Beruflich Dokumente
Kultur Dokumente
Fluoroscopy: Imaging System Used For Monitoring Catheter Based Procedures
Hochgeladen von
Ahallya Jaladeep0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
127 Ansichten19 SeitenThese lectures give a brief into to fluoroscopy
Originaltitel
Fluoroscopy
Copyright
© © All Rights Reserved
Verfügbare Formate
PPTX, PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenThese lectures give a brief into to fluoroscopy
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PPTX, PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
127 Ansichten19 SeitenFluoroscopy: Imaging System Used For Monitoring Catheter Based Procedures
Hochgeladen von
Ahallya JaladeepThese lectures give a brief into to fluoroscopy
Copyright:
© All Rights Reserved
Verfügbare Formate
Als PPTX, PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 19
FLUOROSCOPY
Imaging system used for monitoring Catheter
based procedures.
Fluoroscopy overview
• The principal feature of the imaging chain that
distinguishes Fluoroscopy from radiography is the ability
to produce real-time x-ray images with high frame rates
and a low-dose per image. (5 configurations possible)
• gastrointestinal/genitourinary (GI/GU) systems (with the x-ray tube
under the patient table);
• remote fluoroscopy (over-table x-ray tube);
• interventional vascular (vascular surgery, radiology);
• cardiology and interventional electrophysiology, and
• mobile C-arm confgurations (surgery, pain medicine).
• All confgurations have the same basic components.
Fluoroscopy Imaging Chain
• The basic product of the
Fluoroscopy system is a
projection image.
• Images are acquired at
movie frame rates, so at
30 fps a five minute
session would mean
9000 images.
• So the system should be
capable of obtaining
quality images at low
‘Doses’.
Sensitive Detectors
• Image Intensifiers.
• Flat Panel Detectors.
• Flat panel detectors requires way less radiation than standard
radiography detectors for producing acceptable quality images. For
instance fluoroscopy requires 9-17 nGy incident upon the detector
per image while a X-ray CT or CAT scanner detector requires 5-9
microGy.
Image Intensifier based Fluoroscopy
Image Intensifier Image Intensifier
• (a) a vacuum housing to keep
air out and allow unimpeded
electron flow,
• (b) an input layer that converts
the absorbed incident x-rays
into light, which in turn releases
electrons,
• (c) an electron optics system
that accelerates and focuses
the electrons emitted by the
input layer onto the output
layer, and
• (d) an output phosphor that
converts the accelerated
electrons
Image Intensifier..
• Incident x-rays pass through
the antiscatter grid (not shown),
and must also pass through the
vacuum support window (1 mm
Al) and the support of the input
phosphor (0.5 mm Al).
• X-rays are absorbed by the CsI
input phosphor, producing green
light that is channeled within the
long CsI needles to the
antimony-trisulfide
photocathode, which then ejects
electrons into the vacuum space
of the II.
• Scanning electron micrographs
illustrate the crystalline needle–
like structure of CsI.
Image Intensifier….
CsI
• 400 micron tall and 5 micron wide columnar structures on
top of a substrate, channels light with minimal spreading
as compared to other phosphors.
• For a 60-keV x-ray photon, 3000 light photons are
emitted.
• Photocathode is a thin layer of antimony and alkali metals
that emits electrons when struck by visible light.
• 10-20% conversion efficiency, 400 electrons are released
per 60keV X-ray photon captured in the CsI.
Image Intensifier…
Electron Optics
• The electrons are accelerated by a large electric fielld
created by a high voltage- between the anode and the
cathode.
• Focusing the electrons is accomplished by the several
electrodes in the electron optics chain.
• The kinetic energy of each electron is dramatically
increased by acceleration due to the voltage difference
between the cathode and anode, resulting in electronic
gain.
• The spatial pattern of elec-trons released at the
photocathode is maintained at the output phosphor, albeit
minified
Distortions due to electron optics
Output Phosphor
• The output phosphor is made of zinc
cadmium sulfide doped with silver
(ZnCdS: Ag), which has a green (530 nm)
emission spectrum.
• The ZnCdS phosphor particles are very
small (1 to 2 micron), and the output
phosphor is quite thin (4 to 8 micron), to
preserve high spatial resolution.
• The anode is a very thin (0.2 mm) coating
of aluminum on the vacuum side of the
output phosphor, which is electri-cally
conductive to carry away the electrons
once they deposit their kinetic energy in
the phosphor.
• Each electron causes the emission
approximately 1,000 light photons from
the output phosphor.
The ‘Intensification’
• A 15-35 cm diameter input image is focused to about 2.5cm circle
• To preserve a resolution of 5 line pairs/mm at the input plane, the output
phosphor must deliver a resolution of about 70 lp/mm or better.
• The reduction in image diameter also leads to light intensity amplification,
since, for example, the electrons produced on the 410 cm2 area of the
23-cm-diameter input phosphor are concentrated onto the 5-cm2 output
phosphor.
• By analogy, a magnifying glass collects light over its lens surface area
and can focus it onto a small spot, and in the process, the light intensity
is amplified enough to start a fire using only sunlight.
• The so-called minification gain of an II is simply the ratio of the area of
the input phosphor to that of the output phosphor.
• The ratio of areas is equal to the square of the ratio of diameters, so a
23-cm-diameter II (with a 2.5-cm-diameter output phosphor) has a
minification gain of (22.8/2.55)^2 =81, and in 17-cm mode it is 49.
Optical coupling to video camera
• The lenses used in fluoroscopy systems focus light
emitted at the output phosphor onto the focal plane of the
video camera.
• The lens assembly includes an adjustable aperture,
basically a small hole positioned between individual
lenses. Adjusting the size of the hole changes how much
light gets through the lens system.
• By lowering the gain of the II and associated optics by
increasing the f-number, a higher x-ray exposure rate will
result, producing lower noise images and higher patient
dose. Increasing the gain reduces the x-ray exposure rate
and lowers the dose, but reduces image quality.
Brightness Gain
• The brightness gain is the product of the electronic and
minification gains of the II. The electronic gain of an II is
roughly about 50, and the minification gain is variable
depending on the electronic magnifcation
• . For a 30-cm (12-inch) diameter II, the minification gain
is 144, but in 23-cm (9-inch) mode it is 81, and in 17-cm
(7-inch) mode it is 49.
• The overall brightness gain, therefore, ranges from about
2,500 to 7,000. As the effective diameter (FOV) of the
input phosphor decreases (increasing magnification), the
brightness gain decreases.
Automatic Exposure rate control
• The purpose of the automatic exposure rate control
(AERC) circuit is to keep the signal-to-noise ratio (SNR) of
the image constant when possible.
• The ABC or AERC sensor constantly monitors the
intensity of the X-ray photons and strives to keep the
photon fluence level at the detector, constant.
• Whenever X-ray fluence is lowered a signal is sent to the
X-ray generator to increase the incident fluence level. This
can be done by changing the mA and kV.
• Some AERC systems manipulate the lens aperture once
the regulatory limit is reached.
Fluoroscopy modes of operation
• Continuous fluoroscopy
• 33 ms integration time
• Variable frame rate pulsed fluoroscopy
• 3-8ms integration time
• High frame rates not necessary all the time, can be used in fast
moving anatomy ie pulsative blood vessels.
Field of View and Magnification Modes
• IIs are available in 23,30,35 and 40cm field of view.
• For GI and GU large FOV is required
• 23cm is adequate for cardiac imaging
• Magnification modes are obtained by changing the
voltages applied to the electrodes in II, resulting in
electron focusing from a smaller area of the II input
screen.
• When the magnification mode is engaged the X-ray
collimator automatically adjusts to match the X-ray beam
dimensions to the smaller field of view.
Flat panel detector based fluoroscopy
systems
• Comprised of thin film transistors arrays of individual
detector elements, each element of the array ie pixel has
its own circuitry to record the X-ray photon intensity
incident on that pixel.
• Each pixel has a capacitor for storing the charge induced
(charge scales linearly with the incident energy of the X-
rays) and a transistor which acts as a switch
• Direct and Indirect X-ray detection.
• Direct detection- X-ray induced charge in Selenium is directly
detected.
• Indirect detection- X-ray is converted to light and then detected.
Photodiodes convert light to charge, which is then stored in a
capacitor attached to each pixel element.
Resolution
• The limit is set by the size of the detector element.
• For a system with 30cm fOV and 1024^2 imaging matrix,
the nyquist sampling frequency is 1.7 lp/mm
corresponding to a detector element size of 0.29mm.
Fluoroscopy Suites
• Genitourinary
• Remote fluoroscopy rooms
• Peripheral angiography suites
• Cardiology catheterization suites
• Cardiology electrophysiology laboratory
• Biplane aniographic systems
• Mobile fluoroscopy C-arms
Das könnte Ihnen auch gefallen
- Fluoroscopy & Digital FluorosDokument33 SeitenFluoroscopy & Digital Fluorosprathinam11Noch keine Bewertungen
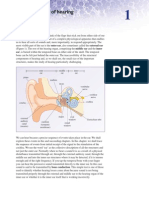
- The Mechanics of HearingDokument13 SeitenThe Mechanics of HearingDustin NguyenNoch keine Bewertungen
- 160 KV & 200 KV: Operator and Service ManualDokument52 Seiten160 KV & 200 KV: Operator and Service ManualJuan Castillo100% (1)
- FluorosDokument85 SeitenFluorosRoshan Chaudhary100% (1)
- Unit 4 MIT Notes SendDokument32 SeitenUnit 4 MIT Notes SendEr Amit VermaNoch keine Bewertungen
- Reference Book: Stewart C.bushong: by Huzaifa Atique Sir Syed University of Engineering & TechnologyDokument20 SeitenReference Book: Stewart C.bushong: by Huzaifa Atique Sir Syed University of Engineering & TechnologyquickdannyNoch keine Bewertungen
- X-Ray Basics: A Concise Guide to X-Ray Production and Image FormationDokument43 SeitenX-Ray Basics: A Concise Guide to X-Ray Production and Image Formationakshaykohli7890Noch keine Bewertungen
- Hardware CircuitryDokument22 SeitenHardware CircuitryMahesh NeelarapuNoch keine Bewertungen
- Health Problem Addressed: Umdns GMDNDokument1 SeiteHealth Problem Addressed: Umdns GMDNYechale TafereNoch keine Bewertungen
- 950 PLUS 12kW HF Generator with 1MP CameraDokument2 Seiten950 PLUS 12kW HF Generator with 1MP CameraWael Fuad AL-MaktariNoch keine Bewertungen
- 1.0 The Origin of Biopotentials (New)Dokument62 Seiten1.0 The Origin of Biopotentials (New)Hanif HussinNoch keine Bewertungen
- Biomedical Instruments Classification GuideDokument53 SeitenBiomedical Instruments Classification GuideSujith SurendranNoch keine Bewertungen
- 500ma X-Ray Machine: Radiography System: 500ma Standard CompositionDokument3 Seiten500ma X-Ray Machine: Radiography System: 500ma Standard CompositionMohsin LatifNoch keine Bewertungen
- Service Manual For X-Ray Collimator: Model Nk202Dokument17 SeitenService Manual For X-Ray Collimator: Model Nk202Wael Fuad AL-MaktariNoch keine Bewertungen
- Resident Physics Lectures: Digital Subtraction Angiography (DSA)Dokument41 SeitenResident Physics Lectures: Digital Subtraction Angiography (DSA)Lajja Parikh PatelNoch keine Bewertungen
- T 670Dokument2 SeitenT 670istenicNoch keine Bewertungen
- Blood Gas AnalyzerDokument13 SeitenBlood Gas Analyzeranon_708486566Noch keine Bewertungen
- Principles of Nuclear Medicine 1Dokument34 SeitenPrinciples of Nuclear Medicine 1damaliso nyirongo2Noch keine Bewertungen
- SAFE LIGHTING FOR PHOTOGRAPHY DARKROOMSDokument13 SeitenSAFE LIGHTING FOR PHOTOGRAPHY DARKROOMSShahid ShdNoch keine Bewertungen
- CT ANGIOGRAPHY - DETAILED IMAGES OF BLOOD VESSELSDokument1 SeiteCT ANGIOGRAPHY - DETAILED IMAGES OF BLOOD VESSELSsreejithNoch keine Bewertungen
- Semiconductor Diode and Transistor FundamentalsDokument355 SeitenSemiconductor Diode and Transistor FundamentalsEman AlbinoNoch keine Bewertungen
- Whole Blood Coagulation Analyzer PDFDokument1 SeiteWhole Blood Coagulation Analyzer PDFmorton1472Noch keine Bewertungen
- FLUOROSDokument14 SeitenFLUOROSAddisu Amare Zena 18BML0104100% (1)
- Physics X-Ray PDFDokument30 SeitenPhysics X-Ray PDFRT Windy100% (1)
- X-Ray Generator Circuits GuideDokument24 SeitenX-Ray Generator Circuits GuideThyago MangueiraNoch keine Bewertungen
- PHCM SeminarDokument87 SeitenPHCM SeminarLalaine De JesusNoch keine Bewertungen
- Defibrillator Waveforms and Cardioversion Procedures ExplainedDokument28 SeitenDefibrillator Waveforms and Cardioversion Procedures ExplainedAnish H DaveNoch keine Bewertungen
- Analytical Equipment: High AccuracyDokument6 SeitenAnalytical Equipment: High AccuracyMartin ClydeNoch keine Bewertungen
- Slide Ultrasound Part 1Dokument27 SeitenSlide Ultrasound Part 1Hanif HussinNoch keine Bewertungen
- Audiometers OK PDFDokument81 SeitenAudiometers OK PDFErnesto AcostaNoch keine Bewertungen
- Biological Safety CabinetsDokument4 SeitenBiological Safety CabinetsHedy Mae Bautista100% (1)
- Evaluation FormDokument6 SeitenEvaluation FormJames Magno100% (1)
- Biological Safety Cabinet SafesolDokument9 SeitenBiological Safety Cabinet SafesolnatrajangNoch keine Bewertungen
- Whatdoyouknowhba1c PDFDokument20 SeitenWhatdoyouknowhba1c PDFahmet keserNoch keine Bewertungen
- Detector System in CTDokument11 SeitenDetector System in CTKuber NeupaneNoch keine Bewertungen
- Phys 162: Solid State Devices Chapter 2 Diode Applications HW 2Dokument5 SeitenPhys 162: Solid State Devices Chapter 2 Diode Applications HW 2Ishamael SnookNoch keine Bewertungen
- L14 UltrasoundDokument27 SeitenL14 UltrasoundLinh PhanNoch keine Bewertungen
- Bon Slitlamp SL-72, SL-75 - User Und Service ManualDokument21 SeitenBon Slitlamp SL-72, SL-75 - User Und Service ManualRobertoNoch keine Bewertungen
- Ivc FilterDokument15 SeitenIvc FilterashishNoch keine Bewertungen
- PLX101D High Frequency Mobile X-Ray Equipment: Nanjing Perlove Medical Equipment Co. LTDDokument3 SeitenPLX101D High Frequency Mobile X-Ray Equipment: Nanjing Perlove Medical Equipment Co. LTDRaditya Desta100% (1)
- Irfan Nurhady (P1337430117060) Dyah Ayu Dwi W (P1337430117061) Kelas Ib Muhamad Fakhrul Rivan (P1337430117062) Galih Tama Ramadhani (P1337430117063)Dokument31 SeitenIrfan Nurhady (P1337430117060) Dyah Ayu Dwi W (P1337430117061) Kelas Ib Muhamad Fakhrul Rivan (P1337430117062) Galih Tama Ramadhani (P1337430117063)Nancy Vidya AgustineNoch keine Bewertungen
- Generation of XraysDokument45 SeitenGeneration of Xraysmuli ani100% (1)
- Diathermy: Presented By: Jignasha PatelDokument84 SeitenDiathermy: Presented By: Jignasha PatelBhargavNoch keine Bewertungen
- Hospt Equip 10 PDFDokument1 SeiteHospt Equip 10 PDFMohammed Al-shamiriNoch keine Bewertungen
- Ultrasonic Imaging SystemDokument20 SeitenUltrasonic Imaging SystemSai SridharNoch keine Bewertungen
- Procedure On Ventricular Septal Defect (VSD) Device ClosureDokument9 SeitenProcedure On Ventricular Septal Defect (VSD) Device ClosurejaganjaggiNoch keine Bewertungen
- Radiographic ExposureDokument70 SeitenRadiographic ExposurechowhkNoch keine Bewertungen
- X-Ray Grids: Dr.R.Raja Singh First Year PG in Radio Diagnosis Madurai Medical CollegeDokument18 SeitenX-Ray Grids: Dr.R.Raja Singh First Year PG in Radio Diagnosis Madurai Medical CollegeFreni FreniNoch keine Bewertungen
- 1.5 Image Sampling and QuantizationDokument17 Seiten1.5 Image Sampling and Quantizationkuladeep varmaNoch keine Bewertungen
- HSB Human Humalyzer Calibration Toolset 001 PDFDokument2 SeitenHSB Human Humalyzer Calibration Toolset 001 PDFluisoft88Noch keine Bewertungen
- Imaging With X-RaysDokument107 SeitenImaging With X-RaysNipunNoch keine Bewertungen
- English M23 Battery Charger, Choke, Condencer, Diode and Transformer (SSEE6)Dokument16 SeitenEnglish M23 Battery Charger, Choke, Condencer, Diode and Transformer (SSEE6)john powerNoch keine Bewertungen
- Fluoro QaDokument16 SeitenFluoro Qahema0% (1)
- Hospital Training: Single Positron Emission Computed TomographyDokument24 SeitenHospital Training: Single Positron Emission Computed TomographyshruthiNoch keine Bewertungen
- Ultrasound Machine: Unit C18.6 Maintaining Medical Imaging Equipment 18.6.7 Maintain An Ultrasound MachineDokument17 SeitenUltrasound Machine: Unit C18.6 Maintaining Medical Imaging Equipment 18.6.7 Maintain An Ultrasound Machineanwar1971Noch keine Bewertungen
- RT 255 Dsa Fluoro 2010 11Dokument54 SeitenRT 255 Dsa Fluoro 2010 11IBRAHIM100% (1)
- Lecure-5 The Origin of Biopotentials - 2Dokument34 SeitenLecure-5 The Origin of Biopotentials - 2Noor Ahmed100% (1)
- Basics of AutoclaveDokument53 SeitenBasics of AutoclaveWallace GualandiNoch keine Bewertungen
- FLUOROSDokument28 SeitenFLUOROSNaty SeyoumNoch keine Bewertungen
- 13 - FluorosDokument30 Seiten13 - FluorosSinan Erol TektaşNoch keine Bewertungen
- LS NI: Jack Leigh, 1939-2008Dokument54 SeitenLS NI: Jack Leigh, 1939-2008Ahallya JaladeepNoch keine Bewertungen
- Module2 PDFDokument406 SeitenModule2 PDFAhallya JaladeepNoch keine Bewertungen
- Coote Gsas - Harvard 0084L 11632Dokument176 SeitenCoote Gsas - Harvard 0084L 11632Ahallya JaladeepNoch keine Bewertungen
- IABP Class FinalDokument166 SeitenIABP Class FinalAhallya Jaladeep100% (1)
- Module5 PDFDokument115 SeitenModule5 PDFAhallya JaladeepNoch keine Bewertungen
- Gate Test Series 4 Chemistry PDFDokument16 SeitenGate Test Series 4 Chemistry PDFAhallya JaladeepNoch keine Bewertungen
- Final Suction Catheters and TubesDokument125 SeitenFinal Suction Catheters and TubesAhallya JaladeepNoch keine Bewertungen
- Surgical Device Heart ValveDokument53 SeitenSurgical Device Heart ValveAhallya JaladeepNoch keine Bewertungen
- Aromaticity, Pericyclic Reactions and PhotochemistryDokument181 SeitenAromaticity, Pericyclic Reactions and PhotochemistryAhallya JaladeepNoch keine Bewertungen
- Module 7Dokument127 SeitenModule 7millinagi95Noch keine Bewertungen
- Module3 PDFDokument174 SeitenModule3 PDFAhallya JaladeepNoch keine Bewertungen
- Module 4Dokument158 SeitenModule 4Sreedevi KrishnakumarNoch keine Bewertungen
- Module8 PDFDokument227 SeitenModule8 PDFAhallya JaladeepNoch keine Bewertungen
- Organometallic Chemistry: A Structured Introduc5on To A Complex But Fascina5ng Field!Dokument48 SeitenOrganometallic Chemistry: A Structured Introduc5on To A Complex But Fascina5ng Field!Ahallya JaladeepNoch keine Bewertungen
- Module6 PDFDokument135 SeitenModule6 PDFAhallya JaladeepNoch keine Bewertungen
- Organometallic Module 9Dokument127 SeitenOrganometallic Module 9Ahallya JaladeepNoch keine Bewertungen
- Experimental Study of Humidification AndDehumidifiDokument19 SeitenExperimental Study of Humidification AndDehumidifiHanz GianNoch keine Bewertungen
- Study of Process Parameters in High Pressure Die CastingDokument9 SeitenStudy of Process Parameters in High Pressure Die Castingruben6286Noch keine Bewertungen
- Environmental Pollution Upsc Notes 90 PDFDokument6 SeitenEnvironmental Pollution Upsc Notes 90 PDFSumit ChauhanNoch keine Bewertungen
- Analytical Method Development and Validation For The Test Related Substances of Pomalidomide in Pomalidomide CapsulesDokument8 SeitenAnalytical Method Development and Validation For The Test Related Substances of Pomalidomide in Pomalidomide CapsulesInternational Journal of Innovative Science and Research TechnologyNoch keine Bewertungen
- For Gas Testing Examination. Unit 1 Air CompositionDokument3 SeitenFor Gas Testing Examination. Unit 1 Air CompositionAman KumarNoch keine Bewertungen
- New Microsoft Office Word DocumentDokument26 SeitenNew Microsoft Office Word DocumentAnitha SathishNoch keine Bewertungen
- Surface Vehicle Standard: Rev. MAY97Dokument4 SeitenSurface Vehicle Standard: Rev. MAY97anupthattaNoch keine Bewertungen
- Dumas Method LabDokument5 SeitenDumas Method LabJohn JosephNoch keine Bewertungen
- Heterogeneous CatalystDokument24 SeitenHeterogeneous Catalystlalukalu420Noch keine Bewertungen
- Proses ManufakturDokument5 SeitenProses ManufakturAchmad HabibieNoch keine Bewertungen
- Progress in Starch Modification in The Last Decade PDFDokument7 SeitenProgress in Starch Modification in The Last Decade PDFLau MaRtiinezNoch keine Bewertungen
- (J. Throck Watson, O. David Sparkman) IntroductionDokument253 Seiten(J. Throck Watson, O. David Sparkman) IntroductionAntonio Carlos da SilvaNoch keine Bewertungen
- AnsiDokument4 SeitenAnsiHendrie NMcNoch keine Bewertungen
- 1 IntoductionDokument2 Seiten1 IntoductionAshutosh SinghNoch keine Bewertungen
- Glydant Plus Liquid (I) 2006Dokument2 SeitenGlydant Plus Liquid (I) 2006Enrique Alberto Valenzuela Hernandez0% (1)
- PPTDokument13 SeitenPPTAJINKYANoch keine Bewertungen
- Synthesis of 2-NitropropeneDokument3 SeitenSynthesis of 2-NitropropeneVictor VikeneNoch keine Bewertungen
- A Presentation On ChemistryDokument18 SeitenA Presentation On ChemistryBimal DasNoch keine Bewertungen
- Linear Thermal Expansion of Solid Materials With A Vitreous Silica DilatometerDokument7 SeitenLinear Thermal Expansion of Solid Materials With A Vitreous Silica Dilatometerluis_may22Noch keine Bewertungen
- w4g Book1a r6Dokument99 Seitenw4g Book1a r6milker100% (1)
- Good Laboratory Practise PCR Qsop38 2010Dokument12 SeitenGood Laboratory Practise PCR Qsop38 2010Abu KanshaNoch keine Bewertungen
- Bock PDFDokument147 SeitenBock PDFjuliaNoch keine Bewertungen
- Rankine & Carnot Cycle - Power Plant Questions and Answers - SanfoundryDokument4 SeitenRankine & Carnot Cycle - Power Plant Questions and Answers - SanfoundryShubhendra YadavNoch keine Bewertungen
- Item Bank Grade 9 SciencDokument12 SeitenItem Bank Grade 9 SciencMarjorie BrondoNoch keine Bewertungen
- Smith, P. R., Jardine, R. J., & Hight, D. W. (1992) - The Yielding of Bothkennar Clay.Dokument18 SeitenSmith, P. R., Jardine, R. J., & Hight, D. W. (1992) - The Yielding of Bothkennar Clay.Daniel Felipe Rodriguez RamirezNoch keine Bewertungen
- Effect of Chemical FertilizerDokument4 SeitenEffect of Chemical FertilizerMrithulaVaidyanathanNoch keine Bewertungen
- Offset Inks - BasicsDokument5 SeitenOffset Inks - BasicsforeverhaleemNoch keine Bewertungen
- Effect of Plasticizers On The Properties of Polyvinyl Alcohol FilmsDokument14 SeitenEffect of Plasticizers On The Properties of Polyvinyl Alcohol FilmsJuanNoch keine Bewertungen
- Design and Fabrication of Condenser For A Room Air Conditioning System (Rep1.)Dokument40 SeitenDesign and Fabrication of Condenser For A Room Air Conditioning System (Rep1.)Satish Alan100% (1)