Beruflich Dokumente
Kultur Dokumente
Sni, Nighbouring GP Participation & E1cb
Hochgeladen von
saheedvk0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
277 Ansichten19 SeitenThis document discusses different mechanisms for nucleophilic substitution reactions. It explains that nucleophilic substitution of alcohols with hydrogen halides can produce rearranged products through carbocation intermediates, but reactions with sulfur or phosphorus halides follow an SNi mechanism without rearrangement. The SNi mechanism involves the nucleophile attacking from the same side as the leaving group to give retention of configuration. Neighboring group participation allows some SN2 reactions to proceed with retention of configuration through two inversion steps. The document also describes an E1CB mechanism where a conjugate base is generated through proton abstraction prior to elimination of the leaving group.
Originalbeschreibung:
education
Originaltitel
Sni, Nighbouring Gp Participation & e1cb
Copyright
© © All Rights Reserved
Verfügbare Formate
PPTX, PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenThis document discusses different mechanisms for nucleophilic substitution reactions. It explains that nucleophilic substitution of alcohols with hydrogen halides can produce rearranged products through carbocation intermediates, but reactions with sulfur or phosphorus halides follow an SNi mechanism without rearrangement. The SNi mechanism involves the nucleophile attacking from the same side as the leaving group to give retention of configuration. Neighboring group participation allows some SN2 reactions to proceed with retention of configuration through two inversion steps. The document also describes an E1CB mechanism where a conjugate base is generated through proton abstraction prior to elimination of the leaving group.
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PPTX, PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
277 Ansichten19 SeitenSni, Nighbouring GP Participation & E1cb
Hochgeladen von
saheedvkThis document discusses different mechanisms for nucleophilic substitution reactions. It explains that nucleophilic substitution of alcohols with hydrogen halides can produce rearranged products through carbocation intermediates, but reactions with sulfur or phosphorus halides follow an SNi mechanism without rearrangement. The SNi mechanism involves the nucleophile attacking from the same side as the leaving group to give retention of configuration. Neighboring group participation allows some SN2 reactions to proceed with retention of configuration through two inversion steps. The document also describes an E1CB mechanism where a conjugate base is generated through proton abstraction prior to elimination of the leaving group.
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PPTX, PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 19
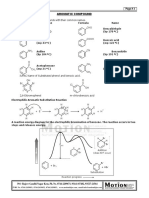
SNi MECHANISM
Nucleophilic substitution of alcohol with HX gives
haloalkane but there is possibilities for the formation of
rearranged products especially when there is formation
of carbocations.
However when sulphur or phosphorous halides are
used , no rearrangement product obtained. It follows Sni
mechanism (Substitution Nucleophilic Internal)
Eg:
R-OH + SOCl2 R-Cl + so2 + HCl
MECHANISM
FIRST STEP:
The lone pair of oxygen attacks electrophilic centre of
SO2Cl2 (S)gives positive charge In oxygen and
negative on other to form an unstable ion pair which
eliminates a molecule of HCl to form Alkyl chloro
sulphite intermediate (similar to SN1)
SECOND STEP
Internal attack of the nucleophile Chlorin to the electrophilic
Carbon from the same side , breaking C-O bond with the
elimination of SO2 molecule
Here Nucleophile attacks from the same side of the
leaving group which leads to retention of configuration
Eg
NIGHBOURING GROUP PARTICIPATION
In some cases the rate of SN2 reactions is found to be
extremely high and the Product is obtained with the retention
of configuration. These reactions follows Nighbouring group
mechanism
Here nighbouring group acts as nucleophile.. Ie one of the
nucleophile is a part of the same molecule and is located at
the C atom or farther from the leaving group
As it involves two steps with Sn2 mechanism two
inversions leads to retention of
configuration
In the first step attack of neighboring group at the
carbon from the backside forming an unstable
intermediate with inversion of configuration ( SN2
reaction )
In the second stage nucleophilic attack takes place to form
the product with inversion of configuration ( Another SN2
reaction) ie two SN2 reactions with inversion of configuration
. Final product with retention of configuration
Eg::
First step
Second step
L-2- BROMOPROPIONATE
L-2- METHOXY PROPIONATE
FEATURES
1)High reaction rate compared to SN2
2)Followsfirst order keinetics (substrate only)
3)Retention as a result of two inversion
4)Also called Anchimeric assistance
E1CB MECHANISM
(UNIMOLECULAR MECHANISM FROM A CONJUGATE BASE)
In E1CB mechanism a proton is initially abstracted to generate
conjugate base at equilibrium which then loses L- giving
elimination products in therate determing step
As the proton has to be removed from a carbon from a neutral
species ( a difficult job)
A strong base is needed for E1CB reaction as in E2 reaction. More
over increase in strength of base and concentration of base
increase the rate of E1CB reaction
If more than one beta hydrogen more acidic hydrogen is removed
Better the leaving group, better will be the rate of E1CB reaction ( if
E1CB is operating) Because rds involve the leaving group otherwise if
the leaving group is so good It will leave before the anion is formed
leading to E2 mechanism
E1CB mechanism operates when the abstraction of proton is very easy
( removal from a highly electronegative atom)and the conjugate base
is resonance stabilised)
E1CB mechanism also operates when the expulsion of leaving
group is difficult ( Leaving group is bad like F-,-OH-,RO- etc or if
leaving group has partial double bond due to resonance)
Eg
Dehydrohaloganation of geminal halohydrin because H+ is
removed from highly
electronegative oxygen
2
Dehydrohalogenation of carbonyl compounds
because resultant carbanion is resonance stabilised
If the leaving group is a better leaving group, E1CB reaction will takes place
But if the leaving group is very good E2 reaction will takesplace
3-
Dehydroflurination of because fluoride is a bad leaving group
4 Dehydrohalogenation when halogen has partial double bond due
to resonance in vinyl halide and aryl halides
5 Dehydration in basic medium
Das könnte Ihnen auch gefallen
- Enol N Ion EnolatDokument39 SeitenEnol N Ion EnolatJulia RahayuNoch keine Bewertungen
- 210 Fa 15 Exam 2 KEYDokument19 Seiten210 Fa 15 Exam 2 KEYdsarathy1Noch keine Bewertungen
- Resonance and Inductive Effects PresentationDokument36 SeitenResonance and Inductive Effects Presentationeagl33yeNoch keine Bewertungen
- Chemistry of Reactive Intermediate FinalDokument38 SeitenChemistry of Reactive Intermediate FinalTefera100% (1)
- Org Chem Sem 3 Paper 2Dokument15 SeitenOrg Chem Sem 3 Paper 2Rohit DeshmukhNoch keine Bewertungen
- Name Reaction 3569Dokument38 SeitenName Reaction 3569Ashish AmbekarNoch keine Bewertungen
- Neighbouring Group Participation or NGP inDokument4 SeitenNeighbouring Group Participation or NGP inbharatbhushansankhya100% (1)
- Lab ManualDokument19 SeitenLab Manualanon_467104036Noch keine Bewertungen
- CN102807528-PREPARATION METHOD OF 10-Methoxy IminostilbeneDokument4 SeitenCN102807528-PREPARATION METHOD OF 10-Methoxy IminostilbeneDipti DodiyaNoch keine Bewertungen
- Retrosynthetic Analysis PDFDokument6 SeitenRetrosynthetic Analysis PDFNoleNoch keine Bewertungen
- Module8 PDFDokument40 SeitenModule8 PDFFaizan AhmadNoch keine Bewertungen
- Organic Chemistry: Stereospecificity and StereoselectivityDokument10 SeitenOrganic Chemistry: Stereospecificity and StereoselectivityNida Adrees100% (1)
- SCH 206-Carboxylic Acids PDFDokument48 SeitenSCH 206-Carboxylic Acids PDFShivani DamorNoch keine Bewertungen
- SGDGDDDokument33 SeitenSGDGDDyopoboy100% (1)
- MSCCH 604Dokument203 SeitenMSCCH 604Gourav Biju100% (1)
- Organic Chemistry 100 Must-Know Mechanisms (Roman A. Valiulin) (Z-Library)Dokument288 SeitenOrganic Chemistry 100 Must-Know Mechanisms (Roman A. Valiulin) (Z-Library)Andrew GrantNoch keine Bewertungen
- Pericyclics-2014 Handout PDFDokument79 SeitenPericyclics-2014 Handout PDFnavchemNoch keine Bewertungen
- Chemistry Syllabus at Iit Kanpur For MSCDokument55 SeitenChemistry Syllabus at Iit Kanpur For MSCRoshayedAliLaskarNoch keine Bewertungen
- Organic Chemistry Chapter 8Dokument41 SeitenOrganic Chemistry Chapter 8채종희Noch keine Bewertungen
- Organic Chemistry ReactionDokument3 SeitenOrganic Chemistry ReactionGAMEPORIUMNoch keine Bewertungen
- Enzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisVon EverandEnzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisBewertung: 4 von 5 Sternen4/5 (2)
- Modern Aldol Reactions, Part2Dokument346 SeitenModern Aldol Reactions, Part2Kybernetikum100% (1)
- Neighbouring Group ParticipationDokument15 SeitenNeighbouring Group ParticipationAbdulMananNoch keine Bewertungen
- Problems On Named ReactionsDokument103 SeitenProblems On Named ReactionsBapu ThoratNoch keine Bewertungen
- Lecture Notes 2 Nano MaterialsDokument21 SeitenLecture Notes 2 Nano MaterialsHuzaifa ShabbirNoch keine Bewertungen
- TerpenoidsDokument17 SeitenTerpenoidsSayli Sawant100% (1)
- Csir Model QuestionsDokument15 SeitenCsir Model QuestionsbaluNoch keine Bewertungen
- Stereochemistry: 4 Edition Paula Yurkanis BruiceDokument43 SeitenStereochemistry: 4 Edition Paula Yurkanis Bruicenrguerrerod100% (1)
- Isomerism in Coordination Compounds PDFDokument7 SeitenIsomerism in Coordination Compounds PDFmf720383270100% (1)
- Huckel Rule of Aromaticity 2 PDFDokument25 SeitenHuckel Rule of Aromaticity 2 PDFUmar Farooq100% (1)
- Class Notes Heterocyclic ChemDokument218 SeitenClass Notes Heterocyclic ChemDeepanshu 1459Noch keine Bewertungen
- RearrangementsDokument64 SeitenRearrangementsBandita Datta50% (2)
- Module 2.3 NGP PDFDokument3 SeitenModule 2.3 NGP PDFIshaan ChaturvediNoch keine Bewertungen
- Functionalization of Indole and Pyrrole Cores Via Michael - Type AdditionsDokument61 SeitenFunctionalization of Indole and Pyrrole Cores Via Michael - Type AdditionsSilvina CancianNoch keine Bewertungen
- CH 4 Part 2 Quinoline and IsoquinolineDokument18 SeitenCH 4 Part 2 Quinoline and IsoquinolineJ'na Jamuna50% (4)
- Redox RxnsDokument30 SeitenRedox RxnsJolaine ValloNoch keine Bewertungen
- Reaction IntermediateDokument20 SeitenReaction IntermediateSiddarth Singh100% (2)
- Reaction MechanismDokument68 SeitenReaction MechanismSiddarth Singh73% (11)
- Aromaticity CompleteDokument104 SeitenAromaticity Completewahidalwahdi100% (1)
- The Negishi Coupling: An Update - Aldrichimica Acta Vol. 38 No. 3Dokument48 SeitenThe Negishi Coupling: An Update - Aldrichimica Acta Vol. 38 No. 3Sigma Aldrich Chemistry100% (1)
- Hetero-Cyclic CompoundsDokument69 SeitenHetero-Cyclic CompoundsNaveed SajidNoch keine Bewertungen
- Carboxylic AcidDokument21 SeitenCarboxylic AcidShalsabila NHNoch keine Bewertungen
- SET-NET Pericyclic ReactionsDokument61 SeitenSET-NET Pericyclic ReactionsBapu ThoratNoch keine Bewertungen
- Heterocyclic Chemistry Guidance Note and ProblemsDokument13 SeitenHeterocyclic Chemistry Guidance Note and ProblemsGhadeer M HassanNoch keine Bewertungen
- Chemistry Chapter 11 Alcohol, Phenol and EtherDokument32 SeitenChemistry Chapter 11 Alcohol, Phenol and EtherVidyakulNoch keine Bewertungen
- Name Reactions-IDokument33 SeitenName Reactions-ISatya KamNoch keine Bewertungen
- Chap 01 Some Basic Principles of Organic ChemistryDokument13 SeitenChap 01 Some Basic Principles of Organic ChemistryParth JainNoch keine Bewertungen
- 4.1.1 Protic Vs Aprotic SolventDokument36 Seiten4.1.1 Protic Vs Aprotic SolventDawit BirhanuNoch keine Bewertungen
- Functional Grour TransformationDokument60 SeitenFunctional Grour TransformationDipankar ArjyaNoch keine Bewertungen
- 04 Reactive IntermediatesDokument115 Seiten04 Reactive IntermediatesMuhammad ArsalanNoch keine Bewertungen
- Conformational Analysis: Carey & Sundberg: Part A Chapter 3Dokument53 SeitenConformational Analysis: Carey & Sundberg: Part A Chapter 3Dr-Dinesh Kumar100% (1)
- CARBONYL CONDENSATION REACTIONS 2 (10 Mei 2013)Dokument34 SeitenCARBONYL CONDENSATION REACTIONS 2 (10 Mei 2013)Mammy Nya AllyaNoch keine Bewertungen
- Aromaticity With Huckle's RuleDokument7 SeitenAromaticity With Huckle's RuleSk ZNoch keine Bewertungen
- Organic Synthesis. Functional Group InterconversionDokument57 SeitenOrganic Synthesis. Functional Group InterconversionJennifer Carolina Rosales NoriegaNoch keine Bewertungen
- Octant Rule, Axial Haloketone Rule GoodDokument11 SeitenOctant Rule, Axial Haloketone Rule Goodsaheedvk50% (4)
- Polymers & Plastics NotesDokument4 SeitenPolymers & Plastics NotessaheedvkNoch keine Bewertungen
- Octant Rule, Axial Haloketone Rule GoodDokument11 SeitenOctant Rule, Axial Haloketone Rule Goodsaheedvk50% (4)
- Drugs Affecting The Central Nervous SystemDokument78 SeitenDrugs Affecting The Central Nervous SystemsaheedvkNoch keine Bewertungen
- Drugs: Ideal Institute of TechnologyDokument42 SeitenDrugs: Ideal Institute of TechnologysaheedvkNoch keine Bewertungen
- Strategy Learning-01-Ashish Hattangdi, Atanu Ghosh PDFDokument14 SeitenStrategy Learning-01-Ashish Hattangdi, Atanu Ghosh PDFEmha RamadhanyNoch keine Bewertungen
- Illegal Drugs: Their Types and EffectsDokument35 SeitenIllegal Drugs: Their Types and EffectssaheedvkNoch keine Bewertungen
- Carbon 13 SpectrosDokument53 SeitenCarbon 13 SpectrossaheedvkNoch keine Bewertungen
- Everyone Can Help Keep Antibiotics Working: - by Using Antibiotics Only When Needed - Following Good Hygiene PracticesDokument35 SeitenEveryone Can Help Keep Antibiotics Working: - by Using Antibiotics Only When Needed - Following Good Hygiene PracticessaheedvkNoch keine Bewertungen
- Medication Administration: BY Thomas PetriciniDokument204 SeitenMedication Administration: BY Thomas PetricinisaheedvkNoch keine Bewertungen
- TUTORIAL 3 - Heterocyclic CompoundsDokument2 SeitenTUTORIAL 3 - Heterocyclic CompoundsSuhada SutajyNoch keine Bewertungen
- SN1 Vs SN2Dokument1 SeiteSN1 Vs SN2nurhanieyNoch keine Bewertungen
- ACS Review 12 Reactions of Arenes - Electrophilic Aromatic SDokument12 SeitenACS Review 12 Reactions of Arenes - Electrophilic Aromatic SMohamad HabbabaNoch keine Bewertungen
- Sharpless Asymmetric EpoxidationDokument3 SeitenSharpless Asymmetric EpoxidationvgvijuNoch keine Bewertungen
- Chem 210 CH 07 SN1 SN2 E1 E2Dokument11 SeitenChem 210 CH 07 SN1 SN2 E1 E2Krish Vora100% (1)
- Alcohol-Preparation of Grignard ReagentDokument4 SeitenAlcohol-Preparation of Grignard ReagentTida TpNoch keine Bewertungen
- ACS Review 18 Enols and EnolatesDokument11 SeitenACS Review 18 Enols and EnolatesJana BazziNoch keine Bewertungen
- Iminium Ion and Enamine Activation of CarbonylsDokument27 SeitenIminium Ion and Enamine Activation of CarbonylsPriya GoleNoch keine Bewertungen
- L LabsDokument7 SeitenL LabsHoodRomelNoch keine Bewertungen
- Substitution Versus Elimination Reactions: Presented By: Hina Javed Punjab Group of Colleges Rwp/IsbDokument15 SeitenSubstitution Versus Elimination Reactions: Presented By: Hina Javed Punjab Group of Colleges Rwp/IsbRaja Haris JavedNoch keine Bewertungen
- Ugi ReactionDokument11 SeitenUgi ReactionNavnath HatvateNoch keine Bewertungen
- Aromatic CompoundsDokument3 SeitenAromatic CompoundsRonak GurJarNoch keine Bewertungen
- Cannizaro ReactionsDokument3 SeitenCannizaro ReactionsvrchemieNoch keine Bewertungen
- Socl N Ag R-Cooh - R-Cocl - R-Cochn - R-CH Conu Ether, Et N Nucleophile (Nu)Dokument37 SeitenSocl N Ag R-Cooh - R-Cocl - R-Cochn - R-CH Conu Ether, Et N Nucleophile (Nu)Shanmugam RameshNoch keine Bewertungen
- Organic Chemistry Sn2 Sn1 E2 E1Dokument4 SeitenOrganic Chemistry Sn2 Sn1 E2 E1vishve2015Noch keine Bewertungen
- Dr. Sheppard CHEM 4201 Fall 2014 Exam I Study Guide: Max MaxDokument4 SeitenDr. Sheppard CHEM 4201 Fall 2014 Exam I Study Guide: Max MaxIbrahim MNoch keine Bewertungen
- Olefination ReactionDokument65 SeitenOlefination ReactionTùng SuNoch keine Bewertungen
- Steglich EsterificationDokument5 SeitenSteglich EsterificationDuc Anh NguyenNoch keine Bewertungen
- Name ReactionsDokument32 SeitenName ReactionsM.NandabalanNoch keine Bewertungen
- Aldehydes and Ketones (Questions)Dokument27 SeitenAldehydes and Ketones (Questions)Dhruv KuchhalNoch keine Bewertungen
- Indole and Pyrrole Synthesis PDFDokument19 SeitenIndole and Pyrrole Synthesis PDFAnuar HernandezNoch keine Bewertungen
- Class Notes Heterocyclic ChemDokument218 SeitenClass Notes Heterocyclic ChemDeepanshu 1459Noch keine Bewertungen
- Quinoline SynthesisDokument6 SeitenQuinoline SynthesisFrancesco TutinoNoch keine Bewertungen
- Reagent ListDokument5 SeitenReagent ListAditya VermaNoch keine Bewertungen
- Coupling ReactionDokument2 SeitenCoupling ReactionAreesha FatimaNoch keine Bewertungen
- SN E Flowchart PDFDokument1 SeiteSN E Flowchart PDFElaina JosephNoch keine Bewertungen
- Chem 215 Myers: Sharpless Asymmetric Dihydroxylation ReactionDokument4 SeitenChem 215 Myers: Sharpless Asymmetric Dihydroxylation ReactionPrabhakar S AchantaNoch keine Bewertungen
- Substitution and EliminationDokument79 SeitenSubstitution and Eliminationjana srutiNoch keine Bewertungen
- Sharad Pratap Singh's Answer To Is It Necessary To Memorise The Name of The Organic Reactions For JEE or Is The Mechanism and Reagents Enough - QuoraDokument7 SeitenSharad Pratap Singh's Answer To Is It Necessary To Memorise The Name of The Organic Reactions For JEE or Is The Mechanism and Reagents Enough - QuorasumitNoch keine Bewertungen
- 1-16Dokument5 Seiten1-16Ayu MilineaNoch keine Bewertungen