Beruflich Dokumente
Kultur Dokumente
Surface Chemistry Assignment PDF
Hochgeladen von
Akshat PandeyOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Surface Chemistry Assignment PDF
Hochgeladen von
Akshat PandeyCopyright:
Verfügbare Formate
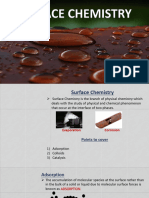
Surface
Chemistry
Key Concepts
Surface chemistry is the branch of chemistry that deals with Water vapour
the properties and processes occurring at the surface or inter-
face that is the boundary separating two bulk phases.
A B
ADSORPTION
A fi Silica Gel (Adsorption)
B fi Anhydrous CaCl2 (Absorption)
The phenomenon of attracting and retaining the particles of
a substance on the surface of a solid or a liquid resulting in If the process of adsorption and absorption both occur si-
a higher concentration at the surface rather than the bulk is multaneously then the process is known as sorption.
known as adsorption. For example, Chalk dipped in Ink.
Æ The substance of the surface of which the process of
adsorption takes place is known as adsorbent (s/l). Mechanism of Adsorption _________________
Æ The substance which is gating adsorbed on the surface The particles present in the bulk and at the surface of a sub-
is known as adsorbate (s/l/g). stance are under different conditions. The forces acting on the
Æ The removal of adsorbate from the surface of adsorbent bulk particles are mutually balanced as they are surrounded
is known as desorption. by the same type of particles from all sides but the particles
Æ The adsorption of gases on the surface of a metal is on the surface have unbalanced or residual attractive forces
known as occlusion. as they are not surrounded by same type of particle from all
sides as a result they have a tendency to attract and retain the
Adsorption Absorption
particles of other substance on their surface.
Æ In the process of adsorp- Æ In the process of absorption
tion the concentration of the concentration of the Adsorbate S/L/G
Surface particle Adsorbent
the substances increases substances is uniformly
only at the surface of ad- distributed throughout the Fnet π 0 solid or liquid
sorbent. bulk of the solid or liquid.
Bulk particle
Æ It is a surface phenom- Æ It is a bulk phenomenon. Fnet = 0
enon.
Thermodynamic approach for Adsorption
Æ The rate of adsorption de- Æ The rate of absorption is
DH adsorption = –ve [Due to decrease in surface energy]
creases with time. uniform through out the
DS adsorption = –ve [Due to decrease in degree of move-
process.
ment]
For example, NH3 on charcoal For example, NH3 in water
IIT JEE PC-V2_05.indd 1 5/12/2017 6:30:11 PM
5.2
As we know, Note: I
DG = DH – TDS The process of physical adsorption occuring at a low tem-
= –DH + TDS perature can be converted to chemical adsorption at a high-
\ DGadsorption = –DH + TDS er temperature.
(i) At low temperature
For example, adsorption of Hydrogen on the surface of Ni
DH > TDS
to form metal hydride.
\ DG = –ve
Adsorption is spontaneous
(ii) At high Temperature FACTORS EFFECTING
DH < TDS ADSORPTION
DG = +ve
Adsorption is non-spontaneous 1. Surface Area of Adsorbent
Surface area μ Extent of Adsorption
DH
(iii) When T = 2. Nature of Adsorbate
DS (i) In chemical adsorption, the particles of the adsor-
then TDS = DH bate adsorb on the surface of adsorbent only if they
\ DG = 0 are able to make chemical bonds.
Hence, equilibrium is attained in between adsorption For example, Oxygen is adsorbed on the surface of
and desorption. metals to form metal oxides.
Adsorption For example, Hydrogen is adsorbed on the surface
Adsorbate + Adsorbent Adsorbed
Desorption of transition metals to form metal hydrides.
Types of Adsorption (ii) In physical adsorption critical temperature (Tc)
1. Physical Adsorption Æ Also called physisorption. Tc μ Ease of Liquification μ Extent of Adsorption
For example, H2 N2 CO CH4 CO2 HCl NH3 SO2
Æ When the particle of adsorbate are held to the sur-
If Tc increases, Ease of liquification increases Ex-
face of adsorbent by the weak Van der Waal forces.
tent of adsorption increases.
Then, the process is called physical adsorption.
3. Effect of Temperature
2. Chemical Adsorption Æ Also called Chemisorption.
Æ When the particles of the adsorbate held to the sur- Ê xˆ
fi Extent of Adsorption Æ Á ˜
face of adsorbent due to the formation of strong Ë m¯
chemical bonds then, the process is called chemi- It is defined as the mass of adsorbate adsorbed per unit
cal adsorption. mass of adsorbent.
Æ The bond may be covalent or ionic and it requires x fi Mass of adsorbate
activation energy. Hence, it is also known as acti- m fi Mass of adsorbent
vated adsorption. At constant Pressure At constant Pressure
x
Physical Adsorption Chemical Adsorption m x
m
Æ Not specific in nature Æ Specific
Æ Activation energy is not Æ Activation energy is
required required Temperature fi Temperature fi
Æ It arises due to weak Van Æ It arises due to formation (A) Physisorption (B) Chemisorption
der Waal forces of strong chemical bonds
Æ These curves are called adsorption of isobars.
Æ Heat of adsorption is low, Æ Heat of adsorption is high,
(A) fi The extent of physical adsorption decreases
i.e., from –20 to –40 kJ i.e., from –80 to –240 kJ
with increase of temperature because the strength
mol–1 mol–1
of Van der Waal forces decreases with increase in
Æ Reversible in nature Æ Generally irreversible in temperature and also as the process is exothermic.
nature (B) fi The extent of chemical adsorption firstly in-
Æ Multi layer of adsorbate Æ Mono layer is formed on creases with temperature because it requires acti-
are formed on the surface the surface of adsorbent vation energy for the bond formation and after that
of adsorbent on increasing the temperature the extent of chemi-
cal adsorption decreases because the process is
Æ Low temperature is fa- Æ Favourable at high tem-
exothermic.
vourable for adsorption perature and extent of
4. Effect of Pressure
and extent of adsorption adsorption decreases with
The variation in extent of adsorption with change in
decreases with the in- increase in temperature
pressure at constant temperature can be explained with
crease in temperature
the help of some graphs called adsorption isotherms.
IIT JEE PC-V2_05.indd 2 5/12/2017 6:30:12 PM
Surface Chemistry 5.3
(A) Freundlich adsorption isotherm: For physisorption (B) Langmuir Adsorption Isotherm: For chemical adsorp-
Æ He explained the variation in adsorption due the tion
change in pressure graphically and mathematically Postulate–1 He postulated that, the particles of adsorbate
as follows. can only form a single layer in depth on the
Æ Adsorbate – Gas surface of adsorbent.
Æ Adsorbent – Solid Postulate–2 There is no interaction in between the adjacent
adsorbed particles.
C D Postulate–3 There is a dynamic equilibrium in between the
x
m process of adsorption and desorption.
B
Postulate–4 All surface sides have equal tendency to at-
tract and retain the particles of adsorbate.
A x aP
P = where, a and b are Langmuir
m 1 + bP
Case-I At low pressure (A Æ B)
constant.
x
μP (i) At low P,
m
1 + bP 1
Case-II At high pressure (C Æ D)
x
x So, = aP
μ P∞ m
m
Case-III At intermediate pressure (B Æ C) Èx ˘
\ Í μ P˙
x Îm ˚
μ P1/n [where n = 1 to ] (ii) At high P,
m
1 + bP bP
x
The resultant condition = KP1/ n x aP a
At low P fi n = 1 m So, = =
m bP b
At high P fi n =
At intermediate P fi 1 < n < \ Èx ˘
ÍÎ m μ P∞ (constant) ˙˚
\ the value of (1/n) ranges from 0 to 1.
Here x fi Mass of adsorbate * As we know,
m fi Mass of adsorbent x aP
=
P fi Pressure of adsorbate gas m 1 + bP
K and n fi Constants that depends on the nature of ad- It can be written as
sorbate and adsorbent. 1 1 + bP 1 b
= = +
Note: 1 ( x /m ) aP aP a
For adsorption of solute in the solution, the expression for 1 1 1 b
Freundlich adsorption isotherm can be given as: \ = ¥ +
x /m a P a
x ∫ y = mx + C
= KC1/ n where, C fi Concentration of solution.
m
Slope = 1
a
Logarithmic form of Freundlich adsorption Isotherm
1
x/m
Slope = 1
n q
x
log m b
q Intercept = a
Intercept = log K 1/P
log P fi Note: 2
As we know Exception,
H2 + Glass (Endothermic)
x
= KP1/ n
m (II) Colloids Æ Colloids are heterogeneous system in
Taking log on both sides which a substance (dispersed phase) is dispersed as
x 1 very fine particles in dispersion medium.
log = log K + log P
m n
IIT JEE PC-V2_05.indd 3 5/12/2017 6:30:13 PM
5.4
Æ Colloid have two parts: 7. Ex- NaCl, KCl in Starch, Gum, Sand Iron
(i) Dispersed phase fi The substance which is pres- H2O Protein in hails in H2O
ent in lesser amount just like solute in true solution H2O
is known as dispersed phase.
(ii) Dispersion medium fi The substance which is * Classification of Colloids:
present in greater amounts just like solvent in true (A) On the basis of physical state of dispersed phase and
solution is known as dispersion medium. dispersion medium.
* Classification of solution on the basis of size of
particles. Dispersed Dispersion Colloids Examples
Phase medium
Properties True Solution Colloids Suspension
Solid Solid Solid Sol Gem Stones
1. Size of Less than 1 between Greater then
Solid Liquid Sol Paints
particle nm 1-1000 nm 1000 nm
Solid Gas Aerosol Dust
2. Nature Homogenous Heterog- Heterog- Liquid Solid Gel Cheese, Jellies
enous enous Liquid Liquid Emulsion Milk, Hair Cream
3. Visibility Not visible Visible under Visible with Liquid Gas Aerosol Fog, Cloud
microscope naked eye Gas Solid Solid Sol Pumice Stone
4. Separation Gas Liquid Foam Froth
(i) Filter paper ¥ ¥ ÷
(B) On the basis of Dispersion Medium
(ii) Membrane ¥ ÷ ÷
5. Tyndall Do not show Shows May or may Dispersion Medium Colloid
effect not show
H2O Hydrosol OR Aquasol
6. Settling Do not settle Does not set- Settles
Benzene Benzosol
down of tles but under down
Air Aerosol
particles ultra centrif-
Alcohol Alcosol
ugation they
can settle
(C) On the basis of Interaction in between dispersed phase and dispersion medium
Lyophilic (Solvent loving) Lyophobic (Solvent lating)
(Hydrophilic)
(i) When the particles of Dispresedphase have a greater (i) When the particles of Dispresedphase have a lesser
the colloids are known as Lyophilic. colloids are known as hyophobic.
(ii) Easily prepared just by mixing the dispersed phase (ii) It requires special methods for preparation.
with dispersion medium.
(iii) More stable (iii) Less stable (requires, stabilising agent)
(iv) Reversible in nature (iv) Irreversible in nature
(v) Coagulation is not so easy. (v) Easily coagulated
(vi) Higher degree of solvation or hydration (vi) Lower degree of hydration or solvation
(vii) Viscosity is more and surface tension is less as com- (vii) Viscosity and surface tension are nearly the same as
pared to that of dispersion medium that of dispersion medium.
For example, generally of organic nature like sols of protein, For example, generally of inorganic nature like sols of met-
starch, etc. als metal sulphides, etc.
(D) On the basic of type of particles of dispersed phase size lies in the colloidal range such type of systems are
(i) Multi-molecular colloids known as multi-molecular colloids.
On dissolution a large number of atoms or smaller For Example,
An (gold) sol and sulphur solution in sulphur sol
molecules aggregate together to form particles whose 1 Dispresed phase particle = 1000 or more S8 molecules
IIT JEE PC-V2_05.indd 4 5/12/2017 6:30:14 PM
Surface Chemistry 5.5
(ii) Macro-molecular colloids Methods of Preparation
Macro-molecules (having higher molecular mass) in a 1. Lyophilic colloids
suitable solvent forms solution in which their size lies They are easily prepared just by mixing the particles of
in the colloidal range such systems are known as mac- dispersed phase with the dispersion medium.
ro-molecular colloids. 2. Lyophobic Colloids (intrinsic colloid)
For example, Starch, protein (Natural macro-molecules) These colloids are prepared by the following methods:
Nylon, polyethene (Man made macro-molecules) (A) Dispersion method
(B) Condensation method
(iii) Associated colloids
Æ There are some substances which behave as strong
electrolytes at lower concentration but at higher (A) Dispersion Method
concentration they exhibit colloidal behaviour due In these methods larger particles are broken into smaller par-
ticles.
to formation of aggregates. The aggregate thus
(i) Mechanical Dispersion
formed are known as micelles and the system is
Inlet (Suspension)
known as associated colloids.
Æ The temperature and the concentration above Metal
which the formation of micelles occurs is known plates
as craft temperature and Critical Micelle Concen-
tration (CMC). Outlet Outlet
For example, Surface acting agents (Surfactant’s) like
soap and detergent. Colloidal Mill
(A) Anionic Surfactants In this method, the particles of the suspension are grind-
Sodium stearate fi C17H35COO–Na+ (Soap) sodium lauryl ed to colloidal sized particles by placing it in between
sulphate two metal plates that rotates in opposite direction in a
Colloidal mill. This method is used for preparing the
CH 3 (CH 2 )11SO 4- Na + (Detergent) colloids of paints, varnises, dies, etc.
(B) Cationic surfactants (ii) Electrical dispersion Bridics Arc Method
Catyl Trimethyl ammonium Chloride fi C16H33(CH3)3N+Cl–
Octadecyl ammonium chloride fi C18 H37 NH3+ Cl- Metal electrodes
fi Associated colloids have both lyophilic and hyophobic H2O (Dispersion Medium)
part. A micelle may contain 100 or more ions. +
KOH
Mechanism of micelle formation (stabilising agent)
C17H35COONa Æ C17H35COO– (stearate) + Na+ Ice Bath
or RCOONa and RCOO– and Na+ Æ In this method, on striking an electric arc in be-
Head tween the metal electrodes result in the vapouri-
(Hydrophilic) sation of metal which is then condensed by the
polar effect of ice bath. This metal disperses in water
Tail and hence forms a colloid. This method is used for
(Non polar) preparing colloids of metal like Au, Ag, Pt, etc.
(iii) Peptisation
Fe(OH)3 + FeCl3 h Fe(OH)3|Fe3+
Precipitate Electrolyte Colloid
Precipitate + Dispersion Medium + Electrolyte (Small
amount) fi Sol.
ÈPeptidising agent ˘
Electrolyte used as ÍÍ i.e.,stabilising ˙˙
(a) Arrangement of stearate (b) Micelle formation on ÍÎ agent ˙˚
ions on the surface of at CMC. (Non-polar tails
water before CMC. towards centre. Polar on Æ The process of converting a freshly prepared
surface of sphere. Precipitate into a colloidal solution by adding elec-
trolyte is known as peptisation.
IIT JEE PC-V2_05.indd 5 5/12/2017 6:30:14 PM
5.6
Æ The electrolyte used in this process are known as 2. Electrodialysis: This method is used when the impuri-
peptising agents. ties are electrolytes.
Æ This process involves the adsorption of suitable 3. Ultrafiltration:
ion (common ion) on the surface of Precipitate Ultra filter paper = Filter paper + Collodion solution
provided by the added electrolyte. As, a result the [4% Nitro – Cellulose is a mixture of alcohol and ether]
charged particles repell each other and thus forms
a colloid. Impure Colloidal
Ultrafiltres
paper Solution
(B) Condensation Method
In these methods smaller particle are aggregated together to
the colloidal size.
(i) Chemical Method after some time
double
Æ As 2O3 + 3H 2S æææææÆ
decomposition
As 2S3 (sol) + 3H 2O
Oxidation Pure
Æ H 2S + Br2 ææææÆ 2HBr + S(sol)
Colloidal
Reduction
Æ 2AuCl3 + 3SnCl2 ææææÆ 2Au(sol) + 3SnCl4 particle
Hydrolysis
Æ FeCl3 + 3H 2O ææææÆ Fe(OH)3 (sol) + 3HCl Impurities
Æ In chemical reactions the molecules form aggre-
gate together to form particles that lie in the col- 4. Ultra centrifugation: In this method, the tube is ro-
loidal range. tated at a very high speed as a result of which pure col-
(ii) Physical Methods loidal particles settle down and the impurities remain
(a) By exchange of solvent: In this method, a true so- in the solution. These pure colloidal particles are col-
lution is mixed with an excess of solvent in which lected and mixed with the dispersion medium to form
the solute is less soluble and the solvent is com- colloidal solutions.
pletely miscible.
ÈSulphur in C2 H 5OH ˘ ÈSulphur in Alcoholic H 2O ˘ PROPERTIES OF COLLOIDAL
Í True solution ˙ + H 2O = Í ˙ SOLUTIONS
Î ˚ Î Colloidal ˚
(b) By excessive cooling: In this method on excessive 1. Heterogeneous nature:
cooling, the molecules of a substance condense to- 2. Visibility: Colloidal particles are too small to be seen
gether to form particles of the colloidal range. with naked eye, but they become visible as bright spots
against dark background when viewed through an ultra
On excessive
H 2O + CHCl3 æææææ
Cooling
Æ ice + CHCl3 fi Colloidal microscope due to scattering of light caused by them.
3. Filterability: The size of solute particles is smaller
(c) By condensing vapors: In this method the vapour than the pore size of filter paper, and therefore, they
of sulphur or Hg are passed through cold water can readily pass through ultra filters, parchment paper,
containing ammonium nitrate.
or animal membrane.
4. Surface tension and viscosity: The surface tension and
PURIFICATION OF COLLOIDS viscosity of lyophobic sols are not very different from
those of the dospersion medium. On the other hand,
1. Dialysis: lyophilic sols show higher viscosity and lower sur-
face tension in comparison to the dispersion medium.
H2 O + 5. Colligative properties: very low due to high molar
Dialysing dissolved mass.
membrane Impure Impurities
Colloidal 6. Brownian movement: The colloidal particles of a col-
solution
loidal solution when viewed through an ultramicroscope
Inlet
Dialyser show a constant zig – zag motion. This type of motion
was first observed by robert brown and is known as
Æ This process involves the diffusion of dissolved
brownian movement. This motion is independent of the
impurities through the membrane and leaving be-
hind a pure colloidal solution. nature of the colloid but depends on the size of the par-
IIT JEE PC-V2_05.indd 6 5/12/2017 6:30:15 PM
Surface Chemistry 5.7
ticles and the viscosity of solution. Smaller the size and The direction of the flow of water (dispresion
lesser the viscosity, faster is the motion. The Brownian medium)depends on the charge of the colloid. For
movement is due to the unbalanced bombardment of the positively charged sols, the medium is negatively
particles by the molecules of the dispersion medium. charged, and hence the flow will take place from
7. Optical Properties: Tyndall effect: The scattering of one side to other. On the other hand, for negative-
ly charged sols, the reverse will occur.
light by the colloidal particles in a colloidal solution
9. Coagulation or precipitation or floculation of col-
is known as tyndall effect as it was first observed by
loids:-
Faraday and later studied in detail by Tyndall. Tyndall
The process of settling of colloidal particles is called
effect is caused by the scattering of blue part of light by
coagulation or Precipitation of the sol. The coagu-
the colloidal particles. lation of the lyophobic sols can be carried out in the
Tyndall effect is observed only when the following following ways.
two conditions are satified: (a) By electrophoresis
The diameter of the dispersed particles is not much (b) By mixing two oppositely charged sols
smaller than the wavelength of the light used. The re- (c) By boiling
fractive indices of the dispersed phase and the disper- (d) By persistent dialysis
sion medium must differ greatly in magnitude. (e) By addition of electrolytes
Zsigmondy:- Tyndall effect to set up an apparatus Colloids Around US
known as ultramicroscope. (a) Blue colour of the sky
8. The electrical properties of colloidal solutions are re- (b) Fog, mist and rain
lated with two phenomena: (c) Food articles :- Milk, butter, halwa
Reservoir (d) Blood :- It is a colloidal solution of an albuminoid sub-
stance.
(e) Soils :-
Cathode
(f) Formation of delta:-
Anode
Application of colloids
(a) Electrical precipitation of smoke
(b) Purification of drinking water
(c) Medicines
Initial level (d) Tanning
(e) Cleasing action of soaps and dettergents
Water Colloidal (f) Photographic plates and films
(dispersion solution
medium) (g) Rubber industry
(h) Industrial products
Zeta potential:
The potential difference between the fixed layer and the dif-
Stop cock fused layer of opposite charges is called electro kinetic poten-
tial or zeta potential.
(i) Electrophoresis: The existance of charge on col- AgI I– K+
loidal particles is determined by electrophoresis. – +
AgI I K
The phenomenon of the movement of col- Mobile layer
AgI I– K+
loidal particles under an applied electric field is
called electrophoresis. The positive charged par- AgI I– K+
ticles move towards the cathode while negatively Fixed layer
charged particles move toward the anode.
(ii) Electro – osmosis: Electro – osmosis may be de- Hordy sulzi rule: Greater the valency of the active ions or
fined as a phenomenon in which the molecules of flocculating ion greater will be its flocculating power
the dispersion medium are allowed to move under For example,
the influence of an electric field whereas colloidal (i) Al+3 > Mg+2 > Na+ for –ve sol.
particles are not allowed to move.
(ii) [Fe(CN)6 ]-4 > PO 4-3 > SO 4-2 > Cl- for + ve sol
IIT JEE PC-V2_05.indd 7 5/12/2017 6:30:15 PM
5.8
Gold Number: The protective power is measured in terms Positively charged colloid Negatively charged colloid
of gold number. It is defined as the number of milligrams of Hydrated metallic oxide Metals Cu, Ag, Au, Sol
a lyophilic colloid that will just prevent the precipitation of Al2O3. xH2O, CrO3. xH2O, Metalic sulphides As2S3,
10 mL of a gold sol on the addition of 1 mL of 10% NaCl Fe2O3. xH2O Sb2S3, CdS sol
solution Basic dye stuffs methylene Acid dye stuff eosin, congo
1 blue sol, red
Protective Power μ Haemoglobin (blood)
Gold Number
Oxide TiO2 Sol Sols of starch, gum gelatin,
clay
Solved Examples
1. For a linear plot of log (x/m) versus log P in a Freun- x 1 =1
dlich adsorption isotherm, which of the following Sol. (d) log = log K + log P n
m n x
statements is correct? (k and n are constants) log m
(a) 1/n appears as the intercept. y = +c + mx
tan 45 = 1 log K
(b) Only 1/n appears as the slope.
(c) log (1/n) appears as the intercept x
log = log 10 + 1 log (0.5) log P
(d) Both k and 1/n appear in the slope term. 1
Sol. (b) log x = log 10 + 1 ¥ log [5 ¥ 10–1]
x log x
1 log x = 1 + (–1) + .6990 = .6990 = log 5
= Kp n m
m \ x = 5 gram
1
x 1 n 4. Gold number is a measure of
log = log K + log P (a) stability of colloidal system.
m n
(b) coagulating power of a colloid.
y = + C + mx log P
(c) size of colloidal particles.
2. The coagulation values in mil- (d) efficiency of the protective colloid.
limoles per litre of the electrolytes used for the coagu- Sol. (d) Smaller is the gold number, higher is its protecting
lation of As2S3 are given below: power.
I. (NaCl) = 52 II. (BaCl2) = 0.69 Gelatin – .005 to .01
III. (MgSO4) = 0.22 Haemoglobin – 0.03
The correct order of their coagulating power is Gum – 0.15
(a) III > I > II (b) I > II > III Egg albumin – .08–.10
(c) II > I > III (d) III > II > I Starch – 25–40
Sol. (d) Gold No. The minimum amount in milligram of pro-
1 tective colloid which must be added to 10 mL gold sol
Coagulation value μ
Coagulating power in order to prevent its coagulation against 1 mL 10%
NaCl (52) BaCl2(0.69) MgSO4(0.22) NaCl solution.
(I) (II) (III) Ê xˆ
5. Graph between log Á ˜ vs log P is provided for ad-
\ coagulating power for As2S3 Ë m¯
MgSO4 > BaCl2 > NaCl sorption of NH3 gas on metal surface.
III > II > I Calculate weight of NH3 gas adsorbed by 50 g of metal
3. Plot of log ÊÁ ˆ˜ against log P is a straight line inclined
x surface at 2 atm pressure.
Ë m¯
at an angle of 45°. When the pressure is 0.5 atm and
Freundlich parameter (K) is 10, then the amount of sol-
ute adsorbed per gram of adsorbent will be (log 5 = 45°
log (x/m)
0.699): A
(a) 1 g (b) 6.99 g 0.3
(c) 3 g (d) 5 g
log P
IIT JEE PC-V2_05.indd 8 5/12/2017 6:30:15 PM
Surface Chemistry 5.9
(a) 100 g (b) 75 g DS = –ve
(c) 200 g (d) 50 g DG = –ve
1 DSurr. = 0
Sol. (c) slope = = tan 45∞ = 1 7. Fe (SCN)n3–n absorb colour because of the complex
n
Fe(SCN)n3–n only, whereas Fe3+ and SCN– both are co-
slope = 1
n = tan 45° = 1 lourless. In the given table, the equilibrium concentra-
tion of Fe3+ and SCN– and corresponding absorbance
45° are given–
x
log m
log K = 0.3 = log 2 Exp. No. [Fe3+] [SCN–] Absorbance
1 3 ¥ 10 (M)
–3
(10 M) 3
log P
2 3 ¥ 10–3(M) (10)2/3(M) 0.3
Considering absorbance proportional to concentration
x 1 of the complex, the value of n is
= KP n (a) 3 (b) 6 (c) 4 (d) 2
m
3+ – ææ Æ 3–n
x 1 Sol. (a) Fe + nSCN ¨æ æ Fe(SCN)n
log = log K + log p
m n
[Complex]
1
= log 2 + log 2 K form =
n [Fe3+ ][SCN - ]n
x { [complex] = Kform [Fe3+][SCN–]n
log = log 2 + (1) log 2
m
\ log [complex] = log Kform + log [Fe3+] + n log [SCN–]
x
=2¥2 [Fe3+] is constant, hence
m
x [Complex]1 [SCN – ]1
=4 log = n log
50 [Complex]2 [SCN – ]2
x = 200 g
10
6. Which of the following parameter is correct regarding \ log 3/0.3 = n log
adsorption of gases over solid? (10) 2/3
(a) DSsystem> 0 (b) DSsurrounding> 0 1
(c) DG > 0 (d) DH > 0 1= n ¥
3
Sol. (b) Adsorption
DH = –ve or n=3
IIT JEE PC-V2_05.indd 9 5/12/2017 6:30:16 PM
Das könnte Ihnen auch gefallen
- Chapter 3-1Dokument4 SeitenChapter 3-1Anirudh CNoch keine Bewertungen
- COLLOIDSconcise Notes OptDokument6 SeitenCOLLOIDSconcise Notes Optaleena'Noch keine Bewertungen
- Surface ChemistryDokument44 SeitenSurface ChemistrySuman SainiNoch keine Bewertungen
- Surface ChemistryDokument50 SeitenSurface ChemistryUDAY PanthNoch keine Bewertungen
- CY8151-Engineering Chemistry-431878289-Unit - 2 PDFDokument49 SeitenCY8151-Engineering Chemistry-431878289-Unit - 2 PDFHari Prakash100% (2)
- Surface Chemistry TheoryDokument23 SeitenSurface Chemistry TheorySara PatilNoch keine Bewertungen
- Surface ChemistryDokument15 SeitenSurface ChemistryAtharvJoshiNoch keine Bewertungen
- Surface Chemistry Resonance NoteDokument8 SeitenSurface Chemistry Resonance NoteSomya Kumar SinghNoch keine Bewertungen
- Surface ChemistryDokument18 SeitenSurface ChemistryTHONGNoch keine Bewertungen
- Surface ChemistryDokument22 SeitenSurface ChemistryPRanavNoch keine Bewertungen
- 01-Surface Chemistry-Theory-Final-E PDFDokument13 Seiten01-Surface Chemistry-Theory-Final-E PDFelve0% (1)
- 01-Surface Chemistry-Theory-Final-E PDFDokument13 Seiten01-Surface Chemistry-Theory-Final-E PDFNara TallaNoch keine Bewertungen
- Med RM Chem SP 2 CH 13 Surface ChemistryDokument24 SeitenMed RM Chem SP 2 CH 13 Surface Chemistrykrish masterjeeNoch keine Bewertungen
- Surface ChemistryDokument10 SeitenSurface ChemistryDivyanshi KatiyarNoch keine Bewertungen
- Surface Chemistry: by General CharacteristicsDokument8 SeitenSurface Chemistry: by General CharacteristicssriNoch keine Bewertungen
- PartitionDokument9 SeitenPartitionUdit GuptaNoch keine Bewertungen
- Surface Chemistry - Practically Study MaterialDokument20 SeitenSurface Chemistry - Practically Study MaterialreddygrNoch keine Bewertungen
- Surface ChemistryDokument48 SeitenSurface ChemistrySai Sasivardhan GampaNoch keine Bewertungen
- Surfaces Chemistry: Adsorption at Solids Solid: Adsorbent Gas/Solute: AdsorbateDokument31 SeitenSurfaces Chemistry: Adsorption at Solids Solid: Adsorbent Gas/Solute: AdsorbateAthinaNoch keine Bewertungen
- (5403) Sheet Surface Chemistry Theory eDokument24 Seiten(5403) Sheet Surface Chemistry Theory eboom rangNoch keine Bewertungen
- Surface Chemistry 2Dokument8 SeitenSurface Chemistry 2SITENDER NARWALNoch keine Bewertungen
- 1686335800Dokument90 Seiten1686335800itspraveenkumar55Noch keine Bewertungen
- A Premium Institute For CBSE, NEET & JEEDokument33 SeitenA Premium Institute For CBSE, NEET & JEEZUHAIB KAMALNoch keine Bewertungen
- Paer III Unit IDokument11 SeitenPaer III Unit IShubhanshu SrivastavNoch keine Bewertungen
- Surface ChemistryDokument6 SeitenSurface ChemistryMukesh Kumar SinghNoch keine Bewertungen
- Adsorption PresentationDokument23 SeitenAdsorption Presentationarun231187100% (1)
- Surface Chemistry: Ssnce DR.S.LDokument54 SeitenSurface Chemistry: Ssnce DR.S.LImthiaz AhmedNoch keine Bewertungen
- 12 - Surface Chemistry (Notes)Dokument27 Seiten12 - Surface Chemistry (Notes)DivyanshNoch keine Bewertungen
- Simultaneous Heat and Mass Transfer (SHMT) : CHE-402 Date: 19/11/2020Dokument17 SeitenSimultaneous Heat and Mass Transfer (SHMT) : CHE-402 Date: 19/11/2020Ali UsmanNoch keine Bewertungen
- UntitledDokument23 SeitenUntitledAnane Lawrence YeboahNoch keine Bewertungen
- Adsorption and Catalysis: Module - 5Dokument16 SeitenAdsorption and Catalysis: Module - 5TeachingTrainingCoaching KnowledgeSharingSessionNoch keine Bewertungen
- Surface Chem 1Dokument1 SeiteSurface Chem 1coding727treeNoch keine Bewertungen
- JEE Advanced 2023 Surface Chemistry Revision Notes - Free PDF DownloadDokument7 SeitenJEE Advanced 2023 Surface Chemistry Revision Notes - Free PDF Downloadhishamkalliyath19Noch keine Bewertungen
- Surface Chemistry NeetDokument13 SeitenSurface Chemistry Neetmanan10jas1529Noch keine Bewertungen
- Lecture Notes ADSORPTIONDokument15 SeitenLecture Notes ADSORPTIONtony frankNoch keine Bewertungen
- Adsorption Isotherms: Dr. K.K. Chahal Jaskirat Kaur (L-2016-BS-265-M)Dokument73 SeitenAdsorption Isotherms: Dr. K.K. Chahal Jaskirat Kaur (L-2016-BS-265-M)Asaad BashirNoch keine Bewertungen
- Adsopsi Pada Permukaan Padatan KatalisDokument13 SeitenAdsopsi Pada Permukaan Padatan Kataliskampus merdeka21Noch keine Bewertungen
- Surface Chemistry-JEE ADVANCEDokument26 SeitenSurface Chemistry-JEE ADVANCESitabai JadhavNoch keine Bewertungen
- Adsorption 1Dokument6 SeitenAdsorption 1Nurul Aqilah Mohd NasirNoch keine Bewertungen
- AdsorptionDokument20 SeitenAdsorptionSwag sprNoch keine Bewertungen
- Surface Chemistry Theory &assignmentDokument36 SeitenSurface Chemistry Theory &assignmentSerious BlackNoch keine Bewertungen
- Fluid-Solid Separation Proces: AdsorptionDokument35 SeitenFluid-Solid Separation Proces: AdsorptionNityantiniNoch keine Bewertungen
- Unit 5 Surface ChemistryDokument18 SeitenUnit 5 Surface Chemistryengineeringchemistry100% (1)
- Sem 2 CHM271 Chapter 7Dokument20 SeitenSem 2 CHM271 Chapter 7happyflowerNoch keine Bewertungen
- Resonance Kvpy ChemistryDokument117 SeitenResonance Kvpy ChemistryHarish MahadevanNoch keine Bewertungen
- Adsorption IsothermsDokument62 SeitenAdsorption IsothermsAsaad BashirNoch keine Bewertungen
- Surface Chemistry Class 12 NOTES CBSEDokument23 SeitenSurface Chemistry Class 12 NOTES CBSERishit KalraNoch keine Bewertungen
- 1205 AdsorptionisothermDokument73 Seiten1205 AdsorptionisothermMohamed KhaledNoch keine Bewertungen
- XLL - Project - AdsorptionDokument12 SeitenXLL - Project - AdsorptionxxxNoch keine Bewertungen
- Surface ChemistryDokument39 SeitenSurface ChemistryDereje BeyeneNoch keine Bewertungen
- KVPY SB 2 SX StreamDokument117 SeitenKVPY SB 2 SX StreamashutoshNoch keine Bewertungen
- Revision Notes On Surface Chemistry - askIITians PDFDokument5 SeitenRevision Notes On Surface Chemistry - askIITians PDFHarsh ShahNoch keine Bewertungen
- 6 2021 09 18!01 01 20 AMttDokument4 Seiten6 2021 09 18!01 01 20 AMttEmran MuhammadNoch keine Bewertungen
- Adsorption (Surface Chemistry)Dokument15 SeitenAdsorption (Surface Chemistry)Salman AshrafNoch keine Bewertungen
- Surface ChemistryDokument22 SeitenSurface ChemistryiycbrthoratNoch keine Bewertungen
- Surface Chemistry: Absorption & Adsorption Section - 1Dokument32 SeitenSurface Chemistry: Absorption & Adsorption Section - 1Gaurav KumarNoch keine Bewertungen
- Surface Chem PDFDokument32 SeitenSurface Chem PDFAdarshNoch keine Bewertungen
- Chapter 7 AdsorptionDokument29 SeitenChapter 7 AdsorptionaliesyaNoch keine Bewertungen
- AdsroptionnDokument31 SeitenAdsroptionnAnonymous S6odqINoch keine Bewertungen
- Time Management PowerpointDokument23 SeitenTime Management PowerpointAkshat PandeyNoch keine Bewertungen
- Mitigating Co From Atmosphere Greenearthers Team Members: andDokument13 SeitenMitigating Co From Atmosphere Greenearthers Team Members: andAkshat PandeyNoch keine Bewertungen
- Chemical Kinetics AssignmentDokument17 SeitenChemical Kinetics AssignmentAkshat PandeyNoch keine Bewertungen
- The Enemy: Pearl S. BuckDokument9 SeitenThe Enemy: Pearl S. BuckAkshat PandeyNoch keine Bewertungen
- Delhi Private School Dubai Chapter 3 Current Electricity Worksheet 1Dokument2 SeitenDelhi Private School Dubai Chapter 3 Current Electricity Worksheet 1Akshat PandeyNoch keine Bewertungen
- Delhi Private School Dubai Chapter 3 Current Electricity Worksheet 2Dokument2 SeitenDelhi Private School Dubai Chapter 3 Current Electricity Worksheet 2Akshat PandeyNoch keine Bewertungen
- Delhi Private School Dubai Chapter 3 Current Electricity Worksheet 3Dokument2 SeitenDelhi Private School Dubai Chapter 3 Current Electricity Worksheet 3Akshat PandeyNoch keine Bewertungen
- Revise Physics in 1days PDFDokument141 SeitenRevise Physics in 1days PDFAkshat PandeyNoch keine Bewertungen
- Solutions - Index QnsDokument7 SeitenSolutions - Index QnsAkshat PandeyNoch keine Bewertungen
- Grade 11 Consortium ADokument7 SeitenGrade 11 Consortium AAkshat PandeyNoch keine Bewertungen
- SCIMA Rare Live Chem GR11Dokument7 SeitenSCIMA Rare Live Chem GR11Akshat PandeyNoch keine Bewertungen
- Gr5 English SA2 Revision Worksheet Answer KeyDokument11 SeitenGr5 English SA2 Revision Worksheet Answer KeyAkshat PandeyNoch keine Bewertungen
- Revise Chemistry in 1day PDFDokument196 SeitenRevise Chemistry in 1day PDFAkshat PandeyNoch keine Bewertungen
- Revise Chemistry in 1day PDFDokument196 SeitenRevise Chemistry in 1day PDFAkshat PandeyNoch keine Bewertungen
- Delhi Private School Dubai Assignment For The Month of October 2018-19 Theme-FoodDokument58 SeitenDelhi Private School Dubai Assignment For The Month of October 2018-19 Theme-FoodAkshat PandeyNoch keine Bewertungen
- GR 5 - Science ASSET Practice WS - 7Dokument6 SeitenGR 5 - Science ASSET Practice WS - 7Akshat PandeyNoch keine Bewertungen
- GR 5 - Asset Practice Ws - 6Dokument8 SeitenGR 5 - Asset Practice Ws - 6Akshat Pandey100% (1)
- Gr5 English SA2 Revision Worksheet Answer KeyDokument11 SeitenGr5 English SA2 Revision Worksheet Answer KeyAkshat PandeyNoch keine Bewertungen
- Why Processed Food Is Going Outdated?: Chemistry RareDokument9 SeitenWhy Processed Food Is Going Outdated?: Chemistry RareAkshat PandeyNoch keine Bewertungen
- Delhi Public School, Jodhpur: Section-ADokument16 SeitenDelhi Public School, Jodhpur: Section-AAkshat PandeyNoch keine Bewertungen
- Glaeser 2nd Chapter OneDokument28 SeitenGlaeser 2nd Chapter OneSheri DeanNoch keine Bewertungen
- LubeDokument14 SeitenLubearondaniel ocampoNoch keine Bewertungen
- XI - Chemistry - Surface Chemistry Module 1 - AssingnmentDokument4 SeitenXI - Chemistry - Surface Chemistry Module 1 - AssingnmentKk StudiesNoch keine Bewertungen
- Last Decade of Research On Activated Carbons As Catalytic SupportDokument57 SeitenLast Decade of Research On Activated Carbons As Catalytic SupportHPCBRASILNoch keine Bewertungen
- Industrial Tribology WearDokument23 SeitenIndustrial Tribology WearAtif HashmiNoch keine Bewertungen
- Industry Guide To The Technion MANLAM 2017Dokument244 SeitenIndustry Guide To The Technion MANLAM 2017V.LNoch keine Bewertungen
- Appendix: Galling Threshold StressDokument8 SeitenAppendix: Galling Threshold StressGarcia RafaelNoch keine Bewertungen
- Physical Chemistry Post Lab - Experiment 3 Surface TensionDokument8 SeitenPhysical Chemistry Post Lab - Experiment 3 Surface TensionGian BanaresNoch keine Bewertungen
- Lubricants: 11 February 2014Dokument2 SeitenLubricants: 11 February 2014Thai TrinhNoch keine Bewertungen
- Price List: SL Product Name & Description Pack Size Price Price (Per Liter) 1 Industrial Gear OilDokument4 SeitenPrice List: SL Product Name & Description Pack Size Price Price (Per Liter) 1 Industrial Gear OilSadid ShahzadNoch keine Bewertungen
- Lecture 1 Part 1 Introduction To Tribology v1Dokument15 SeitenLecture 1 Part 1 Introduction To Tribology v1Aziful AimanNoch keine Bewertungen
- U Mrezi Strasti - Dajana Hamilton - U-Mrezi-StrastiDokument14 SeitenU Mrezi Strasti - Dajana Hamilton - U-Mrezi-StrastiJohnny CrossNoch keine Bewertungen
- Disperse Systems: Tim Dosen Farmasi FisikaDokument73 SeitenDisperse Systems: Tim Dosen Farmasi FisikairmaNoch keine Bewertungen
- Table of Specification Science 6 Quarter 1: Cognitive DomainsDokument2 SeitenTable of Specification Science 6 Quarter 1: Cognitive DomainsGlen Mark MacarioNoch keine Bewertungen
- EmulsionsDokument24 SeitenEmulsionsabdullah2020Noch keine Bewertungen
- Explanation of False Versus True BrinellingDokument7 SeitenExplanation of False Versus True BrinellingHaitham YoussefNoch keine Bewertungen
- Group B1-1 Surface Phenomena Lab Report - FinalDokument25 SeitenGroup B1-1 Surface Phenomena Lab Report - FinalNurnajwa IzzatiNoch keine Bewertungen
- Subject Tribology SyllabusDokument3 SeitenSubject Tribology SyllabusbmdbmdbmdNoch keine Bewertungen
- Emulsions FDA 1694370790Dokument50 SeitenEmulsions FDA 1694370790Amruta JethwaNoch keine Bewertungen
- Spectrochimica Acta Part A: Molecular and Biomolecular SpectrosDokument9 SeitenSpectrochimica Acta Part A: Molecular and Biomolecular SpectrosJesusNoch keine Bewertungen
- Surface Chemistry MCQs - Questions - Paper 1Dokument7 SeitenSurface Chemistry MCQs - Questions - Paper 1krishna prasad ghantaNoch keine Bewertungen
- Journal of Petroleum Science and Engineering: Mohammad Ali Ahmadi, Seyed Reza ShadizadehDokument9 SeitenJournal of Petroleum Science and Engineering: Mohammad Ali Ahmadi, Seyed Reza ShadizadehdsfsfNoch keine Bewertungen
- Coldroom Shelf AaDokument7 SeitenColdroom Shelf Aaanon_622910368Noch keine Bewertungen
- Hydraulic Statement OF THAMSI MANDALDokument3 SeitenHydraulic Statement OF THAMSI MANDALMaheshNoch keine Bewertungen
- CV ModelDokument44 SeitenCV ModelTumuluri SreenivasNoch keine Bewertungen
- Antifoams For Cutting FluidsDokument8 SeitenAntifoams For Cutting Fluidsiskricaman7Noch keine Bewertungen
- Activated Carbon Literature Review-FinalDokument119 SeitenActivated Carbon Literature Review-FinalLuis VilchezNoch keine Bewertungen
- CHEMISTRYDokument27 SeitenCHEMISTRYKhanNoch keine Bewertungen
- Catalogo Rolamento CPMDokument11 SeitenCatalogo Rolamento CPMCesar PimentaNoch keine Bewertungen
- Steric StabilizationDokument2 SeitenSteric StabilizationKrishanu SahaNoch keine Bewertungen
- Hero Found: The Greatest POW Escape of the Vietnam WarVon EverandHero Found: The Greatest POW Escape of the Vietnam WarBewertung: 4 von 5 Sternen4/5 (19)
- Dirt to Soil: One Family’s Journey into Regenerative AgricultureVon EverandDirt to Soil: One Family’s Journey into Regenerative AgricultureBewertung: 5 von 5 Sternen5/5 (125)
- The End of Craving: Recovering the Lost Wisdom of Eating WellVon EverandThe End of Craving: Recovering the Lost Wisdom of Eating WellBewertung: 4.5 von 5 Sternen4.5/5 (82)
- Sully: The Untold Story Behind the Miracle on the HudsonVon EverandSully: The Untold Story Behind the Miracle on the HudsonBewertung: 4 von 5 Sternen4/5 (103)
- The Fabric of Civilization: How Textiles Made the WorldVon EverandThe Fabric of Civilization: How Textiles Made the WorldBewertung: 4.5 von 5 Sternen4.5/5 (58)
- The Beekeeper's Lament: How One Man and Half a Billion Honey Bees Help Feed AmericaVon EverandThe Beekeeper's Lament: How One Man and Half a Billion Honey Bees Help Feed AmericaNoch keine Bewertungen
- The Future of Geography: How the Competition in Space Will Change Our WorldVon EverandThe Future of Geography: How the Competition in Space Will Change Our WorldBewertung: 4 von 5 Sternen4/5 (6)
- Mini Farming: Self-Sufficiency on 1/4 AcreVon EverandMini Farming: Self-Sufficiency on 1/4 AcreBewertung: 4 von 5 Sternen4/5 (76)
- Permaculture for the Rest of Us: Abundant Living on Less than an AcreVon EverandPermaculture for the Rest of Us: Abundant Living on Less than an AcreBewertung: 4.5 von 5 Sternen4.5/5 (33)
- When the Heavens Went on Sale: The Misfits and Geniuses Racing to Put Space Within ReachVon EverandWhen the Heavens Went on Sale: The Misfits and Geniuses Racing to Put Space Within ReachBewertung: 3.5 von 5 Sternen3.5/5 (6)
- Four Battlegrounds: Power in the Age of Artificial IntelligenceVon EverandFour Battlegrounds: Power in the Age of Artificial IntelligenceBewertung: 5 von 5 Sternen5/5 (5)
- System Error: Where Big Tech Went Wrong and How We Can RebootVon EverandSystem Error: Where Big Tech Went Wrong and How We Can RebootNoch keine Bewertungen
- Fire on the Horizon: The Untold Story of the Gulf Oil DisasterVon EverandFire on the Horizon: The Untold Story of the Gulf Oil DisasterNoch keine Bewertungen
- The Intel Trinity: How Robert Noyce, Gordon Moore, and Andy Grove Built the World's Most Important CompanyVon EverandThe Intel Trinity: How Robert Noyce, Gordon Moore, and Andy Grove Built the World's Most Important CompanyNoch keine Bewertungen
- Pale Blue Dot: A Vision of the Human Future in SpaceVon EverandPale Blue Dot: A Vision of the Human Future in SpaceBewertung: 4.5 von 5 Sternen4.5/5 (588)
- The Technology Trap: Capital, Labor, and Power in the Age of AutomationVon EverandThe Technology Trap: Capital, Labor, and Power in the Age of AutomationBewertung: 4.5 von 5 Sternen4.5/5 (46)
- Highest Duty: My Search for What Really MattersVon EverandHighest Duty: My Search for What Really MattersNoch keine Bewertungen
- Faster: How a Jewish Driver, an American Heiress, and a Legendary Car Beat Hitler's BestVon EverandFaster: How a Jewish Driver, an American Heiress, and a Legendary Car Beat Hitler's BestBewertung: 4 von 5 Sternen4/5 (28)
- Transformed: Moving to the Product Operating ModelVon EverandTransformed: Moving to the Product Operating ModelBewertung: 4 von 5 Sternen4/5 (1)
- Restoration Agriculture: Real-World Permaculture for FarmersVon EverandRestoration Agriculture: Real-World Permaculture for FarmersBewertung: 4.5 von 5 Sternen4.5/5 (86)
- The Knowledge: How to Rebuild Our World from ScratchVon EverandThe Knowledge: How to Rebuild Our World from ScratchBewertung: 3.5 von 5 Sternen3.5/5 (133)
- How to Build a Car: The Autobiography of the World’s Greatest Formula 1 DesignerVon EverandHow to Build a Car: The Autobiography of the World’s Greatest Formula 1 DesignerBewertung: 4.5 von 5 Sternen4.5/5 (54)
- ChatGPT Money Machine 2024 - The Ultimate Chatbot Cheat Sheet to Go From Clueless Noob to Prompt Prodigy Fast! Complete AI Beginner’s Course to Catch the GPT Gold Rush Before It Leaves You BehindVon EverandChatGPT Money Machine 2024 - The Ultimate Chatbot Cheat Sheet to Go From Clueless Noob to Prompt Prodigy Fast! Complete AI Beginner’s Course to Catch the GPT Gold Rush Before It Leaves You BehindNoch keine Bewertungen
- The Assassination Complex: Inside the Government's Secret Drone Warfare ProgramVon EverandThe Assassination Complex: Inside the Government's Secret Drone Warfare ProgramBewertung: 4 von 5 Sternen4/5 (55)
- The Weather Machine: A Journey Inside the ForecastVon EverandThe Weather Machine: A Journey Inside the ForecastBewertung: 3.5 von 5 Sternen3.5/5 (31)
- Process Plant Equipment: Operation, Control, and ReliabilityVon EverandProcess Plant Equipment: Operation, Control, and ReliabilityBewertung: 5 von 5 Sternen5/5 (1)
- The Manager's Path: A Guide for Tech Leaders Navigating Growth and ChangeVon EverandThe Manager's Path: A Guide for Tech Leaders Navigating Growth and ChangeBewertung: 4.5 von 5 Sternen4.5/5 (99)