Beruflich Dokumente
Kultur Dokumente
Group Report 1 Analytical Chemistry
Hochgeladen von
sssxxx2ndOriginalbeschreibung:
Originaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Group Report 1 Analytical Chemistry
Hochgeladen von
sssxxx2ndCopyright:
Verfügbare Formate
Group 2 Fransiskus Adithya Ivan Ardianto Puspita Anggreaini William Andreas
Group 2
Group Report 1 Analytical Chemistry
Electrochemistry and Potentiometry
Group Report PBL-1 Analytical Chemistry Group 1
Table of Contents
Problem Definition.............................................................................................................................3 Background Theory ............................................................................................................................3 Electrochemistry ................................................................................................................................3 Oxidation/ Reduction Concept ................................................................................................................. 3 Types of Electrochemical Cells .................................................................................................................. 3 Effect of Concentration on Electrode Potentials The Nernst Equation ................................................. 4 NiCd Battery .............................................................................................................................................. 4 Memory Effect .......................................................................................................................................... 5 Application of NiCd Battery and compared with other types of batteries ............................................... 5 Impact in environment ............................................................................................................................. 6 Potentiometry ...................................................................................................................................6 Direct Potentiometric technique .............................................................................................................. 6 Standard Addition technique .................................................................................................................... 7 Excess standard addition and Direct Potentiometric .............................................................................. 7 Trigger Problem Answers ...................................................................................................................7 Assignment I.............................................................................................................................................. 7 Assignment II........................................................................................................................................... 12 Assignment III.......................................................................................................................................... 14 References....................................................................................................................................... 16 Learning Scheme (Mindmap) of Electrochemistry and Potentiometry.................................................16
2|Page
Group Report PBL-1 Analytical Chemistry Group 1
Problem Definition
1. To understand the concept of batteries in correlation to the electrochemical concept. 2. To understand the concept of potentiometry to determine the concentration of heavy metals, such as copper.
Background Theory
Electrochemistry
Oxidation/ Reduction Concept
1. Oxidation Reduction reactions can be conducted in 2 ways : a. Direct contact between the oxidants and reductans. b. In a reaction in which the reactants do not come in direct contact with one another. 2. Example: Immersing a strip of copper (Cu) in a solution containing silver nitrate (AgNO3). Here, there is an oxidation and reduction reaction happens between the copper and the solution. Reduction (Silver Nitrate) CATHODE Oxidation (Cooper) ANODE Ag+ + e- Ag(s) Cu Cu2+ + 2e-
Oxidation and reactions has an unique characteristic of the transfer of electrons. Note that in the 2nd type of the oxidation reduction, a salt bridge isolates the reactants but maintain electrical contact between the CATHODE and ANODE cells. 3. The voltmeter measures the potential difference between the two metals at any instant, and there is a tendency of the potential decrease approaching 0 V as the reaction approaches the state of equilibrium. When zero voltage is reached, the concentrations of the 2 ions (in this case the Ag (I) and Cu (II) will have values that satisfy the equilibrium-constant expression for the net reduction/oxidation reaction 2 Ag+ + Cu(s) 2Ag(s) + Cu2+ At the equilibrium condition, there is no further flow of electrons will occurs.
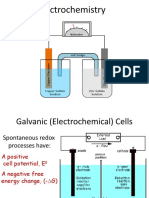
Types of Electrochemical Cells
Electrochemical cells are either galvanic or electrolytic, and can also classified as reversible or irreversible. The table below describes the difference between the galvanic and electrolytic cells. Galvanic (Voltaic) Store electrical energy. Proceed spontaneusly. Electrolytic Need / consume electricity. Occurs at the reverse of the galvanic cell reactions. Cannot be conducted spontaneusly.
3|Page
Group Report PBL-1 Analytical Chemistry Group 1 A reversible cell is a electrochemical cell that can be reversed either in galvanic or electrolytic condition, while an irreversible cell, the cell cannot be reversed because it could cause an entirely different halfreaction to occur at both electrodes.
Effect of Concentration on Electrode Potentials The Nernst Equation
There is a quantitative relationship between concentration and the potential difference value of the electrodes. Consider the reversible half reaction: aA + bB + ... + ne- cC + dD + ... where the capital letters represent chemical formulas for the participating species (atoms, molecules, or ions), e- represents electrons, and the lower case italic letters indicates the number of moles of each species appearing in the half-reaction as it has been written. The electrode potential E for this process is described by the equation [ ] [ ] [ ] [ ] Where Eo = the standard electrode potential, which is a characteristics constant for each half-reaction R = the gas constant 8.314 J K-1 mol-1 T= temperature in kelvins n= number of moles of electrons that appear in the half-reaction for the electrode process as it has been written F= the faraday = 96,485 C (coulombs) Ln= the natural logarithm = 2.303 log Substituting numerical values for the constants, converting to base 10 logarithms, and specifying 25 oC for the temperature give [ ] [ ] [ ] [ ] Both equations above are named before Walther Hermann Nerst, a German physical chemist.
NiCd Battery
The nickel-cadmium battery (commonly abbreviated NiCd or NiCad ) is a type of rechargeable battery using nickel oxide hydroxide and metallic cadmium as electrodes . the components of NiCd battery are: Cathode Anode : Nickel (III) NiO(OH) : Cadmium
4|Page
Group Report PBL-1 Analytical Chemistry Group 1
A separator Electrolytes
: alkaline (KOH)
The reaction in electrodes when NiCd battery discharge:
(at anode)
(at cathode)
The reaction happen from left side to right side when discharge and rechargeable at the opposite.
Memory Effect
NiCd batteries may suffer from a " memory effect " if they are discharged and recharged to the same state of charge hundreds of times. The apparent symptom is that the battery "remembers" the point in its charge cycle where recharging began and during subsequent use suffers a sudden drop in voltage at that point, as if the battery had been discharged. if the device is unable to operate through this period of decreased voltage, it will be unable to get enough energy out of the battery, and for all practical purposes, the battery appears "dead" earlier than normal. The mechanism of memory effect is: the active material cadmium in NiCd Battery is small crystals which cover all the surface of NiCd cells. When memory effect happened, the crystals cover the active materials (Ni,Cd) and cause the drop voltage.
Application of NiCd Battery and compared with other types of batteries
NiCd battery use in many thing, for example the primary cell of NiCd battery usually use in electronic portable and toys. The main application of NiCd battery in cordless telephone and wireless, emergency lamps, and in the plane. Because of the low resistance, the NiCd battery has a high voltage current, and usually use in remote control, and camera, electric vehicles and standby power. Types of Batteries Type of Battery 1.Lead acid Battery Information Comparison with NiCd Battery
Have a higher density than NiCd. NiCd battery is smaller and Anode: Pb; Cathode: PbO2; lighter than lead acid battery Electrolyte : H2SO4. The main application in otomotive Irreversible chemical reaction in NiCd battery has smaller capacity alkaline battery. The voltage of and need high cost to produce.
2.Alcaline Battery
5|Page
Group Report PBL-1 Analytical Chemistry Group 1 alkaline battery was drop when NiCd battery last longer and charge drop. Many of them are keep the constant voltage when disposable battery. Anode: Zn; discharge. cathode: MnO2; electrolyte: Zink cloride 3.Lithium Ion Battery Rechargeable, and will broke NiCd battery when discharge in minimum (rechargeable) voltage. Anode: lithium; cathode and electrolyte: variation. The main application in phonecell, laptop, computer, and camera NiMH has bigger capacity and not toxic because the hydride adsorb the alloy in anode. Low cost to produce. The main applicationin hydride vechicle and prototype humanoid robot. last longer
4. Nickel Hydrida Battery (NiMH)
NiCd battery has low self discharge, 20%/ month (NiCd battery, 30%/month). NiCd not cause voltage decreases. Low resistance cause the high charge flow.
Impact in environment
All of the batteries have chemical materials which cause dangerous impact in environment. The most dangerous materials are heavy metals which carcinogenic, example: mercury and cadmium. It can cause substantial pollution when land filled or incinerated. Because of this, many countries now operate recycling programs to capture and reprocess old batteries. For the people, it can cause cancer, health problem, and died.
Potentiometry
Direct Potentiometric technique
This technique requires only an indicator of potential measurement of the electron when it is dipped in a solution containing an unknown concentration of an analyte and unknown. Indicator electrode is always considered as a cathode and reference electrode as the anode. For the direct potentiometry measurements, the cell potential can be expressed as a potential development by the indicator electrode, reference electrode, and potential functions. It depends on the potential difference measurement of electrodes in a solvent. Potential difference can be measured with pH meters/voltmeter. An electrodes is indicator electrode which use to give the response to the solvents.
6|Page
Group Report PBL-1 Analytical Chemistry Group 1 For calculations, the sign convention for direct potentiometry is the same as the convention mentioned in the general electrochemistry section for standard electrode potential. The indicator electrode is always treated as the right-hand electrode and the reference electrode as the left-hand electrode. The potential difference for the direct potentiometry follows this equation:
After a long derivation from the Nerstian form of the equation, and also considering the activity factor versus the concentration, the final equation that can be used: [ ] This equation effective for the cations. Meanwhile for the anions: [ ]
Standard Addition technique
This technique is commonly used in analytical instrumentation such as in atomic absorption spectroscopy and gas chromatography to find the value of the concentration of the substance (analyte) in a sample of unknown composition by comparison to samples of known concentration.
Excess standard addition and Direct Potentiometric
Calibration and measurement of samples done simultaneously so that the difference in ionic strength and temperature standards and the sample is not too significant. During the process, the electrodes remain immersed in the solution so that there is little change in junction potential solution. Measurement of slope very close to the concentration of the sample shows this method can yield more accurate results in the range of non-linear and can be used with electrodes old or older who was not linear range for the slope is stable.
Trigger Problem Answers
Assignment I
1. According to you why a particular black box on the plane should have its own backup power supply? What do you think are important issues to this topic? Answer: Backup power supply Because simply maintaining the operation of the flight data recorder would not be enough, all of the systems and sensors that require electrical power would also have to 7|Page
Group Report PBL-1 Analytical Chemistry Group 1 remain operative. Now for the cockpit voice recorder, we only need to power the recorder itself and the microphones in the cockpit. An independent power supply here might not be too difficult to implement. Because in the event of engine failure, larger aircraft are also equipped with emergency backup power sources like the auxiliary power generator and ram air turbine to continue operating the black boxes. In addition, we must consider to making a battery mandatory on solid-state recorders to provide an independent power supply in the event of a complete power failure aboard the plane. Black boxes are sometimes never found or too badly damaged to recover some or all of the data from a crash. To reduce the likelihood of damage or loss, some more recent designs are self-ejecting and use the energy of impact to separate themselves from the aircraft. So, we must provide the recorders with a backup battery to operate the devices for up to ten minutes if power is interrupted.
2. Because the paper will be presented in front of the jury who come from different disciplines, you are trying to make the summary of the concept and its relation to the electrochemical battery. What will you write? Answer: A Galvanic cell is an electrochemical cell that derives electrical energy from chemical reactions taking place within the cell. It generally consists of two different metals connected by a salt bridge, or individual half-cells separated by a porous membrane. It is sometimes called a "Voltaic cell", after Alessandro Volta, inventor of the voltaic pile, the first electrical battery. In common usage, the word "battery" has come to include a single Galvanic cell, but a battery properly consists of multiple cells.
8|Page
Group Report PBL-1 Analytical Chemistry Group 1 A Galvanic cell consists of two half-cells. In its simplest form, each half-cell consists of a metal and a solution of a salt of the metal. The salt solution contains a cation of the metal and an anion to balance the charge on the cation. In essence the half-cell contains the metal in two oxidation states and the chemical reaction in the half-cell is an oxidation-reduction (redox) reaction, written symbolically in reduction direction as Mn+ (oxidized species) + n e M (reduced species)
In a galvanic cell one metal is able to reduce the cation of the other and, conversely, the other cation can oxidize the first metal. The two half-cells must be physically separated so that the solutions do not mix together. A salt bridge or porous plate is used to separate the two solutions yet keep the respective charges of the solutions from separating, which would stop the chemical reactions. The number of electrons transferred in both directions must be the same, so the two half-cells are combined to give the whole-cell electrochemical reaction. For two metals A and B: An+ + n e B
m+
A
m+
+me
B n B + m An+
mA+nB
When a metal in one half-cell is oxidized, anions must be transferred into that half-cell to balance the electrical charge of the cation produced. The anions are released from the other half-cell where a cation is reduced to the metallic state. Thus, the salt bridge or porous membrane serves both to keep the solutions apart and to allow the flow of anions in the direction opposite to the flow of electrons in the wire connecting the electrodes. The voltage of the battery is the sum of the voltages of the two half-cells. When a device such as an electric motor is attached to the electrodes, a current flows and redox reactions occur in both half-cells. This will continue until the concentration of the cations that are being reduced goes to zero.
3. From literature you read that type of batteries widely use in aircraft is the type of NiCd batteries. What shorts of thing are associated with this battery? It is true that NiCd batteries have memory effect?
Answer: NiCd Battery
9|Page
Group Report PBL-1 Analytical Chemistry Group 1
The nickel-cadmium battery (commonly abbreviated NiCd or NiCad ) is a type of rechargeable battery using nickel oxide hydroxide and metallic cadmium as electrodes . The components of NiCd battery are: o o o o Cathode Anode A separator Electrolytes : Nickel (III) NiO(OH) : Cadmium : alkaline (KOH)
The reaction in electrodes when NiCd battery discharges:
(at anode)
(at cathode)
The reaction happen from left side to right side when discharge and rechargeable at the opposite. Memory Effect NiCd batteries may suffer from a " memory effect " if they are discharged and recharged to the same state of charge hundreds of times. The apparent symptom is that the battery "remembers" the point in its charge cycle where recharging began and during subsequent use suffers a sudden drop in voltage at that point, as if the battery had been discharged. if the device is unable to operate through this period of decreased voltage, it will be unable to get enough energy out of the battery, and for all practical purposes, the battery appears "dead" earlier than normal. The mechanism of memory effect is: the active material cadmium in NiCd Battery is small crystals which cover all the surface of NiCd cells. When memory effect happened, the crystals cover the active materials (Ni,Cd) and cause the drop voltage. 4. How do you think the development and application of NiCd battery compared to other battery? Do you know about zapping technique in improving battery performance? What do you think about environmental impact of disposable and rechargeable batteries?
Answer:
NiCd battery use in many thing, for example the primary cell of NiCd battery usually use in electronic portable and toys. The main application of NiCd battery in cordless telephone and wireless, emergency lamps, and in the plane. Because of the low resistance, the NiCd battery 10 | P a g e
Group Report PBL-1 Analytical Chemistry Group 1 has a high voltage current, and usually use in remote control, and camera, electric vehicles and standby power. Types of Batteries Type of Battery 1.Lead acid Battery Information Comparison with NiCd Battery
Have a higher density than NiCd. NiCd battery is smaller Anode: Pb; Cathode: PbO2; and lighter than lead Electrolyte : H2SO4. The main acid battery application in automotive Irreversible chemical reaction in alkaline battery. The voltage of alkaline battery was drop when charge drop. Many of them are disposable battery. Anode: Zn; cathode: MnO2; electrolyte: Zink cloride NiCd battery has smaller capacity and need high cost to produce. NiCd battery last longer and keep the constant voltage when discharge.
2.Alcaline Battery
3.Lithium Ion Battery
Rechargeable, and will broke NiCd battery last longer when discharge in minimum (rechargeable) voltage. Anode: lithium; cathode and electrolyte: variation. The main application in phonecell, laptop, computer, and camera NiMH has bigger capacity and not toxic because the hydride adsorb the alloy in anode. Low cost to produce. The main applicationin hydride vechicle and prototype humanoid robot. NiCd battery has low self discharge, 20%/ month (NiCd battery, 30%/month). NiCd not cause voltage decreases. Low resistance cause the high charge flow.
4. Nickel Hydrida Battery (NiMH)
Impact in environment All of the batteries have chemical materials which cause dangerous impact in environment. The most dangerous materials are heavy metals which carcinogenic, example: mercury and cadmium. It can cause substantial pollution when land filled or incinerated. Because of this, many countries now operate recycling programs to capture and reprocess old batteries. For the people, it can cause cancer, health problem, and died.
11 | P a g e
Group Report PBL-1 Analytical Chemistry Group 1 Disposable Battery Primer battery, ex: leclanche cell (1,5 V) Anode: Zn ; cathode: carbon tube ; electrolyte: MnO2 and NH4Cl Reaction : Zn(s)+2MnO2(s)+2NH4Cl(aq)ZnCl2+Mn2O3(s) +2NH3(aq)+H2O Used in telegraf, alarm, and low voltage instruments Rechargeable battery Secunder battery, ex: super iron battery. Cathode: Fe(VII), K2FeO4; anode: zink, electrolide: ammonium chloride and zink chloride. 50% capacity higher than alkaline battery. The new type battery develop in 2004
Assignment II
1. What reactions occur on each electrode in this electrochemical cell if the total reaction is written: ( Answer: In anode, the oxidation process occurs. The half reaction is: ( ) In cathode, the reduction process occurs. The half reaction is: ( ) 2. How to make electrolyte solution in order to obtain batteries with a voltage of 1.5 V at a temperature of 25oC? Answer: Using Nerst Equation: [ ] [ ] [ ] [ ] Standard Electrode Potential of: Mn (Reduction): +1,23 V Zn (Oxidation : +0,763 V Total Electrode Potential: +1,993 V Substituting the Potential and the Potential desired to the Nerst Equation: [ [ ] [ ] ] ) ( )
12 | P a g e
Group Report PBL-1 Analytical Chemistry Group 1 [ [ [ [ [ [ [ [ [ ][ ] ][ ] [ ] ] [ ] ] ] [ ] ] ] [ ] ] ]
Corresponding to the unknown values of the concentration of [Mn3+],[Mn4+] or [Zn2+], the answer is given in ratio of both concentrations. 3. How do you explain the parameters of the battery capacity in amp-hour and battery voltage in V? How to estimate the life of batteries? Answer: Battery voltage is related to the potential difference between the two half reactions occured at the anode (oxidation reaction) and the cathode (reduction reaction). For example, the 1,5 Volt potential difference in common AA and AAA sized battery are derived from the half reaction of their cells, with the respective to their temperature and concentration (following the Nerst equation). The lifetime of batteries can be determined by the Peukerts Law:
Where : Qp : capacity when discharged at the rate of 1 amp. I : the current drawn from the battery. t : time the battery can sustain k : constant with a value about 1,3.
4. Why do you use graphite or carbon bar as electron flow collector on the cathode side nit only the MnO2 Answer: Carbon takes no part in the electrochemical reaction, but it collects the electrical current and reduce the resistance of the manganese dioxide mix.
13 | P a g e
Group Report PBL-1 Analytical Chemistry Group 1
Assignment III
1. How do you determine the possibility of this river was polluted by copper from the industrial waste? Do you know what industries that have copper in their waste?
Answer:
Determination of copper content in the waste industry can be determined by the potential difference through which components such as reference electrode, indicator electrode, and salt bridges. These components can be interconnected to produce a potential difference. From this it can be determined the presence of copper content in the wastewater plant. Industry-industry wastes containing copper, among others. wire mills, metal coatings, pipes and others. 2. In the laboratory you have a pH meter/ volt meter, a standard saturated calomel electrode, and an indicator electrode for copper analysis. Because the committee will assess the project proposal, can you explain in the proposal the method of analysis to determine the content of copper ion in samples taken from the stream using existing equipment? The information is clear enough in terms of both instrumentation and theoretical basic principle of this analytical method.
Answer:
This is the technical set-up for the potentiometry method. The voltmeter is being set in series with the indicator electrode (in this case is the copper metal, because it is a copper concentration analysis) and the reference electrode (the standard saturated calomel electrode). Meanwhile the analyzed solution is poured at the glass beaker where both the indicator and reference cathode is being set. After the setup has been completed, we measure the voltage difference displayed at the voltmeter. The concentration of the analyzed solution can be determined by using the Nerst equation or the derivatives of the equation.
3. By using the direct potentiometric technique, you obtain data such as in figure. How do you determine the concentration of copper in the sample? Answer: From the table, we can collect some data such as: Slope: 0.0302V 14 | P a g e
Group Report PBL-1 Analytical Chemistry Group 1 ECu-ISE: 0.2524V We also take the standard potential data of the reduction reaction of copper: We input the data into the nerst equation: [ [ [ [ ] ] ] ]
4. How do you explain the determination of copper concentration in the sample solution with standard addition technique? How do you explain the difference in direct potentiometric determination technique and standard addition technique?
Answer:
Step in addition standard technique: From the sample, determine the aliquot identical, Vx. adding a number of specific volume variation, Vs. its dilute each solution to a particular volume, Vt. Then calculate concentration on sample, Cu, with equation:
Cu=
Cu= concentration in unknown sample Cs= concentration in standard sample Vs= standard volume Vu= Sample volume E1= electro potential in pure solvent E2= electrode potential in addition m= electrode slope Direct Potentiometric technique This technique requires only an indicator of potential measurement of the electron when it is dipped in a solution containing an unknown concentration of an analyte and unknown. Indicator electrode is always considered as a cathode and reference electrode as the anode. For the direct potentiometric measurements, the cell potential can be expressed as a potential development by the indicator electrode, reference electrode, and potential functions. It depends on the potential difference measurement of electrodes in a solvent. Potential difference can be measured with pH meters/voltmeter. An electrodes is indicator electrode which use to give the response to the solvents. Standard Addition technique 15 | P a g e
Group Report PBL-1 Analytical Chemistry Group 1 This technique is commonly used in analytical instrumentation such as in atomic absorption spectroscopy and gas chromatography to find the value of the concentration of the substance (analyte) in a sample of unknown composition by comparison to samples of known concentration. Excess standard addition and Direct Potentiometric Calibration and measurement of samples done simultaneously so that the difference in ionic strength and temperature standards and the sample is not too significant. During the process, the electrodes remain immersed in the solution so that there is little change in junction potential solution. Measurement of slope very close to the concentration of the sample shows this method can yield more accurate results in the range of non-linear and can be used with electrodes old or older who was not linear range for the slope is stable.
References
1. Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch, Fundamentals of Analytical Chemistry 8th Edition. Saunders College Publishing, New York,2002. 2. http://en.wikipedia.org/wiki/Battery 3. http://en.wikipedia.org/wiki/potentiometry 4. http://en.wikipedia.org/wiki/Ni-Cd
16 | P a g e
Das könnte Ihnen auch gefallen
- Che 1Dokument31 SeitenChe 1dineshsilambam2305Noch keine Bewertungen
- Chemistry For Engineers Laboratory: CHEM 114Dokument8 SeitenChemistry For Engineers Laboratory: CHEM 114Ivyy Joyce BuanNoch keine Bewertungen
- Electrochemical Cells R - Virtual LabDokument3 SeitenElectrochemical Cells R - Virtual LabJosua VivasNoch keine Bewertungen
- Batteries & Fuel Cells Dr. Siju N. AntonyDokument31 SeitenBatteries & Fuel Cells Dr. Siju N. AntonysijunantonyNoch keine Bewertungen
- Need For Alternate Energy SourcesDokument17 SeitenNeed For Alternate Energy SourcesshruniviNoch keine Bewertungen
- Electrochemistry: Assoc. Prof. Jacqui Lou Valenzuela, RCH Chemistry Department Cas, WitDokument53 SeitenElectrochemistry: Assoc. Prof. Jacqui Lou Valenzuela, RCH Chemistry Department Cas, WitJacquiNoch keine Bewertungen
- General Chemistry 2 Quarter 4: Week 7 - Module 7 Standard Cell Potential, Electrochemical Cells and BatteriesDokument21 SeitenGeneral Chemistry 2 Quarter 4: Week 7 - Module 7 Standard Cell Potential, Electrochemical Cells and BatteriesCamille Joves EncarnacionNoch keine Bewertungen
- Chapter 18 ElectrochemistryDokument17 SeitenChapter 18 ElectrochemistryNefliNoch keine Bewertungen
- Batteries: Types of Batteries Primary Batteries Secondary Batteries Fuel Cells / Flow BatteriesDokument24 SeitenBatteries: Types of Batteries Primary Batteries Secondary Batteries Fuel Cells / Flow BatteriesMahek50% (2)
- Battery TechnologyDokument60 SeitenBattery TechnologyKasinathan MuniandiNoch keine Bewertungen
- Electrochemical DevicesDokument91 SeitenElectrochemical DevicesSujalNoch keine Bewertungen
- Chem101 Ho4Dokument4 SeitenChem101 Ho4cyrusryan21Noch keine Bewertungen
- BatteryDokument11 SeitenBatteryHarsha VardhanNoch keine Bewertungen
- Batteries and Fuel CellsDokument31 SeitenBatteries and Fuel CellsTomesh SahuNoch keine Bewertungen
- Chapter6-Electrochemistry (Part 2)Dokument27 SeitenChapter6-Electrochemistry (Part 2)Uswatun KhasanahNoch keine Bewertungen
- Experiment 8 PhyChem IIDokument5 SeitenExperiment 8 PhyChem IIティン ヨロベ100% (1)
- Electrochemistry Chemistry and ElectricityDokument54 SeitenElectrochemistry Chemistry and ElectricityMaria OzaoNoch keine Bewertungen
- B.Tech First Year: Course Name: Engineering ChemistryDokument29 SeitenB.Tech First Year: Course Name: Engineering ChemistryHemant Singh JadounNoch keine Bewertungen
- Chem 131 Lesson 8Dokument5 SeitenChem 131 Lesson 8Denampo Ivan MikhaelNoch keine Bewertungen
- Chapter One: Introduction of ElectrochemistryDokument29 SeitenChapter One: Introduction of ElectrochemistryBayan O. Abu SaadaNoch keine Bewertungen
- Battery PDFDokument7 SeitenBattery PDFCHARITHANoch keine Bewertungen
- CH 3 14Dokument135 SeitenCH 3 14active learning educationNoch keine Bewertungen
- Lecture 4 - Electro Chem PDFDokument49 SeitenLecture 4 - Electro Chem PDFHedric VillenaNoch keine Bewertungen
- BatteriesDokument22 SeitenBatterieshariarun0705Noch keine Bewertungen
- Energy ResourcesDokument6 SeitenEnergy ResourcesAm AsdfghjklNoch keine Bewertungen
- Potentiometry - Without Voice Narration PDFDokument35 SeitenPotentiometry - Without Voice Narration PDFKalyavalla SathyasaiNoch keine Bewertungen
- Electrochemical Energy Systems: Batteries and Fuel CellsDokument42 SeitenElectrochemical Energy Systems: Batteries and Fuel Cellsshubhika guptaNoch keine Bewertungen
- M1 Battery TechnologyDokument13 SeitenM1 Battery TechnologyMalvika RkNoch keine Bewertungen
- ElectrochemistryDokument27 SeitenElectrochemistry22cs103Noch keine Bewertungen
- Chem 17Dokument9 SeitenChem 17Adi SoNoch keine Bewertungen
- Nickel Cadmium 1-WPS OfficeDokument3 SeitenNickel Cadmium 1-WPS OfficeWibi SonoNoch keine Bewertungen
- Experiment 14 - ElectrochemistryDokument10 SeitenExperiment 14 - ElectrochemistryArifmyNoch keine Bewertungen
- Department of Chemical EngineeringDokument12 SeitenDepartment of Chemical EngineeringSheikh AliNoch keine Bewertungen
- Battery Knowledge - : Contact UsDokument4 SeitenBattery Knowledge - : Contact UsSiva MurugesanNoch keine Bewertungen
- Chemistry BeginingDokument5 SeitenChemistry Beginingarchita1072006Noch keine Bewertungen
- Chapter6-Electrochemistry (Part 2)Dokument27 SeitenChapter6-Electrochemistry (Part 2)BagusprPrasetyoNoch keine Bewertungen
- Tutorial Sheet7Dokument5 SeitenTutorial Sheet7Lê Anh QuangNoch keine Bewertungen
- 0005unit V NotesDokument33 Seiten0005unit V Noteskishan kumarNoch keine Bewertungen
- Relating Values of Cell Potential: For General Chemistry 2/grade 12-STEM Quarter 4/week 8.b-cDokument11 SeitenRelating Values of Cell Potential: For General Chemistry 2/grade 12-STEM Quarter 4/week 8.b-cAllona Jane BrionesNoch keine Bewertungen
- Aircraft Electrics - Aircraft BatteriesDokument17 SeitenAircraft Electrics - Aircraft BatteriesErica Zoe BantogNoch keine Bewertungen
- TSSM Topic 5Dokument18 SeitenTSSM Topic 5sudotesterNoch keine Bewertungen
- RSC - ElectrochemistryDokument98 SeitenRSC - ElectrochemistrymokilpoNoch keine Bewertungen
- Lab Activity 7 ElectrochemistryDokument8 SeitenLab Activity 7 Electrochemistryjhunjhun zambranoNoch keine Bewertungen
- Chapter 3Dokument30 SeitenChapter 3Umesh ChandraNoch keine Bewertungen
- Potential of A Galvanic CellDokument6 SeitenPotential of A Galvanic CellRalph Andrew EsperonNoch keine Bewertungen
- Batteries Chemistry ProjectDokument12 SeitenBatteries Chemistry ProjectAbhishek yadav67% (12)
- Unit 12 ElectrochemistryDokument22 SeitenUnit 12 Electrochemistrycream oNoch keine Bewertungen
- Lesson 1 FunctionsDokument7 SeitenLesson 1 FunctionsWaien G. WatamamaNoch keine Bewertungen
- Voltaic Minicell LabDokument6 SeitenVoltaic Minicell LabIvy LongNoch keine Bewertungen
- Chapter Five Introduction To Electroanalytical ChemistryDokument16 SeitenChapter Five Introduction To Electroanalytical ChemistryZekarias LibenaNoch keine Bewertungen
- ElectroDokument13 SeitenElectrodulalsushant3Noch keine Bewertungen
- Step 1: Break Reaction Into Half-Reactions by IonsDokument3 SeitenStep 1: Break Reaction Into Half-Reactions by IonsjoenelNoch keine Bewertungen
- Class 12 Chemistry Revision Notes ElectrochemistryDokument25 SeitenClass 12 Chemistry Revision Notes ElectrochemistrySariska MehraNoch keine Bewertungen
- 5 - Electrochemistry PDFDokument15 Seiten5 - Electrochemistry PDFthinkiit100% (1)
- Electrochemistry - Galvanic Cell NewDokument51 SeitenElectrochemistry - Galvanic Cell NewPink WandererNoch keine Bewertungen
- Electrochemical Cells Revised 12/8/14Dokument7 SeitenElectrochemical Cells Revised 12/8/14SamarpitNoch keine Bewertungen
- Electro ChemistryDokument2 SeitenElectro Chemistryradhikanaveen60Noch keine Bewertungen
- The Stopping and Ranges of Ions in Matter: Handbook of Stopping Cross-Sections for Energetic Ions in All ElementsVon EverandThe Stopping and Ranges of Ions in Matter: Handbook of Stopping Cross-Sections for Energetic Ions in All ElementsNoch keine Bewertungen
- Solid-State Circuits: Electrical Engineering DivisonVon EverandSolid-State Circuits: Electrical Engineering DivisonBewertung: 4.5 von 5 Sternen4.5/5 (4)
- Lite Touch. Completo PDFDokument206 SeitenLite Touch. Completo PDFkerlystefaniaNoch keine Bewertungen
- Dispersion Compensation FibreDokument16 SeitenDispersion Compensation FibreGyana Ranjan MatiNoch keine Bewertungen
- Chapter One Understanding Civics and Ethics 1.1.defining Civics, Ethics and MoralityDokument7 SeitenChapter One Understanding Civics and Ethics 1.1.defining Civics, Ethics and Moralitynat gatNoch keine Bewertungen
- Uptime Elements Passport: GineerDokument148 SeitenUptime Elements Passport: GineerBrian Careel94% (16)
- GearsDokument14 SeitenGearsZulhilmi Chik TakNoch keine Bewertungen
- Pentecostal HealingDokument28 SeitenPentecostal Healinggodlvr100% (1)
- Foxit PhantomPDF For HP - Quick GuideDokument32 SeitenFoxit PhantomPDF For HP - Quick GuidekhilmiNoch keine Bewertungen
- Ghalib TimelineDokument2 SeitenGhalib Timelinemaryam-69Noch keine Bewertungen
- Policarpio Vs Manila Times - Unprotected Speech LibelDokument3 SeitenPolicarpio Vs Manila Times - Unprotected Speech LibelStef BernardoNoch keine Bewertungen
- Exercise No.2Dokument4 SeitenExercise No.2Jeane Mae BooNoch keine Bewertungen
- SHS11Q4DLP 21st CentFinalDokument33 SeitenSHS11Q4DLP 21st CentFinalNOEMI DE CASTRONoch keine Bewertungen
- VtDA - The Ashen Cults (Vampire Dark Ages) PDFDokument94 SeitenVtDA - The Ashen Cults (Vampire Dark Ages) PDFRafãoAraujo100% (1)
- GB BioDokument3 SeitenGB BiolskerponfblaNoch keine Bewertungen
- Fundamental Rights-Right To Equality, Prohibition of Discrimination, Right To Equality of OpportunityDokument76 SeitenFundamental Rights-Right To Equality, Prohibition of Discrimination, Right To Equality of OpportunityutkarshNoch keine Bewertungen
- Approved Chemical ListDokument2 SeitenApproved Chemical ListSyed Mansur Alyahya100% (1)
- Fansubbers The Case of The Czech Republic and PolandDokument9 SeitenFansubbers The Case of The Czech Republic and Polandmusafir24Noch keine Bewertungen
- Proper AdjectivesDokument3 SeitenProper AdjectivesRania Mohammed0% (2)
- ObliCon Digests PDFDokument48 SeitenObliCon Digests PDFvictoria pepitoNoch keine Bewertungen
- Blunders and How To Avoid Them Dunnington PDFDokument147 SeitenBlunders and How To Avoid Them Dunnington PDFrajveer404100% (2)
- Clearing Negative SpiritsDokument6 SeitenClearing Negative SpiritsmehorseblessedNoch keine Bewertungen
- Activity Lesson PlanDokument2 SeitenActivity Lesson PlanPsiho LoguseNoch keine Bewertungen
- A&P: The Digestive SystemDokument79 SeitenA&P: The Digestive SystemxiaoNoch keine Bewertungen
- Spiritual Transcendence in Transhumanism PDFDokument10 SeitenSpiritual Transcendence in Transhumanism PDFZeljko SaricNoch keine Bewertungen
- ESU Mauritius Newsletter Dec 2014Dokument8 SeitenESU Mauritius Newsletter Dec 2014Ashesh RamjeeawonNoch keine Bewertungen
- Progressivism Sweeps The NationDokument4 SeitenProgressivism Sweeps The NationZach WedelNoch keine Bewertungen
- Mathsnacks05 InfiniteDokument1 SeiteMathsnacks05 Infiniteburkard.polsterNoch keine Bewertungen
- Trudy Scott Amino-AcidsDokument35 SeitenTrudy Scott Amino-AcidsPreeti100% (5)
- Developing Global LeadersDokument10 SeitenDeveloping Global LeadersDeepa SharmaNoch keine Bewertungen
- Tugas Conditional Sentences YanneDokument3 SeitenTugas Conditional Sentences Yanneyanne nurmalitaNoch keine Bewertungen
- Final PS-37 Election Duties 06-02-24 1125pm)Dokument183 SeitenFinal PS-37 Election Duties 06-02-24 1125pm)Muhammad InamNoch keine Bewertungen