Beruflich Dokumente
Kultur Dokumente
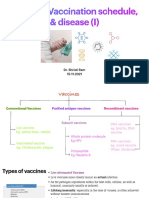
EPI Vaccines: - Inactivated (Killed) Microorganisms - Attentuated Microorganisms Fragments From Microorganisms Toxoids
Hochgeladen von
Irene Grace Balcueva0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
49 Ansichten3 SeitenOriginaltitel
EPI-Vaccines-final
Copyright
© © All Rights Reserved
Verfügbare Formate
PPTX, PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
© All Rights Reserved
Verfügbare Formate
Als PPTX, PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
49 Ansichten3 SeitenEPI Vaccines: - Inactivated (Killed) Microorganisms - Attentuated Microorganisms Fragments From Microorganisms Toxoids
Hochgeladen von
Irene Grace BalcuevaCopyright:
© All Rights Reserved
Verfügbare Formate
Als PPTX, PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 3
EPI Vaccines
• inactivated (killed) microorganisms
• uses the killed version of the microorganism that causes the disease
• attentuated microorganisms
• live microorganisms that have been altered so that they are no longer
pathogenic, but are still antigenic.
• fragments from microorganisms
• contains a small part or piece of the microorganism selected for its
ability to initiate a specific immune response
• toxoids
• made from the microorganism inactivated toxic compound
VACCINE CONTENTS FORM
BCG (Bacillus Calmette Guerine) Live, attentuated bacteria Freeze-dried, reconstituted with a
special diluent
Hepatitis B Vaccine RNA-recombinant, using Hepatitis B Cloudy, liquid, in an auto disable
surface antigen (HBs Ag) injection syringe if available
DPT - HepB - Hib Diphtheria toxoid, inactived pertussis Liquid, in an auto-disable injectio
(Pentavalent vaccine) bacteria, tetanus toxoid, recombinant syringe
DNA surface antigen, and synthetic
conjugate of Haemmophilus
influenzae B bacilli
Oral Polio Vaccine Live, attenuated viruss (trivalent) Clear, pinkish liquid
Anti-measles Vaccine (AMV1) Live, attenuated virus Freeze-dried, reconstituted with a
special diluent
Measles-Mumps-Rubella Vaccine Live, attenuated viruses Freeze-dried, reconstituted with a
(AMV2) special diluent
Rotavirus Vaccine Live, attenuated virus Clear, colorless liquid, in a container
with an oral applicator
Tetanus Toxoid Weakened toxin Sometimes slightly turbid in
appearance; Clear, colorless liquid
Target Setting and Vaccine Requirements
• First specific goal of EPI: 100% immunization of infants/children
against the most common vaccine-preventable diseases
• Vaccine is calculated based on eligible populations.
• Formulas to estimated eligible population:
Estimated number of infants = Total population x 2.7%
Estimated number of 12-to-59 month old children
= total population x 10.8%
Estimated number of pregnant women = total population x 3.5%
Das könnte Ihnen auch gefallen
- Maintaining Potency of EPI VaccinesDokument13 SeitenMaintaining Potency of EPI VaccinesMarylle Joy SullanoNoch keine Bewertungen
- Immunization: For C-1 Med Student DR Muluken JAN/2018Dokument71 SeitenImmunization: For C-1 Med Student DR Muluken JAN/2018Yemata HailuNoch keine Bewertungen
- Expanded Program On Immunization 5Dokument17 SeitenExpanded Program On Immunization 5Tamayo, Shaina AngelaNoch keine Bewertungen
- Bacterial Vaccines: Fluid ToxoidsDokument4 SeitenBacterial Vaccines: Fluid ToxoidsMicha Aguilar ManuzonNoch keine Bewertungen
- Immunization: Expanded Program On Immunization (EPI)Dokument4 SeitenImmunization: Expanded Program On Immunization (EPI)3amabelle arevaloNoch keine Bewertungen
- Unit 3Dokument150 SeitenUnit 3SUBIKSHAA S (RA1911009010233)Noch keine Bewertungen
- PDF Tablet Lepas LambatDokument13 SeitenPDF Tablet Lepas Lambatannisya bubblesNoch keine Bewertungen
- Imunizarea Si Vaccinologia: ImunitateaDokument13 SeitenImunizarea Si Vaccinologia: ImunitateaLoredana CîrlanNoch keine Bewertungen
- Vaccines: Viral Vaccines Bacterial VaccinesDokument3 SeitenVaccines: Viral Vaccines Bacterial VaccinesCharan RebelNoch keine Bewertungen
- Immunological ProductsDokument19 SeitenImmunological ProductsPharma 24Noch keine Bewertungen
- Imunisasi: Bambang MulyawanDokument72 SeitenImunisasi: Bambang MulyawanAnonymous MxxkdtMt6uNoch keine Bewertungen
- IMMUNOPROPHYLAXISDokument30 SeitenIMMUNOPROPHYLAXISAli TARARNoch keine Bewertungen
- Recombinant VaccinesDokument11 SeitenRecombinant VaccinesAshna JoshiNoch keine Bewertungen
- Immunization Program of The PhilippinesDokument142 SeitenImmunization Program of The PhilippinesSophia LalagunaNoch keine Bewertungen
- Vaccine Project - FinalDokument2 SeitenVaccine Project - Finalchelsea garzaNoch keine Bewertungen
- 1 Immunization جمعت كل مهم هناDokument13 Seiten1 Immunization جمعت كل مهم هناReem 10Noch keine Bewertungen
- BscNR20 - Topic 8 - VaccinesDokument6 SeitenBscNR20 - Topic 8 - Vaccinesakoeljames8543Noch keine Bewertungen
- Immunological ProductsDokument35 SeitenImmunological ProductsUMME SADEA RAHMANNoch keine Bewertungen
- MODULE 2: Types of Vaccine and Adverse ReactionsDokument29 SeitenMODULE 2: Types of Vaccine and Adverse ReactionsnandaNoch keine Bewertungen
- HEPADNAVIRIDAEDokument14 SeitenHEPADNAVIRIDAEnur qistina humaira zulkarshamsiNoch keine Bewertungen
- Vaccination PDF.'''''Dokument14 SeitenVaccination PDF.'''''Ompriya SNoch keine Bewertungen
- Vaccines: ImmunityDokument1 SeiteVaccines: ImmunityJohnNoch keine Bewertungen
- Immuniz Ation: Elaine Frances M. Illo, R.M., R.NDokument59 SeitenImmuniz Ation: Elaine Frances M. Illo, R.M., R.NElaine Frances IlloNoch keine Bewertungen
- Bacterial, Viral, Polysaccharides, Recombinant and DNA VaccinesDokument15 SeitenBacterial, Viral, Polysaccharides, Recombinant and DNA VaccinesAshwini_MCCNoch keine Bewertungen
- Imun VaksinDokument62 SeitenImun VaksinLaksmi DwiNoch keine Bewertungen
- EXPANDED IMMUNIZATION PROGRAMDokument6 SeitenEXPANDED IMMUNIZATION PROGRAMIana CastigadorNoch keine Bewertungen
- EDIBLE VACCINES: A COST-EFFECTIVE APPROACHDokument23 SeitenEDIBLE VACCINES: A COST-EFFECTIVE APPROACHVirendra JoshiNoch keine Bewertungen
- Immunization Program of The PhilippinesDokument9 SeitenImmunization Program of The Philippinesnelar MacabalitaoNoch keine Bewertungen
- Different Types of Vaccines: Preparation of Measles VaccineDokument8 SeitenDifferent Types of Vaccines: Preparation of Measles VaccineEkta ManglaniNoch keine Bewertungen
- Community Health Nursing Doh Related Programs: Epi - Types of VaccinesDokument32 SeitenCommunity Health Nursing Doh Related Programs: Epi - Types of VaccinesAngelaNoch keine Bewertungen
- Microorganisms - Lec 6Dokument10 SeitenMicroorganisms - Lec 605 Nalin PrabhatNoch keine Bewertungen
- Vaccine ScheduleDokument17 SeitenVaccine ScheduleMonica SreeNoch keine Bewertungen
- Immunization 2021Dokument67 SeitenImmunization 2021matucojulioNoch keine Bewertungen
- CHN Immunization NotesDokument9 SeitenCHN Immunization NotesJustin AncogNoch keine Bewertungen
- HBB 2306 Notes IIIDokument5 SeitenHBB 2306 Notes IIIALEXNoch keine Bewertungen
- Community Health Nursing Doh Related Programs: Epi - Types of VaccinesDokument32 SeitenCommunity Health Nursing Doh Related Programs: Epi - Types of VaccinesAngelaNoch keine Bewertungen
- NBVHJVDokument20 SeitenNBVHJVThe BestNoch keine Bewertungen
- Updated Immunzation TopicDokument58 SeitenUpdated Immunzation TopicAbbey FritschNoch keine Bewertungen
- Elements of Immunity Acquired ImmunityDokument3 SeitenElements of Immunity Acquired ImmunitySurya AdhikariNoch keine Bewertungen
- 2023 Imunity&Immunization Yr1Dokument73 Seiten2023 Imunity&Immunization Yr1suluckyimonaunguumNoch keine Bewertungen
- IMMUNOPROPHYLAXIS.PPT.pptmDokument21 SeitenIMMUNOPROPHYLAXIS.PPT.pptmprerna12jainNoch keine Bewertungen
- Ant Vaccines WRF Hepatitis B FinalDokument6 SeitenAnt Vaccines WRF Hepatitis B FinalRidhi MurarkaNoch keine Bewertungen
- 3 Vaccines 2022 EnglishDokument28 Seiten3 Vaccines 2022 EnglishVlad SmithNoch keine Bewertungen
- Polio Vaccine: Presented BY: Yallala Shaik Afifa Ruhi, 1 YearDokument15 SeitenPolio Vaccine: Presented BY: Yallala Shaik Afifa Ruhi, 1 YearabuzarNoch keine Bewertungen
- A&P Assignment ActivitiesDokument4 SeitenA&P Assignment ActivitiesGURPARABJOT KAURNoch keine Bewertungen
- WED1 Fuller ScribdDokument30 SeitenWED1 Fuller ScribdNational Press FoundationNoch keine Bewertungen
- Microbiology: Parvovirus, Papillomavirus, and PolyomavirusDokument3 SeitenMicrobiology: Parvovirus, Papillomavirus, and PolyomavirusJustin TayabanNoch keine Bewertungen
- Vaccine types, schedules, and diseases overviewDokument22 SeitenVaccine types, schedules, and diseases overviewSoummyadip RoyNoch keine Bewertungen
- Lecture 10Dokument13 SeitenLecture 10yuki_akitsuNoch keine Bewertungen
- Laws Related To Expanded Program On ImmunizationDokument4 SeitenLaws Related To Expanded Program On ImmunizationSIR ONENoch keine Bewertungen
- Activity 2Dokument11 SeitenActivity 2Sherylou Kumo SurioNoch keine Bewertungen
- Immunization Saves LivesDokument30 SeitenImmunization Saves LivesUqba MishalNoch keine Bewertungen
- Therapeutic Proteins for Blood Disorders and DiseasesDokument28 SeitenTherapeutic Proteins for Blood Disorders and DiseasesXuan HuongNoch keine Bewertungen
- Manual HCVMicrolisaDokument4 SeitenManual HCVMicrolisaShahbaz AhmedNoch keine Bewertungen
- Manual Microlisa HIV Ag AbDokument4 SeitenManual Microlisa HIV Ag AbshanmicroNoch keine Bewertungen
- MON2 Friedland For FellowsDokument21 SeitenMON2 Friedland For FellowsNational Press FoundationNoch keine Bewertungen
- Case Study of Black Death: Our Lady of Fatima UniversityDokument7 SeitenCase Study of Black Death: Our Lady of Fatima UniversityJanna PimentelNoch keine Bewertungen
- Polio VaccineDokument15 SeitenPolio Vaccineamit vermaNoch keine Bewertungen
- Communicable-Diseases 48897647 2Dokument1 SeiteCommunicable-Diseases 48897647 2JULIUS CEZAR QUINAYNoch keine Bewertungen
- Vaccination: How Millions of Lives Have Been Saved - Perhaps YoursVon EverandVaccination: How Millions of Lives Have Been Saved - Perhaps YoursNoch keine Bewertungen
- NCP: Fatigue Related To Decreased Hemoglobin: Rezaba, Vianney Airess R. I I - BDokument8 SeitenNCP: Fatigue Related To Decreased Hemoglobin: Rezaba, Vianney Airess R. I I - BIrene Grace BalcuevaNoch keine Bewertungen
- Assessment:: Functional Health PatternsDokument14 SeitenAssessment:: Functional Health PatternsIrene Grace BalcuevaNoch keine Bewertungen
- Tetanus immune globulin treatmentDokument9 SeitenTetanus immune globulin treatmentIrene Grace BalcuevaNoch keine Bewertungen
- Drug Study AngelaDokument8 SeitenDrug Study AngelaIrene Grace BalcuevaNoch keine Bewertungen
- Discharge PlanDokument11 SeitenDischarge PlanIrene Grace BalcuevaNoch keine Bewertungen
- Celecoxib guide for osteoarthritis, rheumatoid arthritis & moreDokument17 SeitenCelecoxib guide for osteoarthritis, rheumatoid arthritis & moreIrene Grace BalcuevaNoch keine Bewertungen
- NCP: Fatigue Related To Decreased Hemoglobin: Rezaba, Vianney Airess R. I I - BDokument8 SeitenNCP: Fatigue Related To Decreased Hemoglobin: Rezaba, Vianney Airess R. I I - BIrene Grace BalcuevaNoch keine Bewertungen
- Vaccines: by Michelle Marie Therese TeoxonDokument11 SeitenVaccines: by Michelle Marie Therese TeoxonIrene Grace BalcuevaNoch keine Bewertungen
- Anatomy and Physiology OBDokument12 SeitenAnatomy and Physiology OBIrene Grace BalcuevaNoch keine Bewertungen
- BPH Symptoms, Risk Factors, and TreatmentDokument15 SeitenBPH Symptoms, Risk Factors, and TreatmentIrene Grace BalcuevaNoch keine Bewertungen
- Wound Dressing: Group 3 OpdDokument17 SeitenWound Dressing: Group 3 OpdIrene Grace BalcuevaNoch keine Bewertungen
- Penicillin: Libre, Princess Melanie SDokument19 SeitenPenicillin: Libre, Princess Melanie SIrene Grace BalcuevaNoch keine Bewertungen
- COVID 19 prevention tipsDokument4 SeitenCOVID 19 prevention tipsIrene Grace BalcuevaNoch keine Bewertungen
- HypertensionDokument14 SeitenHypertensionIrene Grace BalcuevaNoch keine Bewertungen
- In Consortium With N. Bacalso Ave. Corpanganiban ST., 6000 Cebu City, Cebu, Philippines Tel. Nos. (032) 316-5128/ (032) 4186105 Email AddressDokument2 SeitenIn Consortium With N. Bacalso Ave. Corpanganiban ST., 6000 Cebu City, Cebu, Philippines Tel. Nos. (032) 316-5128/ (032) 4186105 Email AddressIrene Grace BalcuevaNoch keine Bewertungen
- Vicodin Drug Study Que Fransis A.Dokument3 SeitenVicodin Drug Study Que Fransis A.Irene Grace BalcuevaNoch keine Bewertungen
- Herpes Simplex VirusDokument39 SeitenHerpes Simplex VirusWenalyn Grace Abella LlavanNoch keine Bewertungen
- Durg Study: Ascorbic AcidDokument11 SeitenDurg Study: Ascorbic AcidIrene Grace BalcuevaNoch keine Bewertungen
- Cloxacillin: By: Edamarie B. CabasaDokument11 SeitenCloxacillin: By: Edamarie B. CabasaIrene Grace BalcuevaNoch keine Bewertungen
- SEPAK TAKRAW: History of Kick Volleyball in Southeast AsiaDokument11 SeitenSEPAK TAKRAW: History of Kick Volleyball in Southeast AsiaIrene Grace BalcuevaNoch keine Bewertungen
- Nursing Care Plan!: By: James Albert C. MontañezDokument13 SeitenNursing Care Plan!: By: James Albert C. MontañezIrene Grace BalcuevaNoch keine Bewertungen
- Acetaminophen and Naproxen Study for ArthritisDokument9 SeitenAcetaminophen and Naproxen Study for ArthritisIrene Grace BalcuevaNoch keine Bewertungen
- ncp1 2fDokument2 Seitenncp1 2fIrene Grace BalcuevaNoch keine Bewertungen
- SaranilloncpDokument15 SeitenSaranilloncpIrene Grace BalcuevaNoch keine Bewertungen
- Drug Study MiggyDokument2 SeitenDrug Study MiggyIrene Grace BalcuevaNoch keine Bewertungen
- Power Point Discharge PlanDokument8 SeitenPower Point Discharge PlanIrene Grace BalcuevaNoch keine Bewertungen
- Cebu City Medical Center-College of NursingDokument7 SeitenCebu City Medical Center-College of NursingIrene Grace BalcuevaNoch keine Bewertungen
- Oxytocin Induction or Stimulation of Labor BeforeDokument4 SeitenOxytocin Induction or Stimulation of Labor BeforeIrene Grace BalcuevaNoch keine Bewertungen
- Breastfeeding and Formula FeedingDokument4 SeitenBreastfeeding and Formula FeedingIrene Grace BalcuevaNoch keine Bewertungen
- Oxytocin Induction or Stimulation of Labor BeforeDokument4 SeitenOxytocin Induction or Stimulation of Labor BeforeIrene Grace BalcuevaNoch keine Bewertungen
- Cvek Pulpotomy: Report of A Case With Five-YearDokument4 SeitenCvek Pulpotomy: Report of A Case With Five-YearKaren SandovalNoch keine Bewertungen
- Prevention of Surgical Site Infections: Surgical Patient Care SeriesDokument11 SeitenPrevention of Surgical Site Infections: Surgical Patient Care Seriestien duongNoch keine Bewertungen
- Chapter 4 A1 Poster Example 2Dokument3 SeitenChapter 4 A1 Poster Example 2Krisna PamungkasNoch keine Bewertungen
- 2016 - Byk FotoDokument7 Seiten2016 - Byk FotoSyifa IKNoch keine Bewertungen
- Bronchoscopy: Dr. Ravi Gadani MS, FmasDokument17 SeitenBronchoscopy: Dr. Ravi Gadani MS, FmasRaviNoch keine Bewertungen
- AR 40-8 Effective 16 June 2007Dokument10 SeitenAR 40-8 Effective 16 June 2007kbin3838Noch keine Bewertungen
- AIIMS (MBBS) Solved Question Paper 2010Dokument34 SeitenAIIMS (MBBS) Solved Question Paper 2010cbsestudymaterialsNoch keine Bewertungen
- Best Hospitals MethodologyDokument125 SeitenBest Hospitals MethodologyAngeline YeoNoch keine Bewertungen
- OxygenationDokument56 SeitenOxygenationHerald Clarence M. AmbayecNoch keine Bewertungen
- COVID-19 Impact on Nurses' Mental Health and WorkloadDokument6 SeitenCOVID-19 Impact on Nurses' Mental Health and WorkloadJAY LORRAINE PALACATNoch keine Bewertungen
- CTR Master Class CourseDokument15 SeitenCTR Master Class CourseCool N CoolNoch keine Bewertungen
- Jurnal Vitamin CDokument15 SeitenJurnal Vitamin CbethjohNoch keine Bewertungen
- OB Power Point Presentation 002Dokument57 SeitenOB Power Point Presentation 002RitamariaNoch keine Bewertungen
- Jurnal Kesehatan Gigi: Perilaku Dan Keterampilan Menyikat Gigi Terhadap Timbulnya Karies Gigi Pada Anak Di Kota JambiDokument7 SeitenJurnal Kesehatan Gigi: Perilaku Dan Keterampilan Menyikat Gigi Terhadap Timbulnya Karies Gigi Pada Anak Di Kota JambiUntuk TugasNoch keine Bewertungen
- Sonuplus 492 Operator ManualDokument72 SeitenSonuplus 492 Operator ManualShoaib KhanNoch keine Bewertungen
- PharmacovigilanceDokument45 SeitenPharmacovigilancepavani valluriNoch keine Bewertungen
- Pathology Specimens For Gross Exam Only and Exempt From Submission A College of American Pathologists Q Probes Study of Current Policies in 413 InstitutionsDokument7 SeitenPathology Specimens For Gross Exam Only and Exempt From Submission A College of American Pathologists Q Probes Study of Current Policies in 413 InstitutionsbonehssNoch keine Bewertungen
- CasopisiDokument452 SeitenCasopisiGormi DurmiNoch keine Bewertungen
- Manual Procedure Hemoglobin Drabkin Colorimetric MethodDokument1 SeiteManual Procedure Hemoglobin Drabkin Colorimetric MethodAhmad SaeedNoch keine Bewertungen
- EMedicine - Oral Hemangiomas - Article by Steven Brett SloanDokument19 SeitenEMedicine - Oral Hemangiomas - Article by Steven Brett SloanDr Monal YuwanatiNoch keine Bewertungen
- Polyhydra MN IosDokument41 SeitenPolyhydra MN IosAbedelrahmanShekhAhmadNoch keine Bewertungen
- Atrium MedicalDokument12 SeitenAtrium MedicalBo TaoNoch keine Bewertungen
- Human Sexuality and Sexual DisordersDokument42 SeitenHuman Sexuality and Sexual DisordersAbdul Gafoor FahmyNoch keine Bewertungen
- NURSING PRACTICE Test COMMUNITY HEALTH NURSING AND CARE OF THE MOTHER AND CHILDDokument8 SeitenNURSING PRACTICE Test COMMUNITY HEALTH NURSING AND CARE OF THE MOTHER AND CHILDJhannNoch keine Bewertungen
- Stepwise Ventilator Waveform Assessment To Diagnose Pulmonary PathophysiologyDokument8 SeitenStepwise Ventilator Waveform Assessment To Diagnose Pulmonary PathophysiologyPablo Simón Narbona MenaNoch keine Bewertungen
- Gavinos ResumeDokument4 SeitenGavinos ResumeRI NANoch keine Bewertungen
- Tugas Jurnal AppraisalDokument2 SeitenTugas Jurnal Appraisalshabrina nur imaninaNoch keine Bewertungen
- Erectile DysfunctionDokument31 SeitenErectile Dysfunctionrahuldtc100% (1)
- Pathophysiology - PyelonephritisDokument2 SeitenPathophysiology - PyelonephritisFrancis Kevin Sagudo92% (13)
- ImmerciDokument14 SeitenImmercitextilessathishNoch keine Bewertungen