Beruflich Dokumente
Kultur Dokumente
Steel Research - 2016 - SEO - Thermodynamic Assessment of The Al Deoxidation Reaction in Liquid Iron
Hochgeladen von
jukvertOriginaltitel
Copyright
Verfügbare Formate
Dieses Dokument teilen
Dokument teilen oder einbetten
Stufen Sie dieses Dokument als nützlich ein?
Sind diese Inhalte unangemessen?
Dieses Dokument meldenCopyright:
Verfügbare Formate
Steel Research - 2016 - SEO - Thermodynamic Assessment of The Al Deoxidation Reaction in Liquid Iron
Hochgeladen von
jukvertCopyright:
Verfügbare Formate
Process metallurgy
Thermodynamic assessment of the AI deoxidation reaction in liquid iron
Jeong-Do Seo, Seon-Hyo Kim and Kwang-Ro Lee
The deoxidation reaction of aluminum in liquid iron has been investigated thermodynamically using Ab03 crucible at 1873 K under Ar
atmosphere as a fundamental study for the accurate control of inclusions in the ladle refining process. In addition to the equilibrium con-
stant log KAI f0t. the aluminum deoxidation reaction, the first-order and second-order interaction parameters between aluminum and oxy-
gen were experimentally determined in the concentration range of aluminum up to 1 %. The temperature dependence of the equilibrium
constant and the first-order interaction parameter eti was also obtained: log KAI =12.32 - 474001T, e~l =15.57 - 365001T.
The equilibrium relation between aluminum and oxygen contents in the aluminum deoxidized iron by applying interaction parameters and
the equilibrium constant determined in this work satisfies fairly well the equilibrium data over the whole concentration range of aluminum
considered.
Thermodynamlsche Bewertung der AI·Desoxldatlonsreaktlon In Eisenschmeizen. Die Desoxidationsreaktion mit Aluminium in
Eisenschmelzen wurde thermodynamisch untersucht. Grundlegende Studien in einem Ab03- Tiegel bei 1873 K in Ar-Atmosphara sollten
Erkenntnisse iiber die EinschluBkontrolle wahrend der Pfannenbehandlung Iiefem. Erganzend zur Gleichgewichtskonstanten fOr die
Aluminiumdesoxidation, log KAl' wurden auch die Wechse!wirkungsparameter erster und zweiter Ordnung von Aluminiumund Sauerstoff
fUr Aluminiumkonzentrationen bis zu 1% experimentell ermittelt. Die Temperaturabhangigkeit der Gleichgewichtskonstanten und des
Wechselwirkungsparameters erster Ordnung, eti ' lieB sich zu log KAI
. = 12.32 - 474001T, .eti = 15.57 - 36500fTbestimmen.
Die Gleichgewict:ltsbeziehung zwischen Aluminium- und Sauerstoffgehalten in AI-desoxidierten Eisenschmelzen, die mit diesen Wech-
selwirkungsparametern bzw. Gleichgewichtskonstanten bestimmt wurden, geniigt den Gleichgewichtswerten iiber dem gesamten, be-
trachteten Aluminiumkonzentrationsbereich gut.
Aluminum is commonly used as a strong deoxidizer in Experimental
steelmaking process. Its utilization as a deoxidizer, how-
ever, is somewhat limited due to the formation of AI- The equilibrium study was carried out at 1873 K in a
related inclusions in liquid iron. When the aluminum con- LaCr03 resistance furnace equipped with a fused alumina
taining steels are tapped into the cast mould through the tube with a diameter of 50 mm. The tube was sealed at
refractory nozzle, the build-up of non-metallic oxides such both ends with water cooled metal sections. The tempera-
as Ah03 and MgO·Ah03 in the nozzle leads to a break- ture was measured by a thermocouple of PtRh6/PtRh30
down of the whole casting system. Therefore the control of controlled with an accuracy of 2 K. Argon gas was flown
non-metallic inclusions in the refining stage is of great after purifying through H2S04, P20S, Mg(CI04)2 and a
importance to produce high-quality clean steels economi- bed of Ti-granules at 650°C.
cally. The reliable thermodynamic data .of aluminum de- High-purity electrolytic iron ([%0] = 0.0035, [%AI] =
oxidation equilibria should be established to control effi- 0.0003, [%C] = 0.001, [%Si] < 0.001) of 40 g was initially
ciently the formation of non-metallic inclusions associated melted in the Al203 crucible (026x55H) under a deoxi-
with aluminum. Despite the extensively performed previ- dized Ar atmosphere. An appropriate amount of Fe-20%AI
ous works [1...14] on the aluminum deoxidation equilibria, was added into the iron melt. The melt was stirred for
some uncertainty still exists on the equilibrium data. Most about 2 h by an Ah03 rod at 30 min intervals in order to
works have been carried out over the narrow range of very facilitate the deoxidation reaction. To eliminate oxide
low aluminum concentrations' at 1873 K. Furthermore, the inclusions by flotation the melt was maintained under
thermodynamic data previously reported are very incon- unstirring condition for at least 1 h. After reaching equili-
sistent according to the aluminum concentration range bration, the crucible was taken out of the furnace and
considered. quenched rapidly in a He gas stream, followed by water
In this work, the limitation of validity for the first-order quenching. The quenched sample was sliced into a plate
interaction parameter between aluminum and oxygen in shape and then sectioned to small pieces for oxygen and
iron was investigated at 1873 K under the aluminum con- aluminum analysis.
centration range up to 1 %. Not only the first-order and the The total oxygen content of a sample was determined by
second-order interaction parameters between aluminum a method of inert-gas fusion-infrared absorptiometry. In
and oxygen but the equilibrium constant for aluminum the case of oxygen analysis of the samples containing a
deoxidation reaction was estimated at 1873 K by a multi- significant amount of aluminum, it is recognized that the
ple regression method using the published data and the vaporized aluminum from the sample reacts with CO gas
present experimental results. The validity of these values to form the AhO vapour which condenses at lower tem-
was also discussed. peratures. This phenomenon results in the analytical error
of real oxygen contents. In order to eliminate these ana-
Jeong-Do Seo, graduate student; Professor Seon-Hyo Kim; Dr. Kwang- lytical shortcomings, the Sn bath-Ni capsule method cou-
Ro Lee, research assistant, Materials Science and Engineering, Pohang pled with the double graphite crucibles was employed for
University of Science and Technology, Pohang, Korea. the samples containing more than 0.3 % aluminum. The
steel research 69 (1998) No.2 49
1869344xa, 1998, 2, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/srin.199801342 by University Of Wollongong University Of Wollongong Library, Wiley Online Library on [28/02/2024]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
Process metallurgy
analytical method relating to the soluble- or insoluble- Table 1. Chemical composition of metal employed in this work
aluminum concentrations in metal was described in detail Aluminum mass contents in % oxygen mass contents in %
elsewhere [9]. The soluble- and insoluble-aluminum solu- 0,0002 0.0110
tions were prepared individually and then mixed together 0.0875 0.0014
in a 50 ml mass flask in order to determine the total alumi- 0.1300 0.0008
num content in a mixed solution by the inductively cou- 0.2056 0.0007
pled plasma emission spectrometry. 0.2674 0.0008
0.3146 0.0008
Results and discussion 0.3728 0.0018
0.7207 0.0017
The deoxidation reaction for aluminum is expressed by 0.9873 0.0011
Ah03 (s) = 2 [AI] + 3 [0] (1)
Table 2. The previously reported values of interaction parameters
The equilibrium constant, KAI of reaction (l), the appar- between aluminum and oxygen and equilibrium constant of alumi-
num deoxidation reaction at 1873 K
ent equilibrium constant, K~l (= [%AI]2 • [%0]3) and the
AI AI logKAl references
activity coefficient of component i, Ii in liquid iron with eo ro
respect to I % Henrian standard state are correlated as a -4.60 - -13.90 [I]
form of following equation: -1.00 - - [3]
-3.90 - -13.25 [6]
log KAI = 2 10gfAI + 3 logfo + log K~I - log a AIz03 -5.25 - -13.22 [8]
-4.05 - -13.30 [II]
.
= log K~I + 2 (e1\[%i] + e~d%O] + r1\[%Al]2 -5.54 -14.01 [12]
-3.90 1.70 -13.30 [16]
+ r~1 [%0]2 + r~IAI [%O][%AID + 3 (eg [%0]
+ e~1 [%Al] + r~1 [%Al]2 + rg [%0]2 = -e~l {3[%AI] + 2(MAl/Mo)[%0]} - r~l {3[%AI]
+ rg· AI [%O][%Al] ) + 2 ~ (e~1 [%i] + r~ [%i]2 + 2(MAI / Mo)[%O] }2 + r~1 {6[%AI]2 + 2[%0]2
t.]
+ 2(MAl/Mo)[%0][%AI]} + log KAI. (4)
+ r~tl [%i][%Al] + r~{ [%i][%jD
The equilibrium chemical compositions of metal are
+ 3 ~ (eb [%i] + rb [%i]2 + r~o [%i][%O]
t.] given in table 1. The experimental equilibrium data be-
(2) tween aluminum and oxygen at 1873 K are plotted in fig-
+ r& [%i][%jD - log a AIz03
ure 1. In order to have I % Henrian standard state applied,
where i and j represent the components other than oxygen the data selection was only limited to the aluminum con-
and aluminum, respectively. [%i] represents mass content centrations up to 1 %.
of component i. If the second-order interaction parameter terms between
The aluminum deoxidation equilibrium can be finally aluminum and oxygen become abbreviated in equation (4),
written as equation (3) according to the relationship de- the following simplified equation can be expressed:
rived by Lupis et al. [15]:
log KAj - log K~I - 2 ~
, (e~1 [%i] + ell [%Al] 3.5 ....-....---.---.----.-..---r--.--..,.-r--~-..-.....,
3.0
+ r~1 [%i]2) - 3 ~ (e6 [o/oj] + eg [%0] 2.5
]
2.0
+ r6 [o/oj]2) + log a AIZ03 = e~1 {3[%AI]
5' 1.5
+ 2( MAI/Mo )[%O]} + r~1 {3[%AI]2 S.
.!::lI 1.0
+ 2(MAI/Mo)2[%0]2 + IO(MAI/Mo)[%O][%AI]} (3) J 0.5
where M is the atomic mass fraction of the respective
0.0 •
0
PnlIIent
Fruehan[6]
component. -0.5 [] Rhodo[7]
-1.0 A Dimi1rov [12]
Equation (3) can be rearranged as:
0 Suito [9] 1873K
-1.5
log K~I + 2 L, (e~1 [%i] + e11 [%AI] -2.0 L -..........L...--'---L---I_.l.-..a-...I-..........--L---'...........
-5 -4 -3 -2 -I o 1
+ r~1 [%i]2) + 3 L (e6 [o/oj] + eg [%0] log[%Al]
]
Figure 1. Experimental data of aluminum and oxygen equilibrated
+ r6 [o/oj]2) - log a AIZ03 in liquid iron at 1873 K
50 steel research 69 (1998) No..2
1869344xa, 1998, 2, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/srin.199801342 by University Of Wollongong University Of Wollongong Library, Wiley Online Library on [28/02/2024]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
Process metallurgy
log K~l + 2 L (e~l [%i] + e~1 [%Al]
-5 .----.---,.-~--.---.--_r_-~___,
i
+ r~l [%i]2) + 3 L (e6 [%i] + eg [%0]
o j
+ r6 [o/'!i]2) - log aAl203
+ = - e~l {3[%AI] + 2 (MAl/Mo)[%O])}
;!:, g + log KAI. (5)
l es - Calculated
-J o~ • Present
'-' + -15 o FruebaD The relation between the left-hand-side (LHS) of equa-
':;: o tion (5) and 3[%AI] + 3.37[%0] term in the right-hand-
w-es Rhode
N
+
·....0
..
A
<>
Dimitrov
Suito
side is plotted in figure 2 using the values of e~:, eg
:J H~ -20 I...-~_--L --'-_--,-_-'-_"""'----'
along with the contents of aluminum, oxygen, and the
other components from the present experimental results
J ~ 0 234
and previously reported data. A non-linear relationship
3[o/oAI] + 3.37[%0]
exists with an intercept value of log KAI. This result indi-
Figure 2. The relation between the LHS of equation (5) and
cates that the equilibrium between aluminum and oxygen
3[%AI]+3.37[%O] term
in liquid iron can not be explained only by first-order in-
teraction parameters given in the table 2 without consid-
ering the higher order interaction parameters. The interac-
5r----.---,.---.---.---.--...--~___, tion parameters e~l, r~1 and log KAI could be estimated
using equation (3) by a multiple regression analysis as
o follows:
+ log KAI = -12.96 ± 0.14,
':I-. -5
~
.;::
~ -10 e~1 = -4.09 ± 0.41, r~l = 2.67 ± 0.47,
- Calculated
~ • Present
... -15 o FruebaD e~l = -6.90 ± 0.69, r~l = 7.60 ± 1.34,
+ C Rhode
~...
~
-20
A
<>
Dimitrov
Suito r~l,O (r~I'o) = 9.05 ± 1.59 (9.05 ± 1.59). (6)
2 3 4 The curved line indicated in figure 2 was obtained from
3[o/oAl] + 3.37[%0] equation (4) using the values of the equilibrium constant
and interaction parameters obtained in this work.
Figure 3. Relation between the LHS otequatlon (5) plus the sec-
Transferring the second-order interaction parameter
ond-order interaction parameterterms and 3[%AI]+3.37[%O] term
terms in equation (4) to the LHS, the relation between the
LHS and 3[%AI] + 3.37[%0] terms is then reasonably
plotted as a straight line in figure 3. The line is well satis-
fied with the reported data and present experimental results
0 over the concentration range considered.
The relation between aAI(obs) and aAl(cal) is shown in
figure 4. The values of aAI(obs) were determined directly
-1
"......
from the contents of solutes using the available values of
'"
'8 various interaction parameters. On the other hand, the
'-' -2 values of aAI(cal) were calculated from equation (7) using
~ the equilibrium constant and the values of a Al203 and ao
OIl
.s -3 •
0
Present
Fruehan determined in this work:
0 Rhode
/1 Dimitrov
-4 log KAI + log a Al203 - 3 log ao = 2 log aAI (7)
e Suito
-5 Il::.-""---L.......... _ _- - '
--JL....-..i..-...J....--'-~_"----....L.-
where ao and aAI denote the activities of oxygen and alu-
-5 -4 -3 -2 -1 0 minum in liquid iron relevant to the 1 % Henrian standard
log aAl (cal) state, respectively. It can be seen from figure 4 that the
corresponding values of aAI(obs) and aAI(cal) are reasona-
Figure 4. Plotofthe relation between aAI(obs) determined directly
from the contents of solutes as well as the available values of bly in good agreement with each other. This result implies
various interaction parametersand aAI(cal) determined from equa- that the values of interaction parameters experimentally
tion (7) estimated in this work seem to be reliable.
steel research 69 (1998) No.2 51
1869344xa, 1998, 2, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/srin.199801342 by University Of Wollongong University Of Wollongong Library, Wiley Online Library on [28/02/2024]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
Process metallurgy
function of temperature can be derived:
•
0
Present
Fruehan
log KAI = 12.32 - 474001T, (8)
D Rhode
eoAl = 15.57 - 365001T. (9)
A Dimitrov
0 Suito
The values of log KAt and e~t at 1873 K are found to be
-12.99 and -3.92, respectively. The fact that these values
Dca • obtained from equations (8) and (9) agree well with those
determined earlier using equation (3) confirms the validity
-2 of equations (8) and (9).
The equilibrium relations between aluminum and oxy-
gen contents are shown in figure 8, which were calculated
from equation (2) by the iterative method with interaction
0.2 0.4 0.6 0.8 1.0 1.2 parameters and equilibrium constants. It is shown in this
figure that most of the calculated lines from the results of
[o/oAl]
other investigators [6; 8; 11; 12] who only considered first-
Figure 5. Plot of the logarithmic activitycoefficient of oxygen as a order interaction parameters seem to be generally incon-
function of aluminum contentin liquid iron sistent with the experimental data. The calculated line for
the results of Sigworth et al. [16] who considered first- and
The activity coefficient of oxygen, fa is plotted as a second-order interaction parameters fits the experimental
function of aluminum concentration in figure 5. The val- data more closely, but seems to deviate largely from the
ues of log fa show a linear relationship up to 0.2 % AI. data at the high aluminum concentration range of 0.2 <
This result indicates that the consideration of first-order [%AI] < 1 %. However, the calculated line taking into
interaction parameters for the activity calculation could be consideration interaction parameters and equilibrium con-
only valid in the concentration region of [%AI] < 0.2 %, stant determined in this work satisfies fairly well the equi-
where the effect of second-order interaction parameter librium data over the whole concentration range consid-
terms could be negligible. However, at higher aluminum ered. This finding also assumes that equation (6) represents
concentrations the log fa is no longer a linear function of the convincing values of equilibrium constant and interac-
aluminum content and the effect of second-order interac- tion parameters.
tion parameters on the activity becomes more significant.
The determination of temperature dependence of the Conclusions
equilibrium constant, log KAI for the deoxidation reaction
The deoxidation reaction of aluminium in liquid iron
and the first-order interaction parameter, e~1 is graphi- was assessed thermodynamically at 1873K using the pre-
cally shown in figures 6 and 7 used only for the previ- viously reported data and present experimental results. The
ously reported data and present result, respectively. In equilibrium constant, KAt for the aluminum deoxidation
those figures the relatively low-to-high range of aluminum reaction and the values of first- and second-order interac-
concentrations "{ere adapted from the reported data. From tion parameters between aluminum and oxygen in liquid
the slope and intercept values in figures 6 and 7, the equi- iron were derived by a multiple regression method as fol-
librium constant and first-order interaction parameter as a lows:
-6
-7 •
0
Present
FIUeban 0 •
0
Present
FlUeban
-8 D d'Entremont [31 D d'Entremont
D
-I D
A McLean [I] A McLean
-9
0 Kobayashi [5] 0 K.obayashi
D -
-10 v Swisher [4] -2
:(
ll< <0
CU
l'>O -11 -3 e
.!a
-12
-4
-13
log K AI - 12.32 - 47,400rr -5 e~ = 15.57 - 36,500IT
-14
-15 L -........-L-.........---I_"--....L...-..........- - ' - - - - " _ L -.........-1 ·6
4.4 4.6 4.8 5.0 5.2 5.4 5.6 4.4 4.6 4.8 5.0 5.2 5.4 5.6
Iff (x 10 4
) Iff (x 10~
Figure 6. Temperature dependence of equilibrium constants for Figure 7. Temperature dependence of first-order interaction para-
the aluminum deoxidation reaction metersbetween aluminum anq oxygen in liquid iron
52 steel research 69 (1998) No.2
1869344xa, 1998, 2, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/srin.199801342 by University Of Wollongong University Of Wollongong Library, Wiley Online Library on [28/02/2024]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
Process metallurgy
3.0 log KAI = 12.32 - 474001T,
alolp, =1
2.5 Al
eo = 15.57 - 365001T.
2.0 ./
1.5 The equilibrium relation between aluminum and oxygen
~ 1.0
contents in the aluminum deoxidized iron could be ther-
o modynamically well described over the concentration
j; 0.5
o range up to 1 % with the first- and second-order interaction
== l
Experimental data
DO 0.0 parameters as well as the equilibrium constant determined
..s -0.5 in this work .
....... .Dimitrov Calculated
-1.0 (A 01337; received: 05. August 1997)
-_._.- Coo [lIJ values
-1.5 --- Gustafsson [8J References
-2.0 - - Sigworlh [16]
-2.5 l.-.-I----"_....L..---o-_.L.-.-I-----I_-'----I.._.........--' [I] McLean, A.; Bel/, H B.: 1. Iron and Steel Inst, 203 (1965), p.
-4 -3 -2 -1 0 123/30.
[2] Mcl.ean, A.; Ward, R. G.: J. Iron and Steel Inst. 204 (1966), p.
log[o/oAl]
8111.
Figure 8. Comparison of experimental data with thermodynami- [3] d'Entremont, J. C: Guernsey, D. 1.; Chipman, J.: Trans. Met Soc.
cally calculated relation between aluminum and oxygen contents AIME 227 (1963), p. 14/17.
for the equilibrated liquid iron at 1873 K [4] Swisher. J. H: Trans. Met Soc. AIME 239 (1967), p. 123/24.
[5] Kobayashi, K; Omori. Y.; Sanbongi, K: Tetsu-to-Hagane 53
(1967), p. 141/44.
log KAI = -12.96 ± 0.14, [6] Fruehan, R. J.: Metallurg. Trans. I (1970), p. 3403/10.
[7] Rhode, 1. E.; Choudhury. A.; Wahlster, M.: Arch. Eisenhiittenwes.
42 (1971), p. 165/74.
e~1 = -4.09 ± 0.41, r~1 = 2.67 ± 0.47, [8] Gustafsson, s, Mel/berg. r-o.. Scand. J. Metall. 9 (1980), p.
111/16.
[9] Suito, H; Inoue. H; Inoue. R.: ISH Int. 31 (1991), p. 1381/88.
e~1 = -6.90 ± 0.69, r~1 = 7.60 ± 1.34, [10] Holcomb, G. R.; St. Pierre, G. R.: Metallurg. Trans. 23B (1992), p.
789/90.
[II] Cho, S. w.; Suito, H: ISH Int. 34 (1994), p. 177/85.
r~I,O (r11'0) = 9.05 ± 1.59 (9.05 ± 1.59). [12] Dimitrov, S.; Weyl. A.; Janke. D.: steel res. 66 (1995), p. 3/7.
[13] St. Pierre, G. R.: Metallurg. Trans. 8B (1977), p. 215/17.
The temperature dependence of the equilibrium constant [14] Steelmaking Data Source Book, rev. edn. Jap. Soc. Promot. Sci.,
Gordon and Beach Sci. Pub!., New York/London/ParisIMon-
and the first-order interaction parameter, e~1 by using treaux/Tokyo/Melboume, 1988, p. 45/53.
previously reported data and present results were also [15] Lupis, C. H P.; Elliott. 1. F.: Acta Metal!. 14 (1966), p. 529/38.
obtained: [16] Sigworth, G. K; Elliott. J. F.: Metal, Sci. 8 (1974), p.298/310.
steel research 69 (1998) NO.2 53
Das könnte Ihnen auch gefallen
- Grundlagen Der KorrosionDokument38 SeitenGrundlagen Der KorrosionHagen_of_TronjeNoch keine Bewertungen
- Thermodynamics of Antimony, Arsenic and Tin in SteelDokument7 SeitenThermodynamics of Antimony, Arsenic and Tin in SteelNishan GajurelNoch keine Bewertungen
- (1962) The Oxidation of Iron-Chromium Alloys and Stainless Steels at High TemperaturesDokument24 Seiten(1962) The Oxidation of Iron-Chromium Alloys and Stainless Steels at High TemperaturesChitu IulianNoch keine Bewertungen
- 1973 Kinetics of Iron Passivation and Corrosion in Molten Alkali NitratesDokument12 Seiten1973 Kinetics of Iron Passivation and Corrosion in Molten Alkali NitratesAdrian CaraballoNoch keine Bewertungen
- France 1997Dokument11 SeitenFrance 1997KlockerNoch keine Bewertungen
- Abiturprüfung 2023: Chemie, LeistungskursDokument37 SeitenAbiturprüfung 2023: Chemie, LeistungskursSuper NerdNoch keine Bewertungen
- Andrade 1978Dokument5 SeitenAndrade 1978RAGHUNoch keine Bewertungen
- DodecahydrotriphenyleneDokument7 SeitenDodecahydrotriphenylene06林品睿-三色豆冰棒Noch keine Bewertungen
- 1974-Svedung - The Influence of Silicon On The Oxidation Properties of IronDokument10 Seiten1974-Svedung - The Influence of Silicon On The Oxidation Properties of IronDavid Hernández EscobarNoch keine Bewertungen
- TUT - Molare Massen, Stoffmengen Und Chemische GleichungenDokument3 SeitenTUT - Molare Massen, Stoffmengen Und Chemische Gleichungenteipel21Noch keine Bewertungen
- The Mechanism of Stress - Corrosion CrackingDokument14 SeitenThe Mechanism of Stress - Corrosion CrackingDaniel FrancoNoch keine Bewertungen
- ElektrometallurgieDokument19 SeitenElektrometallurgieMatias Garcia LabrañaNoch keine Bewertungen
- Stiller 1984Dokument11 SeitenStiller 1984hp2114bNoch keine Bewertungen
- 2-8 Gr25 SL 2022Dokument10 Seiten2-8 Gr25 SL 2022Sakon UkonNoch keine Bewertungen
- Chemie Heft Raj 2022-2023 9BDokument7 SeitenChemie Heft Raj 2022-2023 9BArdra RajNoch keine Bewertungen
- Hacker Man 1967Dokument12 SeitenHacker Man 1967Piotr PołczyńskiNoch keine Bewertungen
- Einfluß Von Beschichtungen Auf Das Schwingungsrißkorrosionsverhalten Des Chromstahls X20Cr13-SchmittDokument9 SeitenEinfluß Von Beschichtungen Auf Das Schwingungsrißkorrosionsverhalten Des Chromstahls X20Cr13-SchmittAntonioNoch keine Bewertungen
- Elektrometallurgie AluminiumDokument4 SeitenElektrometallurgie Aluminiummonika4512123Noch keine Bewertungen
- smww.3000 METALESDokument140 Seitensmww.3000 METALESfisicoquimica2amfacNoch keine Bewertungen
- t8 f99Dokument14 Seitent8 f99Wanda AfNoch keine Bewertungen
- Chemie 11.12.2023Dokument22 SeitenChemie 11.12.2023TheMorpheasNoch keine Bewertungen
- Korrosion: SchulversuchspraktikumDokument15 SeitenKorrosion: SchulversuchspraktikumasdfasddfNoch keine Bewertungen
- 01 - LK2016 - HT - Redox-Gold Fälschen - LösungDokument9 Seiten01 - LK2016 - HT - Redox-Gold Fälschen - Lösungc0181511Noch keine Bewertungen
- High Temperature Oxidation Behaviour of BoilerDokument171 SeitenHigh Temperature Oxidation Behaviour of BoilerMas ZuhadNoch keine Bewertungen
- Reinhold GKDokument8 SeitenReinhold GKyogolainNoch keine Bewertungen
- Protokoll V09Dokument7 SeitenProtokoll V09Marvin StruveNoch keine Bewertungen
- Coek - Info - Anodic Polarization of Platinum in Sodium ChlorideDokument10 SeitenCoek - Info - Anodic Polarization of Platinum in Sodium ChlorideEdgar HornusNoch keine Bewertungen
- Umweltchemie: Eine Einführung mit Aufgaben und LösungenVon EverandUmweltchemie: Eine Einführung mit Aufgaben und LösungenNoch keine Bewertungen
- DR S Hogewoning Dependence of Hard Burn Potential On Limestone Properties-EnglischDokument17 SeitenDR S Hogewoning Dependence of Hard Burn Potential On Limestone Properties-EnglischHuyentrang NguyenNoch keine Bewertungen
- MetallkundeDokument32 SeitenMetallkundeColumbia GomezNoch keine Bewertungen
- Investigations On Anode Quality in Copper Electrorefining: C. Wenzl, I. Filzwieser, G. Mori and J. PeslDokument6 SeitenInvestigations On Anode Quality in Copper Electrorefining: C. Wenzl, I. Filzwieser, G. Mori and J. PeslNidhiNoch keine Bewertungen
- Hpa-001 1962 35 817 DDokument19 SeitenHpa-001 1962 35 817 DAhmed LaibiNoch keine Bewertungen
- Chemie 2022Dokument16 SeitenChemie 2022Jessica KuhnholdNoch keine Bewertungen
- Galvanotechnik - ReferatDokument10 SeitenGalvanotechnik - Referatsolvalou_Noch keine Bewertungen
- RedoxreaktionenDokument15 SeitenRedoxreaktionenOdranoel RellekNoch keine Bewertungen
- Elektrochemische WasserenthrtunggwfDokument8 SeitenElektrochemische WasserenthrtunggwfArturo Avellaneda TauchertNoch keine Bewertungen
- Baar - 1911 - On The Alloys of Molybdenum With Nickel, Manganese With Thallium, and Calcium With Magnesium, Thallium, Lead, Copper, andDokument44 SeitenBaar - 1911 - On The Alloys of Molybdenum With Nickel, Manganese With Thallium, and Calcium With Magnesium, Thallium, Lead, Copper, andshyamNoch keine Bewertungen
- 0255 2701 (84) 85013 8Dokument10 Seiten0255 2701 (84) 85013 8LTORRESMNoch keine Bewertungen
- H Xi Tillner-Roth Friend 2006 KretzschmarDokument7 SeitenH Xi Tillner-Roth Friend 2006 KretzschmarĐại Đỗ ĐứcNoch keine Bewertungen
- Stöch - Für TestDokument1 SeiteStöch - Für Testsiegfried.ender.martinNoch keine Bewertungen
- Skript Hochtemperaturwerkstoffe Uni SiegenDokument158 SeitenSkript Hochtemperaturwerkstoffe Uni SiegenArnold MasanjaNoch keine Bewertungen
- 12.2 - 4 - Technische VerfahrenDokument2 Seiten12.2 - 4 - Technische VerfahrensmintermanNoch keine Bewertungen
- Koul 1983Dokument9 SeitenKoul 1983Puli SuhasNoch keine Bewertungen
- TSP 231 20241Dokument8 SeitenTSP 231 20241ruben dario guerra chirinosNoch keine Bewertungen
- Gesetz B5 HEAT-GI S2021 LaborberichtDokument7 SeitenGesetz B5 HEAT-GI S2021 LaborberichtScribdTranslationsNoch keine Bewertungen
- Electropolishing of Copper and Copper-Based Alloys in Ortho-Phosphoric Acid PDFDokument8 SeitenElectropolishing of Copper and Copper-Based Alloys in Ortho-Phosphoric Acid PDFmohammadNoch keine Bewertungen
- Nanostrukturierte Aluminiumfluoridschichten: Über das neuartige Niedertemperatur Sol-Gel Verfahren und die charakteristischen EigenschaftenVon EverandNanostrukturierte Aluminiumfluoridschichten: Über das neuartige Niedertemperatur Sol-Gel Verfahren und die charakteristischen EigenschaftenNoch keine Bewertungen
- 015 POTMPH IGF18297 AbschlussDokument2 Seiten015 POTMPH IGF18297 Abschlussmanjumv27Noch keine Bewertungen
- Chimiaadmin,+1994 0444Dokument4 SeitenChimiaadmin,+1994 0444Khoa ĐặngNoch keine Bewertungen
- Reindarstellung, Kristallzuchtung Und Kristallstrukturbestimmung Von IodtrioxidDokument6 SeitenReindarstellung, Kristallzuchtung Und Kristallstrukturbestimmung Von Iodtrioxidrob-1982Noch keine Bewertungen
- 978-3-540-70882-7 - 8 BrennstoffzelleDokument8 Seiten978-3-540-70882-7 - 8 BrennstoffzelleAnedo 1909Noch keine Bewertungen
- Lernheft 2.3 LernendeDokument24 SeitenLernheft 2.3 Lernendevalricrom UnbekanntNoch keine Bewertungen
- Stadtaus 2003Dokument7 SeitenStadtaus 2003Owais AhmedNoch keine Bewertungen
- B040BCD9-1D30-41BE-B20F-F4FCB84AC341 (2)Dokument2 SeitenB040BCD9-1D30-41BE-B20F-F4FCB84AC341 (2)John WongNoch keine Bewertungen
- Kap 07Dokument52 SeitenKap 07DAOUDI nadaNoch keine Bewertungen
- KFK3103Dokument139 SeitenKFK3103Rosendo FazzariNoch keine Bewertungen
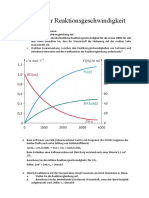
- AB Übungen REaktionsgeschwindigkeitDokument2 SeitenAB Übungen REaktionsgeschwindigkeitphaos85Noch keine Bewertungen
- A970DEDokument8 SeitenA970DEMick JarrNoch keine Bewertungen
- Rheological Properties of Acrylic LaticesDokument4 SeitenRheological Properties of Acrylic LaticesAlex AcevedoNoch keine Bewertungen
- Ets QGDokument2 SeitenEts QGYaretzi RuizNoch keine Bewertungen