Beruflich Dokumente
Kultur Dokumente
Management of Antithrombotic Therapy After Bleeding in Patients With Coronary Artery Disease And/or Atrial Fibrillation
Hochgeladen von
deni2razmoski0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
23 Ansichten20 SeitenOriginaltitel
predavanje jasmina
Copyright
© © All Rights Reserved
Verfügbare Formate
PPT, PDF, TXT oder online auf Scribd lesen
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
© All Rights Reserved
Verfügbare Formate
Als PPT, PDF, TXT herunterladen oder online auf Scribd lesen
0 Bewertungen0% fanden dieses Dokument nützlich (0 Abstimmungen)
23 Ansichten20 SeitenManagement of Antithrombotic Therapy After Bleeding in Patients With Coronary Artery Disease And/or Atrial Fibrillation
Hochgeladen von
deni2razmoskiCopyright:
© All Rights Reserved
Verfügbare Formate
Als PPT, PDF, TXT herunterladen oder online auf Scribd lesen
Sie sind auf Seite 1von 20
Management of antithrombotic
therapy after bleeding in
patients with coronary artery
disease and/or atrial fibrillation
Jasmina Jovanoski
Ohrid 24.3.2017
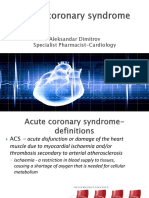
• Bleeding is a frequent complication of the
management of patients with coronary
artery disease (CAD), especially those
presenting with acute coronary syndromes
(ACS) or undergoing percutaneous
coronary intervention (PCI), and of
patients with atrial fibrillation (AF).
• major bleeding of 1–8% at 30 days in ACS
patients,
• 2–5% per year in patients with AF treated
with oral anticoagulants (OACs)
• Patients who are more likely to suffer from
bleeding complications - tend to be at
higher risk of thrombotic events
• Discontinuation of antithrombotic drugs -
increased rate of thrombotic events
(progressive recovery of platelet function
and coagulation activity)
• Bleeding may provoke prothrombotic
responses
• Balancing the risks of further bleeding vs.
potentially fatal thrombotic events is critical
• When the thrombotic risk is higher than
the risk of recurrent bleeding - continuing
antithrombotic therapy.
• When thrombotic risk is in equipoise with
bleeding risk - only brief or temporary
interruption of antithrombotic therapy.
• When bleeding risk outweighs thrombotic
risk - considering, on a case to case basis,
reducing the number and/or dose of
antithrombotic drug(s).
Antiplatelet therapy after
extracranial bleeding
Patients with a recent acute coronary
syndrome and/or percutaneous coronary
intervention (<12 months)
• The risk of stent thrombosis increases with
longer time off treatment, particularly more
than 5 days, and if treatment is stopped
within the 1st month after the procedure.
• newer-generation (everolimus- or
zotarolimus-eluting) drug-eluting stents
(DES) support shortening DAPT duration
in patients at high risk of bleeding
• If major bleeding occurs after 3–6 months of
DAPT, these studies lend support to
discontinuing the P2Y 12 inhibitor and continuing
aspirin alone, particularly in patients without a
prior history of ACS
• risk of stent thrombosis is lowest with the
newest generation DES, the thrombotic risk
appears higher with the new bioresorbable
vascular scaffolds (BVS)
• clopidogrel may be used as P2Y12 inhibitor
after bleeding has occurred during treatment
with prasugrel or ticagrelor, as soon as re-
initiation of antiplatelet therapy is possible.
Patients with stabilized coronary artery disease
(>12 months after percutaneous coronary
intervention or acute coronary syndromes)
• single antiplatelet therapy prescribed for
secondary prevention in stable CAD patients
should be reinitiated within a few days after
bleeding.
• cessation of, or non-adherence to, aspirin were
associated with a three-fold higher risk of major
adverse cardiac events compared to its
continued use , the risk was magnified in
patients with coronary stents.
Resuming antiplatelet therapy
after gastrointestinal bleeding
Consensus summary on antiplatelet
therapy after extracranial bleed
1. For patients at high or very high
thrombotic risk (ACS or coronary stenting
<30 days) who develop minor or major
bleeding - continuation of low-dose aspirin
without interruption. Restarting of the
second antiplatelet agent should be
considered as soon as possible after
stabilization.
• 2. For patients at moderate thrombotic risk (ACS
or PCI with a second generation DES 1–12
months ago) who develop minor or major
bleeding - resumption of low-dose aspirin as
soon as bleeding is controlled (within 3 days)
Restarting a second antiplatelet agent should be
considered if thrombotic risk outweighs recurrent
bleeding risk.
• 3. For patients who develop bleeding while on
DAPT within 3 months of new generation DES
implantation - we suggest resuming DAPT up to
3 months. If patients develop bleeding more than
3 months after new generation DES implantation
and remain at risk of recurrent bleeding -
resumption of only one antiplatelet agent (either
aspirin or clopidogrel).
• 4. Addition of a PPI is recommended in all
cases of upper GI bleeding.
• 5.PPI should also be used in patients on
DAPT at higher than average risk of GI
bleeds.
• 6. The pharmacodynamic interaction
between PPIs (especially omeprazole) and
clopidogrel has not so far been clearly
associated with worse clinical outcome,
but it is preferred to use PPIs with weaker
CYP2C19 inhibition (e.g. pantoprazole)
Oral anticoagulant therapy after
extracranial bleeding
• in the presence of a clear indication, VKA
therapy should be resumed within 1 week
following major GI bleeding
• when (re)starting a NOAC, renal function should
be carefully assessed and monitored to avoid
drug accumulation.
• if an antidote (idaruzicumab) has been used to
reverse the anticoagulant effect of a NOAC, it is
suggested to restart OAC as soon as possible,
preferably within 3–4 days if the individual
bleeding risk allows.
Resuming antithrombotic therapy in patients
with an indication for both antiplatelets and
oral anticoagulant
• stopping either aspirin or clopidogrel for major bleeding
during triple oral antithrombotic therapy.
• VKA vs. NOAC - either can be used in combination with
antiplatelet therapy.
• avoiding the use of prasugrel or ticagrelor as part of
triple therapy
• When NOACs are combined with antiplatelet drugs, the
lowest effective dose for stroke prevention should be
used, meaning dabigatran 110 mg bid (rivaroxaban 15
mg od, apixaban 2.5 mg bid, or edoxaban 30 mg od, only
when criteria for dose lowering are present)
• When PCI-treated patients with non-valvular AF
develop bleeding during triple oral antithrombotic
therapy - stopping either aspirin or clopidogrel
• In patients who develop major bleeding on
double therapy (OAC plus single antiplatelet),
discontinuation of the antiplatelet agent
• We suggest DAPT alone (without OAC) for 1
year after ACS/PCI when a patient with non-
valvular AF and a low risk of stroke
(CHA 2 DS 2 -VASc 1 for men, 2 for women)
develops bleeding during triple or double
antithrombotic therapy.
Antithrombotic therapy after
intracranial haemorrhage
• ICH is one of the most feared
complications of OAC
• Whether and when to restart OAC after an
ICH should be decided on an individual
basis, depending on the patient’s
thrombotic risk and the risk of recurrent
bleeding
• In the absence of mechanical valves,
NOACs - preferable to warfarin after ICH
Das könnte Ihnen auch gefallen
- Acetylsalicyclic Acid 08july2022Dokument6 SeitenAcetylsalicyclic Acid 08july2022Cuong LeNoch keine Bewertungen
- Antithrombotic Treatment of Acute Ischemic Stroke and Transient Ischemic Attack - UpToDateDokument24 SeitenAntithrombotic Treatment of Acute Ischemic Stroke and Transient Ischemic Attack - UpToDateFerasNoch keine Bewertungen
- Antiplatelet Drugs, Anticoagulants and Elective SurgeryDokument5 SeitenAntiplatelet Drugs, Anticoagulants and Elective SurgeryJanice WongNoch keine Bewertungen
- Br. J. Anaesth. 2007 Chassot 316 28Dokument13 SeitenBr. J. Anaesth. 2007 Chassot 316 28Rhahima SyafrilNoch keine Bewertungen
- Treatment of NSTE-ACSDokument4 SeitenTreatment of NSTE-ACSAnuradha NanayakkaraNoch keine Bewertungen
- Acute non-ST-elevation Acute Coronary Syndromes: Early Antiplatelet Therapy (UPTODATE)Dokument27 SeitenAcute non-ST-elevation Acute Coronary Syndromes: Early Antiplatelet Therapy (UPTODATE)kabulkabulovich5Noch keine Bewertungen
- Combining Antiplatelet and Anticoagulant Therapy in Cardiovascular Disease - 2020 - AHADokument7 SeitenCombining Antiplatelet and Anticoagulant Therapy in Cardiovascular Disease - 2020 - AHADrHellenNoch keine Bewertungen
- AFP 2014 12 Clinical RahmanDokument6 SeitenAFP 2014 12 Clinical RahmanmyscribeNoch keine Bewertungen
- Early Antithrombotic Treatment of Acute Ischemic Stroke and Transient Ischemic Attack - UpToDateDokument27 SeitenEarly Antithrombotic Treatment of Acute Ischemic Stroke and Transient Ischemic Attack - UpToDateEduardo QuinteroNoch keine Bewertungen
- QRG AntithromboticDokument6 SeitenQRG AntithromboticthapanNoch keine Bewertungen
- Stroke Clinical PathwayDokument35 SeitenStroke Clinical PathwayLaurencia Leny100% (2)
- Antithrombotic Dilemmas in Stroke MedicineDokument43 SeitenAntithrombotic Dilemmas in Stroke MedicineGopi Krishnan RNoch keine Bewertungen
- WarfarinDokument23 SeitenWarfarinminaNoch keine Bewertungen
- Perioperative Management of Patients Receiving Anticoagulants - UpToDateDokument65 SeitenPerioperative Management of Patients Receiving Anticoagulants - UpToDatejames lxNoch keine Bewertungen
- Perioperative Management of Patients Receiving Anticoagulants. UpToDate. 2017.Dokument40 SeitenPerioperative Management of Patients Receiving Anticoagulants. UpToDate. 2017.radiomedicNoch keine Bewertungen
- Anticoag Antipla in Endoscopic ProceduresDokument14 SeitenAnticoag Antipla in Endoscopic ProceduresAnonymous ZUaUz1wwNoch keine Bewertungen
- Antiplatelet Therapy For Acute StrokeDokument10 SeitenAntiplatelet Therapy For Acute StrokeInstalasi OK RSI JombangNoch keine Bewertungen
- Official Reprint From Uptodate ©2018 Uptodate, Inc. And/Or Its Affiliates. All Rights ReservedDokument43 SeitenOfficial Reprint From Uptodate ©2018 Uptodate, Inc. And/Or Its Affiliates. All Rights ReservedVictor M' PimentelNoch keine Bewertungen
- Acs 11 190524110746Dokument46 SeitenAcs 11 190524110746Obakeng MandaNoch keine Bewertungen
- GI Bleeding in Patients Receiving Antiplatelets and Anticoagulant TherapyDokument11 SeitenGI Bleeding in Patients Receiving Antiplatelets and Anticoagulant TherapyTony LeeNoch keine Bewertungen
- Perioperative Management of Patients Receiving Anticoagulants - UpToDateDokument69 SeitenPerioperative Management of Patients Receiving Anticoagulants - UpToDatePhúc NguyễnNoch keine Bewertungen
- Antiplatelet Therapy For The Secondary Prevention of Ischemic Stroke - UpToDateDokument13 SeitenAntiplatelet Therapy For The Secondary Prevention of Ischemic Stroke - UpToDateSuci WijayaNoch keine Bewertungen
- STD 2Dokument45 SeitenSTD 2Alejandra CastroNoch keine Bewertungen
- Thrombolytic Therapy in Emergency MedicineDokument8 SeitenThrombolytic Therapy in Emergency MedicinemedeviNoch keine Bewertungen
- Acute Coronary Syndrome Oral Anticoagulation in Medically Treated Patients - UpToDateDokument14 SeitenAcute Coronary Syndrome Oral Anticoagulation in Medically Treated Patients - UpToDateBrian VianaNoch keine Bewertungen
- Anticoag GuidelinesDokument22 SeitenAnticoag Guidelinesyusuf100% (1)
- DVT PE ManagementDokument6 SeitenDVT PE Managementகோகுல் தங்கவேல்Noch keine Bewertungen
- Myocardial InfarctionDokument5 SeitenMyocardial InfarctionNikki MacasaetNoch keine Bewertungen
- Manejo Perioperatorio de Pacientes Usuarios de Antiagregantes PlaquetariosDokument9 SeitenManejo Perioperatorio de Pacientes Usuarios de Antiagregantes PlaquetariosErick ToHuNoch keine Bewertungen
- 0718 4026 Rchcir 70 03 0291Dokument9 Seiten0718 4026 Rchcir 70 03 0291Erick ToHuNoch keine Bewertungen
- Farmakologi Kel 3Dokument33 SeitenFarmakologi Kel 3SalsabilaRiyadiniNoch keine Bewertungen
- Dual Antiplatelet Therapy After PCI in Patients atDokument32 SeitenDual Antiplatelet Therapy After PCI in Patients atyamonaelgamNoch keine Bewertungen
- LMWH QML 06Dokument7 SeitenLMWH QML 06pd7qmlNoch keine Bewertungen
- MILgfgsd V1Dokument4 SeitenMILgfgsd V1garywall.ukNoch keine Bewertungen
- Acute Coronary Syndrome: Management of NSTEMIDokument2 SeitenAcute Coronary Syndrome: Management of NSTEMIlonelyhime13Noch keine Bewertungen
- Anticoagulations in Surgery: Sharifah Naiemah BT Syed MansorDokument20 SeitenAnticoagulations in Surgery: Sharifah Naiemah BT Syed MansorSharifah NaiemahNoch keine Bewertungen
- Blood Transfusion.Dokument23 SeitenBlood Transfusion.Madalina TalpauNoch keine Bewertungen
- Myocardial InfarctionDokument20 SeitenMyocardial InfarctionRio Ramon HilarioNoch keine Bewertungen
- Pre and Post Operative CareDokument9 SeitenPre and Post Operative CareSara Abdi OsmanNoch keine Bewertungen
- Nicoumalone Drug Information, ProfessionalDokument62 SeitenNicoumalone Drug Information, ProfessionalSUBHADIPNoch keine Bewertungen
- Perioperative Management of Antithrombotic TherapyDokument40 SeitenPerioperative Management of Antithrombotic TherapyAnonymous KO7A7RcYp2Noch keine Bewertungen
- Articulo - FarmacologíaDokument13 SeitenArticulo - FarmacologíaJhon RVNoch keine Bewertungen
- Aneurysmal Subarachnoid Hemorrhage - Treatment and Prognosis - UpToDateDokument49 SeitenAneurysmal Subarachnoid Hemorrhage - Treatment and Prognosis - UpToDateSerque777Noch keine Bewertungen
- Warfarin Perioperative ManagementDokument8 SeitenWarfarin Perioperative Managementzoran cukovicNoch keine Bewertungen
- Fondaparinux in Acute Coronary Syndromes CA5068 Admin OnlyDokument9 SeitenFondaparinux in Acute Coronary Syndromes CA5068 Admin OnlynaeamzNoch keine Bewertungen
- Hemostatic Strategies For Minimizing Mortality in Major Blood LossDokument26 SeitenHemostatic Strategies For Minimizing Mortality in Major Blood LossJonathan wiradinataNoch keine Bewertungen
- Peri-Operative Management of Patients Receiving AnticoagulantsDokument22 SeitenPeri-Operative Management of Patients Receiving AnticoagulantsCheuk Hei LauNoch keine Bewertungen
- Escalation or De-Escalation AntiplateletDokument33 SeitenEscalation or De-Escalation AntiplateletMangkoksotobanjar Al BanjariNoch keine Bewertungen
- Management of Patients With Compromising Medical ConditionsDokument20 SeitenManagement of Patients With Compromising Medical ConditionsAbdulSamiNoch keine Bewertungen
- ThrombolyticsDokument14 SeitenThrombolyticsJavier Gonzalez, M.D. DABEMNoch keine Bewertungen
- 309 Novel Anti-Platelet Agents and AnticoagulantsDokument13 Seiten309 Novel Anti-Platelet Agents and AnticoagulantsPhani NadellaNoch keine Bewertungen
- Anti Platelet PreopDokument6 SeitenAnti Platelet Preopbrigde_xNoch keine Bewertungen
- Acute Coronary Syndrome 2022Dokument38 SeitenAcute Coronary Syndrome 2022Anonymous100% (1)
- Principles of An-Tiplatelet Therapy: DR Htet Htet Htethtet@Imu - Edu.MyDokument36 SeitenPrinciples of An-Tiplatelet Therapy: DR Htet Htet Htethtet@Imu - Edu.MyAbby LiewNoch keine Bewertungen
- Contraindicaciones Fibrinolisis AcvDokument4 SeitenContraindicaciones Fibrinolisis AcvLudwinNoch keine Bewertungen
- Significant Early In-Hospital Benefit Was Seen. Clopidogrel Is Prefferd ToDokument8 SeitenSignificant Early In-Hospital Benefit Was Seen. Clopidogrel Is Prefferd TogilnifNoch keine Bewertungen
- Unanswered Questions During The Live EventDokument9 SeitenUnanswered Questions During The Live Eventyash_acharya007Noch keine Bewertungen
- Stopp Start ToolkitDokument22 SeitenStopp Start ToolkitRifky IlhamiNoch keine Bewertungen
- Prevention of Atherotrobmotic EventsDokument14 SeitenPrevention of Atherotrobmotic Eventsddandan_2Noch keine Bewertungen
- Bi - Mek Doo: Subject: Market Research For Supply of Equipment Under The Project "Heart SafeDokument11 SeitenBi - Mek Doo: Subject: Market Research For Supply of Equipment Under The Project "Heart Safedeni2razmoskiNoch keine Bewertungen
- Hipertensión Pulmonar (2022)Dokument114 SeitenHipertensión Pulmonar (2022)Mauricio CabreraNoch keine Bewertungen
- b13c - Letterunsuccessful - en LDADokument2 Seitenb13c - Letterunsuccessful - en LDAdeni2razmoskiNoch keine Bewertungen
- b13b - Lettersecond - Best - en (1) MGCDokument2 Seitenb13b - Lettersecond - Best - en (1) MGCdeni2razmoskiNoch keine Bewertungen
- (Journal of Neurosurgery - Spine) Spontaneous Atraumatic Vertebral Artery Occlusion Due To Physiological Cervical ExtensionDokument5 Seiten(Journal of Neurosurgery - Spine) Spontaneous Atraumatic Vertebral Artery Occlusion Due To Physiological Cervical Extensiondeni2razmoskiNoch keine Bewertungen
- Robotic PCI Through The Distal Radial Artery - How and Why? RI Via RoboticDokument29 SeitenRobotic PCI Through The Distal Radial Artery - How and Why? RI Via Roboticdeni2razmoskiNoch keine Bewertungen
- Robotic PCI Through The Distal Radial Artery - How and Why? RI Via RoboticDokument29 SeitenRobotic PCI Through The Distal Radial Artery - How and Why? RI Via Roboticdeni2razmoskiNoch keine Bewertungen
- ShortDokument1 SeiteShortdeni2razmoskiNoch keine Bewertungen
- Vsad, Ma LDGMV LZSDFML BM Embr Awemrb' A RMB Amerb Amdr Blmda Kbal DBM Asldbm Sladbm Asdfm em (Owe (Owbm (Owm4b (Work (Pa Wrmob (LasdfmblsdfbDokument1 SeiteVsad, Ma LDGMV LZSDFML BM Embr Awemrb' A RMB Amerb Amdr Blmda Kbal DBM Asldbm Sladbm Asdfm em (Owe (Owbm (Owm4b (Work (Pa Wrmob (Lasdfmblsdfbdeni2razmoskiNoch keine Bewertungen
- (Journal of Neurosurgery - Spine) Spontaneous Atraumatic Vertebral Artery Occlusion Due To Physiological Cervical ExtensionDokument5 Seiten(Journal of Neurosurgery - Spine) Spontaneous Atraumatic Vertebral Artery Occlusion Due To Physiological Cervical Extensiondeni2razmoskiNoch keine Bewertungen
- List of AbbreviationsDokument6 SeitenList of Abbreviationsdeni2razmoskiNoch keine Bewertungen
- MR Second Za PredevanjeDokument32 SeitenMR Second Za Predevanjedeni2razmoskiNoch keine Bewertungen
- PVI Product Presentation - Oct 2016Dokument52 SeitenPVI Product Presentation - Oct 2016deni2razmoskiNoch keine Bewertungen
- Oajsdfvnasdfopvnaefrasdfvasdfvasdfvaslvnokivawr) Opijnvaw RpovmDokument1 SeiteOajsdfvnasdfopvnaefrasdfvasdfvasdfvaslvnokivawr) Opijnvaw Rpovmdeni2razmoskiNoch keine Bewertungen
- RegalDokument1 SeiteRegaldeni2razmoskiNoch keine Bewertungen
- ByeDokument1 SeiteByedeni2razmoskiNoch keine Bewertungen
- FlugDokument1 SeiteFlugdeni2razmoskiNoch keine Bewertungen
- K UuuuuuDokument1 SeiteK Uuuuuudeni2razmoskiNoch keine Bewertungen
- ByeDokument1 SeiteByedeni2razmoskiNoch keine Bewertungen
- VocabDokument2 SeitenVocabdeni2razmoskiNoch keine Bewertungen
- BokabDokument1 SeiteBokabdeni2razmoskiNoch keine Bewertungen
- LIIIIDokument1 SeiteLIIIIdeni2razmoskiNoch keine Bewertungen
- KokabDokument2 SeitenKokabdeni2razmoskiNoch keine Bewertungen
- KST UrnekDokument1 SeiteKST Urnekdeni2razmoskiNoch keine Bewertungen
- Clan WarDokument1 SeiteClan Wardeni2razmoskiNoch keine Bewertungen
- KLGDokument1 SeiteKLGdeni2razmoskiNoch keine Bewertungen
- Aalsdfka Sdfhmasd Fhma Dmadf, Hma DF, Hmadf, HMDF, MDF Kmdaf KBMDF KBMDF KB Pin PKSRNPKNSFBPKN XCNB PXBNPXKCBN XNCPBX:KLGMM CZBMZ:CBM: CBM Aa S'DF, G As'F.,Gasf .,ga Sdfg.,A Sdfg.,A FG.Dokument1 SeiteAalsdfka Sdfhmasd Fhma Dmadf, Hma DF, Hmadf, HMDF, MDF Kmdaf KBMDF KBMDF KB Pin PKSRNPKNSFBPKN XCNB PXBNPXKCBN XNCPBX:KLGMM CZBMZ:CBM: CBM Aa S'DF, G As'F.,Gasf .,ga Sdfg.,A Sdfg.,A FG.deni2razmoskiNoch keine Bewertungen
- Gingivita Din Ciclul MenstrualDokument10 SeitenGingivita Din Ciclul MenstrualRamonaNoch keine Bewertungen
- IonosonjubileeDokument64 SeitenIonosonjubileePhước Thành Nguyễn CửuNoch keine Bewertungen
- Oncopphyllus) Terhadap Kadar Protein, Serat Pangan, Lemak, Dan TingkatDokument8 SeitenOncopphyllus) Terhadap Kadar Protein, Serat Pangan, Lemak, Dan TingkatIreng PramanaNoch keine Bewertungen
- ADHD Diagnosis and Screening in AdultsDokument4 SeitenADHD Diagnosis and Screening in AdultsGrzegorz Pelutis0% (1)
- CalcitoninDokument38 SeitenCalcitoninArooj WaseemNoch keine Bewertungen
- Assessing General Health Status and Vital SignsDokument1 SeiteAssessing General Health Status and Vital SignsCyril Joy N. FernandoNoch keine Bewertungen
- Endoscopy: Accepted ManuscriptDokument37 SeitenEndoscopy: Accepted ManuscriptShivaraj S ANoch keine Bewertungen
- Vol 26 1 2018 15Dokument10 SeitenVol 26 1 2018 15sind badNoch keine Bewertungen
- OHSMS AwarenessDokument12 SeitenOHSMS AwarenessAbdul MueedNoch keine Bewertungen
- Prostho Summary Manal, Al Dahabi EtcDokument10 SeitenProstho Summary Manal, Al Dahabi EtcDR-RASHEED KHANNoch keine Bewertungen
- Algorithm 0Dokument2 SeitenAlgorithm 0fau fauNoch keine Bewertungen
- Correlation of Procalcitonin Level With NLR in Patient With SIRSDokument1 SeiteCorrelation of Procalcitonin Level With NLR in Patient With SIRSNisa UcilNoch keine Bewertungen
- Dissertation Right To Health LLMDokument32 SeitenDissertation Right To Health LLMSwapnil TembhurneNoch keine Bewertungen
- ASSESSMENT OF THE EAR, NOSE, MOUTH AND THROAT (Chapter 12)Dokument4 SeitenASSESSMENT OF THE EAR, NOSE, MOUTH AND THROAT (Chapter 12)Christianne CapuaNoch keine Bewertungen
- Infectious Diseaes and ModelingDokument46 SeitenInfectious Diseaes and ModelingIntan GeaNoch keine Bewertungen
- Jomo Kenyatta University OF Agriculture & Technology School of Open, Distance and ElearningDokument43 SeitenJomo Kenyatta University OF Agriculture & Technology School of Open, Distance and ElearningKeddie OneJhereNoch keine Bewertungen
- ESMO Guidelines Immunotherapy Toxicities7 PDFDokument24 SeitenESMO Guidelines Immunotherapy Toxicities7 PDFOscar MerinoNoch keine Bewertungen
- Kala Full Chapter FinalDokument72 SeitenKala Full Chapter FinalZANARIAH BINTI SULAIMAN MoeNoch keine Bewertungen
- Naskah Soal BHS InggrisDokument13 SeitenNaskah Soal BHS InggrisYanuar IhsanNoch keine Bewertungen
- Thesis - English (Teen Obesity - THEYAN)Dokument22 SeitenThesis - English (Teen Obesity - THEYAN)nhikkie89% (19)
- Biology of Tooth Movement 2021-2022 2Dokument73 SeitenBiology of Tooth Movement 2021-2022 2luna zeidNoch keine Bewertungen
- Probiotics D Lactic Acidosis Oxidative Stress and Strain SpecificityDokument13 SeitenProbiotics D Lactic Acidosis Oxidative Stress and Strain SpecificityPaul Youn-seok HamNoch keine Bewertungen
- General Information About Nurse Licensure ExaminationsDokument2 SeitenGeneral Information About Nurse Licensure ExaminationsAhmad Deedatt KalbitNoch keine Bewertungen
- Clinical Outcomes of Vertical Bone Augmentation To Enable Dental Implant Placement: A Systematic ReviewDokument13 SeitenClinical Outcomes of Vertical Bone Augmentation To Enable Dental Implant Placement: A Systematic Reviewvanessa_werbickyNoch keine Bewertungen
- IS Case Study DisabilitiesDokument5 SeitenIS Case Study DisabilitiesRuchiNoch keine Bewertungen
- Implementation of Innovative Medical TechnologiesDokument18 SeitenImplementation of Innovative Medical TechnologiesGregorius HocevarNoch keine Bewertungen
- National Tuberculosis Elimination ProgrammeDokument11 SeitenNational Tuberculosis Elimination ProgrammeRekha ChaudharyNoch keine Bewertungen
- Cardiac Asthma - Causes, Symptoms, Diagnosis and Treatment - Tips Curing DiseaseDokument5 SeitenCardiac Asthma - Causes, Symptoms, Diagnosis and Treatment - Tips Curing DiseaseBharat JamodNoch keine Bewertungen
- Candida Krusei TreatmentDokument2 SeitenCandida Krusei TreatmentMohannad AhmedNoch keine Bewertungen
- Dermal Filler AftercareDokument2 SeitenDermal Filler Aftercarelovrata4550Noch keine Bewertungen