toxic Energies That Hijack AuthenticityElisa Romeo83% (12)
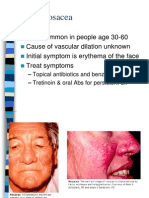
- Acne RosaceaDokument28 SeitenAcne Rosaceanathy_soares100% (1)
- Synopsis de Addiction CenterDokument2 SeitenSynopsis de Addiction CenterNishana100% (1)
- Akhtar, Salman - Universal Interventions - Alterations of The Frame, Method, and Relationship in Psychotherapy and Psychoanalysis.-Karnac (2011)Dokument250 SeitenAkhtar, Salman - Universal Interventions - Alterations of The Frame, Method, and Relationship in Psychotherapy and Psychoanalysis.-Karnac (2011)Pratik BhatuNoch keine Bewertungen
- ISCAID Urinary GuidelinesDokument9 SeitenISCAID Urinary Guidelinesartzain1991Noch keine Bewertungen
- PD F S Abr23 Journal of Endodontics 2012 MoonDokument5 SeitenPD F S Abr23 Journal of Endodontics 2012 MoonMaRii ALeNoch keine Bewertungen
- Food Affects Mood and EmotionsDokument90 SeitenFood Affects Mood and Emotionsarbeta100% (3)
- Introduction To Expert Systems - MYCINDokument8 SeitenIntroduction To Expert Systems - MYCINVictoria OffomaNoch keine Bewertungen
- HfiDokument4 SeitenHfiakalaw3Noch keine Bewertungen
- Bahir Dar University Institute of TechnologyDokument26 SeitenBahir Dar University Institute of Technologytazeb AbebeNoch keine Bewertungen
- Formularium PDFDokument16 SeitenFormularium PDFDewayu NanaNoch keine Bewertungen
- Introduction Cysts of JawsDokument62 SeitenIntroduction Cysts of JawsEnass Alhadi50% (2)
- Comprehensive Exam NCM 145Dokument17 SeitenComprehensive Exam NCM 145Adrian Dela CruzNoch keine Bewertungen
- Trichiasis: Characteristics and Management OptionsDokument6 SeitenTrichiasis: Characteristics and Management Optionsaspal butonNoch keine Bewertungen
- Uses of TanninsDokument1 SeiteUses of TanninsMike SalcedoNoch keine Bewertungen
- CSP - Groin LumpDokument39 SeitenCSP - Groin LumpAnnisa SakilaNoch keine Bewertungen
- Removable AppliancesDokument32 SeitenRemovable AppliancesmegamarwaNoch keine Bewertungen
- WarfarinDokument10 SeitenWarfarinMar Ordanza100% (1)
- Montano NCP TbiDokument6 SeitenMontano NCP TbiKarl Angelo MontanoNoch keine Bewertungen
- Hospital Block: Key PlanDokument1 SeiteHospital Block: Key PlanKripa SriramNoch keine Bewertungen
- Direct Anterior Total Hip ArthroplastyDokument13 SeitenDirect Anterior Total Hip Arthroplasty21701101006 Rosita SariNoch keine Bewertungen
- Occupational Identity Disruption After Traumatic Brain Injury - An Approach To Occupational Therapy Evaluation and TreatmentDokument13 SeitenOccupational Identity Disruption After Traumatic Brain Injury - An Approach To Occupational Therapy Evaluation and Treatmentapi-234120429Noch keine Bewertungen
- Documentation Techniques PDFDokument24 SeitenDocumentation Techniques PDFPooja varmaNoch keine Bewertungen
- KK Tunnel Route MapDokument1 SeiteKK Tunnel Route MapHonolulu Star-AdvertiserNoch keine Bewertungen
- Anesthesia For TurpDokument5 SeitenAnesthesia For Turpdrkadiyala2Noch keine Bewertungen
- Pielago NCPDokument2 SeitenPielago NCPtaehyungieeNoch keine Bewertungen
- Antibiotics Guide 2013Dokument30 SeitenAntibiotics Guide 2013Stefani NoviliaNoch keine Bewertungen
- What Are AntibioticsDokument31 SeitenWhat Are Antibiotics1211988610Noch keine Bewertungen
- The Assessment Interview in Clinical NeuropsychologyDokument33 SeitenThe Assessment Interview in Clinical NeuropsychologyNohora Johanna Rondón100% (1)
- CS URTIs BSN3AG3Dokument21 SeitenCS URTIs BSN3AG3GLORY MI SHANLEY CARUMBANoch keine Bewertungen