Beruflich Dokumente
Kultur Dokumente
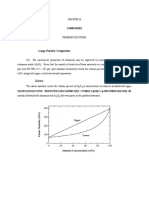
Band Theory of Soids5!1!13
Hochgeladen von
Ravi Kumar BanalaOriginalbeschreibung:
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Band Theory of Soids5!1!13
Hochgeladen von
Ravi Kumar BanalaCopyright:
Verfügbare Formate
Introduction
Bloch stated this theory in 1928. According to this theory, the
free electrons moves in a periodic field provided by the lattice.
The Energy band theory of solids is the basic principle of
semiconductor physics and it is used to explain the differences
in electrical properties between Metals, Insulators and
Semiconductors.
Band Theory of Solids
Electron in a periodic potential
( Bloch Theorem )
A crystalline solid consists of a lattice which is composed of a
large number of positive ion cores at regular intervals and the
conduction electrons move freely throughout the lattice.
The variation of potential inside the metallic crystal with the
periodicity of the lattice is explained by Bloch theorem.
The periodic potential V (x) may be defined by means of the
lattice constant a as V (x) = V ( x + a )
One dimensional periodic potential in crystal.
a
+ + + +
0
V
+ + + + + +
+ + + + + +
Periodic positive ion cores Inside metallic crystals.
electron the of motion the of state the represents k where
a) (x U (x) U
lattice. crystal a of y periodicit with periodic a is (x) U Where
) (x)exp(ikx U (x)
equation r Schrodinge the of solution
l dimensiona one the shown that has Bloch
0 a)] V(x [E
h
m 8
dx
d
0 V] [E
h
m 8
dx
d
equation r wave Schrodinge From
k k
k
k
2
2
2
2
2
2
2
2
+ =
=
= + +
= +
Kroning Penney Model
According to Kroning - Penney model the electrons move in a
periodic potential field provided by the lattice.
The potential of the solid varies periodically with the periodicity of
space lattice.
Consider Schrdinger equation for this case, we can find the
existence of the energy gap between the allowed values of energy
of electron.
For one dimensional periodic potential field.
0 )] ( [
8
2
2
2
2
= +
t
x V E
h
m
dx
d
+
+
+ +
+
V
0
0
a -b
1
2
0 ] [
8
) 1 (
2
2
2
2
= +
t
E
h
m
dx
d
region f or
0 ] [
8
) 2 (
0
2
2
2
2
= +
t
V E
h
m
dx
d
region f or
) 2 .....( 0
) 1 .....( 0
2
2
2
2
2
2
=
= +
|
dx
d
dx
d
2
2
2
2
2
0
2
8
8
[ ]
where
mE
h
m
V E
h
t
o
t
|
=
=
0 , ) (
0 , 0 ) (
0
< < =
< < =
x b v x v
a x x v
.....(a) (x)e U (x)
ikx
k
=
) (x).....(b U a) (x U
k k
= +
.....(c) (x)e Na) (x
ikNa
k k
= +
According to Bloch, the solution of a Schrodinger equation
Where U
k
(x) is the periodicity of the lattice i.e,.
According to Bloch theorem
By using above a, b, and c Bloch conditions, the solutions of
equations (1) & (2) becomes
2mE
h
2
strength Barrier called is b V
barrier potential the of power scattering the is p
b V
h
ma 4
where..P
a cos
a
a sin
P coska
0
0
2
2
=
=
+ = o
o
+1
-1
+1
-1
a o
a cos
a
a sine
P o
o
+
Conclusions
1. The motion of electrons in a periodic lattice is characterized
by the bands of allowed energy separated by forbidden
regions.
2. As the value of a increases, the width of allowed energy
bands also increases and the width of the forbidden bands
decreases. i.e., the first term of equation deceases on the
average with increasing a .
3. Let us now consider the effect of varying barrier strength P. if
V
0
b is large ,i.e. if p is large ,the function described by the left
hand side of the equation crosses +1 and -1 region as shown
in figure. thus the allowed bands are narrower and the
forbidden bands are wider.
a o
0
p
If P tends to infinite the allowed band reduces to one
single energy level
0 p
a o
4. If P tends to zero
No energy levels exist:
all energies are allowed to the electrons.
2
2
2
2 2
2
2
2
2
2
2
2
2
2 2
2 2
2
1
2
)
2
(
1
)
2
(
)
2
)(
8
(
)
2
(
2
cos cos
mv
m
p
h
p
m
h
E
m
h
E
m
h
E
k
m
E
mE
k
k
k
ka a
= = =
=
=
=
= =
=
=
=
t
t
o
o
o
o
Brillouin Zones
The Brillouin zone is a representation of permissive
values of k of the electrons in one, two or three
dimensions.
Thus the energy spectrum of an electron moving in
the presence of a periodic potential fields is divided
into allowed zones and forbidden zones.
Allowed
bands
Energy gap
First
Brillouin zone
E
k
Energy gap
a
t
a
t 2
a
t 3
a
t
a
t 2
a
t 3
E-k Diagram
Origin of Energy band formation in Solids
When we consider isolated atom, the electrons are tightly
bound and have discrete, sharp energy levels.
When two identical atoms are brought closer the outer most
orbits of these atoms overlap and interact.
If more atoms are brought together more levels are formed and
for a solid of N atoms , each of the energy levels of an atom
splits into N levels of energy.
The levels are so close together that they form an almost
continuous band.
The width of this band depends on the degree of overlap of
electrons of adjacent atoms and is largest for outer most
atomic electrons.
N energy levels
N atoms
E
E
1
E
2
E
3
E
2
E
1
E
1
The energy bands in solids are important in
determining many of physical properties of solids.
The allowed energy bands
(1) Valance band
(2) Conduction band
The band corresponding to the outer most orbit is
called conduction band and the next inner band is
called valence band. The gap between these two
allowed bands is called forbidden energy gap.
Classifications of materials into Conductors,
Semiconductors & Insulators:-
On the basis of magnitude of forbidden band the
solids are classified into insulators, semiconductors
and conductors.
Insulators:
In case of insulators, the forbidden energy band is
very wide as shown in figure.
Due to this fact the electrons cannot jump from
valance band to conduction band.
In insulators at 0
0
k and the energy gap between
valance band and conduction band is of the order.
Forbidden gap
Valance band
Conduction band
INSULATORS
Forbidden gap
Valance band
Conduction band
SEMI CONDUCTORS
Valance band
Conduction band
CONDUCTORS
SEMI CONDUCTORS
In semi conductors the forbidden energy ( band ) gap is very small
as shown in a figure.
Ge and Si are the best examples of semiconductors.
Forbidden ( band ) is of the order of 0.7ev & 1.1ev.
CONDUCTOS:
In conductors there is no forbidden gap. Valence and conduction
bands overlap each other as shown in figure above.
The electrons from valance band freely enter into conduction band.
Das könnte Ihnen auch gefallen
- Band Theory of SolidsDokument22 SeitenBand Theory of Solidsmunku05100% (1)
- Band Theory of SolidsDokument12 SeitenBand Theory of SolidsFitrianiNoch keine Bewertungen
- UNIT - 4 - ppt-1Dokument49 SeitenUNIT - 4 - ppt-1Neha YarapothuNoch keine Bewertungen
- Band Theory of SolidsDokument18 SeitenBand Theory of SolidsSachi SinghNoch keine Bewertungen
- Band Theory of Solids: Christian Bergfjord & Selda EkizDokument44 SeitenBand Theory of Solids: Christian Bergfjord & Selda EkizAzhar MahmoodNoch keine Bewertungen
- Chapter # 21 Nuclear PhysicsDokument7 SeitenChapter # 21 Nuclear PhysicsAsif Rasheed RajputNoch keine Bewertungen
- Dielectric Properties of SolidsDokument40 SeitenDielectric Properties of SolidsHannan MiahNoch keine Bewertungen
- Band Theory of Solids: Brajesh Tiwari and R. E. AmritkarDokument9 SeitenBand Theory of Solids: Brajesh Tiwari and R. E. AmritkarkapilNoch keine Bewertungen
- Applications of EMF Sem-6Dokument57 SeitenApplications of EMF Sem-6Mahmood YacoobaliNoch keine Bewertungen
- Introduction To Energy Band For EngineersDokument4 SeitenIntroduction To Energy Band For EngineersDerbew Gahaw100% (1)
- APPLIED PHYSICS Bonding in Solids by CHARIS ISRAEL ANCHADokument17 SeitenAPPLIED PHYSICS Bonding in Solids by CHARIS ISRAEL ANCHACharis Israel Ancha100% (1)
- 2-Crystal Symmetry and Classification-2Dokument7 Seiten2-Crystal Symmetry and Classification-2Cazimir BostanNoch keine Bewertungen
- ELECTRON CONFIG-WPS Office - RecoverDokument50 SeitenELECTRON CONFIG-WPS Office - RecoverLyca RealNoch keine Bewertungen
- Free Electron TheoryDokument8 SeitenFree Electron TheoryNeelam KapoorNoch keine Bewertungen
- Electrical Properties of SolidsDokument54 SeitenElectrical Properties of SolidsAnthonio MJNoch keine Bewertungen
- Thermal Properties of SolidsDokument16 SeitenThermal Properties of SolidsParie PerdanaNoch keine Bewertungen
- Electrostatics DPP 02 Manish Raj Sir Neet Crash Course RelaunchDokument2 SeitenElectrostatics DPP 02 Manish Raj Sir Neet Crash Course Relaunchrajsharmma63Noch keine Bewertungen
- Magnetism in SolidsDokument11 SeitenMagnetism in SolidsNeelam KapoorNoch keine Bewertungen
- Chapter 4 (Periodic Table)Dokument46 SeitenChapter 4 (Periodic Table)Tunku Hilman Al-nordinNoch keine Bewertungen
- XRAYS Chapter PDFDokument11 SeitenXRAYS Chapter PDFAdeel MajeedNoch keine Bewertungen
- Intro To SemiconductorsDokument27 SeitenIntro To SemiconductorsRolando CelesteNoch keine Bewertungen
- UNIT I and II - ESSAY AND SHORT QUESTIONS PDFDokument8 SeitenUNIT I and II - ESSAY AND SHORT QUESTIONS PDFSãkëth RèddièNoch keine Bewertungen
- Imperfections in SolidsDokument10 SeitenImperfections in Solidsshubhamgautam56_64970% (1)
- Chapter 8: Band Theory of Solids Concept of Free Electron Theory: Hour 1Dokument25 SeitenChapter 8: Band Theory of Solids Concept of Free Electron Theory: Hour 1Vivek kapoorNoch keine Bewertungen
- Liquid Drop Model and Bohr-Wheeler Theory of Nuclear FissionDokument6 SeitenLiquid Drop Model and Bohr-Wheeler Theory of Nuclear FissionAmogh BorgaveNoch keine Bewertungen
- Solar CellDokument24 SeitenSolar CellanupamanupamNoch keine Bewertungen
- Thin Film Growth StepsDokument45 SeitenThin Film Growth Stepshazbi2011Noch keine Bewertungen
- Vsepr TheoryDokument7 SeitenVsepr TheoryAashiqueNoch keine Bewertungen
- Eme 1166 Materials Science: DiffusionDokument55 SeitenEme 1166 Materials Science: DiffusionJONATHAN RAJ A/L SEMANNoch keine Bewertungen
- Ferro ElectricityDokument6 SeitenFerro Electricity33noe100% (1)
- Crystal Field TheoryDokument26 SeitenCrystal Field TheorySahil Qaiser100% (1)
- The Steady State Approximation Is A Method Used To Estimate The Overall Reaction Rate of A MultiDokument6 SeitenThe Steady State Approximation Is A Method Used To Estimate The Overall Reaction Rate of A MultiMubashar ShakeelNoch keine Bewertungen
- Unit 2 NotesDokument10 SeitenUnit 2 NotesG.MUNEER BABA,EEE18 Vel Tech, ChennaiNoch keine Bewertungen
- Test-03 Quantum Numbers: B. C. D. E. F. GDokument2 SeitenTest-03 Quantum Numbers: B. C. D. E. F. GNishali Sam100% (1)
- SuperconductorDokument33 SeitenSuperconductorCrisanta GanadoNoch keine Bewertungen
- D AND F BLOCK ELEMENT NotesDokument5 SeitenD AND F BLOCK ELEMENT NotesM AroNoch keine Bewertungen
- Dec 1st - 2015 L10c Diffusion Ficks Laws 2Dokument11 SeitenDec 1st - 2015 L10c Diffusion Ficks Laws 2abo1510Noch keine Bewertungen
- Structure and BondingDokument12 SeitenStructure and BondingNisha JodhanNoch keine Bewertungen
- Steady State Approximation: Supplementary Notes For The Course "Chemistry For Physicists"Dokument5 SeitenSteady State Approximation: Supplementary Notes For The Course "Chemistry For Physicists"Rishav DugarNoch keine Bewertungen
- Coordination Compounds Short Answers Type QuestionsDokument5 SeitenCoordination Compounds Short Answers Type Questionskrishna kumar bhardwajNoch keine Bewertungen
- ch16 Advance CompositesDokument52 Seitench16 Advance CompositesAnonymous 3TpOzKPj5100% (1)
- A Brief Introduction To Molecular Orbital Theory oDokument4 SeitenA Brief Introduction To Molecular Orbital Theory oBheim LlonaNoch keine Bewertungen
- Chapter - 1 Crystal Field Theory in Octahedral Complexes NotesDokument20 SeitenChapter - 1 Crystal Field Theory in Octahedral Complexes NotesMohit KambojNoch keine Bewertungen
- Vector Model For Orbital Angular MomentumDokument6 SeitenVector Model For Orbital Angular MomentumAgrupación Astronomica de Alicante100% (2)
- Chapter 5: Surface Area To Volume Ratios in Nanoscience: Table ofDokument3 SeitenChapter 5: Surface Area To Volume Ratios in Nanoscience: Table ofamanNoch keine Bewertungen
- Young's Modulus by Cornu's Method PDFDokument7 SeitenYoung's Modulus by Cornu's Method PDFAnurag singhNoch keine Bewertungen
- (#3) Direct, Indirect, Ek Diagram, Effective Mass PDFDokument6 Seiten(#3) Direct, Indirect, Ek Diagram, Effective Mass PDFMas RoorNoch keine Bewertungen
- Dielectrics: Unit V Dielectric MaterialsDokument20 SeitenDielectrics: Unit V Dielectric MaterialsSpyx MeniNoch keine Bewertungen
- Energy Bands For Electrons in Crystals (Kittel Ch. 7)Dokument39 SeitenEnergy Bands For Electrons in Crystals (Kittel Ch. 7)sabhanNoch keine Bewertungen
- Revision Notes - Basic Nuclear PropertiesDokument10 SeitenRevision Notes - Basic Nuclear PropertiesPankaj BiswasNoch keine Bewertungen
- Electrical Properties of Semiconductors PDFDokument2 SeitenElectrical Properties of Semiconductors PDFApril Wilson100% (1)
- SuperconductivityDokument48 SeitenSuperconductivityGiuseppe AromatarisNoch keine Bewertungen
- Kirkendal Effect Paper PDFDokument4 SeitenKirkendal Effect Paper PDFAnonymous DmKpKA6wHNoch keine Bewertungen
- Mat Sci Slides 2Dokument29 SeitenMat Sci Slides 2abcdNoch keine Bewertungen
- Debye ShieldingDokument2 SeitenDebye ShieldingAmir AbdellaNoch keine Bewertungen
- Electrostatics NotesDokument29 SeitenElectrostatics NotesUrja MoonNoch keine Bewertungen
- Lect-4 Electron ConfigurationDokument33 SeitenLect-4 Electron Configurationroman leeNoch keine Bewertungen
- A Collection of Problems on Mathematical Physics: International Series of Monographs in Pure and Applied MathematicsVon EverandA Collection of Problems on Mathematical Physics: International Series of Monographs in Pure and Applied MathematicsNoch keine Bewertungen
- Structure and Properties of Inorganic Solids: International Series of Monographs in Solid State PhysicsVon EverandStructure and Properties of Inorganic Solids: International Series of Monographs in Solid State PhysicsNoch keine Bewertungen
- Neusource Entreprneurial Guide: Entrepreneurial SpiritDokument23 SeitenNeusource Entreprneurial Guide: Entrepreneurial SpiritRavi Kumar BanalaNoch keine Bewertungen
- Memory Allocation Functions: C Gives Us Two Choices When We Want To Reserve Memory Locations For An ObjectDokument22 SeitenMemory Allocation Functions: C Gives Us Two Choices When We Want To Reserve Memory Locations For An ObjectRavi Kumar BanalaNoch keine Bewertungen
- 14Dokument61 Seiten14Ravi Kumar BanalaNoch keine Bewertungen
- Transient StabilityDokument16 SeitenTransient StabilityRavi Kumar BanalaNoch keine Bewertungen
- Dowsil 51 AdditiveDokument5 SeitenDowsil 51 AdditiveYến HoàngNoch keine Bewertungen
- Technical ManualDokument48 SeitenTechnical ManualStewart BenNoch keine Bewertungen
- Mercedes Panaromic Roof Lubrication GuideDokument1 SeiteMercedes Panaromic Roof Lubrication Guidehappy_jonNoch keine Bewertungen
- Linear Swell TestDokument3 SeitenLinear Swell TestAhmer AkhlaqueNoch keine Bewertungen
- Javascript and JquerylearningDokument13 SeitenJavascript and Jquerylearningilias ahmedNoch keine Bewertungen
- History of Information Technology: PremechanicalDokument5 SeitenHistory of Information Technology: PremechanicalMichael RamogaNoch keine Bewertungen
- MyanmarDokument30 SeitenMyanmarempty87Noch keine Bewertungen
- Package Power JackDokument5 SeitenPackage Power JackMae MegumiNoch keine Bewertungen
- Rancang Bangun Alat Pendeteksi Tingkat Kematangan Buah Durian Dengan Metode Naïve BayesDokument10 SeitenRancang Bangun Alat Pendeteksi Tingkat Kematangan Buah Durian Dengan Metode Naïve BayesMega Erika ErnawatiNoch keine Bewertungen
- Delphi Com API Do WindowsDokument21 SeitenDelphi Com API Do WindowsWeder Ferreira da SilvaNoch keine Bewertungen
- CQ BalancingDokument4 SeitenCQ Balancingapi-218999959Noch keine Bewertungen
- Yoshimura Rs3 Slip On Exhaust Honda Cbr600 f4 I20012006Dokument4 SeitenYoshimura Rs3 Slip On Exhaust Honda Cbr600 f4 I20012006Ahmed WahidNoch keine Bewertungen
- Giaonx, Bai 24 - P27-28.5. Vinh-Dat Vuong-Revised-1Dokument9 SeitenGiaonx, Bai 24 - P27-28.5. Vinh-Dat Vuong-Revised-1Phước Trương VĩnhNoch keine Bewertungen
- Montague Gold MineDokument6 SeitenMontague Gold MineChrispen MachipisaNoch keine Bewertungen
- Soalan Fizik Chapter 9Dokument10 SeitenSoalan Fizik Chapter 9Yusfalina Mohd YusoffNoch keine Bewertungen
- Olycryl Olycryl: Acrylic Based Waterproofing CoatingDokument2 SeitenOlycryl Olycryl: Acrylic Based Waterproofing CoatingVaittianathan MahavapillaiNoch keine Bewertungen
- 13bentonit SlurryDokument24 Seiten13bentonit SlurrykesharinareshNoch keine Bewertungen
- CenturionDokument28 SeitenCenturiondan.gonzalez2573Noch keine Bewertungen
- Royal 77Dokument131 SeitenRoyal 77victorNoch keine Bewertungen
- Geometric Dimensions & TolerancesDokument96 SeitenGeometric Dimensions & TolerancesPriyadarshi SinghNoch keine Bewertungen
- Barrett Firearms - MRAD - Operators-Manual-18697 18052Dokument23 SeitenBarrett Firearms - MRAD - Operators-Manual-18697 18052Ricardo C TorresNoch keine Bewertungen
- Fabricante de HPFF CableDokument132 SeitenFabricante de HPFF Cableccrrzz100% (1)
- Masters in Business Administration: Bhilai Steel Plant (Sail)Dokument62 SeitenMasters in Business Administration: Bhilai Steel Plant (Sail)संदीप द्विवेदीNoch keine Bewertungen
- GoedhartVCI P 2012 en Version1 IDokument24 SeitenGoedhartVCI P 2012 en Version1 IRodrigo GonçalvesNoch keine Bewertungen
- 6R80L 6R100 Zip inDokument12 Seiten6R80L 6R100 Zip inYisus 616Noch keine Bewertungen
- 18 - Sikafloor - 264 - SGDokument6 Seiten18 - Sikafloor - 264 - SGPaul ManiquisNoch keine Bewertungen
- Classification of MissileDokument4 SeitenClassification of MissileMoaz Hussain100% (1)
- Industrial Training ReportDokument19 SeitenIndustrial Training ReportRam PandeyNoch keine Bewertungen
- CKD Sab SVB Catalogue en Cc770a PDFDokument58 SeitenCKD Sab SVB Catalogue en Cc770a PDFvanNoch keine Bewertungen
- Fasson 0828 DatasheetDokument2 SeitenFasson 0828 DatasheetGregory Alan Francisco IINoch keine Bewertungen